Abstract
Individual research results from population-based genetic and genomic research are traditionally not disclosed to research participants. Current practices of non-disclosure are, however, being challenged by an increasing number of scientists, ethicists and policy-makers who make arguments in favour of disclosing at least individual results of potential health or lifestyle significance to research participants. Simultaneously, research participants are expressing greater interest in accessing their results. This article first provides an overview of main arguments for and against the disclosure of individual research results to research participants. Next, it discusses the need for a move from the current ethical framework under which researchers operate, which is rather protective of research participants and does not encourage disclosure, to a new ethical framework that would better cope with current developments within the field. Comparative analysis of existing and potential ethical frameworks favours implementation of an ethical framework aimed at enhancing the autonomy of research participants through the disclosure of individual research results. Finally, this article explores practical models of disclosure that could be used under such a new framework. A model of disclosure that takes into account the two key criteria of communicability of research results and the research participant’s preferences to determine which results to disclose is found to comply best with the selected ethical framework.
Introduction
Participants enrolling in population-based genetic and genomic research projects are usually informed that no individual benefits can be expected from their participation in the research, which means in practice that neither individual research results nor clinical care are provided. Policies of non-disclosure of individual research results are widespread and have for many years been accepted by research participants, Institutional Review Boards and the scientific community (Beskow et al., 2001). These policies are now being questioned by a number of scientists, ethicists and policy-makers, who make strong arguments in favour of disclosing at least individual results of potential health or life-style significance to research participants (Fernandez et al., 2003; Fernandez and Weijer, 2006; Miller et al., 2008; Ravistsky and Wilfond, 2006; Shalowitz and Miller, 2005; 2008; Sharp and Foster, 2006). The aim of this article is three-fold: first, it provides an overview of main arguments for and against the disclosure of individual research results to research participants. Second, it discusses the need to move from the current ethical framework under which researchers operate, which does not encourage disclosure, to a new ethical framework that would better cope with current developments within the field. Two alternative ethical frameworks are explored: 1) an ancillary care framework based on a leading principle of beneficence, which provides for the disclosure of individual research results when these results can be actively used to rescue individuals from harm; and 2) a promotive ethical framework based on a leading principle of respect for persons, in which individual research results are provided as an action of enhancing the participants’ autonomy. Comparative analysis of existing and potential ethical frameworks is conducted to determine which ethical framework may be best adapted for population-based genetic and genomic research. Finally, the article investigates two potential models of disclosure that could be used under the selected ethical framework: 1) a result-evaluation model which emphasizes scientific and contextual criteria of disclosure; and 2) a communicability model which emphasizes criteria of communicability of research results and the research participant’s preferences to determine which results to disclose.
Main arguments for and against disclosure
Many arguments exist against the disclosure of individual research results to research participants in population-based genetic and genomic research:
First, it is argued that the role of research, in contrast to clinical care, is to produce ‘generalizable knowledge’ consisting of, for example, theories, principles, methods and research results that may benefit society as a whole, rather than to directly benefit individuals or provide care (Bredenoord et al., 2011). Any attempt to blur the line between research and clinical care by disclosing individual research results to research participants is considered inappropriate and even misleading to research participants, who may wrongly expect clinical care from researchers or underestimate the risks associated with participation in research (Clayton and Ross, 2006; Meltzer, 2006).
Second, it is argued that disclosure of individual research results, which are often of experimental nature, varying quality and low predictive value, and are difficult to translate into useful clinical applications, can be hazardous due to the potential to provoke unnecessary distress and anxiety among research participants (Meltzer, 2006; DeCamp and Sugarman, 2004).
Third, disclosure of individual research results is often impossible, owing to common practices of data anonymization, which make attempts to track back individuals challenging.
Fourth, it is argued that the research participants are not educated enough to understand the nature and significance of the results, nor do the researchers have the necessary skills and resources to provide such information in a responsible and useful manner (Bredenoord et al., 2011).
Fifth, researchers often lack the necessary resources in terms of infrastructure, administration and counselling to disclose individual research results (Bredenoord et al., 2011; Forsberg et al., 2009).
Finally, expectations of disclosure set a false priority, as research participants enrol in research for altruistic reasons and not to receive individual benefits (Hallowell et al., 2010).
However, a number of arguments have recently been made by scientists, ethicists and policy-makers in favour of disclosure, which deserve closer attention:
First, population-based research involving genetics and genomics has gone through an unprecedented technological revolution, with the development of molecular biology, bioinformatics and automated genotyping technologies, which enables researchers to produce accurate results quickly and at a tremendously reduced cost (Collins, 2009). Such results potentially constitute information that may impact the health decisions of research participants and their relatives, for instance when severe monogenic conditions such as Huntington’s disease are discovered or when increased risks of developing conditions such as cancer or diabetes are identified.
Second, the emergence of new research designs recruiting individuals by genotype rather than by disease outcome may require that participants be informed about their genotype, which is contradictory to the current ‘one size fits all’ practice of non-disclosure of research results (Beskow et al., 2010).
Third, growing evidence indicates that research participants want to have access to their genetic risk information even if this information has no clinical or personal relevance (Beskow and Smolek, 2009; Kaufman et al., 2008; Meulenkamp et al., 2010; Murphy et al., 2008). Some researchers themselves also express that they would like to disclose certain individual results to research participants, especially in the case of unanticipated findings which may have clinical significance and for which treatment could be provided or which could affect major life decisions related to, for example, reproduction choices (Miller et al., 2009; Ruiz-Canela et al., 2009).
Fourth, an increasing number of national and international policies support a structured and organized method of feedback of research results (Knoppers et al., 2006).
Finally, the emergence of disciplines such as personalized medicine and pharmacogenomics, which aim to develop treatments and drugs adapted to the genetic profile of patients (Collins, 2009), and the successful development of commercial genetic testing companies (Borry et al., 2010), which makes genetic information more widely available, suggest increased interest in disclosure.
Moving towards more flexibility in terms of disclosure
The current ethical framework under which most population-based genetic research is traditionally subsumed is referred to herein as the medical ethics framework. The main tenets are that researchers conduct research and protect research participants from harm rather than focus on disclosure of results. However, advancements in genetics and genomics are reshaping the scientific and ethical landscape as described above and prompting the issue of disclosure to be revisited and revised. Moving to a new ethical framework that is better adapted to the current landscape and which could satisfactorily meet the wishes of both research participants and researchers in terms of disclosure may be needed. Two new ethical frameworks that provide for some degree of disclosure have recently been proposed. First, the ancillary care framework, based on the leading principle of beneficence, proposes disclosure of individual research results to research participants when these results can be actively used to rescue individuals from harm. Second, the promotive ethical framework, based on the leading principle of respect for persons, advocates that individual research results should be provided to participants because it enhances the participants’ autonomy. These alternative frameworks will be explored and compared to the medical ethics framework to identify which framework is most suitable for population-based genetic and genomic research. It should be noted that this comparative analysis is restricted to the issue of disclosure of individual research results to research participants and is not meant to identify an ethical framework to address all issues related to the conduct of biomedical research. The main characteristics of the different ethical frameworks are summarized in Table 1.
Main characteristics of ethical frameworks
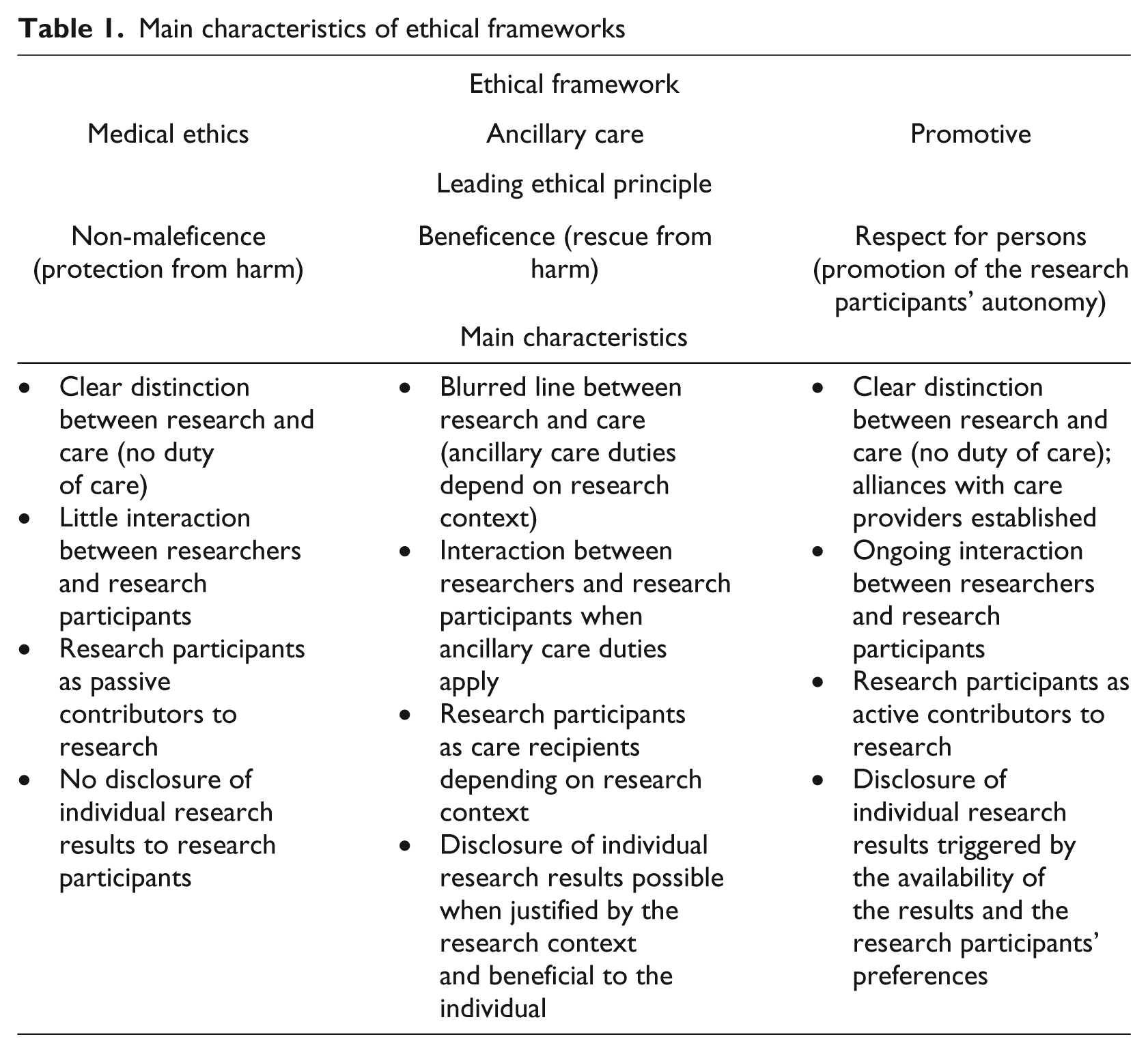
Protecting research participants from harm: The traditional medical ethics framework
The medical ethics framework is founded in medical practice and clinical research and is widely reflected in current national and international guidelines and policies (CIOMS, 2002; Office of Human Subjects Research, 2009; Trials of War Criminals, 1949; UNESCO, 2005; WMA, 1964). Under this framework the principle of non-maleficence is central, although other principles also apply (Cambon-Thomsen, International Data Sharing Conference, 2010). Researchers have an obligation to protect research participants from any potential harm, be it physical, psychological or societal, that may arise during or after the conduct of the research. Disclosure has the potential to be harmful and is neither encouraged nor given priority. Despite arguments explaining that public health research is different from other types of medical research and therefore requires a different ethical framework (Kahn and Mastroianni, 2007), the medical ethics framework is traditionally applied to population-based research involving genetics and genomics. In this framework, research participants are generally viewed as passive contributors to research, their role being to simply provide biological samples and health data. This framework is now being challenged in three important ways: 1) the nature of the information produced by researchers is more ‘personalized’ and potentially has significance for research participants; 2) research is evolving in a way which makes it difficult to preclude any form of disclosure; and 3) participants are increasingly expecting to have some type of interaction or communication from researchers. This raises the question whether the medical ethics framework is compatible with contemporary biomedical research. The ethical frameworks described below have been recently proposed (Belsky and Richardson, 2004; Beskow and Burke, 2010; Shalowitz and Miller, 2005) and address some of the shortcomings encountered with the prevailing medical ethics framework.
Bridging research and care: The ancillary care framework
The ancillary care framework, based on a principle of beneficence, proposes to bridge research and care by requiring that researchers provide research participants with ancillary care under specific circumstances (Belsky and Richardson, 2004). Ancillary care typically includes medical follow-up of research participants or guidance for future health care when conditions have been detected during the conduct of the research. Criteria that trigger ancillary care duties include the extent to which research participants entrust researchers with their health or the nature of the relationship between researchers and research participants – for example, a more familiar relationship developed due to regular meetings between the researcher and participant over a period of time. The level of dependence, vulnerability and gratitude of research participants towards researchers, and the strength of arguments made for or against providing care also come into consideration. An application of the ancillary care framework has recently been proposed for genetic research projects, arguing that the disclosure of individual research results to research participants should be enabled in these projects when the criteria described above are fulfilled (Beskow and Burke, 2010). For instance, according to the ancillary care model, researchers should disclose individual research results to participants in genetic family-based studies when the results cannot be obtained otherwise than through participation in research, and when these results indicate a high probability of developing a serious condition for which treatment can be provided. In such a case, the disclosure of individual research results would be justified by the participants’ high degree of dependence on the researchers conducting the study and by the significance of the research results. In other research contexts such as biobank research using leftover biological samples from clinical care where no direct relationship is established between researchers and research participants, the researchers’ duty to provide ancillary care would be much weaker according to this framework.
The ancillary care framework attempts to bridge research with health care when it can be directly useful for research participants. As we know, such bridging has often been considered inappropriate. The ancillary care framework also acknowledges the research participants’ right to access information about their own health when it can rescue them from harm. However, its application within the realm of population-based genetic and genomic research may be problematic for several reasons:
First, it may create some unjustified or unfair limitations to the disclosure of individual research results by requiring that research participants be highly dependent on researchers and entrust them with their health. However, such a level of dependence and entrustment is not common in many population-based research studies where most research participants have access to health care independently of participation in research and can choose to obtain genetic information through commercial channels.
Second, it requires a deep relationship between researchers and research participants, which is often not the case in population-based research in which large-scale projects including typically thousands of participants are conducted.
Third, the application of the ancillary care framework may lead to the creation of different classes of research participants: those who are considered to be dependent and vulnerable enough to ‘deserve’ access to their individual research results and those who are not sufficiently dependent and vulnerable to deserve such services. However, the nature and significance of individual research results should not depend on the degree of dependence or vulnerability of research participants towards researchers but rather on other criteria such as the scientific validity or clinical and personal relevance of the results (Dressler and Juengst, 2006).
Fourth, this framework implies that research participants are given access to their individual research results only in situations when doing so will ‘rescue’ them from potential harm, i.e. when findings of high health value are made for which medical follow-up can be provided. However, with the exception of a few rare lethal genetic conditions, individual research results currently being generated will often provide information of low predictive value for which no immediate action can be taken. Findings that may seem trivial to researchers could, however, be considered important to the research participants. Similarly, research results showing no increased genetic risk could be meaningful for research participants as it could potentially relieve them from anxiety, for instance when research results show that a research participant with a family history of breast or ovarian cancer is not a carrier of the BRCA1 and BRCA2 genes that are associated with the development of such forms of cancer (Kristman and Kreiger, 2008). In such situations, the act of disclosing individual research results does not constitute a ‘rescue’ of the individual per se but rather an act of reassurance which may be equally valuable for the research participant.
Finally, the ancillary care framework may perpetuate confusion that often arises regarding the role of researchers versus the role of health care providers as it will require that researchers modify their role depending on their power relationship with their research participants and the research context in which they operate. For these reasons the ancillary care framework may not be the most suitable ethical framework for population-based genetic and genomic research.
Empowering research participants: The promotive ethical framework
The promotive ethical framework relies on the principle of respect for persons by proposing that the disclosure of individual research results to research participants takes place on a more systematic basis, that is not only when research participants can be rescued from harm and therapeutic action can be taken, but also in situations where no therapeutic action can be taken but the results generated can be of interest for research participants (Shalowitz and Miller, 2005). Under this framework, research participants are not denied access to their genetic information as a measure of protection as is the case in the medical ethics framework. Furthermore, access to personal results does not depend on the nature of the relationship between the researcher and the participant as is the case in the ancillary care framework. Rather, access depends on the availability of the information itself and on the research participant’s willingness to receive such information (Shalowitz and Miller, 2005). This framework includes several interesting aspects:
First, it fully acknowledges the research participants’ right to access information about their own health independently of the nature or the utility of those results.
Second, it is the framework that may best accommodate the wishes of research participants who are interested in accessing their results (Beskow and Smolek, 2009; Kaufman et al., 2008; Meulenkamp et al., 2010; Murphy et al., 2008].
Third, this framework would be best suited when research designs requiring some disclosure, e.g. studies recruiting participants by genotype, are conducted.
Fourth, this framework allows researchers to show some gratitude towards research participants who have spent time and energy participating in research by providing them with individual research results (Shalowitz and Miller, 2005).
Fifth, it may help to clarify the role of researcher versus the role of health care provider since researchers would not be required to provide information as a part of an action of care but as an expression of their respect for the research participants’ right to self-determination. The role of ‘generalizable knowledge’ provider would still be held by researchers, while the role of health care provider would be held by other professions.
The promotive ethical framework is controversial for several reasons. For instance, it can be argued that the principle of respect for persons, which is the basis of this framework, can be achieved through other means than disclosure, e.g. by simply informing the participants about the terms of the research (Ossorio, 2006). Research participants would then choose to waive their right to access their results because they understand that providing such results is too demanding for researchers (Ossorio, 2006). Indeed, a clear policy of non-disclosure of research results may be more respectful than providing results of varying quality and interest (Clayton and Ross, 2006; Meltzer, 2006). However, if there is a fundamental right to know, i.e. a right to access results regarding one’s health, it is then difficult to understand why research participants should act against their own interest by waiving their right to access potentially useful information. The promotive ethical framework may also be accused of being incompatible with the research participants’ right not to know, which is as important as the right to know (Andorno, 2004). However, under the promotive ethical framework, disclosure is not mandatory but rather is an offer that research participants can choose to decline (Shalowitz and Miller, 2005). A potential pernicious effect of the promotive ethical framework could be that an offer to access individual research results leads research participants to wrongly expect clinical care from researchers even when the conditions for this type of disclosure have been clearly described in the informed consent. This confusion between when an individual participates in research and when she receives care – a phenomenon known as the therapeutic misconception – is argued to be reinforced by the disclosure of individual research results to research participants and to weaken the research participants’ rights (Clayton and Ross, 2006). For instance, research participants may wrongly believe that individual research results should always be made known to them as a component of their health care information, even when they would prefer not to access such information. Researchers themselves may feel an obligation to compensate their research participants for wrongful expectations by providing them with health care, even when they have neither the resources nor the competency to do so (Forsberg et al., 2009). However, the therapeutic misconception, a phenomenon mostly observed among patients participating in clinical research, may not occur as frequently as feared in population-based genetic and genomic research where healthy individuals are primarily recruited (Jonsen and Miller, 2008). Even if such misconception exists, it could be reduced to a minimum, as we will see when discussing models of disclosure, if the proper information and education mechanisms are put in place. Other arguments against the promotive ethical framework exist, as described before: the application of this framework to population-based genetic and genomic research would not necessarily provide participants with useful information because translating results from these studies into information of clinical relevance is often not practicable. Examples from genetic studies where attempts have been made to disclose some results have indeed raised more questions than provided good solutions (Richards et al., 2003; Wensley and King, 2008). However, designing governance structures which are flexible enough to foresee potential practical challenges may be an efficient way to deal with such issues (Caulfield et al., 2008). The harmful character of disclosure, as described earlier in this article, is another important argument. Regardless of the ethical framework model that is applied, researchers should not be exempt from protecting their research participants from physical, psychological or informational harm. However, as we will see in the remaining part of this article, innovative mechanisms exist to evaluate and minimize the risks of harm associated with disclosure.
To sum up so far, comparative analysis of the medical ethics based framework, the ancillary care framework and the promotive ethical framework reveals that, for the specific issue of disclosure in population-based genetic and genomic research, the promotive ethical framework is preferred. Although disclosure is still much debated, it is important to note that policies of non-disclosure are gradually changing in favour of some disclosure (Ethical Advisory Group of the UK10K Project, 2010; UK Biobank Ethics and Governance Council, 2010). This could mean that the practice of disclosure is on its way to being endorsed by the research community. Practical models of disclosure are needed, which rely on specific sets of criteria that can be systematically used to determine the terms of disclosure. Two potential models of disclosure have been recently proposed in the literature: 1) a result-evaluation model which emphasizes the scientific and contextual criteria of disclosure; and 2) a communicability model which emphasizes criteria of communicability of research results and the preferences of research participants to determine which results to disclose. The components of these two models are described and analysed below, with the objective to select the model which would best comply with the promotive ethical framework.
Models of disclosure
Results stemming from genetic and genomic research span a broad spectrum ranging from raw sequence data to known risk factors. Researchers need to determine which results have reached a sufficient level of maturity to be ‘disclosable’. They also need to identify results which are worth disclosing because the diagnostic, therapeutic, reproductive or personal utility of results varies significantly. In addition, they need to take incidental findings into consideration which were neither expected nor directly related to the purpose of the study. Researchers should have access to models of disclosure to help them systematically determine which results to disclose according to specific sets of criteria. Criteria that have traditionally been used to determine the disclosure value are typically classified into three categories: scientific, contextual and individual criteria. Scientific criteria refer to the analytic validity, clinical validity and clinical utility of the research results and are primarily meant to guarantee the scientific quality and utility of research results (Fernandez and Weijer, 2006; Ravitsky and Wilfond, 2006). Results are analytically valid when they are accurate and reliably identify ‘a particular genetic characteristic, such as a nucleotide sequence or a gene expression profile’ (Ravitsky and Wilfond, 2006). Results are clinically valid when enough empirical evidence is gathered to determine a specific association (e.g. increased or reduced risk of developing a condition) between a genotype and a particular clinical outcome (Ravitsky and Wilfond, 2006). Finally, results have clinical utility when they are analytically and clinically valid and can be used to improve an individual’s well-being through access to appropriate interventions (Bredenoord et al., 2011; Ravitsky and Wilfond, 2006; Grosse and Khoury, 2006). Contextual criteria refer to the researchers’ ability to communicate research results and take into consideration the context in which the research was conducted; individual criteria are related to the research participants’ preferences regarding disclosure and their ability to understand the disclosed results (Ravitsky and Wilfond, 2006).
Two recently proposed models of disclosure combine criteria of disclosure in different ways. The result-evaluation model places strong emphasis on scientific and contextual criteria (Ravitsky and Wilfond, 2006), while the communicability model favours individual criteria and additionally proposes criteria of communicability of research results to determine which results to disclose (Kohane and Taylor, 2010). The criteria sets for disclosure used in these two models are also shown in Figure 1.
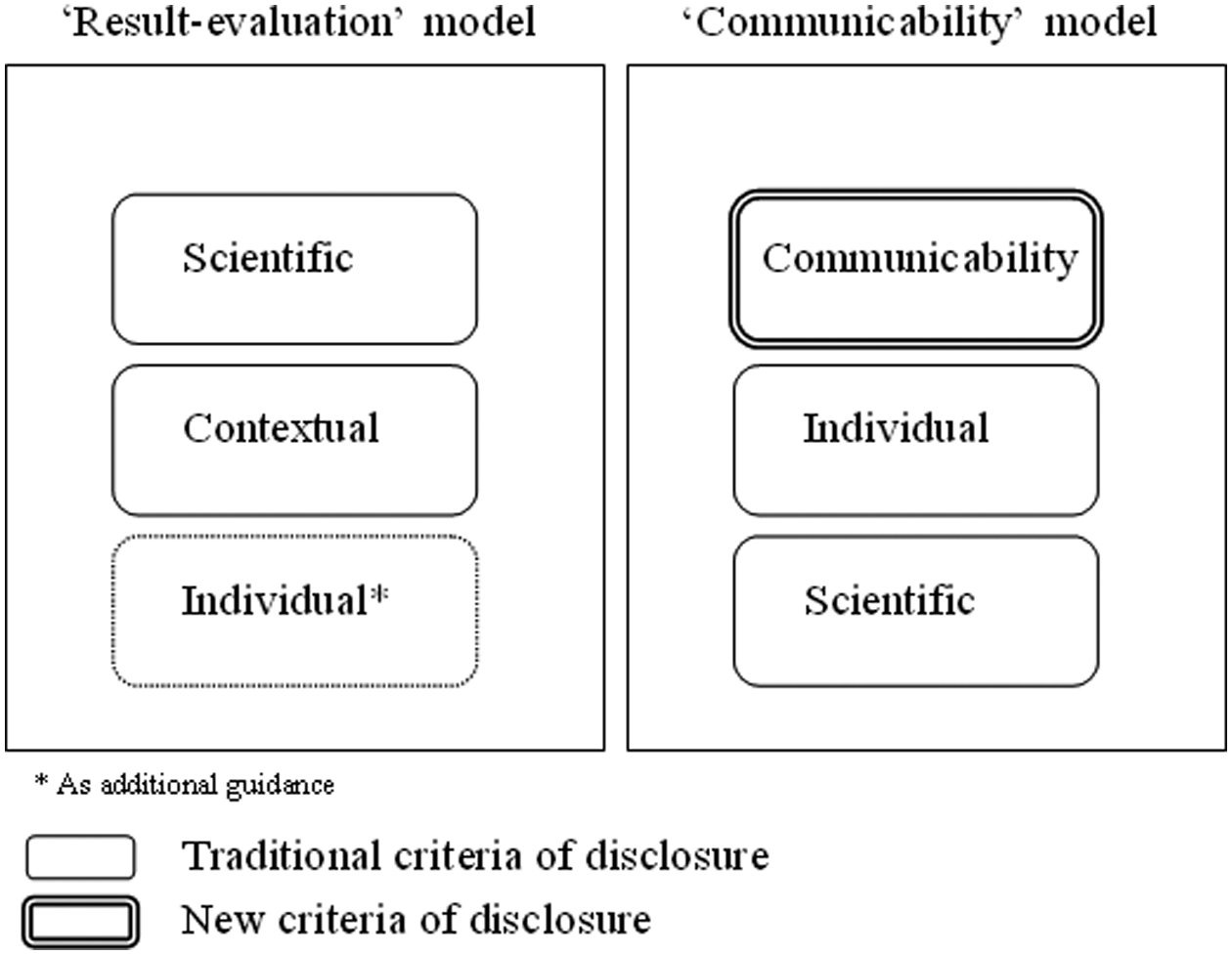
Criteria of disclosure used in models of disclosure
A scientific approach: The ‘result-evaluation’ model
The result-evaluation model prioritizes scientific and contextual criteria to determine which results to disclose. Research results can be disclosed if they are analytically valid and clinically useful and when their clinical validity is proven. Contextual criteria such as the researchers’ professional and financial capacity to disclose or the nature of the relationship between researchers and research participants may influence the disclosure outcome. For instance, according to the result-evaluation model, investigators may be committed to develop the necessary capabilities to offer results if the research participants do not have the possibility to access the results otherwise than through participation in the research. Similarly, researchers may not be required to disclose individual research results if they do not have the necessary financial and logistical resources to do so, or if their relationship with research participants is of a sporadic nature. Individual criteria are also considered as additional guidance. For instance, a participant’s strong preference for accessing results may, in some cases, compensate for a lack of clinical utility of the results (Ravitsky and Wilfond, 2006).
The result-evaluation model seeks to guarantee the scientific quality of research results and simultaneously allows contextual criteria and, to some extent, individual criteria to influence the decision process regarding disclosure. Despite its apparent flexibility, the result-evaluation model may be difficult to apply in a promotive ethical framework, for several reasons:
First, by requiring that the analytical validity and clinical utility of the research results be proven, this model may unnecessarily limit the disclosure of individual results in a way that is even more restrictive than what is normally encountered in other parts of clinical research, creating some form of genetic exceptionalism (Dressler and Juengst, 2006; Fernandez and Weijer, 2006; Sharp and Foster, 2006).
Second, this model assumes that common standards exist for defining and determining acceptable levels of analytical validity and clinical utility. However, such standards do not currently exist and even laboratory certification does not systematically provide sufficient proficiency testing, which makes it difficult to rely on one specific standard (Dressler and Juengst, 2006).
Third, the result-evaluation model uses contextual criteria that may not be the most appropriate. For example, it requires that researchers have sufficient communication skills and budget allocations to allow disclosure. However, as explained earlier, the disclosure of research results should not depend on criteria which are not intrinsically related to the significance and utility of the research results (Dressler and Juengst, 2006; Fernandez and Weijer, 2006).
Fourth, the result-evaluation model, by putting strongest emphasis on scientific criteria, implies that researchers are the only ones who can determine which results to disclose. Research participants cannot influence the disclosure process. This old-fashioned view of the role of research participants is not appropriate in a promotive ethical framework that seeks to empower the research participants by providing them with the necessary information to make their own decisions about their health. The result-evaluation model, despite its qualities, seems therefore to be too restrictive for the promotive ethical framework.
An interactive approach: The communicability model
Rather than relying primarily on traditional scientific and contextual criteria, the communicability model proposes to assess the terms of disclosure by putting emphasis on individual criteria. In addition, it introduces a criterion of communicability of research results which is mainly related to the level of clarity and comprehensiveness of the information provided, i.e. the likelihood that its content will be understood by the research participants (Kohane and Taylor, 2010). For example, according to the communicability model, individual research results should be disclosed to research participants when the research results are sufficiently clear and communicable to be understood by lay people and when the research participants have expressed willingness to receive the results, are willing to accept information about risk and are sufficiently literate to understand the meaning of the information provided. Disclosure is, in this model, required even when the clinical validity of the results is low and no intervention is possible. In contrast, individual research results should not be disclosed according to the communicability model when the research participants have clearly expressed that they do not want to receive results, even if the results have proven scientific validity and clinical utility. Under the communicability model, researchers alone do not determine which results are ‘disclosable’ but are required to interact with the research participants to do so. Researchers are asked to understand the research participants’ preferences in terms of disclosure (disclosure/no disclosure) and also regarding what each participant means by research results, how he or she assesses the information received and interprets its significance, and what he or she determines as being beneficial or harmful information (Kohane and Taylor, 2010). It is through such an interactive process that researchers determine the communicability of research results and evaluate what type of message is comprehensible and appropriate for the research participants based on their health and family history as well as expressed views and values. In the communicability model, the traditional scientific criteria usually defined by researchers are no longer the main determinants to assess disclosure but rather represent one component of the assessment process.
The communicability model is interesting in many ways. First, it offers a more sensitive approach to disclosure. Traditional scientific criteria which are supposed to be neutral (according to scientists) are given less weight and research participants are empowered because they are allowed to determine in co-operation with researchers what type of results are useful to them. Second, the communicability model, due to its design, encourages closer interaction between researchers and research participants, which will be increasingly important for genetic studies (Caufield et al., 2008). Third, this model is not static and allows research participants to change their mind about disclosure. This is particularly useful knowing that the significance and utility of specific research results may change over time. Finally, this model may clarify the role of researchers as research results providers when the research participants regularly interact with researchers and more easily understand the research questions at stake (Sharp and Foster, 2006).
Arguments against disclosure as described in the first part of this article could also be used against the communicability model of disclosure. For instance, it may be argued that the communicability model is harmful to research participants because disclosure creates unnecessary distress and anxiety among research participants. However, this model makes disclosure possible depending on which level of risk, jointly evaluated by researchers and research participants, the research participants are willing to take. A second counter-argument is that the communicability model can be seen as impossible to implement in practice because it requires enormous resources in terms of human power and infrastructure. However, developments in information technologies may help facilitate the implementation of such a model of disclosure. In similar research contexts, web-based patient portals are currently being developed to facilitate interaction and communication between patients and their health care providers 1 or to give research participants more control over their personal information.2,3
Because of its flexibility, its ability to adapt to an evolving research landscape, and its ambition to empower research participants, the communicability model of disclosure may be best fitted to meet the requirements of the promotive ethical framework. However, the application of a communicability model of disclosure will primarily depend on whether the promotive ethical framework is adopted by researchers, ethicists and policy-makers.
Conclusion
A number of arguments have traditionally been made against the disclosure of individual research results to research participants in population-based genetic and genomic research. However, recent developments within the field argue in favour of moving from the current medical ethics framework to a promotive ethical framework that would enhance accessibility for participants to information about their own health. Under such a framework, a communicability model of disclosure emphasizing individual criteria of disclosure could be used. However, the application of such a model will require a number of efforts. For instance, research participants should be sufficiently educated about the nature of genetic and genomic results and their potential significance for themselves and their family members (Sharp and Foster, 2006; Taylor and Johnson, 2007). Responsibilities in terms of disclosure should also be clearly mapped between researchers, ethics boards, genetic counsellors and GPs in order to determine who discloses what to whom (Caulfield et al., 2008). Established processes of disclosure approved by ethics boards should be integrated in the governance structure of research projects to ensure that disclosure is made according to specific criteria that all parties have agreed upon, and that the results are provided through a common channel (Caulfield et al., 2008). Resources in terms of infrastructure and human power should be allocated by funders and researchers to facilitate the implementation of a model of disclosure, and the researchers and their ethics boards should be familiar with local legal requirements in order to avoid unnecessary legal liability (Knoppers, 2001). Hopefully, a move towards implementing a promotive ethical framework will facilitate the conduct of innovative research designs through increased interaction between researchers and research participants and benefit research participants, researchers and society at large.
Footnotes
Acknowledgements
This research was supported through funds from The European Community’s Seventh Framework Programme (FP7/2007-2013), ENGAGE Consortium, grant agreement HEALTH-F4-2007-201413. The author thanks Senior Researcher Jennifer R. Harris, Norwegian Institute of Public Health, Norway, and Professor Jan Helge Solbakk, University of Oslo, Norway, for their comments to the manuscript.
The author declares no competing interests.
