Abstract
Randomized controlled trials (RCTs) are considered the gold standard for demonstrating the efficacy of a treatment or intervention; however, the RCT study design alone is insufficient to prevent all forms of bias. In this paper, we review conflicts of interest and other biases that pose challenges to the methodological rigor of psychedelic RCTs, and explore recommended bias mitigation measures. We focus on biases of special relevance to psychedelic RCTs and methods of mitigating bias that we believe are actionable and urgent practices to implement in psychedelic research: (1) independent data assessment, (2) multisite trials, (3) quantitative bias analysis, and (4) open science practices. Although we advocate for the adoption of these practices in psychedelic research, they are not psychedelic specific. We suggest that the challenges posed by psychedelic research offer an opportunity for researchers to creatively find and implement solutions across the study lifecycle to reduce bias in other fields of clinical research.
Introduction
Randomized controlled trials (RCTs) are considered the gold standard for demonstrating the efficacy of a treatment or intervention. Randomizing participants to experimental or control condition(s) balances both observed and unobserved participant characteristics between groups, in addition to ensuring that participant allocation to each group is unpredictable, such that differences in study outcomes can be causally attributed to the study drug/intervention (Hariton and Locascio, 2018). The re-emerging field of psychedelic 1 research has conducted numerous RCTs testing if various psychedelic compounds can improve symptoms of mental disorders including depression and anxiety (e.g. Raison et al., 2023; Goodwin et al., 2022; Carhart-Harris et al., 2021; Griffiths et al., 2016). Yet despite its considerable strengths, the RCT study design alone is insufficient to prevent all forms of bias, both in psychedelic research and in general.
Bias and its impact on study results recently became the center of an FDA Advisory Committee discussion appraising the safety and efficacy of midomafetamine (MDMA) capsules for PTSD, which the FDA declined to approve (Reardon, 2024). Methodological problems in the sponsor’s clinical trials included functional unblinding, as 90% of participants randomized to MDMA were able to guess their study group assignment (MDMA or placebo). As a result, biases such as detection bias (when groups in a trial differ in the way outcomes are collected or verified), performance bias (differences that occur due to knowledge of intervention allocation), and expectation bias (when people’s expectations about the experiment can influence the results) may have affected study results (FDA, 2024; Mustafa et al., 2024; van Elk & Fried, 2023). Furthermore, concerns were raised that investigators, therapists, and participants allegedly held strong prior beliefs about the benefits of MDMA-assisted therapy. It was alleged that some trial therapists had reportedly discouraged negative reports by participants, and encouraged positive ones, thereby potentially introducing demand effects and resulting in skewed reports of harms and benefits (Mustafa et al. 2024). These, and other issues, have led some to doubt the validity of the data and the accuracy of reported harms and benefits from the sponsor’s trials (Mustafa et al., 2024). It has proven frustratingly difficult to understand whether such biases were present, and if so, their scope, magnitude, or effect on the data.
Concerns about bias in clinical trials are, of course, neither new nor specific to psychedelic research. Jacques Normand, study director of an Institute of Medicine panel that investigated needle exchange programs, noted that three conditions may contribute to concerns around bias in clinical research: “(a) a sensitive topic (e.g. something illegal or controversial); (b) ideological differences concerning the outcomes being examined; and (c) the research contains uncertainty” (Bachrach and Newcomer, 2002). Psychedelics may fall under many, if not all of these conditions, as these substances were outlawed in the United States by the Controlled Substances Act of 1970 and demonized by the media in the 1960s and 1970s (Siff, 2008; Siff, 2015). Uncertainty, Normand argues, may also be present if for “ethical or logistical reasons, the science is unable to implement the gold standard of a double-blind, randomized controlled trial,” (Bachrach and Newcomer, 2002) and de-blinding is known to occur in psychedelic studies (Marks, 2024; Muthukumaraswamy et al., 2021). A number of other factors (e.g. financial conflicts of interests, investigator biases, intellectual conflicts of interests, etc) can also impact data collection, rigor, and research integrity.
Researcher-level biases in clinical trials are important to address, as they violate the principle of equipoise. Theoretical equipoise (also known as personal equipoise) requires the investigator to be in a state of genuine uncertainty about the comparative effectiveness of each arm of the trial (Fried, 1974), while clinical equipoise argues that the condition of equipoise is satisfied if the medical community is in a state of genuine uncertainty (Freedman, 1987). Violations of equipoise can lead to bias, that in turn may account for a proportion of the effect of the intervention (i.e. a proportion of the effect seen is due to bias and not the intervention itself) (Cook and Sheets, 2011). Equipoise is held as an ethical justification for randomization in clinical trials, and if a trial demonstrates a consistent strong effect of the intervention (entailing that there may no longer be equipoise), all participants must be unblinded and receive the treatment (Deichmann et al., 2016). By reducing researcher-level biases, equipoise can be better maintained in clinical trials.
To address the issue of researcher-level bias, a number of measures have been proposed for clinical trials, and more specifically for psychedelic clinical trials, but have generally been unevenly applied (Buchman et al., 2024; Hovmand et al., 2023; Bradley et al., 2020). Some potential solutions include Open Science practices (methods that are aimed at introducing transparency into the research process, increasing accessibility of the data and sharing of methodology), and the application of bias-mitigation policies and regulations that the pharmaceutical industry is subject to. Uptake of Open Science practices have been steadily increasing, but are not yet the norm in many fields (Armeni et al., 2021; Christensen et al., 2020). Similarly, pharmaceutical clinical trials in the United States are subject to certain regulations that may help to address bias (Cashin et al., 2023). However, these practices are rarely combined in academic research settings—in this paper, we advocate for their application, using academic psychedelic clinical trials as an example.
We focus here on researcher-level biases that have been recently discussed as posing challenges to the methodological soundness of psychedelic clinical trials. Using adverse events reporting as a case study, we examine the impact of biases on psychedelic science. We then review measures that have been previously recommended in the literature to address these factors, then argue for four methods of reducing bias and conflicts of interest that we believe are particularly actionable and urgent practices to implement: (1) independent data assessment, (2) multisite trials, (3) quantitative bias analysis, and (4) greater use of Open Science practices. Crucially, we believe that these practices will help respond to growing calls to promote independence and insert independent assessment practices within the scientific process (Klein and Kesselheim, 2024; van Elk and Fried, 2023). We detail the rationale for these and current clinical trial bias-reduction standards and then analyze their application specifically in psychedelic research contexts. We advocate for the adoption of these measures, along with improved science communication and transparency. Overall, we aim to increase awareness of the real and perceived sources of bias in psychedelic clinical trials, and articulate ways that they can be addressed in their design, conduct, and reporting.
Researcher biases and conflicts of interests in clinical trials
Recent work in the research ethics literature has pointed to several factors that can bias the analyses and reporting of clinical trial data. Financial conflicts of interests (COIs) (see Table 1 for an overview of various COIs) have long been recognized as a potential factor leading to unethical, illegal, or unprofessional behavior on the part of the researcher (Field and Lo, 2009), with COIs in drug and treatment studies sponsored by pharmaceutical companies strongly associated with the reporting of positive findings (Friedman and Richter, 2004). In psychiatric clinical trials, financial COIs are common, with nearly half of all studies reporting at least one author with a COI (Perlis et al., 2005). Within psychedelic clinical trials, recent analyses suggest that financial relationships with industry and philanthropic funders may similarly risk the autonomy and integrity of the research (although such a risk is applicable to all research) (Buchman and Rosenbaum, 2024). Given the predominance of funding from philanthropic sources, industry, and individual donors, and the historic lack of federal funding for psychedelic research, financial COI may be of special relevance in psychedelic science (Petranker et al., 2020).
Types of conflicts of interest.
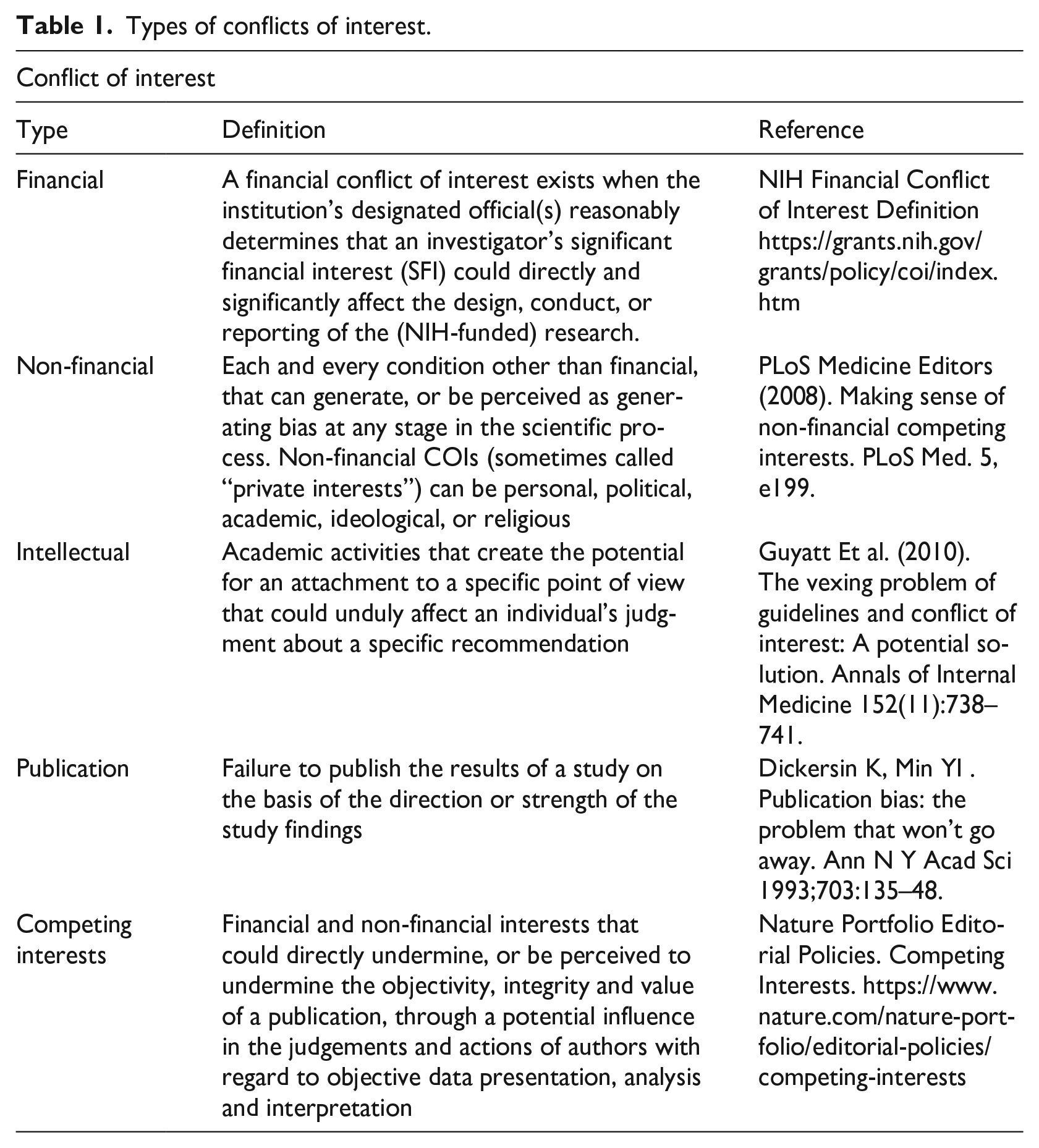

More attention has recently been paid to non-financial COIs—such as personal relationships, political activity, or the desire to please a funder—and the harms that they can cause (Grundy, 2021; Wiersma, 2018; Yusuf & Bosch, 2006). Financial and non-financial COIs are frequently intertwined; for example, pharmaceutical companies may encourage collaborations with physicians through both financial incentives and non-financial ones (for example, public recognition or endorsement of an individual’s expertise). Some effort has been made to assess the impact of non-financial COIs on biomedical research: Viswanathan and colleagues, in their investigation of strategies to mitigate non-financial COIs in systematic reviews, argue that ignoring non-financial interests may cast doubt on the impartiality of a review (Viswanathan et al., 2014). In academic clinical trials, flawed or inaccurate results may arise due to unduly strong affinity for a specialty, to a scientific theory, or to academic appearance (Østengaard et al., 2020).
A key subset of non-financial COIs are intellectual COIs. Although it may be difficult to delineate when a viewpoint becomes an intellectual COI, or whether all interests are conflicts of interest (see Bero and Grundy, 2016 for discussion), preliminary evidence has pointed to the potential bias associated with intellectual COIs. For example, one study found that primary authors of studies included in a meta-analysis were more likely to interpret the results of a meta-analysis as indicating a strong association compared to independent methodologists (Panagiotou and Ioannidis, 2012). In the context of clinical practice guideline development, Norris and colleagues found that the recommendations given for mammography screening guidelines may be affected by the specialty and intellectual interests of the guideline’s authors (Norris et al., 2012).
It is important to note that a risk of bias can still be present even if researchers do not deliberately manipulate or misrepresent their data (i.e. commit fraud), as even when trying to be objective, researchers’ judgements may still be subject to unconscious biases (Dana and Loewenstein, 2003). One source of bias for all researchers is their disciplinary perspective and the theoretical models embedded within, especially when research starts with a narrow conceptual framework. Multidisciplinary collaborations may be needed to see how researchers’ perspectives influence their work (Bachrach and Newcomer, 2002). Environmental influences may additionally be a subtle source of investigator bias (e.g. if an investigator’s university has particularly close ties with industry) (Bachrach and Newcomer, 2002). Other factors that may affect clinical trials include a number of subconscious biases such as confirmation bias—the tendency to believe what one wants or expects to believe—and anchoring bias, or the tendency to cling to a belief despite accumulating contrary evidence (Kahneman, 2011; Tversky and Kahneman, 1974).
In psychedelic research (and other fields), bias can stem from sources other than the primary investigator, such as from industry sponsors and trial therapists. As with other fields, industry involvement in psychedelic clinical trials may impact safety outcome data and trial results in a variety of ways, for example, through the selective reporting of data and the favoring of study designs that may not detect adverse events (Koning et al., 2024). Regarding non-financial COIs, as one example, the MDMA trials reviewed by the FDA (discussed above) were sponsored by a single advocacy organization, raising concerns about the influence of ideological biases on trial results (Bedi et al., 2023). Various stakeholders in psychedelic research, such as trial therapists, may also hold intellectual COIs, for example, by believing in the benefits of psychedelics to the extent of ignoring all contradictory evidence (Petranker et al., 2020). As one such case, allegations of trial therapists holding strong positive beliefs about MDMA-assisted therapy in the FDA-reviewed trials were raised, which may have led to inaccurate reporting of adverse events and outcomes (Marks, 2024). Some allege that personal use of psychedelics by trial therapists can also threaten objectivity and may be fairly common, with one survey of facilitators associated with a Phase II psychedelic trial finding that 88% had experience with at least one kind of psychedelic (although limited by a small sample size, n = 32) (Aday et al., 2023). Although personal experience with psychedelics may have benefits (Rosenbaum et al., 2024), it may also bring with it a risk that the therapist might introduce personal beliefs or interpretations to the participant which may then bias study results (Johnson, 2020).
At the researcher-level, concerns have been raised that personal use of psychedelic substances by psychedelic researchers may compromise researcher objectivity and research integrity, with one study finding that researcher associations with psychedelic culture may decrease public confidence in research quality and validity (Forstmann et al., 2021; Kious et al., 2023) 2 . Other worries involve the coupling of research with advocacy (e.g. researchers supporting the legalization of psychedelics), which some argue may also present a threat to equipoise (Poppe and Repantis, 2024).
Finally, functional unblinding, or the discerning of the assigned treatment by the researcher or participant, is an oft-encountered source of bias within psychedelic clinical trials due to the drug’s distinctive psychoactive effects (Muthukumaraswamy et al., 2021). This threat to equipoise may be compounded by other sources of bias that may be present, for example, if researchers already hold strong convictions about the beneficial effects of psychedelics.
In attending to the various biases and conflicts of interest in psychedelic clinical trials, it is important to recognize the diverse sources from which they may originate. In Box 1, we explore one potential consequence of ignoring biases in research that is of particular interest and importance in psychedelic trials: the inaccurate reporting of adverse events, efficacy, and other outcomes. Case Study: Inaccurate Adverse Events Reporting - A Risk of Ignoring Biases in Clinical Trials, and Proposed Solutions. In the following section, we review measures that have been proposed to mitigate bias in clinical trials.
Brief review of measures proposed to reduce bias in clinical trials
Various measures have been proposed and implemented to mitigate these biases in general, particularly within the context of industry-sponsored trials (for a select summary, see Table 2). In recognition of the biases potentially introduced by financial COIs, the majority of biomedical journals and funders (estimated to be over 90% of journals) have adopted financial COI policies and guidelines for researchers (Resnik, 2023). For example, the Public Health Service and the National Science Foundation in the United States requires funded institutions to adopt written policies for the disclosure and managing of funded investigators’ financial interests (National Science Foundation [NSF], 2005; National Institutes of Health [NIH], 2022), whilst many journals have modeled their policies on those published by organizations like the International Committee of Medical Journal Editors (ICMJE) (Resnik, 2023; International Committee of Medical Journal Editors [ICMJE], 2024). Moreover, the ICMJE has recently updated its policies to classify the intentional non-disclosure of COIs as a form of scientific misconduct (International Committee of Medical Journal Editors [ICMJE], 2018). Guidelines for disclosing financial COIs within the context of clinical trials and research have also been proposed and developed (Gottlieb, 2020). However, it has been suggested that COI disclosures may not be the most effective method of avoiding biases, and instead provide a false sense of comfort as they may not actually achieve their goals of preventing actual or apparent bias, nor of helping the reader to assess the influence of the COI present (Dunn et al., 2016; Goozner, 2010; Tresker, 2022; Yusuf and Bosch, 2006). De Melo-Martin and Intemann, drawing upon evidence showing that COI disclosure policies do not discourage researchers from forming financial relationships with industry, argue that COI disclosures do not help readers and reviewers identify bias in published articles (de Melo-Martin and Intemann, 2009), pointing to the need for other measures to help mitigate the impact of financial COIs.
Select summary of measures proposed to address biases.
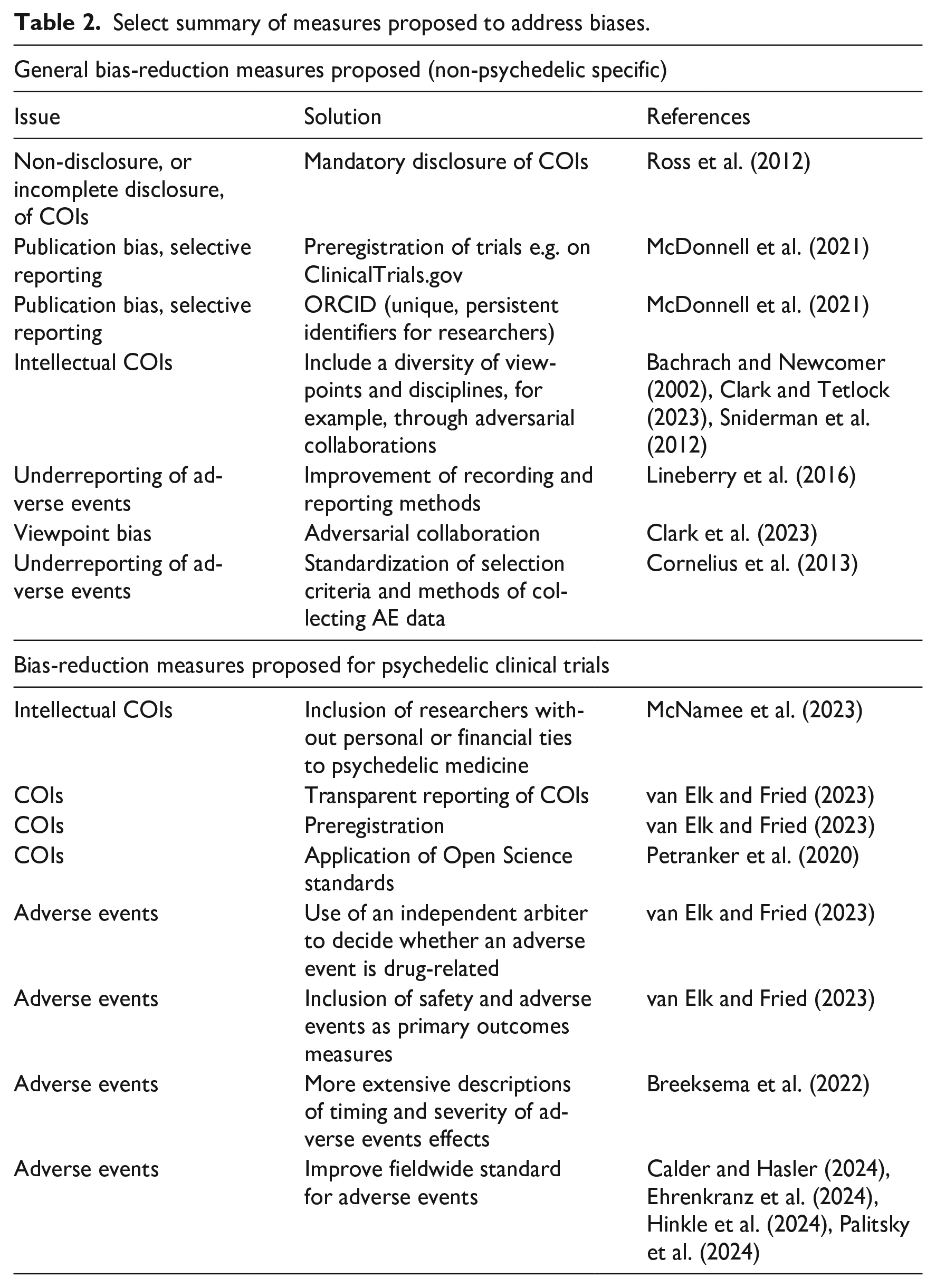
A number of policies for scientific grant review and other research activities have been created to address non-financial COIs, while an increasing number of journals have begun to adopt policies regarding non-financial COI disclosures (Resnik, 2023). For example, the ICMJE’s guidelines on COI disclosure explicitly mention that interests such as personal relationships, academic competition or intellectual beliefs may represent or be perceived as COIs, and should be disclosed (International Committee of Medical Journal Editors [ICMJE], 2024). Similarly, in 2018, Nature began requiring authors to disclose non-financial conflicts of interest, including unpaid memberships in advocacy organizations and acting as an expert witness (Nature, 2018). More generally, Sniderman et al. and Bachrach and Newcomer both advocate for the inclusion of a diversity of viewpoints to help combat intellectual COIs, arguing that fostering an open contest amongst different perspectives is one of the best protections against biases stemming from our viewpoints (Bachrach and Newcomer, 2002; Sniderman et al., 2012).
Adversarial collaborations may be one particularly well-suited method to accomplish this (Mellers et al., 2001). Engaging in adversarial collaboration restricts a researcher’s ability to design a study that preemptively favors their desired outcome (Clark and Tetlock, 2023). However, the adoption of measures within clinical trials to mitigate non-financial COIs, (e.g. employing an independent trial statistician or establishing an additional independent data safety and monitoring board) can be varied.
There have also been recent proposals in the literature specific to the psychedelic clinical trial context that aim to address COIs and researcher biases by inserting independence within the research process. McNamee and colleagues call for researchers “without personal or financial ties to psychedelic medicine” to, amongst other suggestions, join the research teams of clinical trials (McNamee, Devenot & Buisson, 2023), while Barnett and colleagues advise being “dispassionate and objective in the study of psychedelics, avoiding the quasi-religious stance that has come to characterize some elements of the ‘psychedelic movement’” (Barnett et al., 2024, p. 2). Elk and Fried similarly argue for the inclusion of industry-independent experts in all stages of the clinical trial process for industry-sponsored trials (e.g. design, data collection, writing).
To address bias from functional unblinding, employing dose-response or active comparator study designs, or using active placebos, can help mitigate this issue. Additionally, symptoms could be assessed by independent raters who would be ideally unaware of the treatment’s nature (Butler et al., 2022; Wen et al., 2024). In addition, recording participants’ and facilitators’ past experience with psychedelics, or limiting the proportion of participants with prior psychedelic use, may be helpful. Improving reporting procedures for blinding, and doing so in a quantitative and systematic fashion, will also be important for the field (Wen et al., 2024).
Other suggestions to help address COIs in psychedelic science include the transparent reporting of COIs in a publicly accessible manner (e.g. not behind a paywall in journals, upfront at conferences), and the implementation of measures that can help safeguard against COIs, such as preregistration and blinding of group membership during analysis (van Elk and Fried, 2023). Preregistration, adherence to Open Science principles, increased replication efforts and reporting constraints on generalizability to limit hype are also called for by Petranker et al. (2020). Finally, we call for increased federal funding in the psychedelic research field to help mitigate both financial and non-financial COIs for researchers.
Proposals to safeguard and insert independence within the scientific process
In the following section, we detail four methods that we argue will help respond to the above-described calls to insert independent assessment within the scientific process (e.g. for adverse events reporting and for the review of prior research) (Klein and Kesselheim, 2024; McNamee et al., 2023; van Elk and Fried, 2023; Petranker et al., 2020). The methods described below introduce independent assessors or sites to help reduce the influence of researcher-level biases, with quantitative bias analysis and Open Science measures adding an additional stage where the potential influence of bias can be checked and mitigated.
Independent assessors and sites— if sufficiently resourced, free from conflicts of interest and separate from the institution running the trial/experiment— can add a valuable layer of oversight to trial procedures, especially those which include a subjective component (Dodd et al., 2008). Their use can promote objectivity in the running and analysis of the trial, reduce bias, and protect participant safety, all of which may contribute to promoting the trial’s credibility and trustworthiness (Schöffski, 2021)—one survey from the Pew Research Center (2019) found that open access to data and independent review increased Americans’ trust in research findings. Although more work is needed to describe and standardize the concept of independence and how it can be established for the assessor/site, such methods can contribute to improving the rigor of research conducted not just in psychedelic science, but in all fields.
Proposal 1: Independent assessment of clinical trial data
Independent/external assessments of clinical trial data (e.g. of primary outcomes, adverse events) are one method that can help combat researcher biases and conflicts of interest, improve clinical trial data’s rigor and accuracy, and ensure objectivity (Ross et al., 2012). Such an assessment could consist of “an independent evaluation of the data conducted by professionals with no ties to, or financial compensation from, the sponsor or its surrogates. . .” and “accompany the published abstract in medical journals” (Haines and Miklos, 2011, p. 10). As one example of this in practice, JAMA requires industry-sponsored studies to have data analysis conducted by an independent biostatistician (Deangelis and Fontanarosa, 2010). Specific calls from psychedelic researchers for the injection of more independence include suggestions to hire independent auditors to investigate and share results from clinical trials to help rebuild trust, and for independent arbiters to determine whether adverse events are related to the treatment in question (Marks, 2024; van Elk and Fried, 2023).
Independent assessment and oversight methods are not new, and have been implemented for clinical trials in various medical areas. Independent oversight of research frequently occurs via Institutional Review Boards (IRBs), while Independent Data Monitoring Committees (IDMCs) are commonly used in both academic and industry-sponsored trials to help protect the interests of the patient, and to ensure participants are not being exposed to unnecessary risk. IDMCs may additionally analyze interim data for large studies, with a focus on the safety of the patients enrolled (Sartor and Halabi, 2015; Walters, 1993). In response to concerns about investigator bias and honest reporting of adverse event data in industry-sponsored trials, Auerbach and colleagues propose that an Independent Clinical Events Committee (CEC) could help to review and adjudicate clinical events data accordingly (Auerbach et al., 2013). They found that for an industry-sponsored spine surgery clinical trial, an independent CEC reclassified 37% of adverse events (the large majority being an update in severity)—such a committee thus could potentially help to mitigate investigator bias and facilitate an accurate assessment of safety data (Auerbach et al., 2013). Another study found that for a Phase III oncology clinical trial, investigators tended to overestimate response rates when compared with an Independent Review Committee, pointing to the benefit of their inclusion for reducing investigator bias (Tang et al., 2010).
For psychedelic clinical trials, implementing an independent assessment of data post-publication could take the form of making more data publicly available, following the principles of Open Science, with appropriate measures to secure participant confidentiality, thereby allowing independent researchers to perform their own analysis. Pre-data publication, independent assessment could entail having an independent panel or committee analyze interim data (including video recordings of experimental procedures) at prespecified time points to ensure that data is being collected accurately and that patient safety is not compromised. This analysis and review could also be conducted by a Contract Research Organization. Following van Elk and Fried (2023) and Auerbach and colleagues, an independent committee could also make decisions regarding the relatedness and severity of adverse events.
Proposal 2: Multisite trials
Multisite trials are another method that may reduce the risk of bias and improve the rigor of clinical trial data. Such trials can reduce personal biases from researchers in running the study, given the variety of sites and researchers (Das, 2022). Moreover, they provide opportunities to draw upon various methodological, statistical and clinical expertises and perspectives, given the participation of a larger number of investigators (Fuller et al., 1994). There may also be a decreased risk of data manipulation and fishing, while having diverse sites also allows for comparison across site groups and the detection of variation (Das, 2022). Multisite trials additionally may lead to more diverse study samples, as they offer opportunities for subject recruitment from broader and more representative sampling pools or geographic regions, and may lead to more rapid recruitment of participants (Flynn, 2009).
In psychedelic clinical trials, multisite studies may reduce the likelihood of researcher-level bias as each site could be run by different investigators (e.g. academic sites and industry sites), who may have different backgrounds and training and would be unlikely to share the same potential biases or COIs. Lack of diverse ethnoracial inclusion is common in psychedelic studies (and clinical research generally), but multisite trials provide more opportunities to recruit and enroll larger samples of diverse, representative participants (Hughes et al., 2024; Thrul et al., 2021). Currently, a variety of therapeutic and therapeutic support modalities are used in psychedelic clinical trials (Cavarra et al., 2022), but conducting a multisite psychedelic study would create an additional need and motivation to standardize therapeutic procedures and reduce variability between sites. Comparing results and adverse events between sites can help identify any issues with study procedures and implementation, and support the development of best practices that can be shared publicly to benefit future studies. The independence of each site would be a key component to reducing bias, and criteria must be established in order to define and ensure the independence of each site.
Proposal 3: Quantitative bias analysis
Quantitative bias analysis (QBA) is a set of methods that can be used to evaluate the sensitivity of study results to systematic error (i.e. bias) and to assess the effects of bias on the direction and magnitude of effect estimates (Fox and Lash, 2020). QBA can address multiple forms of bias, including information bias (e.g. misclassification), selection bias (e.g. differential loss to follow up between treatment arms), and confounding (Lash et al., 2009). To address bias, QBA can be performed at any stage of study design or analysis (Fox and Lash, 2020). Conducting QBA is of particular importance when seeking to understand causal relationships and conducting analyses that may inform policy decisions or clinical practice (Lash et al., 2009).
Especially given the concerns around bias influencing the results of psychedelic clinical trials, QBA may help address any bias that may have occurred during the conduct or analysis of a study, including investigator-level cognitive biases. Specific examples include using QBA to address underreporting of adverse events, overly positive outcome scores, and non-representative participant demographics. Including QBA during the study design phase may help design an internal validity substudy to address bias and/or determine which data will be most important to collect because of the likely effect on study results (Lash et al., 2009). Conducting QBA once study results are known can delineate the magnitude and direction of bias on the results, thereby providing an understanding of what the effects would have been had there been no bias. Pre-registering and publishing QBA plans and results demonstrates transparency and can promote informed discussion within the field about reducing bias in clinical trials.
Proposal 4: Open Science in clinical trials
Discussions of bias in clinical trials and their accompanying mitigation strategies have generally proceeded separately to discussions about Open Science practices and the “replicability crisis” (that published research findings frequently cannot be replicated). Although no formal definition exists, characteristics of Open Science include transparency at all stages of the research process (Ramjoué, 2015), accessibility of data, results and publications (Bisol et al., 2014), sharing of methods and intermediate stages of the research (Grubb and Easterbrook, 2011), and being collaboratively developed (Hormia-Poutanen and Forsström, 2016). The importance of independent replication of results and reproducibility has also been re-emphasized by the Open Science movement (Schooler, 2014).
Several Open Science practices have been proposed, with the goal of reducing bias and improving the methodological rigor of clinical trials and in research more generally. Pre-registration, the sharing of clinical trial data and code, and registration at ClinicalTrials.gov promote the replication of analyses and greater transparency. In the United States, the mandatory registration of certain clinical trials (i.e. at ClinicalTrials.gov) by the FDA Amendments Act of 2007, along with primary and secondary outcomes, has helped to establish a record of research and to prevent publication bias (Bradley et al., 2020); however, many other non-drug related academic studies do not use this platform and registration can be incomplete (e.g. not uploading informed consent forms within the mandated period, see Axson et al., 2024). In some other countries, Sunshine Acts, which require the disclosure of physicians’ financial interests, have been implemented (Fabbri et al., 2018). Other suggestions aimed at reducing bias in research include establishing an accessible and regularly updated database of COIs from researchers, institutions, physicians and patient advocacy groups (Bradley et al., 2020).
We suggest that, if not already implemented, psychedelic clinical trials in academic settings could benefit from greater use of Open Science practices (e.g. preregistration, open sharing of data, and sharing study materials, replication), in addition to adopting the methods discussed above of reducing bias such as the independent assessment of data. These bias reduction methods should be acknowledged as related to and contributing toward Open Science, as they aim to reduce researcher-level and other biases in research, improve transparency, and help to establish trust in research. One approach to encourage the use of such practices may be to create Badges (akin to Open Science Badges) (Figure 1—Open Science badges) that acknowledge and incentivize the use of these methods (Kidwell et al., 2016). Another promising avenue for independent replication of psychedelic research are programs such as the National Institutes of Health’s recently established Replication Initiative, which provides funding for researchers to replicate their results with a third-party Contract Research Organization (NIH, 2025).
Badges to acknowledge open practices, Center for Open Science, CC-by attribution 4.0 international, https://osf.io/tykzf. While Open Science badges exist for preregistration, open data and open materials, we suggest additional badges be available for those who choose to implement the methods we describe, such as a badge for independent statistical analysis.
Implementing certain Open Science methods within psychedelic research may be difficult, given the sensitive nature of the data involved (e.g. video recordings of dosing sessions, psychiatric profiles). Without careful consideration and thought, open data sharing could potentially violate participants’ safety, privacy, and confidentiality. While the open sharing of video recordings of dosing sessions may not be feasible without violating the privacy of participants, the sharing of other study materials, such as protocols and summary data, may also contribute toward the goal of transparency while preserving participants’ privacy. In general, assessing the risk of disclosure and re-identification will be crucial in deciding which forms of data to share (Alter and Gonzalez, 2018).
Conclusion: Summary and recommendations
In the previous sections, we have reviewed various types of researcher-level biases and their specific application to psychedelic research. To help facilitate an in-depth exploration and discussion, we focused here on measures that may help to address real or perceived researcher biases and conflicts of interest—however, it is outside the scope of this paper to discuss other issues that may affect the accuracy of study results such as fraud, poor study design, and the external validity of study results. Other approaches will be needed to improve the rigor and validity of the research conducted, and to help ensure equipoise is maintained.
We described and advocated for four specific measures that can be implemented to reduce the risk of biases (both real and perceived) in psychedelic clinical trials: (1) independent assessment of the data, (2) multisite trials, (3) quantitative bias analysis, and (4) increasing the uptake and use of Open Science practices. Crucially, many of these potential solutions rely upon the concept of independence and independent review, which requires commitment and effort to introduce and maintain. Although we suggest that these methods be applied to psychedelic clinical trials, they are not psychedelic-specific measures and may be good practice for all clinical trials. We acknowledge that such practices require varying amounts of time, effort, and resources to incorporate, and some may be unfeasible given the resources and funding available. These barriers do present obstacles to their implementation: nonetheless, conducting rigorous, internally valid and trustworthy research is critical to good science and advancing the field of psychedelics. Establishing these practices as norms for the field and increasing investment in these practices will be essential to future psychedelic science.
Finally, while our discussion of these issues were motivated by the current challenges within psychedelic research, they are not unique and are present in many other scientific fields. By looking to and learning from both Open Science and methods that have been used to reduce bias in clinical trials (in industry-sponsored and academic research settings), psychedelic research can work toward improving its rigor and transparency in addition to suggesting improvements that could be more widely adopted in science more generally. We believe that the challenges in the psychedelic field offer an opportunity to creatively find and implement solutions that can then be applied to improve the rigor of clinical research more generally.
Footnotes
Acknowledgements
We are grateful to the two anonymous reviewers for their comments that greatly improved the content of the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
D.B.Y. reported research funds from the National Institutes of Health (NIH) and Heffter Institute as well as support support through the Hopkins Center for Psychedelic and Consciousness Research funded by Tim Ferriss, Matt Mullenweg, Craig Nerenberg, Blake Mycoskie, and the Steven and Alexandra Cohen Foundation. He has delivered paid lectures to the Integrative Psychiatry Institute.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Ethics approval
The authors declare that research ethics approval was not required for this study.
