Abstract
Controlled Human Infection studies are a new and uncommon way of conducting clinical research in low resource settings. Regardless of the supposed importance of human infection studies in advancing clinical research, there is still limited literature on community perceptions of this field of research, particularly in low-resource settings. An exploratory qualitative study involving 107 community members organized into 16 focussed groups was conducted in Kampala city and Buikwe district in central Uganda between September 2023 and March 2024. The groups comprised market vendors, secondary school teachers, university students, media professionals/journalists, mosques/church attendants and Community Advisory Board members. Data were analysed using a systematic inductive analysis approach. While community members across all deliberative focus group discussions acknowledged the potential of human infection studies to accelerate the development of therapeutics for infectious diseases that disproportionately affect low-income countries, the majority viewed deliberate infection method as difficult to accept at the moment. Several would not be willing to participate in controlled human infection study due to the perceived fear of death and associated long term consequences, infrastructural challenges, mistrust of scientists, cultural and religious beliefs. Deliberate effective engagement with the community may enhance community acceptability of controlled human infection studies. Such engagement should use contextually appropriate messages to create awareness and augment acceptability of the human infection method in low resource settings.
Introduction
Generally, research plays a pivotal role in advancing knowledge and societal progress. Specifically, medical research informs health care practice, human biology and behaviour (Kandi and Vadakedath, 2023). In sub-Saharan Africa (SSA), where high burdens of disease exist, clinical research is of particular importance for advancing our knowledge of disease, developing novel remedies and improving disease management (Piantadosi, 2024). Randomized controlled trials (RCT) are known as the most scientifically rigorous method of hypothesis testing and have traditionally been the gold standard for evaluating the efficacy and safety of new interventions (Saturni et al., 2014; Victora et al., 2004). However, conventional RCTs, particularly phase II and phase III trials could be challenging since they require large sample sizes, high costs for implementation and are time-consuming (Balasingam et al., 2014; Roestenberg et al., 2019; Sessler and Myles, 2020; van der Graaf and Cheah, 2019). Clinical research sponsors and investigators are increasingly adopting modifications to conventional trial designs and methodologies that introduce flexibility and efficiency (Harvin et al., 2020). Controlled Human Infection (CHI) method is one of the alternative clinical trial designs and methods used in clinical research to address some of the challenges posed by traditional RCTs in terms of flexibility and efficiency (Harvin et al., 2020; Roestenberg et al., 2019; Sessler and Myles, 2020; van der Graaf and Cheah, 2019).
Controlled Human Infection studies are clinical studies designed to deliberately expose research participants to specific pathogens also known as challenge agents (for instance bacteria, viruses, parasites) in controlled environments (Dholakia, 2018; Jamrozik and Selgelid, 2020a, 2021b; Vaz et al., 2021). The intention is to study how the infection progresses in humans, develop challenge agents, observe the body’s immune response and test efficacy of therapeutics and or vaccines (Balasingam et al., 2022; Vaz et al., 2021). In a CHI study, a pathogen whose biological properties, behaviour and effects have been extensively studied and understood is intentionally given to carefully selected adult participants, in controlled settings (Raymond et al., 2019; Roestenberg et al., 2019; Sciences, 2018; WHO, 2021). The objective is to understand the pathogenesis and transmission, prevention and treatment of diseases (Balasingam et al., 2022; WHO, 2021).
Although CHI studies play a vital role in the development of vaccines and or therapeutics for infectious diseases and have contributed to answering important scientific and public health questions (Balasingam et al., 2022; Raymond et al., 2019; Roestenberg et al., 2018a; Sztein and Booth, 2022), they raise unique ethical, safety, environmental, social and scientific challenges (Jamrozik and Selgelid, 2020a). For instance, the concept of intentional infection, risks and burdens, benefit sharing, informed consent and education, undue influence, risk benefit evaluation, compensation of participants, right to withdraw, religious and cultural beliefs, CHI research expertise, inadequate facilities to monitor and treat adverse events, inadequate facilities to produce and store infection materials, third party risks and relevant guidelines/regulatory frameworks (Gordon et al., 2017; Jamrozik and Selgelid, 2020b; van der Graaf and Cheah, 2019). Such challenges may even be heightened in low resource settings.
Controlled Human Infection studies have made important contributions to the treatment and prevention of infectious diseases of global health importance (Balasingam et al., 2022; Raymond et al., 2019; Roestenberg et al., 2019). However, the concept of intentional infection of healthy research participants with disease-causing pathogens is still thought-provoking, unconventional, creates puzzling questions and raises eyebrows, among community members, particularly in low-resource setting countries where CHI studies are new (Njue et al., 2018). Currently, there is an ongoing CHI study in Uganda however, this type of research is not only misunderstood, but it has also raised several ethical and regulatory concerns that warrant deeper exploration (Egesa et al., 2022; Elliott et al., 2018; Koopman et al., 2019). The perceptions of ordinary people, as well as the acceptability of CHI studies remain unknown. Earlier studies (Egesa et al., 2022; Elliott et al., 2018; Koopman et al., 2019) have highlighted the ethical and scientific considerations for the establishment of a CHI for schistosomiasis in Uganda However, they do not report on the opinions of the general public regarding the deliberate infection method. Our study therefore set out to explore the perceptions of ordinary people here referred to as community members. This paper aims to voice the views of community members regarding the CHI method and augment acceptability of the human infection method in low resource settings.
Materials and methods
Study design and approach
A cross sectional exploratory qualitative study of community members’ perception of CHI studies was conducted in central Uganda. The research was guided by the constructivist paradigm, this approach aims to understand “the world of human experience,” and proposes that reality is socially constructed (Mackenzie and Knipe, 2006; Mertens, 1998). Data was generated through dialogue between the researcher and research participants. The design and methods used were deemed appropriate because the study aimed to gain a deeper understanding of the perceptions of the community members on the CHI study method.
Study setting and participants
Participants were purposively selected from Kampala City (urban) and Buikwe District (rural). While being urban or rural was the basis for these choices, Buikwe (rural) in particular was selected due to its proximity to Kampala (urban) and likelihood of having research naïve populations. The study involved ordinary people belonging to specific categories such as, community advisory boards (CAB), market vendors (MV) with petty business, university students (UNI-ST) with background in both sciences and arts courses, secondary school teachers (SST) from private and government owned schools, media professionals/journalists (MP) and church/mosque attendants/leaders (RGL). Participants were identified from the different corresponding venues, research institutions, markets, government and private owned universities, secondary schools, media stations (TV, radio, newspaper etc) and religious places (churches/mosques). Prior to engagement with the individual participants, administrative clearance and/or permission was sought from the respective institutional head/designee.
Study procedures
Potential participants were approached, identified and purposively selected on an individual basis. The informed consent process and deliberative focus group discussions (dFGD) were conducted either in English or Luganda (a commonly spoken language in the study areas) based on the participants’ preference. The informed consent process and dFGDs lasted 2–3 hours approximately. Light refreshments were provided to participants during the sessions.
Data collection
The dFGD was the method for data collection. Deliberative focus group discussion approach was used because CHI is a relatively new and complex health topic in Uganda. The dFGD method is instrumental in instances where the research is gathering information on complex health topics (Fishkin, 2009; Rothwell et al., 2016). In dFGDs, the investigator identifies innovative ways to educate and inform potential participants about the topic of interest prior to the focus group discussion (Rothwell et al., 2016). For our research, we used a power point presentation and video drama skit as the innovative ways to highlight the design and conduct of CHI studies. This was done to educate, discuss and inform the participants about CHI method prior to the actual data collection. The educational materials are provided as Supplemental Documents.
Phases of the deliberative focus group discussions
The dFGDs comprised of two sessions, an educative session and group discussion. Materials used in the educative session contained information on the brief history of CHI studies, what they are, what makes them different from the usual RCTs, countries that have conducted CHI studies, some of the types of infections/diseases that have used the CHI method, the value of the CHI studies, and a video drama skit giving a scenario of a malaria CHI study. Balanced information that incorporated both the pros and the cons of CHI studies was presented to the research participants prior to group discussion. The principal investigator (WN) participated in all dFGDs for consistency and as an expert. The educative sessions enabled participants to become more conversant with the topic of discussion, comfortable and reflective. This culminated in to more informed and thoughtful responses, thus enriching the quality of the data collected. A team of six comprising of the principal investigator a social scientist and bioethics expert and five research assistants who are experienced in qualitative research conducted the dFGDs. The research team received training on the protocol, study tools and basic research ethics before conducting any study activities. Data was collected between September 2023 and March 2024.
Ethical considerations
The study was reviewed and approved by the Makerere University School of Biomedical Sciences Research Ethics Committee (SBS-2023-307) and the Uganda National Council for Science and Technology (SS 1925ES). Written informed consent and permission to audio record was obtained from all participants prior to enrolment into the study. All participants were assured of confidentiality.
Data analysis
A total of 16 transcripts for the dFGDs were analysed. The data analysis utilized an inductive approach. The transcribed dFGDs were thoroughly reviewed multiple times to internalize the content. Subsequently, a coding framework was developed based on four transcripts (25%), that were manually reviewed and coded to establish the initial set of codes. These codes were then crosschecked iteratively by three coders trained in qualitative data analysis to enhance reliability. Any discrepancies in coding were resolved through discussions and mutual consensus. All transcripts were imported into NVivo version 12 for open coding. The codes were further categorized and themes were identified. Illustrative quotations for each emerging theme were chosen for the results presentation. These are illustrated in the Results section.
Results
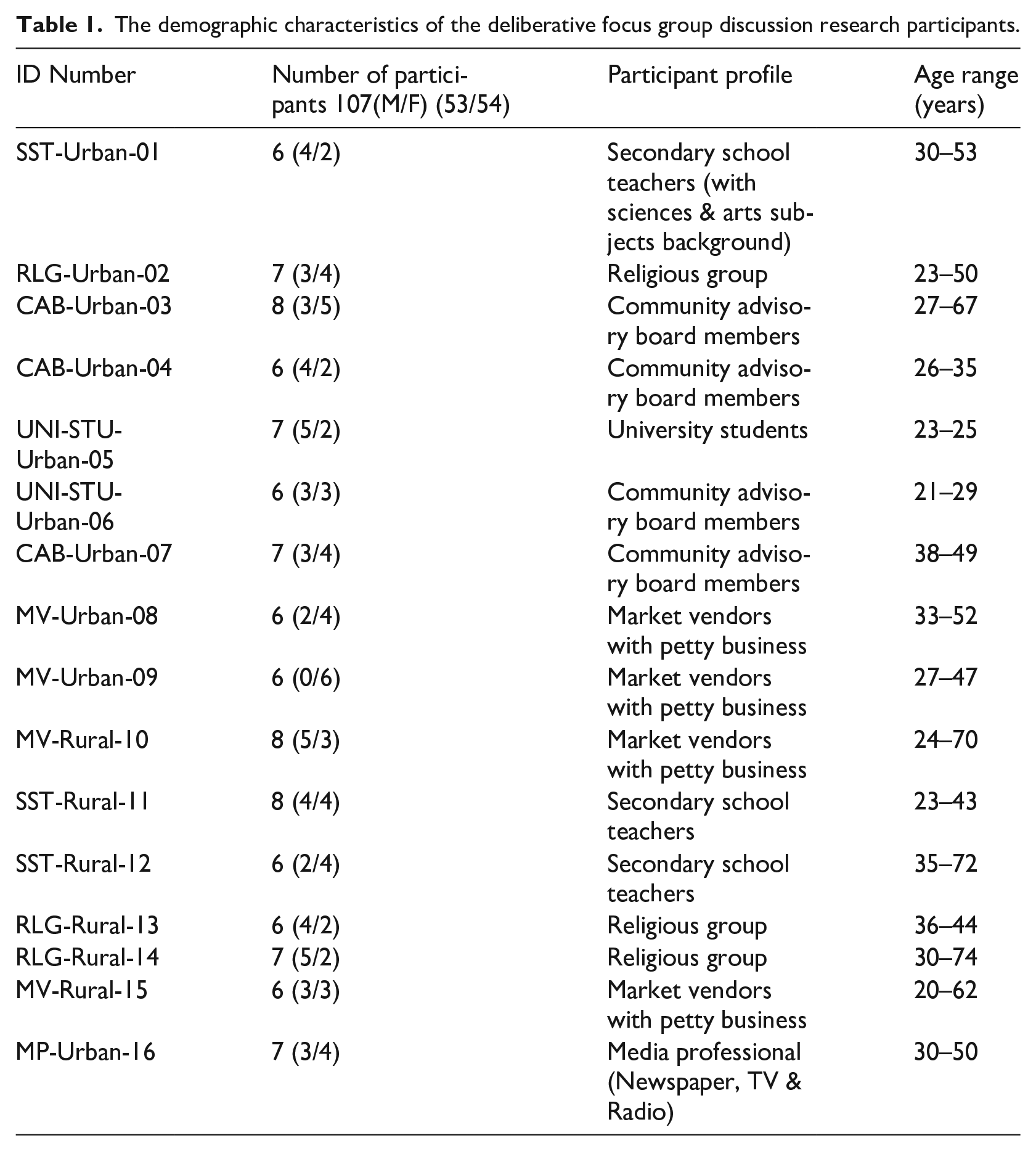
A total of 107 participants in 16 dFGDs took part in this exploratory qualitative study. Each group comprised of 6–8 participants; the majority were above 35 years of age. Demographic characteristics of the research participants are presented in Table 1
The demographic characteristics of the deliberative focus group discussion research participants.
Summary findings on the opinions of community members regarding CHI study methods are reported in this section. Table 2 below presents the themes and summary key findings.
Themes and summary key findings.

Perceptions of community members on CHI method
In this section we present study findings under four headings. First, we describe the positive perceptions. Second, we report on the negative perceptions and or reasons given by community members for not willing to participate in CHI study. Third, we illustrate views on acceptability and feasibility of CHI studies in Uganda. Fourth we report on factors likely to motivate community members to participate in a CHI study. The most common codes from the theme are discussed below.
Theme 1: Positive perceptions/perceived relevance of CHI study method
We asked participants for their views on the usefulness of CHI method for infectious diseases, such as malaria, SARs COV (COVID-19), Bilharzia. Controlled Human Infection studies were perceived relevant by majority of the participants across all focus group discussions. The reasons for viewing CHI studies as useful are discussed in the illustrative quotes below.
Accelerate the development of new effective drugs, vaccines and or therapeutics
Several community members perceived CHI studies as a valuable tool for accelerating development of novel drugs and vaccine technologies, “It will help us to come up with new ideas of treatments, new medicines, . . . . . . if medical personnel develop a vaccine against malaria . . . . just like measles and other diseases. . . .”-UNI-STU-Urban-06. Development of efficacious and effective drugs was citied in almost all focus group discussions. “. . . I feel like in terms of making effective medicine CHI studies are better”-CAB-Urban-04. Therapeutics for diseases that disproportionately affect LICs such as malaria were favoured. Also, participants perceived CHI research as useful because it would lead to the development of numerous treatment options. “. . . having alternative well proven medication for endemic diseases.”-SST-Rural-11. This was perceived as advancement in science and technology. “..whenever you have treatment options, and you pick an option that you’re comfortable with, chances of healing faster are high. . . I think it will be a very big advancement to science. . .”-CAB-Urban-04. Controlled Human Infection studies were perceived to offer a broad array of therapeutics which can enable individuals to select the most suitable approach for their conditions and personal preferences, eventually leading to more improved disease management, patient satisfaction and better overall health outcomes.
Build sense of ownership and trust in the products developed
Some participants viewed CHI research as useful in building public trust and mutual ownership of the research outputs.“. . . . . .when you tell people that we are going to be using treatments and that research was done in the USA or China, they feel like that is something different, but if research was done in Masaka and Mbale [districts in Uganda] and intervention showed that it worked, they will be like this person was like me. So, they will feel a sense of ownership . . . .”-UNI-STU-Urban-06. Participants perceived a sense of ownership and confidence in the products developed if CHI research is carried out in country and on Ugandans.
Enhance clinical research and expansion of new knowledge
More so, community members opined that CHI studies are likely to have positive outcomes and impacts that go beyond individual participants to benefit future generations. “. . . this research is going to help our relatives in the future to develop science & technology fast . . .” -MV-Rural-15. Human infection studies were viewed as likely to contribute to advancement of science and technology leading to improved quality of life for broader populations. “. . . . Some participants might not benefit directly out of these [CHI studies] but it shall help the coming generations . . . .” -RLG-Rural-14. The benefits of CHI studies were perceived to extend from development of vaccines and treatments to broader societal impacts. University students and media professionals mentioned that CHI studies would be valuable for obtaining new knowledge about the pathogens and history of disease. “. . . . . . CHI studies should be done in Uganda because it will give new avenues to study disease progression, which I think will be a plus for managing diseases. . . . .” -MP-Urban-16. “. . . . this type of research helps us to get new information. . . it helps them [researchers] to monitor the way the germs behave in different bodies. . .” -UNI-STU-Urban-07. The data indicates that CHI studies offer unique opportunities to uncover valuable insights that can improve researchers’ understanding of infectious diseases, their genesis, transmission and potential interventions.
Require small sample sizes and are fast compared to individual RCTs
Human infection studies were perceived as a cost-effective alternative by media professionals and CAB members (2/16 FGDs), as compared to traditional RCTs. “..seen as being time-saving because we’ve seen research taking seven years to get results, but I believe now this one because we are using the infection itself, we are going to get results very fast. it requires very few numbers of people, that means costs are going to be lower. As compared to these other studies where you need a good number of people, we use a lot of money to get the results, I believe it is one of the best of its kind. . . .” -CAB-Urban-03. Research that deliberately exposes humans with pathogens allows scientists to monitor the infection and treatment response over a much shorter period making it possible to gather results in months rather than years. Also, trials are normally done on a smaller number of participants as compared to phase II RCTs.
Theme 2: Negative perceptions
Many participants across all dFGDs had negative views towards intentional infection of healthy individuals. Some opined that CHI research is ethically challenging and felt that such research should be conducted in animals and not humans. “. . .we have very many animals in our country, we have rats, we have dogs. . .Why don’t they use animals instead of playing with the life of a human being. . . .”-MV-Urban-09. Participants reacted in disbelief and viewed the method as inappropriate. “. . . . I still see a problem there, all the previous methods we have had are for finding treatments, but now we have the medication and we are infecting a healthy person to learn from them. . . . , according to me I don’t see very much use there. . . .” -CAB-Urban-03. Although CHI studies provide valuable information on pathogenesis, vaccines and treatments, there were a number of negative views and challenges associated with the method.
Fear of the unknown and long-term consequences of human intentional infection
Participant’s concerns were majorly influenced by fear of the unknown. They expressed wary for the long-term consequences of intentional infection, physical harm and the resultant disruption of daily living. “. . . . . the concern is what if I become lame?”-SST-Urban-01. “. . . . . . . in case a participant God forbid lost life; is there anything that takes into account the trauma caused to that individual’s family and friends? beside health risk, the last one is usually death. . . .”-UNI-STU-Urban-07. The fear of death due to failure of treatments was eminent and the majority of participants almost in all the dFGDs mentioned it. “. . . . . .because even though malaria has treatment, a lot of people are still dying of malaria. if they infect you and you get the complicated strain of malaria the drugs will not respond, and you end up dying. . . . ”-MP-Urban-16. Study findings reveal the fear of death as an eminent concern. Some participants were afraid of the new mutations that could arise after intentional infection and the risk of failure to control the experiments. “. . . . there are lots of risks if you can’t control them, you’re finished. If there are new elements that come up, new either mutations or new whatever it is, it might cause a lot of harm. . .” -CAB-Urban-05. The costs of treatment in case participants are inadvertently harmed after inoculation of infectious pathogens during the CHI study was another concern from some community members. “. . . . . . the cost of treating that person, if that person falls sick, the stages grow from bad to worse. . . . since we don’t have adequate resources . . . .”-MP-Urban-016. “. . . . . . are there some budgets that have been put in place in case of things go wrong? I mean, in case some compensations to be done?. . . . . . . .” -SST-Urban-01. For a few participants, models of infection that are not using the natural route of infection (actual vectors). There was wary whether what they are to be infected with is the actual infectious agent under study. “. . . . ., is it true that what they are telling me they are infecting me with is the actual pathogen or it is something different? . . .”-SST-Urban-01. Conduct of CHI studies is associated with number of risks, oversight by RECs and adherence to risk management strategies are essential to ensure risk reduction and safety of participants.
Myths about the motives of CHI research in LICs
Some participants were suspicious about the true intentions behind CHI studies given these studies involve intentional exposure of disease-causing pathogens to healthy participants “. . . . there is something which this white man is trying to test for something behind this [CHI method]. . .”-MV-Urban-09. Also, human intentional infection method was viewed as a concept from the global North with a hidden agenda that is not in the best interest of the Africans. “. . .we have heard of conspiracies, theories saying they [the white man] want to kill Africans or to reduce our lifespan or fertility. . . . . . for fear that somebody may take advantage like they have done in the past. . .”-UNI-STU-Urban-07. Participants opined that ordinary remedies were favoured, and they did not trust interventions that involve deliberate infection. “. . .I don’t trust in [CHI studies]. A black African has to follow the old ways whereby they give you Chloroquine [medication] after getting sick with malaria but not first infecting with germs for malaria and then afterwards test their medicines on you, that one is not good at all. . .”-MV-Urban-09. Myths about research generally in LICs often stem from misunderstanding and misinformation regarding research practices. This leads to distrust, fears and concerns about exploitation. Addressing the fallacies is important for fostering trust with the local communities.
Beliefs that do not favour CHI method
Some community members viewed deliberate infection as unnatural and religiously inappropriate. They opined that their religious beliefs would not favour participation in a CHI study. “. . . . . when it comes to mosques, let them not waste time to come because with the mosques they [Muslims] are not going to accept. . . . . . .people will say these [researchers] have come with a trick of killing us . . . .”-RLG-Urban-02. Also, some viewed CHI studies as assuming divine prerogative and tempting God by intentionally infecting individuals with pathogens. “. . . . . disease comes and God helps you to get healed, I can’t tempt God to put a disease in my body. . . . . . . . even when you are swallowing tablets you always say, “God heal me” So why should I test God to insert a disease into my body and then ask God to heal it . . .I can’t risk and there is no compensation amount which I want. . .”-RLG-Rural-14. “. . . we are praying against sickness. . . It’s hard to accept for a person to get intentional infection, that you go and get what? infected!!. . . . I will not because that’s the fraternity that I’m representing. . . .”-CAB-Urban-04. They advised that for religious issues be addressed for people to accept deliberate infection as they commonly expressed; “. . .Talk to them [religious people] if they accept this procedure. . . .”-CAB-Urban-03. Cultural and religious beliefs about health and illness shape perceptions towards human infection method and may influence willingness to participate in CHI studies. Moreso, few community members commented on the definition of a “healthy” participant in CHI studies indicating that it is not just absence of disease. “. . . . some people might be healthy, but we know that the real definition of health is not just absence of disease but a complete state of mind and others, but if we are to go by the real definition of health, you will find that almost no one is healthy. . .”-UNI-STU-Urban-06. Participants questioned whether scientists consider other aspects of health. “. . . I think this method [CHI] wouldn’t be bad but as people are having many types of illnesses. . .”-MV-Rural-10. According to the World Health Organization (WHO) health is not simply defined in physical terms as absence of disease or disability but also includes mental and social dimensions.
Theme 3: acceptability and feasibility of CHI method in a limited resource country
Participants were asked for their views on the acceptability of the deliberate infection method by the general public. The majority of the participants indicated that the CHI method may not be acceptable at the moment. “. . . . will this be accepted in Uganda? I see with time we shall accept it. Most people, most communities are at different levels, but like I already hinted earlier on, it needs a lot of preparation. . .” -SST-Urban-01. Participants noted that the low acceptability of CHI studies may be attributed to the negative perceptions presented in theme 2 above. The other factors mentioned that could impact the acceptability of CHI method pertain to the novelty of human challenge studies in these settings, the lack of trust in the government and scientists, limited resources and absence of ethical and regulatory frameworks. These factors are expounded in the next few paragraphs.
Unfamiliarity with the concept of intentional infection
Most participants were unfamiliar with the concept of deliberate human infection, due to this some said no to CHI studies from the onset. “. . .am saying no, . . .for those things which they have [models of infection] let them use them on animals other than using human beings. . . .”-MV-Urban-09. Some participants opined that acceptability of CHI studies to the public may take a long time due to fallacies about human intentional infections. “. . . . . . . . .people may take long to accept, they have myths and misconceptions about the method. . . . . .”-CAB-Urban-03. Due to the fear of death and physical harm, some participants thought that CHI studies would be resisted and not accepted by the public. “. . . . . . . . I don’t think it will be acceptable. . . . I think even the shortest discussion we’ve had; my biggest fear is death. What if I die? What if I participate and I die? So, I think most people will have resistance. . . .”-MP-Urban-16. The lack of awareness about CHI method fuelled fears based on misinformation and misinterpretation of the goals of human infection studies.
Doubts about information from local governments and scientists
Some community members perceived that CHI research may not be acceptable to the public due to misgiving about the truth in the information from the government and scientists. “. . . . . . . . I think the acceptability of that method will really depend on how much we trust our scientists or even the government. . . . . . . and even how much information we are given. . . .”-MP-Urban-16. Other participants expressed mistrust and suspicions about the government “. . . I don’t know whether people mistrust government or what, . . . . . . . people are so suspicious of the government. . . .”-SST-Urban-01. For a few participants, the history of exploitation in medical research contributed to the distrust of scientists. “. . . . we have heard stories in the US of doctors giving black men/women infections. . . so even when we trust scientists, we know that there is a history of dishonesty. . .”-MP-Urban-16. Mistrust of government and the history of exploitation in medical research influence the negative perceptions about CHI and its acceptability in LICs. For CHI studies to be accepted in Uganda, participants opined that, sensitization and engagement of communities is required. “. . .It’s going to take a lot of community engagement, teaching, education, for them to understand. Just like even us here, we were first resistant, we are beginning to accept. . ., because of what we are looking at as the results from CHI studies. . .it’s going to take more community involvement, engagement. . . .”-CAB-Urban-03. Acceptability of CHI studies in LICs will take some time and requires respectful and transparent engagement with local communities.
Limited financial resources, legal framework and appropriate technology
Regarding feasibility, community members citied logistical challenges such as inadequate financial resources as a deterrent to the conduct of CHI studies. “. . . . . .because of the funding part of it that is struggling. . . . . . . the government doesn’t have money. . .”-MP-Urban-16. Participants opined that Uganda being a LIC would not be able to fund CHI research, more resources and preparations are required. “. . . . financial constraints. we are a developing country, one of the challenges, the financial. . . .”-SST-Urban-01. Some participants disqualified implementing these studies in Uganda, due to the lack legal frameworks specific to CHI studies. “. . . . they shouldn’t conduct these studies . . . . . . . how are the researchers going to work with the law? so right from that, you can see that it’s criminal, can’t they find other means to try and do the research without doing that [intentional infection]?”-MP-Urban-16. “. . .are there some legal provisions for this?. . .”-SST-Urban-01. A few participants believed that Uganda currently does not have the essential infrastructure for the effective conduct of CHI studies unlike in the developed countries. “. . . I don’t support that method [CHI studies] because I see it being used in the developed countries, . . . . . . our country is not yet developed to that level of those developed countries. For them they have enough resources to use to treat those participants but for us we have not yet reached such a level. . . . .”- MV-Rural-10. For CHI research to be effectively implemented in LICs, participants recommended more resources in terms of infrastructure, legal frameworks and capacity are required. Though our research participants raise some concerns about acceptability of CHI studies, they are not extraordinary, and do not violate any of the fundamental ethical principles.
Theme 4: Reasons for willingness to participate in CHI study
Although, majority of the participants were not willing to participate in a CHI study due to perceived risks, fear of the unknown and age. “. . . . for me my age cannot manage such things [CHI studies]. . . I cannot participate. . . .”-SST-Rural-12. They suggested reasons that would influence others to take part in a human infection study as presented below.
Economic gains
The majority of the participants across all focus group discussions (16/16) mentioned monetary benefits as a key motivator for participation in CHI studies, “. . . . the financial benefits!! that is number one . . . .” --CAB-Urban-03. Provided there is money, “. . . . . . . if researchers include money in it [CHI studies], people understand money. Even some of us here, if at all you said that N1 [participant number 1], I would like to take you for a CHI study, she will not think about the pregnancy she has got [all laughed] she just goes. . . .”-MV-Urban-08. Others thought that the younger adults would be more interested because of economic gains. “. . . . . . I still think you’ll get people who are interested in participating, but I’m seeing they might be young people, in my opinion. . ., their interest . . . would depend on the compensation they are going to get. . .”-MP-Urban-16. Study findings reveal monetary gains as a key motivator for participation in CHI studies, even for those that had reservations about the concept of intentional infection.
Adequate and clear participant information about the CHI method
A few participants noted that if researchers provide clear information and explanations to the research communities about the CHI method, this may influence individuals and or communities to participate in a CHI study. “. . . . . . a clear explanation of what he/she is going to be involved in, could make a person participate. . . .”-CAB-Urban-03. Assurance that there are experts available to conduct CHI studies. “. . . . . the confidence that researchers are not gambling, these are controlled [CHI studies]. . . .” -SST-Urban-01. Several participants noted that researchers need to ensure transparency and adequate information sharing “. . . . . . . We [CAB members]. .have to explain these things [CHI method] to them [community people] until they are fully understood, you know whenever we conceal some information from the people get different information. . .”-CAB-Urban-04. Transparency, education and simplifying the risks will demystify CHI method and bridge the knowledge gap. “. . .bridging the knowledge gap so that you show the advantages of participating in a CHI study and explaining exactly what is going to be done from the first procedure up to the end, any risks related to health, economic, social and mental and how they are going to be managed so that he makes an informed decision. . . . .”-UNI-STU-Urban-06. To facilitate participation in CHI studies, provision of adequate and clear information about CHI studies and risks to potential participants/research communities is essential.
Preferences for some disease conditions and model of infection in a CHI study
The pathogen, disease under study, site of administration of the infection were raised as key determinates on willingness to participate. A number of participants cited being comfortable participating in a CHI study for a disease condition that they have previously suffered from, “. . . . . . . If it is malaria, I have been suffering from malaria and I have been getting okay. I can participate but if it is bilharzia which I have never suffered from, I cannot at all, imagine a snail!! . . . .”-UNI-STU-Urban-07. Also, a CHI study investigating a disease that has a short recovery period was preferred, “. . . . . . if it’s for malaria I may participate because I know that it takes short time, it’s not something that takes many months or weeks, and then you’re back to your normal work and lifestyle . . . . . .” -CAB-Urban-03. If the illness under study has proven treatment, “. . . . . .people would be willing to take the risk if there is assurance that the diseases they are infecting people with can be cured and some have gone through it and have been healed, with no major problem. . . .”-SST-Urban-01. For a few, CHI studies for viral infections seemed scarily since viruses tend to metamorphose. “. . . . . . . viruses tend to mutate and comes out as another variant that is highly virulent and gives me most severe of the conditions. So, for mine; if there is anything, I fear more viral conditions under study. Those will be so scaring. . . .”-UNI-STU-Urban-06. “. . . . . . . I would honestly say, it depends on the pathogen they are giving me. I think I can take malaria. You don’t tell me come for COVID-19 CHI study and I come, it is a total no there!!. . . .”-SST-Rural-11. For others, the route of administration of the infection was a point to consider, “. . . . the issue of being infected maybe I can accept it but the idea of bringing mosquitoes to bite me, I don’t support it. . . .” -MV-Rural-10. Some participants opined that if a disease is prevalent in a given area, that may influence community members to participate in a CHI study. “. . . malaria is too prevalent in the central part of Uganda, so when you bring a malaria CHI study, people may come on board. . . .”-SST-Rural-12. The malaria CHI model was favoured since majority had previously suffered from malaria, got cured and it has a short recovery period.
Altruism and willingness to help others
Several participants cited altruism and patriotism as reasons why some people may accept to participate in a CHI study. “. . . . . .I think it can involve patriotism because people wish to fight for humanity, those people who say, I’m ready to fight for my country. . .”-CAB-Urban-03. “. . . . I have a feeling that there are those people who are really good-hearted, patriotic. They can say, let me sacrifice . . . . .ahaa.”-MP-Urban-16. The sense of allegiance, pride and commitment to one’s country is likely to inspire. For others the selflessness and desire to help others “. . . . . the love for helping other people. . . . a person can participate because he wants to save other people’s lives. . . .” -MV- Rural-10. Study findings reveal that some community members may feel a sense of duty to their country, viewing their participation in a CHI study as a form of national service and a contribution to the greater good.
Finally, participant mentioned the need for adequate time to ponder about the information provided as well as consult close relatives before making a decision. “. . . if I am given enough time, I can think about it and decide either to participate or not. . . .”-MV-Rural-15. Others opined consultation with family heads and close relatives “. . . . . . before CHI studies are carried out, it is good to first consult the people who own homes [family heads] . . . .” -MV-Urban-08. A handful of participants were willing to take part in a CHI study, if there is assurance of adequate clinical care and safety, “. . .as long as I am sure that there is good medical treatment to enable me heal, I can participate . . . .” -MV-Rural-15. Provision of sufficient time and the opportunity to consult family members and friends aligns with the ethical principles of respect for autonomy that ensures that participant’s rights and well-being are prioritized in the research process.
Discussion
In Uganda and other LICs, the acceptability of CHI studies as well as community members’ views regarding the concept of intentional human infection remain unknown. Yet understanding community opinions is crucial for assessing the feasibility and acceptability of CHI studies (Jamrozik and Selgelid, 2020a; Kaewkungwal et al., 2019; Vaz et al., 2021). Study findings reveal a poor understanding of the CHI method among community members, with both positive and negative perceptions expressed. On one hand, a number of participants viewed CHI studies as relevant in accelerating research and development of novel therapeutics. On the flip side, substantial concerns were raised about the possible risks associated with participation in CHI studies, and unacceptability of the deliberate human infection model. Important to note, was the perceived fear of death and associated long-term consequences. The main messages emerging from this exploration of community perceptions regarding CHI studies, divulge several opinions from market vendors, CAB members, secondary school teachers, university students, media professionals/journalists and mosques/church attendants in a rural and urban setting in Uganda.
Controlled Human Infection studies were viewed as a valuable tool for accelerating development of therapeutics, particularly for diseases that disproportionately affect LICs such as malaria. Also, participation in CHI studies was viewed as an opportunity to gain direct access to therapeutics, healthcare monitoring and early treatment options for endemic illnesses that are otherwise unavailable or unaffordable. While participants perceived usefulness of CHI studies in terms of tangible benefits such drugs, this may not be the case for most CHI studies. Particularly those conducting early phase research, developing of models of infection and not intended to lead to immediate development and approval of needed palpable therapeutics (Choy et al., 2022; Higham et al., 2024; Wildfire, 2022). In addition, sentiments that research ought to be responsive to the needs of local communities are common and plausible (Folayan et al., 2019; Khanpoor et al., 2024; London, 2008). Sharing outcomes and benefits from international collaborative research with research participants/local communities is usually cited as a moral prerequisite for research (Bedeker et al., 2022; Hurst, 2017; Lairumbi et al., 2011, 2012). How the requirement for benefit sharing is understood and perceived by local communities has important implications for acceptability and justification of CHI studies in LICs. Understanding and managing the expectations of the local populations and individual research participants is essential during community engagement (CE) and research processes. Prior and during research implementation, articulating the long term expected benefits from CHI research programmes as a whole as well as the short term and immediate benefits from solo human infection studies is necessary (Jamrozik and Selgelid, 2020b). Human infection studies were perceived as dangerous, risky and unsafe since they involve deliberately exposing “healthy” individuals to disease causing agents. The fear of death and physical harm were the most cited reasons for reluctance to participate in CHI studies. Such fears are valid and are not unique to our study. Research by Vaz et al. (2021) highlights similar concerns by members of the public. Though human infection studies have a murky history (Jamrozik and Selgelid, 2021b; Wildfire, 2022) in the recent past, CHI studies have evolved as a new methodology in modern infectious disease research that is well regulated (Balasingam et al., 2022). Rigorous ethical and scientific review by RECs and other regulatory bodies as well as safety protocols guarantees that potential harm to participants is minimized (Bambery et al., 2016; Toomey et al., 2025). Although adverse events occur, reviews by (Adams-phipps et al., 2023; Balasingam and Wilder-Smith, 2016; Toomey et al., 2025) show that a number of infection studies have been conducted safely with no deaths and few SAEs reported. Like any other research, informed consent is fundamental in CHI research. Detailed participant information about the potential risk, benefits and harm would aid in addressing the perceived fears. The availability of research expertise, world class laboratory facilities and healthcare resources to care for CHI participants provides additional assurance for risk minimization (Jamrozik and Selgelid, 2020a) though, this may not be the case in LICs. Lack of adequate healthcare systems and well financed research infrastructure are significant barriers to conducting CHI studies in LICs (Jamrozik and Selgelid, 2020a; Kaewkungwal et al., 2019). Fears on whether local facilities can provide the necessary medical care for CHI participants are not unique to this study (Kapumba et al., 2020). Development of state of the art research infrastructure as well as using residency protocol designs where necessary (Hodgson et al., 2015) can be used to reduce risks to CHI participants in LICs. Similarly, addressing the perceived fears requires a combination of compassion, education, respect of autonomy and building confidence in the research communities regarding the ability of the research facilities to ably manage adverse events that may arise in relation to participation in CHI research.
The concept of intentional infection of human participants generally was queried and rejected from the onset by community members. The pathogen under study, site/route of administration of the infectious agent were also stated as concerns likely to deter participation in CHI studies. The notion and definition of a “healthy” volunteer in CHI studies was questioned indicating that it is not just absence of disease. Queries on what scientists mean by a “healthy” participant were tenacious. Several participants raised concerns on what “healthy participants” really means in the context of CHI studies. Understanding what “healthy” means is crucial, particularly in selecting participants for clinical research. In the context of CHI studies, the aim is to gain insights into how pathogens infect human hosts and cause disease, understand immune responses to infection, or assess the safety and efficacy of new interventions in a controlled environment, and in the absence of confounding factors. By definition therefore, in a clinical study a “healthy participant” is a “person who does not have the disorder or the disease being studied, but may have other disorders that are not addressed in the specific setting of the research” (Marchesini et al., 2017). These issues are normally considered and addressed in the design of CHI research and are part of the ethical framework. We therefore opine that participants in CHI research should be in “good health” to minimize adverse effects (Binik, 2020; Darton et al., 2015).
Community members were sceptical of researchers’ motivations, fearing that they would be exploited for the benefit of others. As a result, there is suspicion about the true intentions behind CHI studies the fact that these studies involve intentional exposure of disease-causing pathogen to human beings. Distrust of scientists and government is rooted in historical abuses of biomedical research most of which involved intentional infection of vulnerable groups (Bambery et al., 2016; Franklin and Grady, 2001; Miller and Moreno, 2021; Schaefer, 2004). Although, early experiments provided valuable insights, the unethical, illegal and unconsented events of deliberate infection have marred public perceptions of CHI studies (Frieden and Collins, 2010; Jamrozik and Selgelid, 2021a). Research communities are suspicious about CHI research due to the profound scars and harm inflicted in the past. Research that is exploitative, condones equity and disrespects local cultural values is likely fuel mistrust. For CHI studies to gain popularity, it’s essential that scientists demonstrate commitment to the basic ethical principles of justice, respect of autonomy, non-maleficenc and beneficence (Drolet et al., 2023). Addressing distrust requires that researchers and governments prioritize CE, transparency and accountability. More so, effective CE and local partnerships are important for building trust, refining study designs, improving the willingness of individuals to participate in CHI studies and averting controversies that could undermine the importance of CHI research in LICs.
At the moment, CHI studies are not yet understood and may be difficult to accept by most community members. They viewed deliberate human infection as unnatural, unrealistic and religiously inappropriate. Majority are unfamiliar with the idea of human infection and uncertain about how CHI studies are conducted or what participation in a CHI study would involve. This leads to suspicions based on misinformation and misinterpretation of the goals of CHI studies. Conduct of CHI studies in LICs could undermine local confidence in research and public health more generally if suspicions, cultural and religious beliefs are not addressed. It’s important to note that these studies are new and not yet well appreciated in LICs (Vaswani et al., 2020). Despite being in existence for decades in HICs, it is only of recent that they have been introduced in LICs (Jamrozik and Selgelid, 2021b; Metzger et al., 2019; Oxford et al., 2024). Acceptability of CHI studies will take some time and requires intentional effective CE that goes beyond CABs. Even if the CHI study design might be acceptable to the local regulatory bodies including the REC in terms of risk/benefit assessment and other aspects, community acceptance should be appropriately and formally assessed. Understanding the local environment, cultural beliefs and religious contexts is crucial for ensuring that CHI studies are both acceptable and respectful of local beliefs. Community engagement is an indispensable part of setting up and maintaining CHI studies’ programme in LICs. Scholars (Karris et al., 2020; Lo et al., 2015) argue that one key lesson from HIV research is that involving communities early enhances the quality of the research and fosters a shared sense of ownership between researchers and community members. Hence, promoting community support for the research process that later translates to opportune participant recruitment, improved informed consent process, adherence to study procedures as well as facilitating the uptake of research outcomes. Researchers and advocates of intentional human infection research programmes can draw inspiration from the conduct of HIV research in which CE enhanced the quality of research and promoted a sense of mutual ownership.
The study findings are subject to limitations; research participants were unfamiliar with the concept of deliberate human infection. Prior to the group discussions educational sessions on CHIS were held. This promoted a more informed group and collection of context valuable data. Although the education materials used in this study were aimed to edify research participants about the CHI platform, information we provided could have biased the discussion and/or the study results towards the desired outcomes of the research.
Conclusion
Controlled Human Infection studies are a new and uncommon way of conducting clinical research in LICs. Although CHI studies offer valuable opportunities for advancing clinical research, they are not void of negative perceptions and contentious positions. Perceptions of CHI research by community members in low resource settings are multifaceted. Though community members across all group discussions acknowledged the relevance and potential of CHI studies to accelerate the development of therapeutics for infectious diseases, majority viewed the deliberate infection method as difficult to accept and risky at the moment. Many were not willing to participate in a CHI study due to several negative views. However, financial compensation and incentives was seen as key motivators for participation in CHI studies, even for those that had reservations about the risks. Acceptance of the human infection model by community members will take some time and would require intentional effective engagement with the community. In addition, ethical acceptability of CHI studies depends on adherence to the fundamental ethical principles of respect for autonomy, beneficence and justice. The World Health Organization (WHO, 2022) provides further guidance on the ethical conduct of CHI research, emphasizing the principles. The principles are key in ensuring that CHI studies are conducted ethically, particularly in LICs. Overall, CHI studies are acceptable, provided there is effective engagement of the community, appropriate risk benefit assessment, effective informed consent process, continuing research ethics education and a good ethical review framework in the country. Effective implementation of CHI studies in LICs also requires, improvement of local capacity (research expertise, regulatory frameworks) and research infrastructure (production, storage of infection material, medical care facilities), increasing skills and providing wider career opportunities and exposure of research stakeholders (researchers and regulators) to well established CHI programmes.
Supplemental Material
sj-pptx-1-rea-10.1177_17470161251355985 – Supplemental material for How community members in Uganda perceive controlled human infection studies
Supplemental material, sj-pptx-1-rea-10.1177_17470161251355985 for How community members in Uganda perceive controlled human infection studies by Winfred Nazziwa, Stella Neema, Alison Elliot, Samson B. Kaboko, Julius Ecuru and Erisa Mwaka in Research Ethics
Footnotes
Acknowledgements
The research was supported by a grant from the Fogarty International Center of the National Institutes of Health under Award Number D43TW010892. The contents are however, solely the responsibility of the authors and do not necessarily represent the official views of the National Institutes of Health. We are grateful for the time and invaluable contributions of research participants of this study.
Ethical considerations
The study was reviewed and approved by the Makerere University School of Biomedical Sciences Research Ethics Committee (SBS- 2023-307) and the Uganda National Council for Science and Technology (SS 1925ES).
Consent to participate
Written informed consent and permission to audio record was obtained from all participants prior to enrolment into the study.
Consent for publication
Not applicable.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The research was supported by a grant from the Fogarty International Center of the National Institutes of Health under Award Number D43TW010892.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
The datasets generated during and or analysed during the study are not publically available due to legal restrictions and confidentiality issues but are available from the corresponding author on reasonable request.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
