Abstract
Although well established in developed countries, Ethics review boards in the academia, and specifically for social and behavioral sciences (SBS) research, is a relatively new, and still a controversy inducing endeavor. This study explores the establishment and functioning of ERBs in Israeli academia, serving as a case study for the challenges and progress made in ensuring ethical research practices in non-medical related spheres. A purposeful sample of 46 participants was selected, comprising ERB current or past members and SBS researchers, who each interacted extensively with ERB’s evaluation processes. The participants came from all eight research universities of Israel, as well as seven large public and private academic colleges. Open, semi structured In-depth interviews were conducted in order to facilitate a critical reflective stance among the various participants, exploring their experiences, behaviors and the meanings they assign to ERB processes. Our research revealed two major themes. The first is the developmental trajectories in the construction processes of ERBs in Israel, comprised of the following stages: Initiation, expansion, opposition, and acceptance and assimilation. The second was a typology of participation throughout the ERBs’ construction process. Inspired by Merton’s strain theory, this typology includes conformists, ritualists, ideologists, control-freaks, and insurgents. The findings expose a unique perspective into the initiation, establishment, and development of ERBs, which can shed light on the role of ERBs in general, as well as point out how resistance to ERBs, as well as the different reasons for championing them, affected not only ERBs form and their legitimacy, but also their interaction with researchers.
Introduction
Ethics review boards in the social sciences are known and well established in various developed countries. However, and unlike ERBs in the medical and clinical fields, they are a much more recent development. Initially this development was conceived as simple or even natural, as part of strengthening the scientific rigor of the social sciences’ methodological approach. It was also part of the growing understanding that research in social sciences can potentially harm participants and therefore ethical regulation is needed. However, this new ethical regulatory change encountered resistance and criticism. Various studies and scholarly writings have started to both conceptually question the justification of this development, as well as empirically question its effectiveness.
In this article we attempt to add to the existing knowledge in this field by providing a qualitative-historical analysis of the Israeli experience. The case study is about the story of the creation of committees for the ethical review of research with human participants in Israel. As will be argued, through the specific case study and historical narrative, a better understanding can be gained, framing the existing debate regarding the need, effectiveness, and success of ERBs in the social sciences.
Literature review
A short history of ERBs in the social sciences
The regulation and administration of formal bodies that control and supervise the ethics of biomedical experiments and research are considered a relatively recent and modern development (Baker et al., 1999; Hedgecoe, 2009; Jonsen, 2000; Rubin, 2011; Rubin and Koren, 2003; Stark, 2007, 2010). Moreover, the extension of these institutional regulatory norms to research in the Social-Behavior Sciences (e.g. social work, sociology, law, economics, etc., referred to hereinafter as
ERBs were not mandated for reviewing SBS research until many years after they first appeared. This is not surprising, as in many cases, the potential risk inherent in SBS research is relatively low compared to its clinical counterpart: SBS studies do not commonly employ dangerous substances and any harm possibly resulting from studies—usually emotional distress—is usually minor or reversible. Moreover, the type of risks common to SBS research can be understood and assessed more easily by the research participants themselves (Dingwall, 2008), thus diminishing the problematic power relationship between researcher and participant and the need for external protection of participants’ wellbeing.
In effect, ERB regulation of SBS research dates to 1975 when the US Department of Health and Human Services (HHS) began requiring IRB 2 approval of research proposals as a precondition for the release of HHS funds. Later, other US federal agencies, institutions, and foundations followed suit (Ceci et al., 1985), and in 1991, the Code of Federal Regulations’ “Common Rule” (Title 45, Part 46, Subpart A) was legislated, which most federal agencies, including those that fund SBS research, now follow (Israel and Hay, 2006).
The need to perform collaborative research with US-based academics and to receive US federal funding gave rise to a growing demand for the facilitation of ERB procedures in academic SBS settings in other countries as well, such as Canada (Haggerty, 2004) and the UK (Hedgecoe, 2009; Shaw et al., 2005; Slowther et al., 2006), which also adopted systematic regulation of SBS research review. In other countries, academic institutions began to voluntarily establish their own ERBs, independent of a guiding central authority; in most cases, their review processes copied the US model or adapted it to specific scientific journal requirements or research proposals calls.
The regulation of research ethics review in Israel
Israel has developed its research ethics review process in a unique manner. The formal institutional supervision and regulation to ensure ethical medical human experimentation began only in 1980 through the Israeli Public Health Regulations (clinical trials on humans). The regulations were drown in response to the 1975 addition to the Helsinki Declaration, of the requirement that all experimental protocols “should be transmitted to a specially appointed independent committee for consideration, comment and guidance.” In accordance, the regulations stipulated that all human research performed in clinical settings in Israel must be approved by an institutional ERB, named “the Helsinki Committee” (Landau, 2008; Landau and Shefler, 2007; Rubin, 2010, 2011; Rubin and Koren, 2003). The regulations also detailed the committee’s structure and membership 3 and adopted the 1975 version of the Helsinki Declaration as a guideline for the committee’s review.
It is important to note, that the regulations were drown by the Minister of Health, based on his authority, set out in article 33 of the Public Health Act of 1940, “to establish regulations regarding the opening, registration, management and supervision of hospitals and clinics.” Thus, both the regulations and their pursuing Ministry of Health’s (here by: MoH) director’s guidelines were, and still are to this day, legally relevant only to hospital settings and required only hospitals to establish a local ERB. Israeli health funds (HMOs) have later voluntarily adopted this regulatory scheme into their settings, established their own ERBs and see themselves as bound by both its procedural and substantial requirements.
In 2006, the MoH’s director’s guidelines clearly stipulated that the approval of a Helsinki Committee is a pre-requisite for performing not only clinical research, “involving physiological, chemical, biological, pharmacological, or radiological intervention,” but also SBS research, performed in medical settings. This was incentivized by the expansion of SBS research in hospitals in the early 2000s, especially by social work based and nursing-based researchers. The guidelines also established a specific Helsinki sub-committee for SBS research proposals.
ERBs in the academia in Israel
Due to the lack of legal requirements for research ethics review outside of the health sphere, most Israeli universities and academic institutions originally did not establish any formal process for ethical review or approval for their studies (Rubin, 2011). Consequently, most academic research has not been ethically reviewed well into the 2000s. The exception to this were classic clinical research projects, performed in academic settings, that in the absence of academic-set ERBs were submitted for review, under different pretenses, 4 to hospital Helsinki committees, and in some cases kept on being submitted there even after an ERB was established in the relevant academic institution.
The grass root need for ethical approval, regardless of a lack of regulatory requirement, rose out of the unique context of the Israeli academia. Israel is a small country, with few researchers and few academic journals. In order to flourish, Israeli science cannot exist in a bubble, and is very much based on international collaborations. The personal promotion of academic researchers in Israel encourages, and sometimes requires, the performance of a post-doctorate abroad and the publication of articles in journals with a high impact factor—most of them foreign, usually in English. Similarly, many of the evaluators for academic promotion purposes come from abroad. For example, in the social sciences—the common expectation at Israeli universities is that two-thirds of the evaluators (a total of six) will be from abroad. Many of the journals as well as research and evaluation partners are American, due to the Israeli preference for the English language.
The incentive and institutional reward model of the Council for Higher Education in Israel is also based on the same indicators, and therefore Israeli researchers largely adapt themselves to international and especially American expectations and standards—ethical review standards included.
As will be depicted later in this paper, academia set ERBs were only established in recent years, and the review of SBS research and experiments, performed in and by institutions outside clinical settings, has slowly set root and expended with it. For example, the University of Haifa’s Faculty of Social Welfare and Health Sciences voluntarily established its own ERB, and faculty members were encouraged—but not mandated—to submit their as well as their students’ research proposals for approval. This initiative was formally adopted in 2010 by the university’s senate (Haifa, 2011).
Most other Israeli universities have also formed their own ERBs since then, resulting in a “cultural” shift. Yet, both the establishment of ERBs and the review of SBS research in the academia are still not required by any formal or statutory code, and are performed on a voluntarily, non-regulated basis, with their structure, procedures, and requirements varying between institutions.
It is important to note that the Israeli Ministry of Health made several attempts over the years to coherently regulate research ethics, in a manner that disregarded the research’s setting—whether medical, academic, or other. This started with a 1997 first draft that after 8 years evolved into the Medical Experiments on Humans’ Draft Law (2005) (State Comptroller's Report 55B, 2004). It did not mature into law, yet was re-animated once every few years (either as a ministerial or a private bill). None of these attempts ever came to fruition. This was mainly due to severe criticism—amongst other things, over its non-disciplinary-adaptable and medically centered perspective. This type of criticism intensified over the years, correlating with the increasingly central role of empirical research in the SBS.
Our research was initiated at the end of 2012, on the backdrop of one of the more assertive of those regulatory attempts and its correlative spike in academic backlash. With its empirical components eventually executed in 2015–2016, our research both documented a turning point in the development of research ethics review in the Israeli academia and, as will be described at the end of our paper, served as an engine for change and progress into the current last stage in its development.
As academia-set ERBs were constructed with no central guiding hand, little is known of their reception, performance, and effectiveness (Koren, 2003; Rubin, 2011). Moreover, unlike ERBs in the USA or the UK, there has been almost no empirical research of their acceptance and institutional role.
The debate concerning ERBs in the social sciences
Over the years, the scientific literature has seen various critiques of ERBs in the SBS (Bosk and De Vries, 2004; DuBois and James, 2004; Ferraro et al., 1999; Hayes et al., 1995; Kancelbaum, 2002; Lincoln and Tierney, 2004; Malouff and Schutte, 2005; Pritchard, 2002; Silberner, 1998), including accusations that ERBs may be guilty of any one or more of the following: (a) favoring the protocols of selected colleagues; (b) concealing conflicts of interest; (c) making unreasonable requests to changing protocols; (d) imposing excessive bureaucratic requirements; (e) staffing reviewers who lack training in qualitative methods; or (f) being incompetent.
Cohen (1998), at a summit meeting titled “ERBs: All Checks and No Balances,” cited specific concerns: censorship of research topics, inconsistent decision-making, harassment, lack of accountability, biases, ineffective communication with investigators, lack of knowledge about research, unrealistic assessment of risks, lack of appeal mechanisms, wide variations in the interpretation of the federal regulations, and a focus on participants’ rights—leading to neglect of scientific merit. Adding to this list, other scholars noted excessive time to assess protocols, rudeness, justifying idiosyncratic rulings with the pretext that “local standards” overrode standard evaluation criteria, and being overly conservative and protective of the institution and the research participants (Bell and Wynn, 2023; Edwards et al., 2004; Keith-Spiegel and Koocher, 2005; Wainwright and Saunders, 2004).
The critics of ERBs also point out the inappropriateness of importing biomedical ethics into SBS, which, it is claimed, has resulted in perverse consequences (American Association of University Professors [AAUP], 2006; Caeymaex et al., 2023; Dingwall, 2008). In medical research ethics, continue the critics, a knowledge gap favoring researchers is assumed, and with it the need to predetermine hypotheses and procedures prior to the research, an interventional purpose for every study, and a weighty ethical duty to do no harm. SBS research, on the other hand, shares determinately different characteristics: Much of SBS research starts with no particular hypotheses or procedures in mind; many participants, rather than the researchers, actually enjoy the upper hand in the information imbalance; much of SBS research is observational and not interventional; and last, but not least, not all SBS codes of conduct place the same weight on the duty to do no harm. Much more weight is placed instead on the duty to seek and communicate the truth (Lincoln and Tierney, 2004; Peled-Raz et al., 2021; Schrag, 2010).
Finally, critics of the ERBs in SBS argue that falsely attributing biomedical research characteristics is a type of censorship, which infringes on the basic human right of free speech (Hamburger, 2004). From this perspective, ERBs practice a form of speech licensing, which in effect, is the censorship of ideas. Moreover, the critics further argue, the social costs of such scientific censorship are far greater and more harmful than the benefits; it interferes with legitimate scientific research, which can result in the unnecessary loss of lives (see, e.g. Dingwall, 2008).
Despite the wealth of criticism, most has not been based on empirical evidence, but rather on anecdotal or personal experience (De Vries et al., 2004). It is also interesting to note that the qualitative studies have shown that there is a dynamic and historical context to the ERBs way of work. As pointed out by Hedgecoe (2008, 2012) and Stark (2013), ERBs attitudes and perceptions are not static but dynamic. For example, their approach toward qualitative research (as opposed to quantitative ones) has changed throughout the years as both the membership and the expertise changed. Their work has also been marked by local and individual experiences of personal trust and first-hand/face-to-face knowledge of the researchers they regulate and assess. These findings emphasize the significance of the specific historic, contextual, and local framework of the ERBs operations for understanding their outcomes and actual impact.
Since Israeli ERBs in SBS are still evolving and being constructed, they are uniquely suitable for studying the social and interactional processes by which ethics regulations are defined, implemented, negotiated, and established; in this sense, ERB members and researchers serve as primary stakeholders in the ERB process.
Methodology
The methodology presented hereby is part of a much broader, mixed-methods study on ERBs in Israel, funded by the Israel Science Foundation. This paper reflects part of the qualitative stage of the study which was aimed at studying the participants’ experiences and behaviors and the meanings that the participants assign to the ERB process without superimposing predetermined categories (Kvale and Brinkmann, 2009; Rubin and Rubin, 2011). It’s a processual and relational examination that, although referencing it, does not explore the ethical basis, nature, and development of ERBs’ actual decision making.
This portion was based on in-depth interviews in order to facilitate a critical reflective stance among the various participants, which will allow them to relate to their experiences from both an experiential position and a reflective one (Ben-Ari and Enosh, 2011).
Sampling and participants
The study focused on ERB members, researchers, and Deans of Research at Israeli research universities and academic colleges. 5 A purposeful sample of 46 participants was selected, comprising ERB current or past members and SBS researchers, who each interacted extensively with ERB’s evaluation processes. The participants came from all eight research universities of Israel, as well as seven large public and private academic colleges.
Procedure and Instrument
All participants were directly approached (via phone or email) by the research team. After receiving full information regarding the study, and once an informed consent was given, a meeting was set up for the interview, according to the time and place convenient to the participant. The interviews were conducted, during the period of 2015–2016, in various locations (e.g. university offices, libraries, cafes, private homes), and commonly took between 60 and 120 minutes. All interviews were audio recorded and transcribed into text.
All interviews were conducted based on an open, semi-structured interview guide. The interview guide focused on the participants’ perceptions of ethical conduct of research, the need (or lack there-of) for ethical regulation of research, the ERB procedures in their institution, the guidelines they use to evaluate proposals, and their perceptions of the interaction between ERB and researchers.
Qualitative analysis
The analysis of interviews was based on grounded theory principles (Charmaz, 2006; Strauss and Corbin, 1998), using Charmaz’s (2006) clear guidelines for separating, sorting, and synthesizing data with qualitative coding. The process may be described as consisting of three stages: open coding, axial coding, and integration (Strauss and Corbin, 1998). Open coding refers to the initial reading of raw data, during which the researcher marks comments and questions in the text. In the axial coding stage, the researcher connects emergent categories, while trying to find relationships between these and the points expressed by the participants and to identify subcategories. In the final stage, integration, connections are made between various themes across the different data sources and general categorizations are formulated, leading to the findings that will be presented below.
Limitations
Our dataset was collected between the end of 2015 to mid-2016. As such, it did not document the last of the phases in the development of ERBs in the Israeli academia.
Nevertheless, given that this paper analyzes historical processes and typologies of institutional interaction, the data remains valuable for illustrating core stages in the evolution of academic ERBs, offering insights worthy of exploration and learning.
Ethics, trustworthiness, and credibility
The research team consisted of five members; three males and two females; three were themselves ERB members (one of them—Chair of an ERB); all had extensive previous personal experience with submitting research proposals to ERBs; and three had ethical-legal background. The study received the ethical approval of the ERB at the University of Haifa. From a risk perspective, this study did not include any specific risk. However, looking at potential conflict of interests and biases, this research was more demanding, as three out of the five investigators, are members of ERBs themselves. Self-reflection was therefore crucial for the research team. The team conducted regular reflection meetings discussing both administrative and personal aspects of the research.
Qualitative research is focused on achieving trustworthiness and credibility about the study subject, without making claims to finding an absolute truth (Hammersley, 1995), as opposed to quantitative research, in which validity means truth (Angen, 2000). In this study, the use of extensive quoted interview excerpts in the analysis aimed to enable the readers to evaluate whether different aspects of the data that were collected create consistently rich and thick descriptions (Lieblich et al., 1998; Lincoln and Guba, 1985; Padgett, 1998). In addition, our qualitative research did not attempt to generalize; since the variables of each study are uniquely derived from the context and the data collected from the interviewees, there is no expectation to replicate identical findings (Schofield, 1993).
Findings
Analyzing participants’ experiences revealed two major themes. The first is the developmental trajectories in the construction processes of ERBs in Israel. The second was a typology of participation throughout the ERBs’ construction processes. We will present the key sub-themes of the developmental process and highlight the “typical” ways of participating and reacting to it.
Institutional development of ERBs
Stage 1: The initiation stage
The earliest stage of the establishment of ERBs in Israel was characterized by “individual enthusiasm.” It reflected the notion that there was a need for ethical overseeing of research behavior—something that was missing and was not much discussed or debated by researchers in Israel at the time. As described by one of the participants: [The person who initiated the committee] was an American guy. He was the first chairman. . . he took it seriously, that is, he established the committee, the guidelines, the requirement that there be a representative of every faculty on the committee, the administrative array, as well as the principled, conceptual structure of the committee, he is the one who initiated it. [Interview 24—ERB member]
Another participant, shared his experience as an ERB initiator: When I came back [after studying for PhD abroad], I wasted long years trying to “break out of the ghetto” [of being a westernized country without established ERBs]. I talked with the dean of social sciences, talked with the heads of the sociology department, and of anthropology, talked with the heads of some other programs, convened meetings [about establishing ERBs] . . . and yet for many years nothing happened. [Interview 3—ERB chair]
As implied in this quote, it took “the enthusiasts” quite a while to get the official recognition and cooperation in establishing ERBs in their universities.
Internationally imported enthusiasm later grew local branches, as exemplified by this quote: In the beginning we brought the tradition of ethical examination, as graduates of X University where there was a requirement for an ethics committee. And then we started in this kind of rather partisan way of doing it, we took the X University’s model, its forms and basically faculty members in the department judged other faculty members’ research. It was at the beginning. There was no defined committee. [interview 22—ERB chair]
Stage 2: The expansion—Enthusiasm needs perseverance
Once the enthusiasts were successful in their initial introduction of ERBs to the social sciences, their next step was to expand the scope of their activity, both in terms of authority and subject matter.
Then we started to settle a bit officially but only in the psychology department. In other circles it remained in the old, partisan model. And at the end the committee at the psychology department became fully institutionalized, parallel to the processes of ERBs institutionalization throughout the university. [interview 22—ERB chair]
Since the establishment of ERBs did not include an official mandate of submission of research proposals to ERBs—but rather left it optional, the next step was to try to make it mandatory. The idea was that it was not enough only to introduce the concept of ERB, but to mainstream it, mandate it, and make it an integral part of the research process within the universities. At the beginning, these attempts to broaden the scope were rejected, as described by one participant: We tried in our department to require researchers and students to present their studies for [ethical] approval, and it was rejected in the departmental meeting. . . .. A year later I was smarter, so I asked to oversee only the students’ proposals. This got almost unanimously approved. . . This experience kept repeating itself. When I asked to have everybody go through an ethical expertise exam, it didn’t go through. When I suggested that only new faculty members must go through this exam, it was unanimously approved. [Interview 3—ERB chair]
As this example shows, the process of expanding the scope and scale of ERBs’ operation, had to be done slowly, and incrementally. The actual expansion was diverse and differed from one institution to another. For example, at one of the universities, it started with an ERB only at the Department of Psychology, which later led to the establishment of a university-wide ERB. For several years, those ERBs had hardly any proposals submitted to them and served only those researchers who had cooperative projects with US or European based universities that asked for local ERB approval.
As described by interviewer 1, several years later, the chair of the University-wide ERB, started advocating for a formal University Senate decision compelling all faculties and schools to establish their own ERBs. Despite this formal decision, only two faculties followed through immediately (the Faculty of Social Sciences and the Faculty of Health Sciences). Several years later, the chair of the University-wide ERB led a second Senate decision that mandated every research project to be submitted to an ERB approval, and that every researcher of the university should undergo some formal training in research ethics. Yet it took several more years for most of the faculties to establish their own ERBs.
Similar processes were described in other institutions, with some variations. Some institutions started with a psychology-based committee, which was later encompassed into a university wide committee [Interview 15—ERB member; 34—ERB member]. Others only established an institutional-wide ERB; some tend to delegate the ethics review process in relevant cases to external medical ERBs in hospitals, and otherwise ignore the issue, while others did not undergo any formal establishment of ERBs till only after the commencement of our research project.
In some cases, an institutional decision was made to require a form of ERB mechanism, yet several years had passed before researchers were all aware of it, and it was widely enforced: According to the [University’s] Senate decision, we were required to have ethical approvals, but I did not know about this decision, as many did not. This decision had no teeth. They simply decided, put it in a drawer and continued working as before. So, this decision was binding, but it was simply not enforced, and now it is enforced. [interview 2—ERB chair]
It was gradual. I remember a period when there was no committee in the faculty. I wanted an ethics approval. The university had one institutional committee. And they didn’t understand what I was talking about. I don’t know who the members of the committee were - historians or something like that. . . but then little by little there was a change and expansion. What drove the change, in my opinion, was that the journals started to require an ethics committee review confirmation. [interview 41—researcher]
In some institutions the process of ERBs’ assimilation is still ongoing: We are heading in the right direction, but it is not yet fully integrated. Those who use the ERBs are those who already know [that its mandatory] and know us and are constantly submitting. Yet, I am sure that we have a few individuals who are not yet aware, no, do not want to know that there is an ethics committee. [interview 21—ERB chair]
In most institutions the lack of coherent integration was discipline based: It didn’t occur to them at all, it’s not part of their professional culture. It’s a scandal, right? I don’t know what happens in other sociology departments in other places, it would be interesting to check it, in contrast to anthropology where research ethics is an extremely hot topic. . . [interview 25—ERB member]
Stage 3: Opposition
The last two quotes indicate, among other things, the existence of some passive internal opposition to the conduct of ethical review. Opposition existed throughout the previous two stages and was voiced either explicitly or, most often, implicitly. One of the participants who was “on board” the process of establishing ERB, described how at an early stage of the ERB development, he was told that: “There are many good things in the USA – I really like their hamburgers; but their attitude regarding ethics is not something we would like to import. . .” [Interview 3—ERB chair]
As reflected in this experience, for some, the “importation” of ERBs to Israel was viewed, yet again, as simply “copying” of either the “American way” of doing things, or the “medical way” of doing things, which do not necessarily reflect the Israeli culture nor the appropriate way of conducting research in non-medical academic disciplines. The opposition became much more significant and visible once ERBs were formally in place and on their way to becoming mainstream.
There was an over-medicalization, attempting to solve issues that simply do not exist in the social sciences. . .but I think this is a built-in tension, because as I said, doctors look at things differently from people in the behavioral sciences and I think this is a training-based perspective. There is nothing to be done, a doctor has a very clear prism regarding human beings while researchers in the behavioral sciences have a different prism, and this creates tension that is always present. [Interview 36—ERB member]
Some interviewees have described other reasons for researchers’ opposing stance toward ERBs in Israel—either their own, or through observing others: They come in advance with some anti-committee stance. . .I’ll tell you why, because the Ethics Committee is criticizing them. This is its prerogative and people do not want to hear criticism and they think they are the experts and “who are you to tell them that their research is not done or designed well” [interview 22—ERB chair] They view it as legalization, formalization, “Nonsense-zation” et cetera, it is only perceived in the negative, focusing on the disturbing and bothersome aspect of it, which of course is there. [Interview 25—ERB member] I think these committees are completely unnecessary, so we need to look at it from a slightly broader perspective. I think these committees are without goals. They have negative effects on research. First, they delay, at best only in time, the conduct of research. . . .I don’t think that the promotion of science is foremost in the minds of the ethics committee members. Part of the ethics of research should be the advancement of science. I think this is a negligible item on the agenda of ethics committees. [Interview 37—researcher]
Stage 4: Acceptance and assimilation
It took several years for ERBs to be naturally assimilated in their institutions’ research protocols and accepted by the researchers. Even though not all institutions have incorporated ERBs in the same manner and with the same level of investigators’ support, it has become an integral part of conducting research in all of the sampled institutions, As described by the participants: The assimilation (of ethical review in SB research research) was achieved. . ., achieved with difficulties but in the end successfully. [Interview 22—ERB chair] And I already feel it, because this process is happening. It’s quite new, it took a long time for people to understand and agree, it’s just a little, a little work, even mentally, for some, but the applications I receive now are different from those I received even a year ago, certainly before two years – “Cleaner.” I see that there is a growing ethical awareness. The applications require fewer comments from the reviewers. So here we are. [Interview 2—ERB chair] At first, they thought it was nonsense, as well as a nuisance. Like – “this is only a small thing I’m doing. . . why do I need someone to approve it?,” But I think that today it has changed, that people understand the potential harm, understand the importance of protecting participants. . . [Interview 15—ERB member]
Typology of participation in ERB Processes
So far, we have described the stages of establishing ERBs in Israeli Academia. The second theme that emerged from our study was the versatile typology of participation and reactions to the processes of constructing and implementing ethical reviews. It should be noted that those types of reactions are not separate from the processes, but rather were inherent to them and one could not be understood without the other as contextual background.
The following behavioral patterns characteristic to both researchers and committee members, were classified into a typology that was inspired by Merton's (1957) strain theory, although not identical. Whereas strain theory was developed within criminological context and may raise questions of relevance when applied to “normative” researchers and members of ERBs, the analogy is easily followed. In developing strain theory, Merton posited that society presents its members with goals that are considered to be normative, and with normative means to achieve those goals. By relating to the demands thus set up by society, its members are put under regulatory strain. Thus, they must either accept the goals set up by society, work to achieve them, and do it by the “approved” means; or they may reject the goals, or the means, or both, in order to relieve the strain. Accordingly, the resulting typology Merton presented related to the various ways by which ordinary persons either accept or reject such goals and means. Similarly, from our perspective, the construction of ERBs and the regulation of research behavior, has put new regulatory strains on researchers and ERB members, defining the normative ways by which researchers should conduct their research behavior. The choices reported by our interviewees regarding their acceptance or rejection of the new definitions of either the goals of ERBs, or the means by which those ERBs operate, gave rise to the following typology. Our final typology included conformists, ritualists, believers, centralists, and insurgents.
Type 1: The Conformist
Conformists are defined by Merton as those who accept both the goal and the means of the social order, in this case, the ERBs as an institution. Conformist researchers and ERB members are “good organizational citizens.” As such, they either volunteer, are being pushed or agree to cooperate with the new ERB regulations. They accept its necessity, submit the proper forms and yield to the committee instructions. An example of this can be found in the following quote: Some researchers understand that it is more than just a nuisance. . . That there is a need for another set of eyes reviewing what I intend to do. That it is worth looking at to see whether I unintentionally do some harm. (Interview 11—ERB chair)
Type 2: The Ritualist
According to Merton, ritualists reject the goal but accept the means. In our context, we define ritualists as those who lack the deep understanding of the essence of ethical considerations and instead either conform to the procedure of ethical approval or even sanctify it. An example of such an approach can be found in the following quotes: I believe that for some of the researchers, it is only a formal procedure: “There is no choice, they force me.” (Interview 11—ERB chair) I think it’s already passed a stage, we’re already on the automaton part. You know? It’s like when you are being careful driving and stop at red light: Is it because of fear of law enforcement? Or are you already used to stopping at a red light. . . . You just know that this is the norm. Researchers have been educated to submit to the committee. [interview 12—ERB member] I don’t know if the committee is doing its job “successfully.” We are doing something bureaucratic, and we manage to do this bureaucracy well. . . .I guess it improves the research a bit. But not hugely. [interview 21—ERB chair] . . .I have to, because they told me it was necessary. Not that I understand what it is for. It’s not fruitful no. they deal with such small nuances. Fix such a word or another word. OK, you want us to do it? We will do what you want. Just give us permission. [interview 41—researcher]
Type 3: The Believer
Believers approach the ethical review process as a means of serving their ideological ends and find their way into ERB committees in order to further those. They practice ideological political correctness, through reviewing not only the ethics of the research protocol but also the legitimacy of asking the researched question. They attempt to extend the accepted ethical principles beyond the common scope of respecting participants’ rights and researchers’ safety and apply the process to goals that serve their own wider sense of justice. One example for this approach can be found in the following quote: We had a committee member who claimed that it would be unethical to review a proposal dealing with possibility of ADHD among male batterers, because, as she claimed, such findings may later serve as a justifying factor in domestic abuse trials. [Interview 14—ERB chair]
Misuse of research outcomes may of course be of ethical concern, yet the ideological goal of protecting victims of domestic violence served in this case as the justification for a desk-rejection of a research proposal, muting a legitimate research question, instead of for a demand for the introduction of precautionary measures to minimize misinterpretation of result.
Type 4: The “Centralist.”
The opportunity to regulate and control others seems to attract some persons with a need to control and use a centralist approach to ERB-leadership. Those appear to either enjoy the position or feel that no other can perform better than them in the role. They conduct a very centralized ERB process. In some cases, centralist control has become a hallmark of the committee-chair, to the point of distrust and resistance of researchers, and even of committee members.
There was a committee member who felt that the committee had a lot of power, and there was a feeling that he took full advantage of it. . . . Being a member of an ethics committee is a position of power, and someone can sometimes take it a little too far. . . . I think this is a place where you have to be very careful. [Interview 9—ERB member]
One institutional ERB’s chairperson, described a most centralized format of review, insisting that nothing would be authorized without his personal examination, while ignoring all other members of his so-called committee: There were departmental committees [before I started my role as chair of the institutional ERB], and they would have authorized research without any further review. They thought they can
Type 5: The “Insurgent” (innovator)
Merton identifies “innovators” as those who accept societal goals but have few legitimate means to achieve those goals, thus they innovate (design) their own means to get ahead. Although Merton mentions mainly criminal acts as such means, we identified innovative means as attempts to bypass procedures or certain committee demands.
Some resisting researchers look for ways to avoid submission of proposals to the ERB: The committee is very strict, and this makes things much harder, and people get upset. They tell me – “in another university I would not have had such problems.” [interview 22—ERB chair]
In some cases, this is followed by a “committee shopping” technique: I know of a situation where a faculty member was angry that he had to submit for review of a specific committee. He said, “I don’t know if they will approve it here, I have a partner in sociology, I can pass it through them.” [Interview 34—ERB member]
There are also preemptive innovators, who offer to construct their own committees with their own “ethical rules,” to avoid being subordinated by what they perceive as external or non-relevant ethical requirements. This is well exemplified by two of our interviewees, both referring to the occasional regulatory attempts by the Ministry of Health: There was a departmental discussion where I suggested that we should be prepared. It’s breathing down our necks (. I wanted to say something like – “let’s create our own criteria, otherwise they will slap us with something worse.” [interview 32—ERB chair] I understood that the committee was established in response to some bill that proposed to transfer all research to the approval of ethics committees in hospitals. . . and the fear of such an unreasonable requirement. This resulted in the establishment of a well-organized internal institutional committee. [interview 29—ERB chair]
Discussion
What can one learn from the Israeli experience? The findings expose a unique perspective into the initiation, establishment, and development of ERBs, which can shed light on the role of ERBs in general. We found great value in the accounts of the founding fathers and mothers of ERBs in Israel, who shaped not only their modus operandi, but also their overall acceptance in Israeli academy. Furthermore, we can point out how resistance to ERBs, as well as the different reasons for championing them, affected not only ERBs form and their legitimacy, but also the relationships between researchers and ERB members.
Understanding the emerging typology of process
Figure 1 presents the types of behaviors and actions enfolding in regard to the development of ethical review in Israel. We can discern between those driven by an ideology, and those who are driven by other concerns. The ideologists include both the original entrepreneurs who initiated the process of constructing ethical review systems, who believe and accept the need for ethical review and serve out of acceptance of the need for balance; the other ideological group, the prompters, is composed of those who see research ethics as part of wider ideological or moral system and use the ethical review process to advance or dictate their ideological belief system.
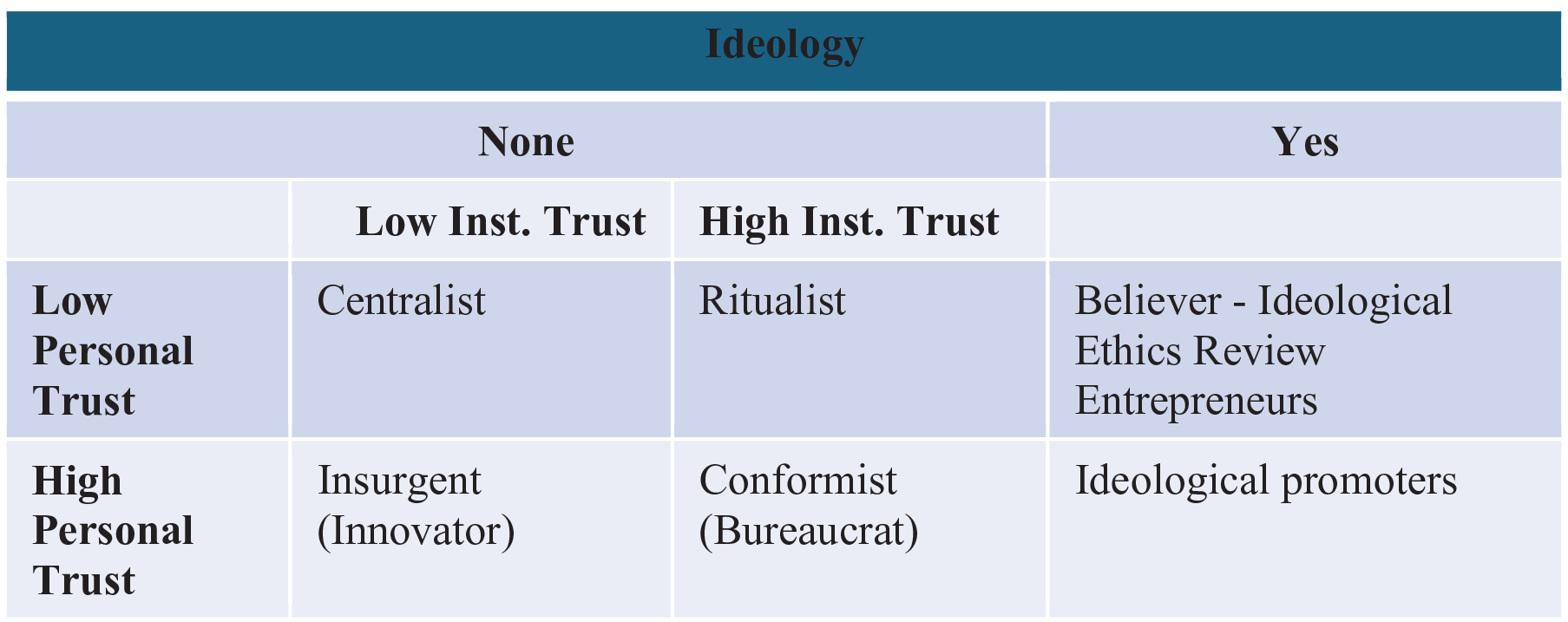
Typology mapping.
Other processes relating to the ethical review, as emerging from our findings, were driven by non-ideological concerns. As presented in Figure 1, Those may be sub-divided according to their trust in the system and in others within the specific context. Thus, the “centralists” may be described as ones who have little trust in the system or in other persons, therefore insisting on tightly controlling the review process and micro-managing it. “Let not the cat guard the cream.” The ritualists, like good bureaucrats, on the other hand, have high trust in the system, and low trust in other researchers, thus insisting on keeping every nuance of the process as tightly according to the existing dictates of the system, without allowing for any degrees of freedom nor the fitting of process to specific contexts. The conformists trust both system and others. They follow the system, and perceive others as reliable, thus act as good and functional community members. Finally, the insurgents tend to trust themselves and other researchers, while less trusting the system.
The paradigmatic shift of ethical review of research in social sciences
The developmental process described above can be seen as a very typical or “normal” process of incremental organizational change. Lewin (1951) described three stages of change in organizations or social systems in general. First, a “defrosting” of old patterns of behavior, attitudes, and values. Second, a transformation stage, at which new patterns of behavior values and attitudes as well as institutional arrangements emerge and are being reorganized and modified. Third, the Frosting stage, of establishing the new order, with changes then being aligned and settled.
Yet, the typology of reactions to the process, indicates that the type of change researchers had to live with, was of a different order. An experience of misalignment emerged between the process and the magnitude of change demanded of individual researchers. While the process was incremental, the level of change was radical, of second order, affecting the very nature of researchers’ work. Although the structural change was of minor magnitude, just another committee of many already existing in any academic institute; still, from the perspective of the “workers” (researchers), the nature of the change was dramatic, and called for redefinition of their work habitus.
ERBs in Israel evolved from an initiation stage (that started in the 1990s) through a stage of persuasion and search for legitimacy, to an institutionally regulated and mandated stage.
In most of the institutions we examined, the initiation stage involved a firm push by an “ethics entrepreneur.” The entrepreneurship of ERB formation in Israel could be a result of two trends that characterize higher education in Israel. The first is the “Americanization” of Israeli Academy. The constant gap between the number of PhD graduates and academic positions in Israel created a very competitive academic job market. To prevail, many researchers use a postdoctoral period of research abroad, mostly in the United States, to gain an advantage amongst their peers. This common stage in the academic development of researchers in Israel brought with it new research practices, such as the need for ethical review for non-medical studies. Upon their return and assimilation in the Israeli academy, these faculty members initiated similar practices in their institutions.
The second trend evolved from the first one. The lack of protocols for ethical review of non-medical research led these entrepreneurs to “import” practices from the one that already existed—the medical research ethics framework, and the medical ERBs (“Helsinki committees” in Israel) protocol. Despite the incompatibility of this Biomedical-ethical framework, it was the only one that was known and perceived as well grounded at the time. The entrepreneurs used the Medical ERB protocol as basis for the newly developed non-medical ERBs, causing much resistance. Many of our study’s participants referred to this trend as the “medicalization” of their study and expressed disapproval and resentment to it.
The persuasion stage often involved working through much resistance, due to the need to break away from traditional norms, that did not recognize the need for ethical review of social science research. It also required the formation of a new disciplinary-specific SBS research ethics framework. This allowed for the transition from the recognition of the general goal behind the ERB requirement, and the identification with its means of achieving better fitted SBS goals.
It wasn’t until the final consolidation and acceptance stage that ERBs became an integral part of the new norm of research, a stage that in many Israeli institutions is still in process.
This process is akin to the paradigmatic shifts described by Kuhn (1962), in which an established thinking paradigm, including basic assumptions, practices, and scientific language, is encountering difficulties and contradictions, yet is not discarded until a new paradigm, with different assumptions, practices and scientific language arises to replace it. Such replacement is never smooth nor taken for granted, rather it always encounters resistance by the “old guard” who find it hard to adapt to those new ways of approaching their substance and discipline. As attributed to Kuhn, an old paradigm is not discarded; rather it dies away with the last of its believers. This has been true for both the general integration of ERBs, as well as for the move from a biomedical ethics governing framework, to a SBS uniquely fitted one.
Epilog—In search of foundational agreement
Having no national binding regulatory framework for the operation of ERBs in the academia in general, and in SBS in particular, each of Israel’s academic institutions have established their own unique ERB framework, resulting in an incoherent and un-unified review “system”.
Moreover, the diverse voices and attitudes toward the existing frameworks, as evidenced in the typology presented in the findings, also added to the theme of incoherence and to the lack of a broad consensus regarding various aspects of the SBS’s ERB.
While still in the process of finding the appropriate balance within its regulation of the SBS’s ERB, a solid foundational agreement seems to have emerged in regard to some aspects of ERBs in the academia in general. This has been aided by our own research project, conducted in the years 2015–2016, which sparked a cross-institutional interest in ERBs’ structure and conduct. While our project included the interviewing of research Deans, ERB members and relevant researchers, our questions initiated farther discussions and many of the interviews were followed by impromptu debriefings, about the background to our questions, and the variety of ERB constructs we’ve encountered.
This culminated first in unofficial inter-institutional discussions, and then, in 2017 an official inter-university committee has been established by all the eight then member universities of the Israeli university presidents’ committee (VERA). 6 The committee was charged with proposing a unified yet institutionally flexible scheme for ethical review of research conducted in the Israeli academia. The resulting committee’s position paper was issued in 2021, and later individually signed by the universities’ research Deans.
As of 2023, this position paper is still in the process of intra-institutional adoption, by the original members of the VERA venture as well as other academic institutions, who recognized it as the guiding document for the ethical review of research in the academia.
This has moved the development of ERBs in the academia in Israel to a new, supra-institutional phase. This phase is mainly concerned with strengthening the coherence of ERBs’ mandates and operation, while maintaining a system nuanced enough so to fit the different disciplines and academical-structures it must serve. Once fully in place, this new phase will necessitate farther research.
Footnotes
Acknowledgements
We would like to thank the Israel Science Foundation for their financial support of the research project at the center of this paper, as well as the interviewees willing to share their experiences and thoughts with us.
Funding
All articles in Research Ethics are published as open access. There are no submission charges and no Article Processing Charges as these are fully funded by institutions through Knowledge Unlatched, resulting in no direct charge to authors. For more information about Knowledge Unlatched please see here: ![]() The research this paper is based on was funded by the Israel Science Foundation (ISF), Research Grant Application no. 1474/15.
The research this paper is based on was funded by the Israel Science Foundation (ISF), Research Grant Application no. 1474/15.
Ethics approval
The research this paper is based on has been approved by the ERB at the University of Haifa, approval No. 187/15.
