Abstract
There exist indications that sense of agency (SoA), the experience of being the cause of one’s own actions and actions’ outcomes, is altered in autism. However, no studies in autism have simultaneously investigated the integration mechanisms underpinning both implicit and explicit SoA, the two levels of agency proposed by the innovative cue integration approach. Our study establishes a first complete characterization of SoA functioning in autism, by comparing age- and IQ-matched samples of autistic versus neurotypical adults. Intentional binding and judgments of agency were used to assess implicit and explicit SoA over pinching movements with visual outcomes. Sensorimotor and contextual cues were manipulated using feedback alteration and induced belief about the cause of actions’ outcome. Implicit SoA was altered in autism, as showed by an overall abolished intentional binding effect and greater inter-individual heterogeneity. At the explicit level, we observed under-reliance on retrospective sensorimotor cues. The implicit-explicit dynamic was also altered in comparison to neurotypical individuals. Our results show that both implicit and explicit levels of SoA, as well as the dynamic between the two levels, present atypicalities in autism.
Keywords
Motor impairments and action control alterations are highly prevalent in autism, and a growing number of researchers are calling for their inclusion as core clinical features of autism (e.g., Bhat et al., 2011; Fournier et al., 2010; Mostofsky & Ewen, 2011; Wilson et al., 2018). With growing evidence that socio-communicative autistic symptom severity is associated with the intensity of motor and action control alterations (Dziuk et al., 2007; Gizzonio et al., 2015; Ohara et al., 2020) and that motor alterations could precede social difficulties in development (Esposito et al., 2009), it has been suggested that sensorimotor dysfunctions (Mosconi & Sweeney, 2015) and motor cognition alterations (Casartelli et al., 2016) could constitute a core cognitive component of autism, underpinning the better-recognised socio-communicative manifestations of autism.
The sense of agency (SoA) refers to the subjective experience of being the cause of one’s own thoughts and actions (Gallagher, 2000; Haggard, 2017) and to the capacity to distinguish their consequences in the external world from the consequences of others’ actions (Balconi, 2010). It is a cognitive function classified as a construct of the sensorimotor system by the Research Domain Criteria framework (National Institute of Mental Health, 2019), and its study offers a conceptual framework creating bridges between internal, sensorimotor processes and interpersonal, social processes. The study of SoA in the general population and in several neuropsychiatric conditions, such as schizophrenia and obsessive-compulsive disorder (OCD), has drawn a growing amount of interest in the last decade and has led to significant advances in theoretical models. Even if the study of SoA offers a promising framework to characterise the autistic cognitive phenotype, allowing to better explore the presence of concomitant socio-communicative and sensorimotor impairments, only a limited number of studies have specifically investigated SoA in autism (see the articles by Lafleur et al., 2016; Zalla & Sperduti, 2015 for detailed reviews). Moreover, the majority of these studies have not been designed based on promising recent theoretical models, such as the cue integration approach (Moore & Fletcher, 2012; Synofzik et al., 2013; Synofzik, Vosgerau, & Newen, 2008).
The cue integration approach postulates that SoA can be divided into an implicit and an explicit level that serve different functions (Synofzik, Vosgerau, & Newen, 2008). The cue integration approach also establishes that different sensorimotor and contextual cues are integrated as a function of their availability and reliability at each level (Moore & Fletcher, 2012; Synofzik et al., 2013) to allow an optimal experience of agency and effective motor control. Although there exists early evidence that SoA is altered in autism (Sperduti et al., 2014; Zalla et al., 2015), a complete characterization of the functioning of the two levels of SoA is yet to be achieved, and the integration mechanisms that may be altered at both levels remain unknown.
The SoA framework also shares interesting bridges with self-other distinction difficulties in autism. Self-other distinction allows to implicitly or explicitly differentiate one own’s sensations, knowledge, and feelings from those of others (Simantov et al., 2021) and has drawn sustained attention in the study of the autistic cognitive phenotype. Self-other distinction is studied in the emotional, cognitive, and sensory-motor domains (Eddy, 2022). At the low-level, sensory-motor level, evidence of self-other distinction difficulties in autism comes from studies showing altered motor imitation abilities (see the artice of Edwards, 2014 for an extensive review), self-touch perception alterations (Deschrijver et al., 2017), and implicit SoA atypicalities (Sperduti et al., 2014). Sensory-motor self-other distinction is thought to be associated with clinical manifestations of autism such as hyper-imitation (Spengler et al., 2010), echopraxia/echolalia (Eddy, 2022; J. H. Williams et al., 2004), and tics/behavioural compulsions (Gadow et al., 2008; Uljarević et al., 2022). At the cognitive, higher-order mentalizing level, manifestations of self-other distinction difficulties include theory-of-mind and perspective-taking deficits (Baron-Cohen et al., 1999; Peñuelas-Calvo et al., 2019; Simantov et al., 2021), altered meta-cognitive aspects of SoA (Zalla et al., 2015), and psychological self-representations (Uddin, 2011). Empathy, dependant on affective self-other differentiation, is also hypothesised to be imbalanced in autism with higher emotional empathy but reduced cognitive empathy (Shalev et al., 2022).
Implicit SoA in neurotypical and autistic individuals
Implicit SoA, which consists of a pre-reflexive representation of action as being caused by oneself or not (Synofzik, Vosgerau, & Newen, 2008), is largely rooted in the sensorimotor system and allows rapid registration of agency, fast motor adaptation (Desmurget & Grafton, 2000), and reduction of noise in the sensory system via sensory attenuation (Blakemore et al., 1998). Implicit SoA is conceptualised from an earlier influential model of SoA (comparator model of SoA; Frith et al., 2000). In this account, the execution of a goal-directed movement relies on the formation of internal models of action. The intention to produce a trained goal-directed action leads to the selection of an inverse model (Kawato, 1999), the motor plan that is the most likely to achieve the selected goal. The inverse model then gives rise to a motor command that is sent to the limbs to execute the movement and to a forward model that contains predictions about the sensory consequences of the movement (Miall et al., 1993). The forward model is then compared to actual sensory feedback in the posterior parietal cortex, likely in the inferior parietal lobule (David, 2010). According to the comparator model of SoA, a match between expected and actual sensory feedback leads one to perceive the action and its outcomes as self-caused. Information issuing from this “comparator” can be seen as a retrospective sensorimotor cue to SoA as it is available after the execution of action.
Self-produced actions are also accompanied by a subjective shift in the perception of the time of occurrence of both the action and the outcome towards one another, a phenomenon coined as intentional binding (IB; Haggard et al., 2002). This robust phenomenon occurs only in the presence of a voluntary movement (Engbert et al., 2007; Moore & Obhi, 2012) and is a commonly used proxy of implicit SoA. It has been shown that, when the action’s outcome is highly predictable, the temporal shift of the action towards the expected outcome still occurs even if the outcome is actually not presented (Moore & Haggard, 2008). This indicates that implicit self-agency can emerge despite a mismatch at the comparator and that signals preceding action execution, such as the forward model containing the sensory predictions, largely contribute to implicit SoA and to the IB effect. Those prospective sensorimotor cues, that is, cues available before the execution of action, include forward models of action but also action-selection signals (Chambon & Haggard, 2012) and free choice over action (Barlas & Kopp, 2018), as they have been shown to increase IB strength. Neurofunctional studies of IB have also identified that temporary disruption of the pre-supplementary motor area (p-SMA), a region thought to participate in translating intentions into motor commands (David, 2010), causes a significant decrease in the strength of the binding (Cavazzana et al., 2015; Moore et al., 2010).
To our knowledge, only two studies have investigated IB in autism: one in both the auditory and visual modalities (Sperduti et al., 2014) and one in the auditory modality only (Finnemann et al., 2020). These studies suggest that the IB effect is abolished in autistic adults in the visual modality but preserved in the auditory modality. The abolition of the IB effect for action with visual outcomes provides early evidence that implicit SoA is altered in autism and is compatible with studies finding alterations of prospective control of visually guided action (Papadopoulos et al., 2012; Schmitz et al., 2003; Wang et al., 2015) and motor planning difficulties (Dziuk et al., 2007; Kaur et al., 2018; MacNeil & Mostofsky, 2012) in autism. Findings of Sperduti et al. (2014) are also coherent with the emerging account of autism as a disorder in predictive coding (Lawson et al., 2014; Sinha et al., 2014) stipulating an imbalance between the precision ascribed to sensory evidence and prior beliefs in different domains such as sensorimotor and social predictions. Although alterations in sensorimotor predictive mechanisms gather more and more attention in autism research, it is still necessary to confirm if implicit SoA alterations in autism specifically arise from difficulties with the integration of prospective sensorimotor cues.
Explicit SoA in neurotypical and autistic individuals
The explicit level of SoA can be described as a meta-cognitive, conscious experience of agency in which one attributes the causality of an action and its outcomes to a specific agent (Synofzik, Vosgerau, & Newen, 2008). It is measured by verbal reports that are also called judgments of agency (JoA). The cue integration process at the explicit level integrates previously discussed sensorimotor cues with other contextual cues such as background beliefs about plausible causes of stimuli in the environment, evaluation of performance, and emotional valence of outcome (Synofzik et al., 2013). For example, explicit SoA over one’s own thoughts has been experimentally reduced by placing participants in a mock scientific apparatus allegedly capable of thought insertion (Olson et al., 2016). Explicit SoA over observed movements performed by others have also been elicited by coincidental instructions to move (Wegner et al., 2004). Meta-cognitive JoA are associated with increased activity in the anterior prefrontal cortex, a region linked to self-reflective processing (Miele et al., 2011). Synofzik and colleagues’ cue integration model also implies that the two levels of agency are sequential and that the registration of agency at the implicit level is in itself a cue that can contribute to the integration process at work at the explicit level.
Before the emergence of contemporary theories of SoA, Russell (1996) and Russell and Hill (2001) hypothesised that a deficit in action monitoring, a concept that resembled the actual definition of SoA, could constitute a precursor of autistic socio-communicative difficulties. This hypothesis gave rise to three studies (Grainger et al., 2014; Russell & Hill, 2001; D. Williams & Happé, 2009) using almost identical experimental paradigms, in which autistic children had to discriminate a visual stimulus under their control from distractors that moved randomly. Detection performances of autistic children were equivalent to those of neurotypical children in all those studies. Early work on SoA in autism also include a study by David et al. (2008) conducted with autistic adults and using visual feedback alteration of a joystick trajectory controlled by the participants. SoA was assessed via dichotomic JoA only, and no differences in recognition of altered/unaltered feedback were found. However, as none of these experiments were designed with the influence of the cue integration approach of SoA, these tasks could not untangle the relative contributions of sensorimotor and contextual cues to JoA. Equivalent detection performances could be based on underlying atypical cue integration in autistic individuals. This hypothesis seems supported by a later study by Zalla et al. (2015) exploring explicit SoA in autistic adults but using an innovative experimental task. Participants had to attain X targets with a cursor controlled by a computer mouse. Lag was introduced in the cursor to induce unreliability in sensorimotor signals. Contextual cues were varied by offering feedback on performance, as success to attain an “X” target was followed by a specific sound. The authors found that autistic individuals based their JoA mainly on performance feedback and less on cursor/movement congruency than neurotypical individuals, suggesting an under-reliance on sensorimotor cues in explicit SoA in autism. Interestingly, this tendency was also correlated to lower performances on the Faux-Pas Recognition Task (Baron-Cohen et al., 1999), assessing theory-of-mind abilities. However, although Zalla and Sperduti (2015) have hypothesised alteration of prospective but spared retrospective mechanisms, the relative contribution of prospective/retrospective sensorimotor and contextual cues to explicit SoA have yet to be assessed in an experiment where they can be untangled.
To our knowledge, no studies have investigated simultaneously implicit and explicit levels of SoA in an autistic sample. We believed this lack of investigation arise from the challenge of developing a psychophysics task assessing both implicit and explicit proxies on a trial-by-trial basis. Hence, we were also interested in running an exploratory analysis of the dynamic between those two levels and in evaluating the relative contribution of implicit registration of agency to the explicit level in autistic individuals.
Characterising the autistic cognitive profile for SoA by specifying cue integration processes
The objectives of the present experiment were threefold. First, we sought to provide a first complete characterization of the autistic cognitive profile for SoA using the cue integration approach of SoA. Second, we intended to determine if autistic individuals presented altered cue integration processes at implicit and explicit levels of SoA, by examining the relative contributions of prospective/retrospective sensorimotor and contextual cues. Finally, in an exploratory analysis, we wanted to examine the relative contribution of the implicit registration of agency to the explicit level and to characterise the dynamic between the two levels of SoA in autistic individuals.
To do so, we administered to a sample of autistic adults and to neurotypical matched controls, a psychophysics task simultaneously assessing implicit and explicit SoA for a given action, using IB and dichotomic JoA, respectively. Participants were asked to perform multiple pinching movements on a dynamometer for which we controlled SoA cues’ reliability. Retrospective sensorimotor and contextual cues were respectively manipulated with feedback alteration and inducement of a belief about the number of agents that could be the cause of action’s outcome. For implicit SoA, we expected reduced IB effect in autistic individuals, as a result of under-reliance on available prospective sensorimotor cues. For explicit SoA, we anticipated that autistic individuals would base their JoA to a greater extent on contextual cues than on sensorimotor cues. As our third objective was exploratory in nature, we did not have a priori hypotheses.
Methods
Participants
Twenty-three adults with a clinical diagnosis of either autism spectrum disorder, according to Diagnostic and Statistical Manual of Mental Disorders- Fifth edition (DSM-5; American Psychiatric Association, 2013), or Asperger syndrome or autism according to Diagnostic and Statistical Manual of Mental Disorders – Fourth edition - Text Revision (DSM-IV-TR; American Psychiatric Association, 2000) were recruited from the participants database of a specialised autism clinic and from an online add. All diagnoses were established by expert psychiatrists or expert psychologists based on clinical interviews and clinical questionnaires. A few participants were diagnosed at external clinics, but the research team revised every evaluation report to ensure the validity of the diagnosis. Because certain comorbidities are frequently associated with autism, autistic participants who reported having a diagnosis of attention-deficit/hyperactivity disorder (ADHD), depression or anxiety disorder, and/or the use of antidepressant, anxiolytic, or a psychostimulant medication were included in the study. Potential participants reporting other psychiatric diagnoses and/or the use of other classes of psychoactive medication were excluded.
Twenty-two neurotypical volunteers were also recruited to be part of a control group matched for age, intellectual functioning (IQ), and gender. Neurotypical participants were screened to ensure none had a previous history of psychiatric or neurodevelopmental disorders or were using, on a regular basis, medication that could alter psychological functioning.
Two participants, one autistic and one neurotypical, were excluded from the data analysis after admitting lack of compliance to task instructions, so the final sample included 22 autistic individuals and 21 neurotypical individuals (see Table 1). Independent t-tests and a Pearson’s chi-square test of homogeneity were conducted to verify whether groups differed on age, gender, and intellectual functioning. No significant differences were observed between the two groups on any of those parameters.
Characteristics of the two final samples.
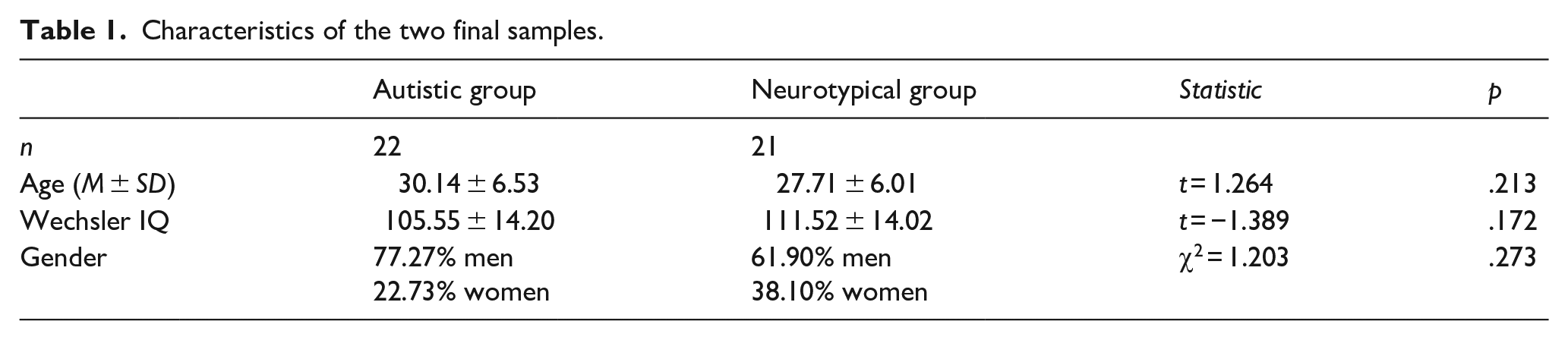
All recruited participants reported being right-handed or ambidextrous, having no diagnosed neurological or motor disabilities, and having a normal or corrected to normal vision. Participants’ intellectual functioning scores were obtained via the participants’ database or were assessed with the Wechsler Adult Intelligence Scale–Fourth Edition (WAIS-IV; Wechsler, 2008) after the completion of the experimental task. All participants had a full-scale IQ score of minimally 80 (Wechsler Intelligence Scale for Children-Third edition [WISC-III], Wechsler Adult Intelligence Scale–Third Edition [WAIS-III], or WAIS-IV). The study was approved by local health authorities ethics committee. Written informed consent was obtained from participants before the study and renewed after the false belief procedure.
SoA task
We used an SoA psychophysics task validated in the general population (Lafleur et al., 2020). Participants were seated in front of a computer screen, with the right forearm laying down in a custom-made arm brace with a 90° flexion. Participants used their right-hand thumb and index to perform pinching movements on a strain-gauge isometric force sensor (hand dynamometer, Vernier Software and Technology, Beaverton, OR). The device was calibrated to participants’ maximal voluntary contraction (MVC). Pinching movements were followed by the apparition of a green flash on the screen, and the height of this flash was determined by the force applied on the dynamometer. Interval reproduction (implicit SoA measure) and dichotomic JoA (explicit SoA measure) were obtained after each trial via computer mouse buttons that were controlled by participants’ left hand. We employed the temporal reproduction method developed by Humphreys and Buehner (2010), consisting in the reproduction of the perceived interval by a sustained mouse click.
The SoA task was preceded by a learning phase that allowed the formation of an internal model of action for pinching movements and their visual outcomes. It consisted of a total of 90 interleaved (3 × 30) trials where participants pressed on the dynamometer to hit a target corresponding to 20%, 50%, and 80% of the participant’s MVC. Participants were asked to press on the dynamometer with a simple, quick pinching movement, and they were informed that a harder press on the dynamometer would result in the flash appearing higher on the screen. On each trial, the green flash appeared on the screen after a random delay of 600–1200 ms at a height corresponding to the force employed. Participants could learn to control the dynamometer by comparing the intended height with their actual performance. The learning phase allowed the participant to integrate the baseline force applied/green flash height relationship present in the no alteration condition.
The main task was then performed by participants under two different social contexts: the One-agent and Two-agent contexts, whose order was counterbalanced across participants. Prior beliefs about agency were manipulated with instructions given to participants before the completion of each context. The main task consisted of a series of trials under four conditions: no alteration, small alteration, large alteration conditions (those three taken together, operant conditions) and an observation condition (see presentation details of the trials in Figure 1). In the operant conditions, participants were asked to freely select a mental target height on the screen and to perform a pinching movement on the dynamometer to cause a green flash to appear after a random delay. The random delay of 600–1200 ms was programmed to start at the time of the force peak and to end with the apparition of the green flash. In the no alteration condition, the green flash appeared on the screen at the height corresponding to the true force applied by participants, according to the force-height relationship of the learning phase. In the small alteration and large alteration conditions, the spatial congruency of the green flash was manipulated by adding or subtracting 20% or 40% of participant’s MVC, respectively. All operant conditions were indistinctively preceded by a black fixation cross, and the beginning of the trial was signalled by the transformation of the cross into a black rectangle. The no alteration condition represented 67% of the operant trials to maintain the baseline force-height relationship learned in the learning phase as the most probable action’s outcome. The small and large alteration conditions were representing a mismatch from this baseline internal model of action. The observation condition was preceded by a white fixation cross, and the beginning of the trial was also signalled by the transformation of the cross into a white rectangle. A predictable white flash of a random duration between 50 and 100 ms was presented at a random height on the screen. After a random delay between 600 and 1200 ms, a green flash appeared at the same position. After each trial of each condition, participants were asked to reproduce the time interval between the end of event #1 (pinching motion or white flash) and the beginning of event #2 (green flash) by holding in one button of the computer mouse. They were also asked to answer whether “Me” or “Not me” caused the green flash.
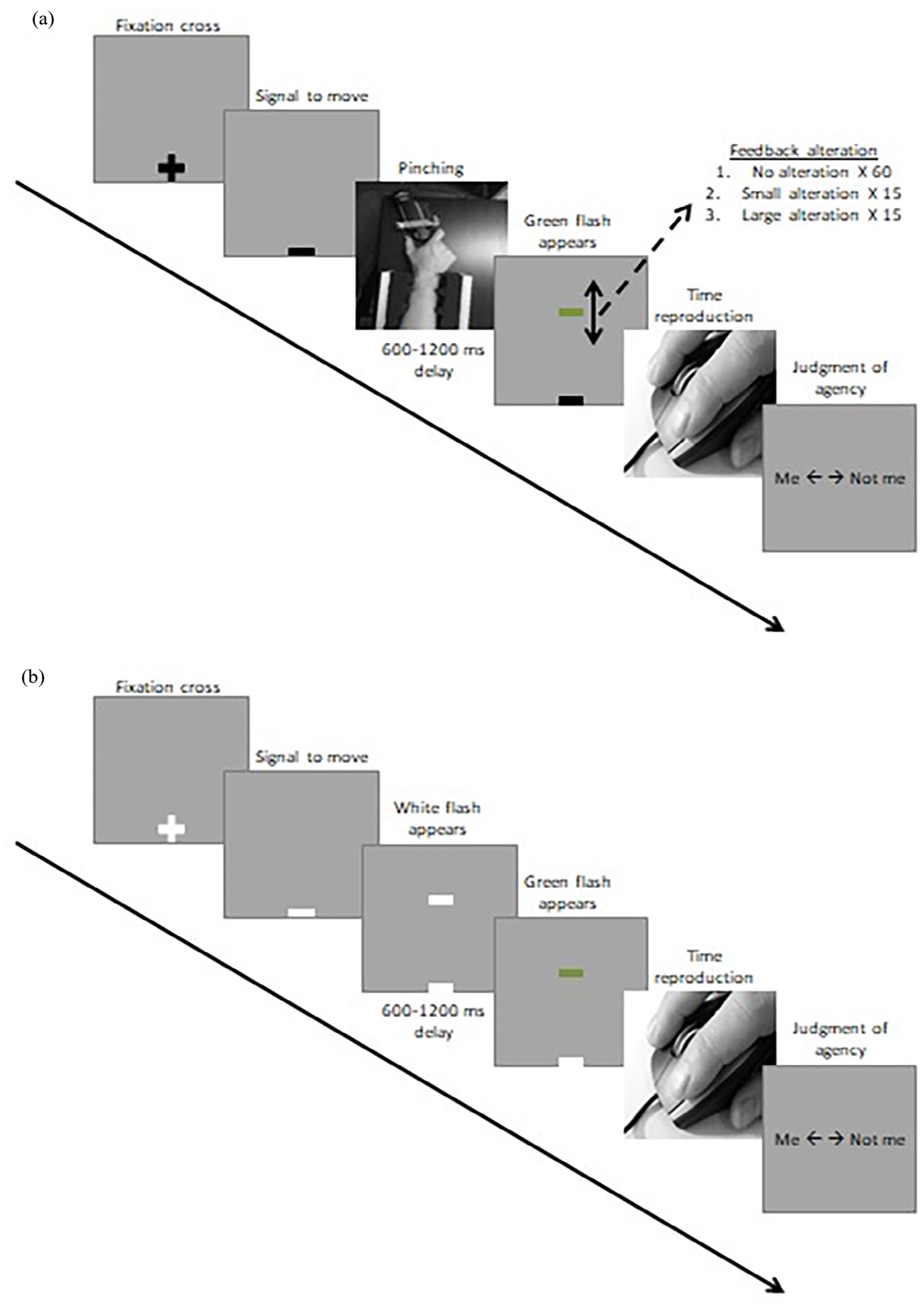
Schematic representation of trials under the operant (a) and observation (b) conditions. Participants were fixating a grey screen on which was displayed a fixation cross. For operant conditions, the black fixation cross turned into a black rectangle signalling to participants that they could perform a pinching movement on the dynamometer with free choice over the force applied. A custom algorithm was programmed to detect deceleration in the movement (acquisition rate of approximately 84 Hz) and to retrieve the maximum force in the movement curve. After a random delay between 600 and 1200 ms starting at the time of maximum force applied, a green flash appeared (1) at the height corresponding to the force/height relationship learned in the learning phase (no alteration, 60 trials), (2) at a height corresponding to the force applied with an alteration of ±20% of participants’ MVC (small alteration, 15 trials), or (3) at a height corresponding to the force applied with an alteration of ±40% of participants’ MVC (large alteration, 15 trials). Trials under each condition were interleaved in a random order in the same block, and participants were not informed of the presence of different feedback alterations conditions. Participants were then asked to reproduce, by holding down the mouse button, the time interval between the end of their pinching movement and the apparition of the green flash. Finally, they were asked to indicate whether they thought the green flash was caused by themselves or someone else. For the observation condition (15 trials), the white fixation cross turned into a white rectangle signalling to participants that they needed to be attentive to the screen. After a fixed, predictable 250 ms delay, a first white flash appeared at a random height. A second green flash followed after a 600–1200 ms random delay. Participants were then asked to reproduce the time interval between the end of the first white flash and the apparition of the second green flash. They were again asked to determine whether they had caused the green flash or not.
The aforementioned task was performed under two contexts. Trial conditions were identical, and the context varied only in instructions given to participants. In the One-agent context, participants were instructed before the start of the block that they would be the only ones controlling the green flashes and that the flashes would appear to the height corresponding to the force/height relationship of the learning phase. Before the Two-agent context, participants were instead instructed that they would control the height of the green flashes for a certain amount of trials only. They were told that the experimenter would be another agent able to control the height of the green flashes on their screen, and this, for an unknown and unpredictable number of trials. In order to induce this false belief in a convincing manner, the experimenter ostentatiously connected a network cable from the experimental device to a second computer located in the same room, but the screen was hidden to participants’ view.
Measures and data analysis
Implicit level: IB
IB was used as an implicit SoA proxy and was measured using a temporal reproduction method. To compare time intervals varying between 600 and 1200 ms, we computed a relative reproduction error (RRE) index for each trial ([subjective inter-events interval − objective inter-events interval] / [objective inter-events interval]). A negative RRE represented the degree to which the objective interval was underestimated by participants. A greater negative RRE on operant conditions than on the observation condition indicated an IB effect. Analyses were run using IBM SPSS statistics v26. We ran a repeated-measures factorial analysis of variance (ANOVA) using a linear mixed model approach (LMM) with Group (autistic, neurotypical), Social context (One-agent, Two-agent), and experimental condition (observation, no alteration, small alteration, large alteration) as independent variables and RRE as a dependent variable. All factors were set as fixed effects, and the variance-covariance structure as compound symmetry.
Explicit level: JoA
We measured the explicit level of agency using a dichotomous verbal proxy (“Me” or “Not me”). We ran a repeated-measures logistic regression using a generalised linear mixed model approach (GLMM) with Group (autistic, neurotypical), Social context (One-agent, Two-agent), and Experimental condition (observation, no alteration, small alteration, large alteration) as independent variables and dichotomic JoA as the dependent variable.
Relationship between implicit and explicit SoA
To assess the contribution of implicit agency to explicit JoA in our exploratory analysis, we ran a repeated-measures logistic regression using a GLMM adding the RRE continuous variable to the other independent factors, that is, Group (autistic, neurotypical), Social context (One-agent, Two-agent), and Experimental condition (observation, no alteration, small alteration, large alteration). Dichotomic JoA were used as the dependent variable. There are contexts, such as effect modification by several factors, where adjusting the α-value to something higher than the conventional 0.05 threshold is justified (Thiese et al., 2016), and it has been used in medical (Lee et al., 2011) and biological (Carranza Martin et al., 2018) research, for example. Indeed, setting an α-value of 0.1 for four-way interaction terms reduces the risk of excessive Type II errors and can reveal relevant effects, especially when the effect size is large. Therefore, and as our third analysis was exploratory in nature, an α-value was set at 0.05 for simple, double, and triple effects and at 0.1 for four-way interactions effects.
Results
Effect of social context and feedback alteration on implicit measure
The linear mixed model analysis for RRE showed significant main effects of the Experimental condition (F(3,8987) = 23.28; p < .001) and Social context (F(1,8987) = 10.19; p < .05), but not of Group (F(1,43) = 3.96; p = .053). More importantly, there were significant Group × Social context (F(1,8987) = 23.44; p < .001) and Group × Experimental condition (F(3,8987) = 34.09; p < .001) interactions. The triple interaction was not significant (F(3,8987) = 0.50; p = .681). Detailed results are presented in Table S1 and Figure S1 (see Supplementary Material).
Different post hoc pairwise comparisons of RRE means were of interest. Alpha values of 0.05 were used and adjusted for multiple comparisons using the Bonferroni correction. As shown in Figure 2, Social context × Group contrast revealed a significant difference between One-agent and Two-agent contexts in the neurotypical group (ΔM = 0.065; p < .001), but not in the autistic group (ΔM = 0.013; p = .238).
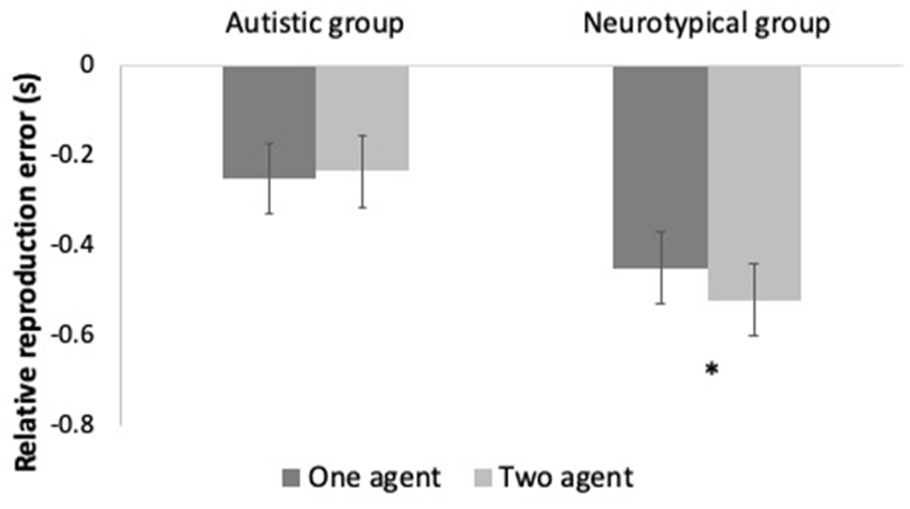
Implicit level of SoA: Relative reproduction error (RRE) in the One-agent (dark grey) and Two-agent (light grey) contexts in autistic (left) and neurotypical (right) groups. Significant differences across contexts represented by *. Error bars represent 95% confidence intervals.
Within-group comparisons of experimental conditions showed that none of the RRE means differed across experimental conditions in the autistic group, as shown in Figure 3. This indicates the absence of temporal binding for action effect. In the neurotypical group, RRE means of all three operant conditions significantly diverged from that of the observation condition. Operant conditions did not differ between one another. Conversely, between-group comparisons of the different experimental conditions revealed that RRE means for the observation condition did not differ across groups (ΔM = 0.077; p = .499). However, RRE means of operant conditions all significantly differed across groups: no alteration (ΔM = 0.272; p < .05), the small alteration (ΔM = 0.292; p < .05), and the large alteration (ΔM = 0.253; p < .05).
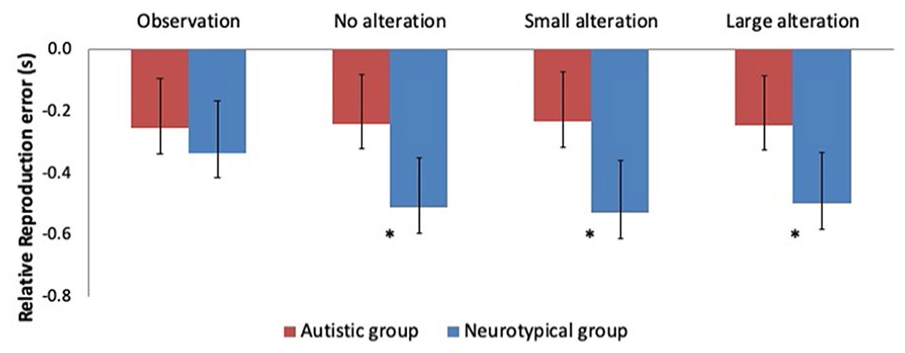
Implicit level of SoA: Between-group comparisons of RRE means across experimental conditions. In the autistic group (red), equivalent RRE means are found in all conditions, whereas a difference between observation and other conditions is found in the neurotypical group (blue). Significant between-group differences represented by *. Error bars represent 95% confidence intervals.
Taken together, those results confirm the presence of an IB effect for pinching movements with visual outcomes in the neurotypical group for all operant conditions. Social context also had a significant impact on binding strength in the neurotypical group. Feedback alteration had no impact on IB strength for that group. No IB effect was detected in the autistic group, as RRE means for all operant conditions were equivalent to the RRE mean in the observation condition. Neither social context nor feedback alteration had an impact on IB strength in the autistic group. The IB effect, which is a robust finding in the general population, can therefore be seen as globally abolished in our sample of autistic participants.
Effect of social context and feedback alteration on explicit measures
The generalised linear mixed model analysis for JoA showed significant main effects of Social context (F(1,9014) = 233.05; p < .001) and Experimental condition (F(3,9014) = 439.18; p < .001), but not of Group (F(1,9014) = 0.57; p = .449). Significant interaction effects were found for Context × Condition (F(3,9014) = 13.05; p < .001), Context × Group (F(1,9014) = 13.50; p < .001), and Condition × Group (F(3,9014) = 12.84; p < .001). More importantly, the three-way interaction effect was significant (F(3,9014) = 3.07; p < .05). Self-attribution levels (or “Me” answer level) for each condition of each context in each group are depicted in Figure 4. The three-way interaction effect was further investigated, with results of prime interest described in the following section. The detailed results of all decomposed effects for self-attribution levels are presented in Table S2 and Figure S2 of Supplementary Material.
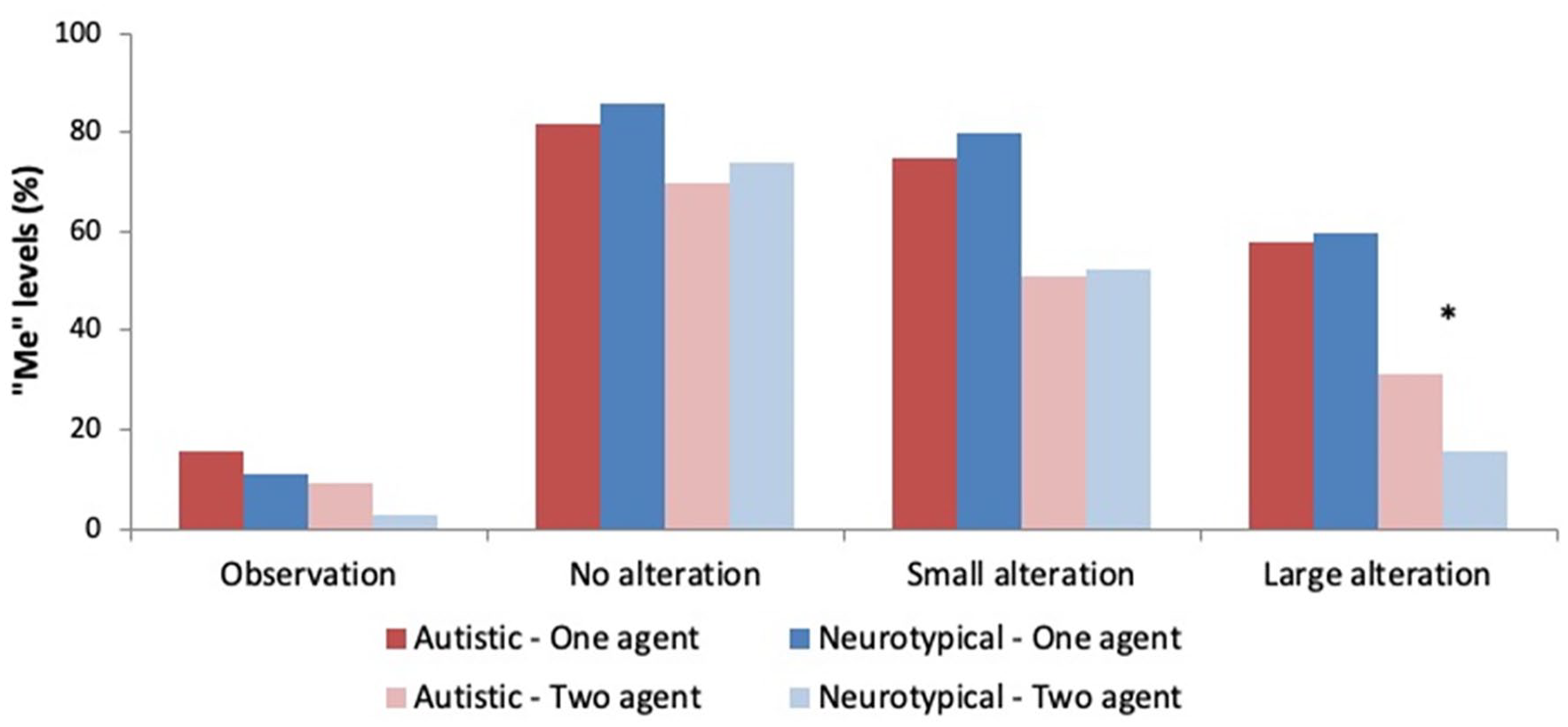
Explicit level of SoA: Percentage of self-attribution in the One-agent (dark colours) and Two-agent (light colours) contexts for autistic (red) and neurotypical (blue) groups. There is a significant Group × Condition × Context interaction. The designator * indicates significant between-group difference in self-attribution in the large alteration condition of the Two-agent context.
Decomposing for social contexts, the Condition × Group interaction was significant for both the One-agent (F(3,4507) = 3.56; p < .05) and the Two-agent (F(1,4507) = 11.43; p < .001) contexts. Within-group pairwise comparisons of all experimental conditions for each context were all significant (Bonferroni correction applied), as presented in Table S2 of the Supplementary Material. Odds ratios were also calculated for every pairwise comparison and are available in Supplementary Material. For example, the chances for an autistic participant to answer “Me” in the One-agent context is 24.39 (1/0.041) times higher in the no alteration condition than that in the observation condition, whereas it was 50 (1/0.02) times higher for a neurotypical participant. Interestingly, in the Two-agent context, autistic participants had 5.034 more chances of answering “Me” in the large alteration condition than in the no alteration condition, whereas neurotypical had 15.168 more chances of answering me in the same conditions.
Decomposing for experimental conditions, the Group × Context interaction was not significant for the observation condition (F(1,1286) = 2.67; p = .102), the no alteration condition (F(1,5156) = 0.66; p = .418), and the small alteration condition (F(1,1286) = 0.84; p = .358). However, the Group × Context interaction was significant for the large alteration condition (F(1,1286) = 16.93; p < .001). It is important to note that self-attribution levels were significantly lower for neurotypical participants in this condition only for the Two-agent context (t(9014) = 2.97; p < .01). An autistic participant had indeed 2.462 more chances of answering “Me” in the large alteration condition of the Two-agent context than a neurotypical participant, suggesting that, in a context where there was ambiguity about the actual cause of a sensory event, neurotypical individuals detected and used in a greater extent the large feedback disparity to attribute the cause of the flash to an external source.
Relationship between implicit and explicit SoA
For our exploratory analysis of the implicit-explicit levels relationship, the generalised linear mixed model analysis investigating the relationship between RRE and JoA showed significant main effect of RRE (F(1,8998) = 6.93; p < .01), Social context (F(1,8998) = 68.38; p < .001), and Experimental condition (F(3,8998) = 439.18; p < .001), but the main effect of Group was not significant (F(1,8998) = 0.73; p = .395). For interaction effects, we focused on interaction terms containing the RRE factor as the relationship between RRE and JoA was the focus of this analysis. Significant double interaction effect was found for RRE × Condition (F(3,8998) = 8.62; p < .001). Triple interaction effects were significant for RRE × Context × Group (F(1,8998) = 8.35; p < .01) and for RRE × Condition × Group (F(3,8998) = 2.95; p < .05). More importantly, the quadruple interaction effect was significant (F(3,8998) = 2.51; p = .057) at the α-value threshold of 0.1 set for four-way interaction in our exploratory analysis.
We further investigated the quadruple RRE × Context × Condition × Group interaction effect by analysing the RRE-JoA relationship separately in each group.
For the autistic group, the generalised linear mixed model analysis revealed no significant triple RRE × Context × Condition interaction effect (F(1,4604) = 0.549; p = .649) and only one significant interaction effect, that is, the RRE × Condition effect (F(1,4604) = 22.23; p < .001). Therefore, we further decomposed the analysis on the RRE factor for each condition. For the observation condition, there was no significant effect of RRE (fixed coefficient of −0.636; t = −1.784; p = .075). For the no alteration condition, fixed coefficient of 0.979 was significant (t = 7.850; p < .001), with odds ratio of 2.662. This means that the chances for autistic participants to answer “Me” in the no alteration condition were 2.662 times more important with every gain of 1 s (or 1000 ms) on the RRE variable. For the small alteration condition, the fixed coefficient of 1.007 was significant (t = 4.342; p < .001), with an odds ratio of 2.736. For the large alteration condition, a fixed coefficient of 0.528 was significant (t = 2.778; p < .05), with an odds ratio of 1.696. Hence, the relationship between RRE and JoA was significant in all operant conditions in the autistic group.
For the neurotypical group, the generalised linear mixed model analysis revealed no significant triple RRE × Context × Condition interaction effect (F(1,4394) = 2.478; p = .059) and only one significant interaction containing the RRE factor, that is, the RRE × Context effect (F(1,4394) = 6.265; p < .05). Therefore, we further decomposed the analysis on the RRE factor for each context. For the One-agent context, a fixed coefficient of −0.658 was significant (t = −4.463; p < .001), with an odds ratio of 0.518. This means that the chances for neurotypical participants to answer “Me” in the One-agent context were 1.931 (1/0.518) times more important with every reduction of 1 s (or 1000 ms) on the RRE variable. For the Two-agent context, a fixed coefficient of −0.326 was significant (t = −2.079; p < .05), with an odds ratio of 0.722. Hence, the relationship between RRE and JoA was significant in the two contexts in the neurotypical group.
Results from the analysis of the functioning of implicit level in autistic individuals show an abolition of the IB effect, while results from this analysis of the correlation between implicit and explicit levels show a positive association between RRE and JoA. This could be explained by the presence of a certain heterogeneity in our autistic sample that is masked by the averaging procedure of group differences analysis. To investigate this possibility, we conducted an exploratory a posteriori analysis of each participant time reproductions at the implicit level to evaluate if there were different clusters of time-estimation patterns. We identified three potential patterns in individual datasets: (1) under-estimation (RRE mean for operant conditions lower than RRE mean for observation), (2) equal estimation (RRE mean for operant conditions within 100 ms of RRE mean for observation), and (3) over-estimation (RRE mean for operant conditions is greater than RRE mean for observation). The results of this a posteriori investigation are represented in Figure 5. A Group (2) × Time estimation patterns (3) Pearson chi-square test was significant (χ2(2, N = 43) = 12.48, p < .01). Post hoc analysis of the adjusted residuals showed group differences for Under-estimation (p = .001) and Over-estimation (p = .005), but not for equal estimation (p = .194) patterns. Bonferroni corrections were applied.
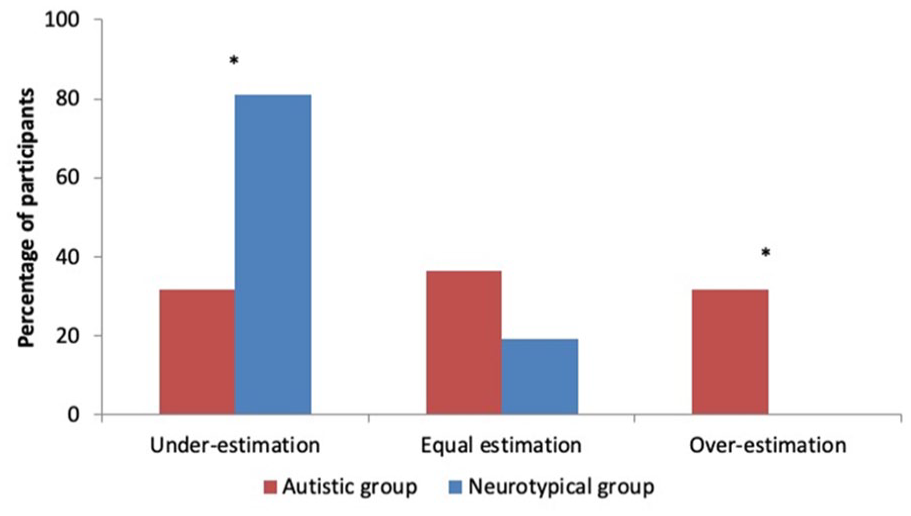
Distribution of time estimation profiles across autistic (red) and neurotypical (blue) groups. Significant between-group differences represented by *.
Discussion
The present study’s objectives were to provide a complete characterization of the autistic cognitive profile for SoA and to determine if autistic individuals display atypicalities in the integration of agency cues at implicit and explicit levels of SoA. Hereafter, we discuss the alterations of SoA processes in autism observed in our study for the (1) implicit level of SoA, the (2) explicit level of SoA, and for the (3) dynamic between implicit and explicit levels.
Implicit SoA: abolished overall IB effect in autism as an indication of altered internal models of action
IB is a robust phenomenon found in the general population (Moore & Obhi, 2012) and consists in the compression of the subjective interval between an intentional action and its outcome (Haggard et al., 2002; Wenke & Haggard, 2009) in comparison to the subjective interval between two external events. As expected, we detected a clear IB effect for the pinching movements in our neurotypical sample, as all subjective interval means in the operant conditions were underestimated in comparison to the intervals mean in the observation condition. However, the IB effect was globally abolished for all operant conditions among the group of autistic participants, as none of the subjective intervals means in the operant conditions differed from the one in the observation condition. Abolition of the IB effect in the autistic group was also highlighted by the higher RRE means in every operant condition compared to the neurotypical group, meaning that autistic participants show a reduced under-estimation bias for action-outcome interval estimation for self-performed actions. Indeed, autistic participants showed more veridical estimations of time in the operant conditions, as their reproductions of the intervals between actions and outcomes were, in average, closer to the objective intervals than those of neurotypical participants. This indicates that autistic individuals are less susceptible to the IB effect, in which a perceived compression of the action-outcome intervals creates a distinction between self-generated actions and outcomes and externally generated actions and outcomes. Using a different IB assessment method, our study confirms the results of Sperduti et al. (2014) who provided first indications that implicit SoA mechanisms were atypical in autism by showing that the IB effect was abolished for visual outcomes. It is to be noted that reproduced interval means in the observation condition did not significantly differ across groups, strongly suggesting that the observed abolition of IB for visual outcomes is not the incidental consequence of time perception alterations in autism. Although it has been suggested that a time perception impairment might be a key characteristic of autism (Allman et al., 2011), a recent meta-analysis (Casassus et al., 2019) concluded that evidence for a low-order time perception deficit in autism, implying processes such as detection, estimation, or reproduction of time intervals for sensory events, is mixed and that no clear consensus can be established as of now. Our results point out that autistic participants actually showed no atypicalities in time perception and reproduction for external stimuli. This is congruent with a study by Price et al. (2012) concluding that Asperger individuals show no alteration of their internal clock.
Our results also confirm that, in the neurotypical population, the IB effect is driven by prospective sensorimotor cues. Indeed, RRE means were equivalent in all three operant conditions in the neurotypical sample, indicating that the manipulation of the reliability of retrospective sensorimotor cues with a gradual alteration of feedback had no effect on IB strength. This implies that signals such as the presence of a forward model of action and action-selection processes suffice to create an IB effect in the neurotypical population, which is also in line with studies showing that the actual identity or intensity of the action’s outcome does not impact IB strength (Beck et al., 2017; Haering & Kiesel, 2014). We also observed a significant effect of social contexts on IB strength for neurotypical participants, with reproduced intervals being less underestimated in the One-agent context. It is compatible with the finding of Desantis et al. (2011) who showed that previous causal belief can impact IB strength. However, the effect we observed was markedly small, with differences between contexts in the range of 65 ms, as opposed to differences in the order of 170 ms between the observation and operant conditions. This observed effect of social context on time reproduction is unlikely to have a major impact on SoA functioning, as the IB effect was still occurring in both social contexts despite the slightly lower under-estimation in the One-agent context. In contrast, in the group of autistic participants, none of the prospective sensorimotor, retrospective sensorimotor, or contextual cues had any impact on reproduced intervals. This indicates that implicit SoA in autism, as assessed with the IB proxy, is not informed by the prospective cues on which neurotypical individuals usually rely. It also confirms that IB for visual outcomes is globally absent in autism and that binding strength is not influenced in autistic individuals by alternative cues, namely retrospective sensorimotor or contextual cues. Therefore, the signature provided by IB, in the neurotypical population, for self-produced actions and outcomes, is not observed in our autistic sample.
Prospective cues to implicit SoA arise from internal models that guide the enactment of actions, such as signals resulting from the selection of the inverse model or the production of a forward model that contains the sensory predictions. When learning a new movement, parietal cortices are involved in acquiring a “body” and a “motor” schemas (Daprati et al., 2010) based on external sensory feedback (Koziol et al., 2012). The cerebellum receives a copy of these schemas (Ito, 2005; Koziol et al., 2012), which results in the formation of an internal model of action (Ebner, 2013; Ishikawa et al., 2016; Wolpert et al., 1998) containing the dynamic sensory and motor information to produce the action. The granule cells of the cerebellum, which receive inputs from multiple sensory fields, are able to perform multimodal integration and are thought to be involved in the production of a forward model for actions (Huang et al., 2013). These internal representations are refined and updated in the cerebellum with repetitions (Ito, 2008; Synofzik, Lindner, & Thier, 2008) in procedural learning, and the most-effective behaviours are relayed to, then memorised by, the motor cortex through cerebro-cerebellar circuitry (Galea et al., 2011; Houk et al., 2007). Pre-SMA and cerebellum are also thought to be involved in the recruitment of internal models in action preparation (Bursztyn et al., 2006).
Our results suggest that the process of formation and/or recruitment of internal models of actions are altered in autism, reducing the reliability and availability of such cues to SoA. There exists robust evidence that the processes of internal models of action are altered in autism. It was showed that autistic children present an over-reliance on proprioceptive feedback, compared to visual feedback, when adapting their movements to unexpected alterations (Haswell et al., 2009), and that this over-reliance on proprioception in motor control is specific to autism (Izawa et al., 2012). Autistic children also show enhanced learning from proprioceptive errors in motor control in comparison to neurotypical children but reduced learning from visual errors (Marko et al., 2015). This suggests that the formation of internal models of action is biased towards proprioceptive information in autism. This is of importance because most skilled gestures required or learned in social interactions imply the visual modality. Indications that the formation of forward models of action is altered in autism also come from a growing number of studies showing that feedforward control of movement is atypical. Examples of this include decreased accuracy in the initial force output to attain a target (Mosconi et al., 2015; Wang et al., 2015), higher error rates in a repetitive aiming task (Papadopoulos et al., 2012), and a lack of anticipatory changes in muscle activity prior to a predictable change in load (Schmitz et al., 2003). Difficulties using visual feedback to adjust movements are also identified (Ament et al., 2015; Mosconi et al., 2015), and visuo-motor difficulties are also found to be linked to autism symptoms severity (Miller et al., 2014). Thus, our results added to those of these sensorimotor control studies seem to indicate that predictive mechanisms in motor control and cognition present significant alterations in autism. This observation is a clear contribution to broader accounts of autism such as the predictive coding account (Lawson et al., 2014; Sinha et al., 2014).
The hypothesis that abolition of IB in autism is an indication of an altered formation of internal models of action is also compatible with an implication of the cerebellum and the cerebro-cerebellar circuitry. Indeed, the cerebellum plays a crucial role in internal model formation, and there exists a consensus that autism is associated with different cerebellar anatomy and cerebellar-related motor and cognitive alterations (Fatemi et al., 2012). Neurofunctional data show that movements of autistic adults are associated with reduced activity in pre-SMA and cerebellar hemispheres (Takarae et al., 2007). Reduced activation of the sensorimotor portions of the cerebellum in autism also correlates with difficulties in using visual feedback to adapt movements (Marko et al., 2015). Finally, asynchrony between the activations of visual and motor systems in movement is correlated to autistic symptoms severity (Nebel et al., 2016).
Explicit SoA: under-reliance on sensorimotor cues to inform JoA in autism
Our results for the dichotomic JoA measure revealed one important atypicality in the way autistic participants established their JoA at the explicit level of SoA. While both autistic and neurotypical groups showed similar global patterns of JoA, as evidenced by reduced general levels of self-attribution in the Two-agent context in comparison to the One-agent context and a gradual reduction in “Me” answers with an increase in feedback alteration, intergroup comparisons did reveal a significant difference in their explicit SoA functioning. Indeed, autistic individuals showed higher levels of self-attribution than neurotypical individuals in the large alteration condition of the Two-agent context. This difference was the only one highlighted between groups.
Reflecting on this effect, we underline that, in the Two-agent context, there existed ambiguity about the plausible cause of the action’s outcome. Therefore, contextual cues provided no reliable information for determining the JoA, and prospective and retrospective sensorimotor cues were the only available cues that would allow to discriminate between self- and externally-produced stimuli. Examining the contribution of retrospective cues in this context, while both groups showed a gradual decrease in self-attribution levels with increased feedback alteration, the extent to which autistic and neurotypical participants used large discrepancies in the predictable vs actual height of the green flashes to inform their JoA significantly differed. Indeed, when feedback was unaltered or was only slightly altered, self-attribution levels were equivalent across the groups. However, as mentioned, self-attribution levels were 2.46 times more likely in the autistic than in the neurotypical group for the large alteration condition. This indicates that autistic individuals in our sample were significantly less likely to use the diagnostic information provided by a large feedback discrepancy to infer external agency. In contrast, when actual feedback was either unaltered or altered only in a subtle way, autistic individuals presented a pattern of JoA identical to that of neurotypical individuals. This important result suggests that autistic individuals display an under-reliance on diagnostic retrospective sensorimotor cues that strongly indicate external agency.
Taken together, our results for both the implicit and explicit levels of SoA allow us to revisit Zalla and Sperduti’s (2015) hypothesis that SoA atypicalities in autism might be characterised by impaired prospective mechanisms and spared retrospective mechanisms. Their proposition ensued notably from their observation of abolished IB effect in the visual modality (Sperduti et al., 2014), which strongly suggests altered prospective sensorimotor cue integration at the implicit level in autism. Our experiment confirms altered integration of prospective sensorimotor cues at the implicit level. However, the contribution of retrospective sensorimotor cues to implicit SoA in autism has to be further tested with implicit proxies of SoA that are sensitive to retrospective processes such as sensory attenuation. We propose that future research solve this issue by assessing sensory attenuation in the more meaningful visual modality.
Zalla’s group also explored the influence of different agency cues on explicit SoA. In the study by Zalla et al. (2015), participants had to attain targets under different conditions that manipulated contextual cues (performance feedback) and sensorimotor cues (alteration of temporal and spatial congruency of the cursor). Their results showed that autistic adults’ explicit feeling of control over the cursor was less sensitive to feedback alterations than the one of neurotypical individuals. We argue that the hypothesis of spared retrospective mechanisms is not in line with the results of this experiment. Indeed, we believe that their experiment about explicit SoA (Zalla et al., 2015) constitutes a strong indication that autistic individuals’ integration process at this level under-rely on retrospective sensorimotor cues, as feedback alteration signals arise from the comparator mechanism and become available only after the completion of the action. The results of our current experiment also support that retrospective sensorimotor cues’ integration process at the explicit level is altered, as diagnostic signals arising from large feedback alteration did not lead to the same reduction of self-attributed JoA in the autistic group.
In addition, our experiment indicates that the autistic functioning of explicit SoA is influenced in the same way as the one of neurotypical individuals by the induced belief that they were the only plausible cause of action’s outcome. This is compatible with the experiment by Zalla et al. (2015), in which they showed that autistic individuals used contextual cues provided in the same fashion as did for neurotypical participants. However, the agency belief that we used in our experiment was a contextual cue that is prospective in nature as it was available before the action, while performance feedback, the contextual cue provided in the experiment by Zalla et al. (2015), is rather retrospective, as it was made available only after the completion of the movement. It leads us to think that the prospective/retrospective distinction cannot by itself explain all the atypicalities encountered in the functioning of both levels of SoA in autistic individuals.
In a nutshell, contrary to early studies about action monitoring in autism that used experimental tasks not allowing to manipulate the availability or reliability of different agency cues (David et al., 2008; Grainger et al., 2014; Russell & Hill, 2001; D. Williams & Happé, 2009), our results clearly show that autistic individuals present an atypicality in explicit SoA functioning. Our results rather confirm Zalla et al.’s (2015) findings of atypical cue integration with an under-reliance on diagnostic sensorimotor cues, specifying however that integration of retrospective sensorimotor cues is the mechanism underpinning the altered explicit SoA in autism.
Relationship between implicit and explicit SoA: a window into autistic heterogeneity in SoA functioning?
Group comparisons allowed us to shed light on the presence of atypicalities in the autistic functioning of SoA at both implicit and explicit levels. However, our experiment was also designed to allow a trial-by-trial exploratory analysis of the relationship between our implicit marker (time reproduction) and the explicit marker (dichotomic JoA). Synofzik and colleagues’ (2013) cue integration model predicts that the registration of agency at the implicit level is in itself a cue that enters the weighing process at the explicit level. So far, even for the general population, only a few studies have investigated the contribution of IB to the explicit level on a trial-by-trial basis. Pyasik et al. (2018) and Imaizumi and Tanno (2019) have found a significant association between IB and explicit JoA. Using the same psychophysics task as the one used in the current study, Lafleur et al. (2020) reported no significant association between their measure of IB and dichotomous JoA in the general population (Lafleur et al., 2020). Yet, in the current study, employing a more powerful statistical model (generalised linear mixed model vs generalised estimating equation for the previous study) allowed the detection of an association between time reproductions and self-attribution levels for the neurotypical group.
Indeed, our present results indicate an association between the IB effect and self-attribution levels at the explicit level in the neurotypical group. A greater under-estimation of the action-outcome interval was associated with a greater chance of answering “Me” at the explicit level in all four conditions of the One-agent context. This association was the same in the Two-agent context for all conditions confounded, but the effect was of a relatively smaller magnitude. Those results are compatible with the Synofzik et al. (2013) conceptualization. Indeed, our results suggest that the registration of agency at the implicit level is integrated at the explicit level in the establishment of a conscious, explicit JoA. We still want to underline, though, that only the integration of several different cues can fully explain the determination of JoA at the explicit level. It is also interesting to note that ambiguity about the possible cause of the action’s outcome seems to reduce the weight of the implicit registration in the integration process at the explicit level, which is an argument in favour of context-dependant weighing in cue integration.
Our results for the autistic group contained notable differences from the results of the neurotypical group. For the autistic group, results showed no effect of social contexts on the strength of the association between time reproductions and JoA, but rather an impact of the experimental conditions. First, there was no significant link between time estimations and JoA for the observation condition. However, all operant conditions saw a positive association between higher time reproduction errors and self-attribution levels. Higher time estimation values were indeed associated with greater probabilities that autistic participants self-determine they themselves were the cause of the visual stimulus. Different cognitive explanations could account for this association between higher values of RRE and higher levels of self-attribution of agency in autistic individuals. In the first place, this trend could imply that subjective over-estimation of the objective time interval between action and outcome could be used by autistic individuals as a marker of self-agency. In this explanation, over-estimation would be an implicit proxy of self-agency that is opposite to the habitual IB effect (involving under-estimation) recurrently found in the neurotypical population. The registration of agency at the implicit level would be attested by over-estimation of the objective intervals, and this registration could constitute a cue entering the integration process at the explicit level. A second alternative explanation would be that the subjective under-estimation of the action-outcome time interval could be used by autistic individuals as a cue of external agency rather than a cue for self-agency.
However, the overall abolition of the IB effect found in our analysis of the implicit level combined with the observation that higher values of time reproductions seem to contribute to higher levels of self-attribution of agency further raise the question of heterogeneity within the autistic spectrum. Indeed, this observation led us to analyse the interindividual heterogeneity in the time estimation patterns found within our implicit marker of SoA. Our results suggest that there exist group differences between the distributions of individuals between different patterns of time reproduction (under-estimation, equal estimation, and over-estimation). Autistic individuals are less likely to show under-estimation, that is, an IB effect, and more likely to show over-estimation, that is, an un-binding effect, than neurotypical individuals. The heterogeneity of the autistic group was also illustrated by the equal distribution of individuals between those three patterns of time estimation. In contrast, neurotypical individuals were predominantly showing under-estimation of the action-outcome interval, with none of them showing over-estimation. This a posteriori analysis suggests that the sensorimotor cognitive architecture that underlay implicit SoA displays variability across the autistic spectrum. We suggest that further research to characterise the autistic cognitive profile for SoA could investigate subgroup differences in SoA functioning and their associations with clinical features using larger sample groups. This variability in cognitive profiles could very well be reflective of the behavioural variability in the sensorimotor domain.
Transdiagnostic considerations on atypical SoA
The cue integration approach of SoA was partly developed to explain different symptoms of schizophrenia, such as delusion of control and delusion of influence (Synofzik et al., 2013; Synofzik, Vosgerau, & Newen, 2008; Synofzik & Voss, 2010). In the present article, we reviewed and provided new data on atypical functioning of SoA in autism using this paradigm that allows to disentangle the contribution of several and varied cues to the two levels of agency. A growing amount of indications point towards altered SoA functioning in several neuropsychiatric conditions such as schizophrenia and autism, but also in OCD, Tourette’s syndrome, Parkinson’s disease, anorexia nervosa, and borderline personality disorder. Along with other researchers (see the articles by Malik et al., 2022 and Moccia et al., 2024 for comprenhensive reviews of SoA in neuropsychiatric disorders), we believe that transdiagnostic accounts of SoA can not only shed light over partly shared phenomenology in some neuropsychiatric conditions but also help provide a clearer neurocognitive explanation of the differences among cognitive and clinical profiles.
The results of our present study highlight the presence of both convergences and divergences between the SoA cognitive profiles in autism and schizophrenia. Several studies have consistently found enhanced IB in individuals with schizophrenia (Moccia et al., 2024) at the implicit level of SoA. This altered implicit SoA has been notably shown to be explained by impaired and unreliable predictive motor signals, such as the forward model containing the predictions of action’s consequences (Synofzik et al., 2009; Voss et al., 2010). At the explicit level of JoA, Synofzik et al. (2009) found that individuals with schizophrenia tend to attribute to themselves actions’ consequences even when they are largely distorted and show an over-reliance on visual, external signal over proprioceptive signals. This tendency to use visual, external feedback to inform JoA was correlated to the severity of delusions of influence symptoms. In our autistic sample, our data suggest that autistic individuals also present unreliable internal models of action. But, as discussed earlier, complementary studies show that, contrary to the findings in individuals with schizophrenia, internal models of action in autism are biased towards the proprioceptive over the visual modality. This could account for the difference found in profiles at the implicit level with enhanced IB in one condition, and overall abolished IB in the other (but see previous sections for the heterogeneity found in our autistic sample). Although there have been studies showing a correlation between over-reliance on proprioception and socio-communicative symptoms of autism (Haswell et al., 2009; Izawa et al., 2012), it would be of interest to further examine the link between IB profiles in autism and the intensity of socio-communicative symptoms, as well as other clinical features of autism such as stimming or compulsive behaviours. Interestingly, the only study on OCD and IB have found that OCD traits are associated with reduced IB strength (Oren et al., 2019).
Limitations and future directions
To our knowledge, our study is the first to assess simultaneously the integration mechanisms of both levels of agency in autistic individuals according to the emerging cue integration approach of SoA. However, the current study faces some limitations that future research could overcome. First, the large characterization of SoA functioning in autism examined the contributions of several agency cues on two measures of SoA, which led to a rather complex experimental design. Given the relatively small sample sizes, it is difficult to affirm whether the findings are representative of a larger autistic population. Second, the current study being partly exploratory in nature, the relationship between implicit and explicit levels of SoA as well as the differences in estimations profiles across groups should be revisited in future studies. Still, the current results provide important provisional information about the autistic functioning of SoA and that it can provide informed direction for future research on this cognitive function in autism. An interesting avenue to explore is the contribution of prospective sensorimotor cues to both levels of SoA by using devices such as transcranial magnetic stimulation for a more direct manipulation of the availability of sensorimotor cues.
Conclusion
In the present study, we sought to establish a complete characterization of implicit and explicit SoA functioning in autism, using cue integration approaches (Moore & Fletcher, 2012; Synofzik et al., 2013). We provide first evidence that SoA functioning in autism coincidently presents atypicalities at both the implicit and explicit levels, as well as peculiarities in the interaction between the two levels. We found that the IB effect was overall abolished in autistic individuals, highlighting reduced weight of prospective sensorimotor cues at the implicit level. This result strongly suggests altered formation of internal models of action in autism. We also demonstrated that the explicit level of SoA in autism was characterised by an under-reliance on diagnostic retrospective sensorimotor cues on which neurotypical individuals usually rely. We also found out that the dynamic between implicit and explicit levels was different in the autistic group, as greater IB strength at the implicit level did not lead to greater self-attribution at the explicit level. Contrary to the neurotypical group, the autistic group showed an overall association between greater time estimation and self-attribution level. The autistic group showed greater heterogeneity. Indeed, there were fewer individuals showing IB and more individuals showing unbinding in the autistic group than in the neurotypical group. We believe these patterns of time estimation at the implicit level of SoA (under-estimation, equal estimation, and over-estimation at the implicit level) can bring more sensitivity to further studies of SoA functioning. Taken together, our results confirm the presence of atypicalities in the autistic functioning of SoA at both levels and re-assert the relevance of studying action control and motor cognition in autism. We believe that the idea socio-communicative competence relies on the development of skilled behaviour (Mostofsky & Ewen, 2011; von Hofsten & Rosander, 2012) and motor cognition constitutes, more than ever, a compelling framework to be further investigated in the cognitive neurosciences of autism.
Supplemental Material
sj-docx-1-qjp-10.1177_17470218241311582 – Supplemental material for Atypical implicit and explicit sense of agency in autism: A complete characterization using the cue integration approach
Supplemental material, sj-docx-1-qjp-10.1177_17470218241311582 for Atypical implicit and explicit sense of agency in autism: A complete characterization using the cue integration approach by Alexis Lafleur, Vicky Caron, Baudouin Forgeot d’Arc and Isabelle Soulières in Quarterly Journal of Experimental Psychology
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This work was supported by career awards from Fonds de recherche du Québec – Santé (FRQS) to IS and BFA. AL received a doctoral award from FRQS. FRQS was not involved in the conduct of study or manuscript preparation.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
