Abstract
Despite their vulnerability, adolescents are often excluded from health research due to ethical concerns about research with minors, especially in low-income regions like Sub-Saharan Africa. We enrolled adolescent girls aged 15–17 years and caregivers of girls of the same age. Using a 25-question Comprehension Score Sheet, we applied a quantitative approach to compare the comprehension of informed consent of 33 adolescent girls and 41 caregivers of adolescent girls aged 15–17 years. The assessments were audio-recorded and reviewed for quality check. The results showed that adolescent girls were significantly better than caregivers in comprehending informed consent information overall and specifically on study procedures, voluntarism and study purpose. This suggests that adolescents can understand informed consent information at the same level as or better than caregivers who are entrusted with providing permission for adolescents to participate in research.
Introduction
Despite their vulnerability to HIV infection (Day et al., 2020; Kudowa et al., 2019; Mabaso et al., 2021; Tucker et al., 2019; United Nations Children’s Fund [UNICEF], 2021), adolescents are often excluded from health research (Loveday et al., 2022; Santelli et al., 2017; Santos, 2012) because of inconsistent or lack of standardized guidelines (Day et al., 2020; Folayan et al., 2014; Marsh et al., 2019), and ethical-legal frameworks that create numerous challenges related to adolescent consent to research participation (Day et al., 2020; Loveday et al., 2022; Singh et al, 2006). Thematic analyses by Munung et al. (2022) and Nkosi et al. (2022) have shown inconsistencies and ambiguities in the existing legal and ethical frameworks across different countries. The existing status underscores the need for consistent and clearer guidance on caregiver waivers and adolescent self-consent. The 2016 version of the International Ethical Guidelines for Health-related Research involving Humans try to address this by providing for circumstances in which caregiver waivers are ethically justified to grant adolescent self-consent. This includes situations where the risk level is minimal and where special protections are put in place to ensure the protection of a minor’s best interests (WHO, 2011).
Researchers often avoid having to deal with the need to obtain caregiver permission for minors below the legal age of consent, and thus have restricted research enrolment to emancipated minors and adults (Marsh et al., 2019; Mutea et al., 2019, 2020) to the extent that research studies are first conducted with adults to obtain data. It is only after confirming the intervention is safe and effective in adults that they are implemented among minors and/or adolescents. For example, 12 trials of the effectiveness of oral pre-exposure prophylaxis (PrEP) 1 were conducted among adult populations (Baeten et al., 2012; Choopanya et al., 2013; Grant et al., 2010; Grohskopf et al., 2013; Thigpen et al., 2012). Once the efficacy of PrEP was ascertained, it was rolled out for implementation science, and demonstration and observational programmes among adolescents aged 15–17 years (Gill et al., 2017; Machado et al., 2017; Mathur et al., 2018; Ministry of Health, Zambia, 2019; Mullins et al., 2017; Saul et al., 2018). Studies on HIV self-test were also first conducted among adults (Mavedzenge et al., 2011; United Nations, 2010; US Food and Drug Administration- Consumer Health Information [US FDA-CHI], 2012) and later on rolled out to adolescents (Hatzold et al., 2019; Inwani et al., 2021; Zanolini et al., 2018).
Comprehension by a research volunteer is critical to the effectiveness of a consent procedure and determines how the participant is empowered to use the information to make an informed decision on whether or not to participate in a research study (Afolabi et al., 2018; Iseselo et al., 2024; Shah et al., 2018). It also determines the applicability of research ethical principles (Tam et al., 2015). Consent forms are often long and complex with wording that exceeds average reading level, and potential participants are likely not to recall critical elements of informed consent (Afolabi et al., 2014; Burgess et al., 2019; Dyer et al., 2022; Foe and Larson, 2016; Neelakantan et al., 2022; Ossemane et al., 2018). The conventional concept of informed consent applies to decisions about research participation made by those with the legal and cognitive capacity to make such choices (Bazzano et al., 2021; Biros, 2018; Kraft et al., 2017). Research ethics guidelines list five critical elements of informed consent; a research volunteer gives informed consent to be enrolled in research if the volunteer is competent to act, receives a detailed disclosure of relevant research information, comprehends and processes the disclosed information, acts voluntarily and consents to participate in a research study (Council for International Organizations of Medical Sciences [CIOMS], 2016; National Committee for Research Ethics in the Social Sciences and the Humanities [NESH], 2022; World Health Organization [WHO], 2011; World Medical Association [WMA], 2013).
Adolescents are minors and considered vulnerable because their level of maturity is likely to compromise their understanding of voluntarism and ability to choose freely, and may therefore not fully comprehend the risk of participating in research compared to adults (Folayan et al., 2014; Grootens-Wiegers et al., 2017; Madden et al., 2016). Therefore, adolescents need special protection under legal guidelines in different countries and in international ethical guidelines when they are to be involved as research participants (Nkosi et al., 2022). Regulatory institutions prescribe the support and protection of their caregivers in order to create a setting in which they can make competently informed decisions (Crane and Broome, 2017; Grootens-Wiegers et al., 2017; McGregor and Ott, 2019). However, the need to obtain caregiver permission prior to involving adolescents in health research has some challenges (Folayan et al., 2014; Santos, 2012). There are concerns that once permission is secured from their caregivers, adolescents may be enrolled in a study without any additional efforts to explain to them the implications of their participation in the research (Okello et al., 2013). Equally central, is the assumption that caregivers understand the studies sufficiently to give informed permission for minors under their care to participate in research.
We explored the understanding of an informed consent document (ICD) prepared for a randomized controlled trial (RCT) on secondary distribution of self-tests to promote HIV testing among partners of sexually active adolescent girls aged 15–19 years (Wango et al., 2023). The purpose of this study was to determine if adolescents in this age group would be able to understand the consent as well as, better than, or worse than their caregivers in order to inform our plan to request for waiver of caregiver permission for the RCT we were designing. In this paper, we quantitatively compare the comprehension of the ICD between adolescent girls ages 15–17 years and caregivers of adolescent girls in the same age bracket, to inform future HIV research involving adolescents.
Methods
Development of consent comprehension assessment tool
Questions for consent comprehension assessment tool were derived from all sections of a standard informed consent document, with more questions on sections that present the reasons for the study, the procedures involved, confidentiality and voluntarism. Trained Research Assistants (RAs) with long experience in consenting participants helped in collating consent comprehension questions from about 40 studies, including over 10 studies with adolescents, that Impact Research and Development Organization (IRDO) – a local organization under which the study was conducted – has implemented in the past. Questions on Purpose, Study Procedures and Risk and Benefits were derived from the consent document to be used in the planned RCT. Based on the RA experience and that of the lead authors (GNW, JO and KA), the team selected the questions to ask and how to frame them.
Participant recruitment
The study partnered with counselling staff in our DREAMS (Determined, Resilient, Empowered, AIDS-Free, Mentored, Safe) programme and other HIV testing and counselling programmes in Siaya County to refer adolescents with negative HIV status to participate in the interviews. DREAMS is a public-private partnership programme, funded by the United States President’s Emergency Plan for AIDS Relief, that provides a comprehensive, multi-sectoral package of interventions to address structural factors, such as gender-based violence, exclusion from economic opportunities and a lack of access to secondary school, all of which increase their vulnerability to HIV (Saul et al., 2018; USAID, 2023; USAID.gov/global-health). HTC Counsellors working in DREAMS and other HTC programmes were given orientation on the study and eligibility criteria and were asked to refer girls reporting at least one sexual act in the previous 3 months, HIV negative, documented as female and aged between 15 and 17 years, to the study for more information and eligibility screening. Recruitment fliers with study information including eligibility criteria and contact details were also posted in the DREAMS safe spaces and issued to DREAMS mentors to give to adolescent girls who were interested. Additionally, information about the study, eligibility criteria and contact details were announced in churches, market places, health facilities, youth groups and other appropriate locations.
Caregivers were recruited through referrals by the adolescent girls who had participated in the study, and through peer referrals by caregivers who were already enrolled. Adolescents were enrolled and interviewed first, then asked if they were willing and comfortable to refer any caregiver of an adolescent aged 15–17 years to the study. They were informed that the caregiver need not be their caregiver but that they were free to refer their own caregivers if they were conformable doing so. They were informed that they did not have to inform those they were referring that they had also participated in the study. Caregivers were not linked to the adolescents who referred them, and were not informed that adolescents had also participated in the same study.
Eligibility criteria
Eligibility criteria for adolescent girls included having been tested for HIV within the past 1 month as evidenced by a referral card from an HIV testing site, residence in Siaya County, having a male sexual partner with whom they intend to meet at least twice in the next 3 months (for an opportunity to offer self-test kits), and willing and competent to provide written informed consent. These eligibility criteria were a replica of those in the planned RCT whose consent was assessed for comprehension. Caregivers were eligible if they were taking care of an adolescent girl aged 15–17 years (not necessarily those enrolled in the study), resident of Siaya County, and willing and competent to provide written consent.
Consenting process
Both adolescents and caregivers were taken through the consenting process in the language of choice by trilingual RAs fluent in English, Kiswahili and Dholuo (local language). Once enrolled, the same RA took them through a second consent document for the planned RCT that was to assess the impact of HIV self-testing on male partner and couple testing. A standard approach was used in administering the informed consent, where literacy was assessed in participant’s language of choice (an independent person witnessed consenting of illiterate or semi-literate participants) and a copy of the consent given to the participant.
The study staff then read out the ICD verbatim as the participant listened and followed along, asking questions and being asked questions about key aspects to assess understanding; they however did not sign this second consent. Participants were not informed ahead of time that their comprehension of the ICD would be assessed later in order to not pay keener attention than they would in a normal consenting session. They were then directed to a second RA for more detailed assessment of comprehension of the second ICD. For uniformity in consenting, the first RA administered all the consents while the second RA assessed comprehension of all participants.
Informed consent comprehension assessment
A 25-question Comprehension Score Sheet was developed with questions assessing understanding of: background and purpose of study, study procedures, voluntarism, risks and benefits, compensation, confidentiality, ethics committee and contacts, among other topics (for details, see Table 1). For each response option chosen, participants were asked to provide a brief explanation of their understanding. To confirm whether participants were confident about their responses and not guessing the multiple choice answers provided, each question was asked three times and participants were informed they could repeat the same answer three times if they were confident of their answer or change their response if they felt the previous response was incorrect. They were assured that asking the question multiple times did not mean their previous response was incorrect. The second RA documented the responses as they were given. The interviews were audio-recorded, transcribed and translated into English where needed.
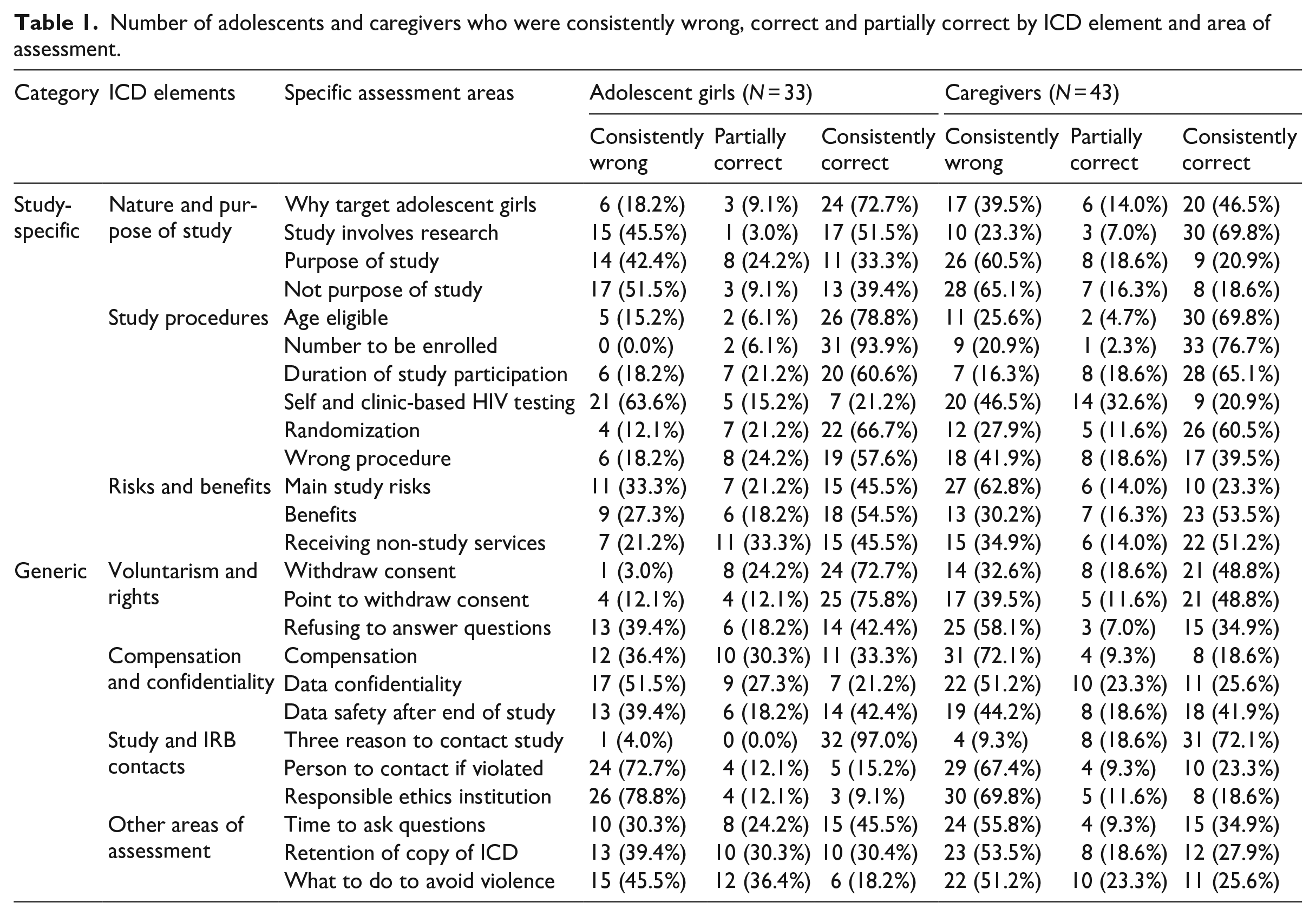
Number of adolescents and caregivers who were consistently wrong, correct and partially correct by ICD element and area of assessment.
Quality and integrity assurance
To ensure quality and integrity of data collected, one investigator listened to all the audio-recordings to ensure the guide was used appropriately and consistently and that the transcriptions were accurate. The lead investigator (GNW), who was blind to the previous scores by the RA, also independently scored the responses using the same score sheet. A team of the senior author, the two RAs who administered baseline consent and consent comprehension (LO and MO), the data officer who transcribed the comprehension sessions (NO), and two other RAs who participated in other phases of the study sat together and reviewed all discrepant scores between the RA and the investigator. For example, for a participant who was scored C, C, B in the three responses to a question by the RA and A, C, B by the investigator, the team reviewed the transcripts and resolved the discrepant scores; where not clear, the team listened to the original recorded response to determine the actual response. For each question, participants were considered to be correct if they were consistently correct in all three attempts and consistently wrong if their responses were consistently incorrect in all three attempts. Participants were considered to be partially correct (unsure) if their responses were correct in one or two attempts and wrong in at least one attempt.
Data processing and analysis
Using the 25-question Comprehension Score Sheet, we applied a quantitative approach to compare the comprehension of informed consent of the adolescent girls and caregivers. The 25 comprehension questions were collapsed into 9 sections grouped into 2 categories including: (1) study-specific elements (nature and purpose of study, study procedures and risk and benefits); (2) generic elements (voluntarism and rights, compensation and confidentiality, study IRB and contacts and other elements). We then computed the number of participants who gave correct, partially correct and wrong responses in each category on the three responses per question. To determine the mean score per category of questions, the responses were first weighted. A correct response at the first attempt scored a weight of 3, at second attempt scored 2 and at third attempt scored 1; wrong responses were scored zero. The scores therefore ranged from 0 to 6 for each question. The average score per category was the mean average score of questions in that category. The average score per subpopulation (adolescents and caregivers) was computed by getting the summation of scores of all participants in each sub-population per category divided by the total number of participants in the sub-population (see Table 2). Descriptive statistics was applied to summarize data. A paired t-test was conducted to compute the overall mean scores for both adolescents and caregivers.
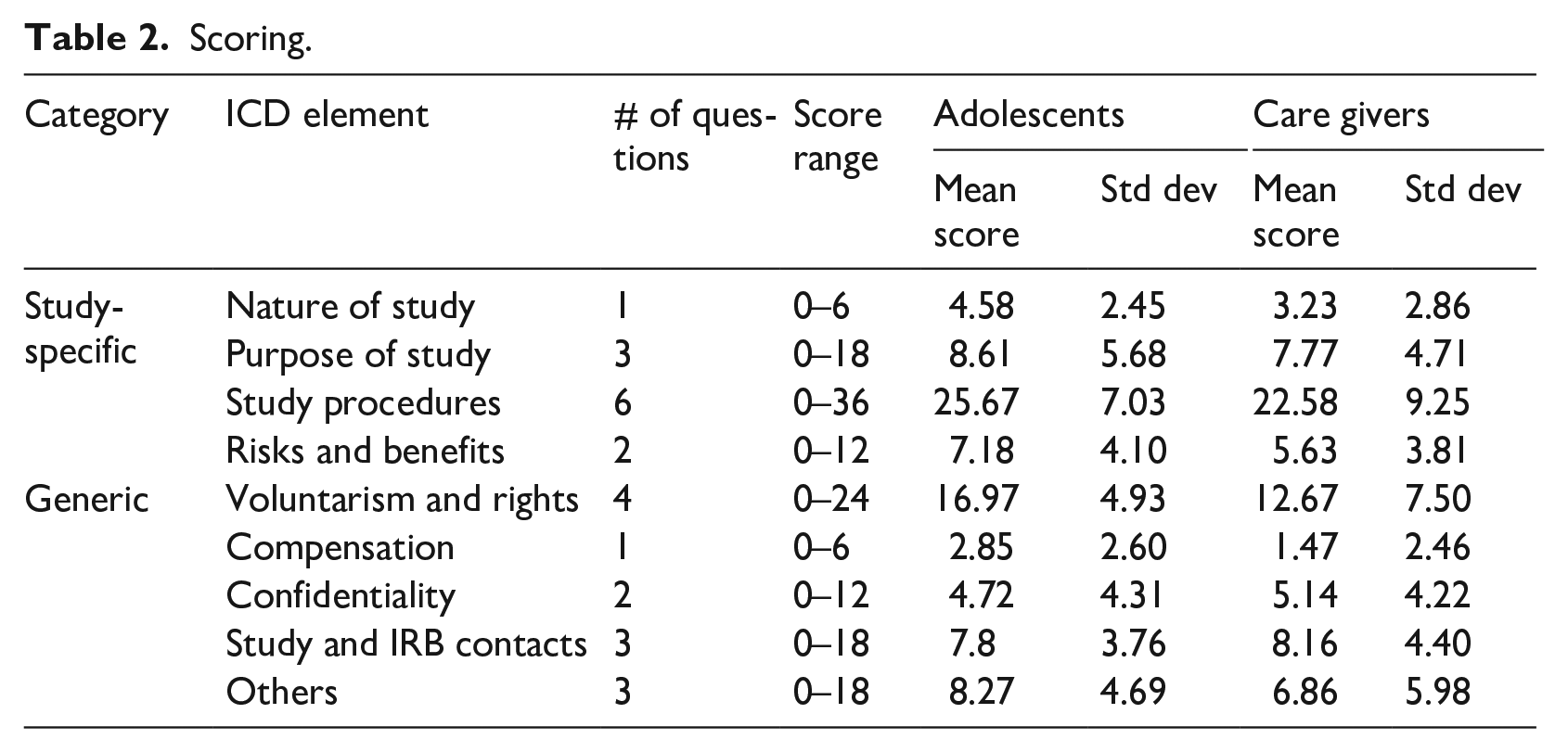
Scoring.
Ethical considerations
Ethical approval for the study was obtained from Maseno University Ethics Review Committee on 29th January, 2016. Due to concerns over potential for inadvertent disclosure of adolescent’s sexual activity to caregivers through reading the ICD, the Ethics Review Committee granted waiver of written consent. All study staff and HTC Counsellors working in IRDO’s DREAMS programmes were taken through the basics of research ethics with humans by the study investigators and all signed a confidentiality agreement. Enrolled participants were assigned unique identification codes which were used instead of their names or any identifying information. The consent document was developed using the equivalent of Grade 6 reading level of English to make the document relatively easy to understand. For girls willing to participate in the study but unable to read and/or write, an independent witness was engaged to sit throughout the consenting process. However, questions on comprehension were asked to the participant after releasing the witness.
Results
Participant demographic information
A total of 33 adolescent girls aged 15–17 years and 41 caregivers aged 23–64 years consented to participate in the informed consent comprehension assessment. The mean age for adolescents and caregivers was 16.5 and 41.9 years, respectively. Slightly more than half of the adolescents (51.5%) had at least some secondary education in comparison to about 19.5% of the caregivers who had at least secondary education. About 97% and 80.5% of the adolescents and caregivers, respectively, had attained at least some primary education.
Background and purpose of study
Adolescents were more consistently correct on why the study targeted adolescent girls, at 72.7% compared to 46.5% of the caregivers. However, caregivers were more consistently correct, at 69.8%, in their understanding of the study as a research undertaking compared to the adolescents, at 51.5%. The overall mean score for adolescents and caregivers on study background was 4.58 (σ = 2.45) and 3.23 (σ = 2.86) out of a total score of 6, respectively. About one-third of the adolescents (33.3%) and one-fifth of the caregivers (20.9%), with mean scores of 8.61 (σ = 5.68) and 7.77 (σ = 4.71), out of the total score of 18, respectively, were able to consistently recall the purpose of the study.
Study procedures
Both adolescents and caregivers tended to be consistently correct on questions assessing their comprehension of study procedures that related to age eligibility, sample size and duration of participation; however, adolescents were more consistently correct in understanding randomization than caregivers. The adolescents’ scores on study procedure ranged from 57.6% to 93.9% compared to 39.5% to 76.7% for caregivers. Only about 20.0% of both adolescents and caregivers were consistently correct on the distinction between oral self-testing and clinic based HIV testing. The overall mean score on questions about study procedures for adolescents and caregivers was 25.67 (σ = 7.03) and 22.58 (σ = 9.25) out of a possible 36 scores, respectively.
Voluntarism and rights to withdraw consent
Whereas caregivers tended to be average (48%–51%) on questions about voluntarism and rights, adolescents were more consistently correct on these questions with scores ranging from 42.4% to 75.7%. The overall mean score for adolescents and caregivers on study voluntariness and rights was 17.0 (σ = 7.5) and 12.62 (σ = 4.93) out of 24, respectively.
Risks, benefits and compensation for time
Almost double the proportion of the adolescents (45.5%) was consistently correct on the question about the main risk of participating in the study compared to the caregivers (23.3%). However, the proportion of adolescents and caregivers who were consistently correct on the benefits of the study was almost same, at 54.5% and 53.5%, respectively. The overall mean score for adolescents and caregivers on main risk and benefits of participating in the study was 7.18 (σ = 4.1) and 5.63 (σ = 3.81) out of 12, respectively. The scores on questions about compensation for both adolescents and caregivers were low. About one-third of adolescents (33.3%) and nearly one-fifth of caregivers (18.6%), with means scores of 2.85 (σ = 2.60) and 1.47 (σ = 2.46) out of 6, respectively, were consistently correct on this question.
Confidentiality and other rights of participants
On questions that assessed confidentiality, about one-fifth of adolescents (21.2%) and a quarter of the caregivers (25.6%), with mean score of 4.72 (σ = 4.31) and 5.14 (σ = 4.22) out of 12, respectively, were consistently correct. Both adolescents and caregivers were assessed on other elements of ICD including: retaining a signed copy of the ICD, asking questions during consenting process and how to avoid intimate partner violence (IPV) in the study that was being proposed. Nearly half of the adolescents (45.5%) were consistently correct on the question on when participants could ask questions compared to 34.9% of the caregivers. Only 30.4% of the adolescents and 27.9% of the caregivers were consistently correct on the question about retaining a copy of the consent/assent form while 18.2% of the adolescents and 25.6% of the caregivers consistently recalled what one would do to avoid IPV.
Study and IRB contact information and roles of IRB
The proportion of adolescents and caregivers who were consistently correct on the question about study and IRB contact information were 15.2% and 23.3%, respectively, with mean scores of 7.8 (σ = 3.76) and 8.16 (σ = 4.40) out of 18, respectively. Noticeably, only 9.1% of the adolescents and 18.6% of the caregivers could consistently recall the IRB as responsible for overseeing the ethical implementation of the study while almost all adolescents (97.0%) and about three-quarters of the caregivers (72.1%) were consistently correct on the main reasons why they may contact the study and IRB about their participation and need for information.
Summary of findings
Overall, there were minimal differences between adolescents’ and caregivers’ comprehension of most of the ICD elements including; purpose of study, benefits, confidentiality and compensation. There was a mean score difference of 1.46 (σ = 1.5) between adolescents and caregivers, with a 95% CI of 0.31–2.61 scores. Using a two sample t-test comparing the overall score for; nature of study, purpose of study, study procedures, risks and benefits, voluntarism and rights, compensation, confidentiality and study and IRB contacts among others, the mean overall score for adolescents was 9.6 while that for caregivers was 8.2. This difference was significant at p-value = 0.02 indicating that the adolescents were significantly better than caregivers in comprehending the ICD.
Discussion
Participants’ understanding of key elements of a research consent document and process is essential to their enrolment in any research with humans, and for minors, caregivers’ understanding is equally important before they can be deemed competent to give permission for minors under their care to participate in research studies. Overall, our results showed that adolescents demonstrated a higher level of understanding of the elements of ICD compared to adult caregivers, perhaps because their higher education level compared to caregivers compensated for their lower chronological age.
Understanding of background and purpose of study
The number of caregivers who were consistently correct on the background and purpose of the study was markedly lower compared to adolescents. Even though greater proportions of the adolescents and caregivers were consistently correct on the study background, their recall of the purpose of the study was low. This finding is akin to the findings of a study conducted in Mozambique that showed that adult participants had a challenge recalling the study purpose and objectives just after a day (Ossemane et al., 2018). The failure to understand the nature and purpose of the research is likely to undermine the validity of consent provided by participants (Wendler and Grady, 2008). It is therefore important for researchers to ensure that participants understand the nature, goal and objectives of research studies for which they are being enrolled.
Understanding of study procedures
Our study showed that the recall of both the adolescents and caregivers on the study procedures including participant recruitment and randomization was considerably high. On average, the adolescents had a higher level of education than caregivers and despite their level of maturity, adolescents were capable of understanding research study procedures just like the caregivers. Understanding research procedures is a precursor to understanding potential risks resulting from these procedures (Burgess et al., 2019; Ndebele et al., 2012). This is likely to influence potential participants’ voluntariness, their understanding of rights to withdraw and options for available alternatives to research participation (Burke et al., 2005). Researchers should ensure that potential participants have at least understood the procedures of research studies they are participating in (Gupta, 2013).
Understanding of voluntarism and rights to withdraw consent
The caregivers in our study tended to be more average compared to the adolescents whose scores were higher on their understanding of voluntarism, and the right to withdraw or decline to answer questions they were not comfortable with. Studies have shown high level of comprehension about voluntarism and right of withdrawal of consent (Afolabi et al., 2014; Atal et al., 2018; Burgess et al., 2019). Similarly, others have also shown that adolescents, regardless of age variation, are capable of comprehending the voluntary nature of research participation (Crane and Broome, 2017) and can make voluntary choices for themselves (McGregor and Ott, 2019).
Understanding of risks, benefits and compensation for time
An unanticipated finding of our analysis was that caregivers were more consistently wrong on the main risk of participating in the study by two-fold compared to the adolescents while the recall of both cohorts of the benefits of participating in the study was at the same proportion, which was above average. Other studies have shown that the least well understood concept in ICD is potential risks of participating in a research study (Afolabi et al., 2014; Burgess et al., 2019; Dyer et al., 2022; Foe and Larson, 2016). A key concern about the quality of informed consent is the fear that potential research participants can be preoccupied by the prospect of financial gain such as compensation for time at the expense other ICD information. This may result in poor understanding of risks, non-monetary benefits and available alternatives (Stunkel et al., 2010), hence the need for the informed consent process to adequately inform prospective participants about the nature of a study and its potential risks and benefits.
Understanding of confidentiality and data protection
Our analysis also showed that there was no difference between the level of comprehension of the adolescents and caregivers on questions about confidentiality and protection of participants’ data. The proportion of adolescents and caregivers who were consistently wrong on the question on confidentiality and safety of collected data was the same. Again, these findings are not unique to our study. Other studies have also shown that both adult and adolescent participants have challenges in understanding the aspect of confidentiality and privacy in research studies they are enrolled in (Afolabi et al., 2014; Neelakantan et al., 2022). The risk of breach of confidentiality and uncertainty about data safety are some of the key aspects in the consenting process and potential participants should be clear about them before enrolling in research.
Recommendations
Adolescent comprehension of ICD is an essential goal that researchers purpose to achieve in their endeavour to improve their health and general well-being. While it is best practice for researchers to obtain caregiver consent in order to enrol adolescents in their research, they often experience a moral conflict over this issue, struggling to determine when to obtain caregiver consent, knowing this may lead to a systematic bias in the sample where the population under study is misrepresented (Liu et al., 2017). Even though our study shows that adolescents can understand the key elements of ICD to the level of caregivers or even better, this may not be enough to undermine the need to address caregivers’ concerns about the safety and well-being of their children when they participate in research studies. If caregivers’ permission is waived, adolescents/minors may be exposed to unanticipated research risks or harm (Bauman et al., 2020). Research ethics guidelines should therefore be framed in a manner that balances protection and inclusion and is considerate of adolescents’ developmental autonomy and decision-making capacity. This may instil confidence among researchers in designing research protocols and ICDs for adolescents. This confidence can be boosted if researchers are protected against possible lawsuits from caregivers when their adolescent children participate in research for which IRB waiver for caregivers’ consent are granted without their permission.
Limitations and strengths
This study was implemented in one county in western Kenya and the results may limit generalizability nationally or globally. The sample size that was assessed was also relatively small. The comprehension assessment questions were not pilot-tested with adolescents and may have been above their cognitive level of comprehension. For example, some of the questions were framed as double negatives, and participants may have found the logic difficult to follow. To confirm whether participants were confident about their responses and not guessing the multiple choice answers provided, each question was asked three times. This can both be a weakness and a strength. This approach may have created doubts and confusion in participants in their responses, and could partially explain the inconsistency in some of the responses.
On the other hand, asking the same questions three times helped to minimize potential guesswork that is often associated with multiple choice responses. The study also had other methodological strengths. We applied the same assessment tool to the adolescents and caregivers and were able to standardize assessment of comprehension. The assessment sessions were also audio-recorded and to ensure quality and integrity of data collected, all the audio-recordings were reviewed by one investigator to ensure that the guide was used appropriately and consistently and that the transcriptions were accurate. Finally, the questions covered all components of an ICD, which allowed us to assess comprehension of the entire document.
Conclusion
Overall, our results suggest that adolescents can understand the elements of an ICD at a level that is significantly higher than that of caregivers who are entrusted with giving them permission to participate in research studies. There was a significant difference between adolescents and caregivers in retention of information about the research study which indicated that adolescents performed better than the caregivers in comprehending the ICD. As such, our data give reassurance that for low-risk research studies with adolescents aged 15–17 years, waiving the requirements for caregivers’ permission may not compromise the ethical requirement of ensuring comprehension of ICD. This will allow adolescents greater access to research that benefits them. However, researchers should ensure that for adequate understanding, the consenting process is meaningful, structured and complete. Given the study limitations, we add to the clarion recommendation that further research is needed to develop a standardized tool which can objectively estimate a quantifiable threshold of ICD comprehension regardless of socio-demographic status of research volunteers. We also suggest that implementing such a study in different regions of the country with participants recruited from urban, peri-urban and rural areas may yield results that are generalizable in Kenya, specifically, and Africa in general. Finally, research is needed to explore ways of improving standards of research conduct and researcher confidence when waiving caregiver permissions.
Footnotes
Acknowledgements
The authors would like to thank the study participants, Ministry of Health leadership in Siaya County who allowed the study to be conducted, and IRDO staff working in the study.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by University of North Carolina at Chapel Hill/University of Pennsylvania [R01MH111602].
Ethical approval
Ethical approval for the study was obtained from the Maseno University Ethics Review Committee (MSU/DRPI/MUERC/00247/15).
