Abstract
The relationship between age-related hearing loss (ARHL) and cognitive impairment (CI) remains intricate. However, there is no robust evidence from experimental or clinical studies to elucidate their relationship. The key unaddressed questions are (a) whether there is a causal effect of ARHL on CI and (b) whether efficacious treatment of ARHL (such as hearing-aid use) ameliorates CI and dementia-related behavioral symptoms. Because of several methodological and systematic flaws/challenges, rigorous verification has not been conducted. Addressing these stumbling blocks is essential to unraveling the relationship between ARHL and CI, which motivated us to undertake this review. Here, we discuss the methodological problems from the perspectives of potential confounding bias, assessments of CI and ARHL, hearing-aid use, functional-imaging studies, and animal models based on the latest information and our experiences. We also identify potential solutions for each problem from the viewpoints of clinical epidemiology. We believe that “objectivity,” specifically the use of more objective behavioral assessments and new computerized technologies, may be the key to improving experimental designs for studying the relationship between ARHL and CI.
Keywords
Because of progressive population aging, cognitive impairment (CI) has become a global public health concern. The identification of sensitive but treatable preclinical biomarkers for CI has been a fundamental task for neurological scientists. The close association between sensory disorders (such as hyposmia, hearing loss, and dysopia) and CI has evoked considerable interest among researchers. Other than the canonical olfactory impairment, age-related hearing loss (ARHL) has gained increasing attention because it is amenable to simple and easy intervention, namely hearing-aid (HA) treatment. ARHL (also termed “presbycusis”) is the most common chronic sensory problem in older adults. According to estimates, ARHL affects > 40% of people over 50 years old and approximately 71% of people over 70 (Slade et al., 2020). ARHL seems to be an ideal symptom for screening and intervention for preclinical CI because of its high prevalence rate and simple intervention. Indeed, several studies have elucidated the association between hearing loss and CI. However, the results have been inconsistent among different cohorts and different laboratories. There is a lack of consensus about two issues: whether there is a causal effect of ARHL on CI and whether efficacious treatment of ARHL (such as HA use) ameliorates CI and dementia-related behavioral symptoms.
Several recent reviews have discussed these issues from multiple perspectives. Loughrey et al. (2018) conducted a systematic review to assess the association of ARHL with cognitive function, CI, and dementia. They found a small but significant association of ARHL with global cognition, executive function, episodic memory, processing speed, semantic memory, and visuospatial ability. Notably, they found no significant association between Alzheimer’s disease (AD) and ARHL (Loughrey et al., 2018). Later, a Japanese group reviewed the mechanisms of ARHL and CI and formulated four hypotheses: the cognitive-load hypothesis, common-cause hypothesis, cascade hypothesis, and overdiagnosis or harbinger hypothesis (Uchida et al., 2019). Bowl and Dawson (2019) summarized the findings from human and animal studies concerning ARHL and CI from the perspectives of genetics, pathology, and etiology. They also identified the promising therapeutic strategies against ARHL from the viewpoints of otolaryngology (Bowl & Dawson, 2019). Most recently, Slade et al. (2020) discussed the association between the ARHL and CI from the viewpoints of neuroanatomy; they focused on the brain areas, including auditory cortex, speech areas, attentional networks, and motor system, that are related to ARHL and CI (Slade et al., 2020). In addition, Mamo et al. (2018) systematically reviewed the effect of HA on CI. Their findings suggested that treatment of ARHL with HA can improve the communication ability and quality of life of patients with CI; however, they could not authentically determine whether HA could benefit the clinical outcomes of CI because of the experimental design of the available studies (Mamo et al., 2018). These studies provided useful knowledge and insights concerning ARHL and CI from different perspectives. However, no review article has discussed the problems that may hamper the elucidation of the actual association between CI and ARHL.
Our laboratory also carried out a series of studies in 2009 regarding sensory loss and CI based on the aging population in Shanghai. During our investigation of ARHL and CI, we also encountered many methodological and technological problems that remarkably affected the experimental design and may have potentially reduced the study quality. A lack of recognition and resolution of these problems is a key barrier to uncovering the association between ARHL and CI. Hence, this review focuses on discussing the stumbling blocks in the study of ARHL and CI. We attempt to identify potential solutions and provide updated “take-home messages” on the basis of the latest evidence and technology. Our synthesis may help improve future investigation of the relationship among ARHL, HA, and CI.
Potential Hypotheses/Theories Regarding ARHL and CI
Several theories have been proposed to explain the association between ARHL and CI. However, these theories have not been rigorously verified because of the experimental difficulties. In this regard, many authors preferred to use the term “hypothesis” (Slade et al., 2020; Uchida et al., 2019). Here, we introduce the predominant theories related to the relationship between ARHL and CI.
Age is the common cause of ARHL and CI
This theory is also referred to as “the common-cause hypothesis,” which holds that ARHL and CI are the parallel results of age-related degeneration. ARHL is a broad term that attempts to cover the effects of hearing loss caused by aging. Age, on the other hand, is an important factor in CI. Aging is known to be associated with various CI-related diseases, including vascular cognitive impairment (Rundek et al., 2022), type 2 diabetes mellitus (Chakraborty et al., 2021), age-related eye disease (Le et al., 2022), mild traumatic brain injury (Ozono et al., 2022), Parkinson’s disease (PD; Charisse et al., 2022), and AD (Buglio et al., 2022). If this theory is valid, there is no causal relationship between ARHL and CI, and age is an underlying confounding factor. Treatment of ARHL may have little effect in ameliorating CI.
Cognitive-load theory
This theory is also called the “information-degradation hypothesis.” According to this theory, subjects with ARHL are in a state of “auditory-input deficiency.” Individuals with ARHL have to spend more cognitive resources for processing the degraded auditory information, causing an “overload” of the memory system. This may subsequently induce structural changes in the brain and finally cause cognitive decline. If the cognitive-load theory is correct, treatment of ARHL such as HA should be beneficial for CI.
Brain-reaction theory
Some authors also refer to this theory as the “cascade hypothesis” (Uchida et al., 2019) or “sensory-deprivation hypothesis”(Slade et al., 2020). This theory advocates that sensory deprivation in subjects with ARHL will directly or indirectly contribute to brain structural changes, such as reduction (Belkhiria et al., 2019) and suppression (Husain et al., 2014) of the anterior cingulate cortex and reduction of the bilateral superior temporal gyri (Peelle et al., 2011). The direct cause is related to the disuse effects of auditory function, and the indirect cause is due to the compensatory cortical reorganization and neural alterations. CI is the final outcome of these direct and indirect brain changes.
These theories appear plausible and are supported in part by experiments. However, the evidence available is empirical and fragmented. Although it is well known that hearing loss has a significant effect on the development of individual intellectual functions in children (Lieu et al., 2020; Niedzielski et al., 2006; van Wieringen et al., 2019), in the case of older adults, no study to date has provided compelling evidence of the casual relationship between ARHL and CI because of some methodological flaws and/or difficulties, which motivated us to undertake this review. Hence, we discuss the known methodological problems in the following sections.
Potential Factors Leading to Confounding Bias
Confounding bias is a statistical term that refers to a systematic bias during the evaluation of the casual relationship between an exposure factor (ARHL) and clinical outcome (CI) caused by mixing of the effects of exposure and the extraneous risk factors. The pathophysiology of CI is quite complex and is associated with many factors. Age, lifestyle factors (such as smoking, alcohol abuse), socioeconomic factors (such as income, occupation, and education level), and cardiovascular risk factors (such as increased body mass index, hypertension, hyperlipemia, and type 2 diabetes) are strongly associated with CI (Box 1). Moreover, once these factors are also closely associated with ARHL (Box 2), they may play a role as a confounding factor. These potential confounding factors make it extremely difficult to judge the relationship among the involved factors, particularly the causal relationship between ARHL and CI.
Box 1.
Factors Closely Associated With Cognitive Impairment
Several studies have suggested the relationship among lifestyle factors, socioeconomic factors, cardiovascular risk factors, and cognitive impairment (CI). These factors are also reportedly associated with age-related hearing loss (ARHL).
However, there are extremely complex relationships and interactions among these three factors. For example, worse lifestyle commonly results in greater cardiovascular risk, and lifestyle modifications reduce the cardiovascular risk. Likewise, lower socioeconomic status is associated with a worse lifestyle, which may lead to greater cardiovascular risk. If these associations are considered in the background of relationships between ARHL and CI, the use of HAs is an increasingly important confounding factor that must be taken into account. Too many factors with complex interactions increase the complexity of the casual chain. Questions such as “Which one is the real risk factor?” and “Which one is the confounding factor?” become ambiguous.
Box 2.
Factors Associated With Age-Related Hearing Loss
It is well known that noise exposure and ototoxic medications are independent causes of hearing loss in people of any age. However, several reports have also documented the relationship between noise exposure and cognitive impairment (CI). No study has investigated the relationship between CI and ototoxic medications.
Age
In terms of the common-cause hypothesis, age is a common cause of CI and sensory impairment. Fischer et al. (2016) found parallel changes in hearing, visual, and olfactory functions along with cognitive function. Despite the independent association of these sensory impairments with CI, 85% subjects with ARHL, 81% subjects with dysopia, and 76% subjects with hyposmia did not develop CI during the 5-year follow-up (Fischer et al., 2016). Eckert et al. (2019) found that the hearing changes did not track with the brain atrophy because these changes were observed in both ARHL and aging (Eckert et al., 2019). These findings seem to support that age, rather than ARHL, is the underlying cause of CI.
Factors associated with CI and ARHL
Boxes 1 and 2 summarize the influence of factors associated with CI and ARHL, respectively. The complex causal relationship among age, factors in Box 1, Box 2, ARHL, and CI (Fig. 1) increase the intricacy of the direct relationship between ARHL and CI. In light of a statistical model indicating the relationship between intervention/exposure, outcome, and confounder (Hernán & Robins, 2020; Vanderweele et al., 2014), ARHL was denoted A in this model presenting the intervention/exposure; CI was denoted Y presenting the outcome; and other factors such as age, factors in Box 1, and Box 2 were denoted C1, C2, and C3, respectively, presenting the confounders when they affect both exposure (A) and outcome (Y; Fig. 2). Because age (C1), lifestyle and cardiovascular risk factors in Box 1 (C2), and noise exposure in Box 2 (C3) are associated with both ARHL (A) and CI (Y), all of these can be common causes of ARHL (A) and CI (Y). From another perspective, all of these may be confounding factors in the investigation of the causal association between ARHL (A) and CI (Y). Moreover, age (C1) is also related to the cardiovascular risk factors (C2); hence, age (C1) may serve as a final confounder (Fig. 2).
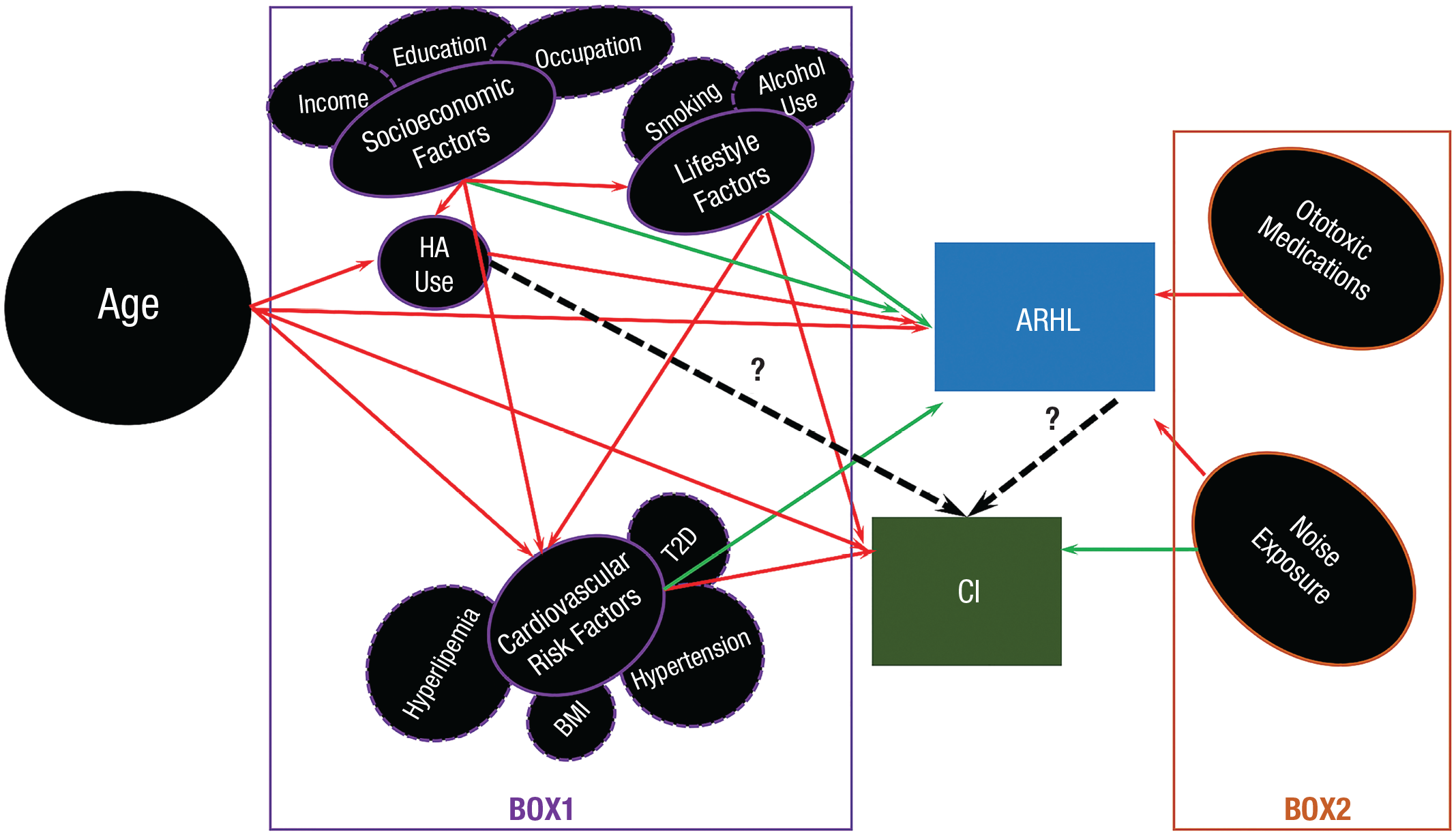
The complex interactions and relationships among age, Box 1, Box 2, cognitive impairment (CI), and age-related hearing loss (ARHL). The red lines represent the casual relationships that have been confirmed, the green lines represent the casual relationships that have been verified by several studies, and the dashed black lines represent the casual relationships that require further verification.
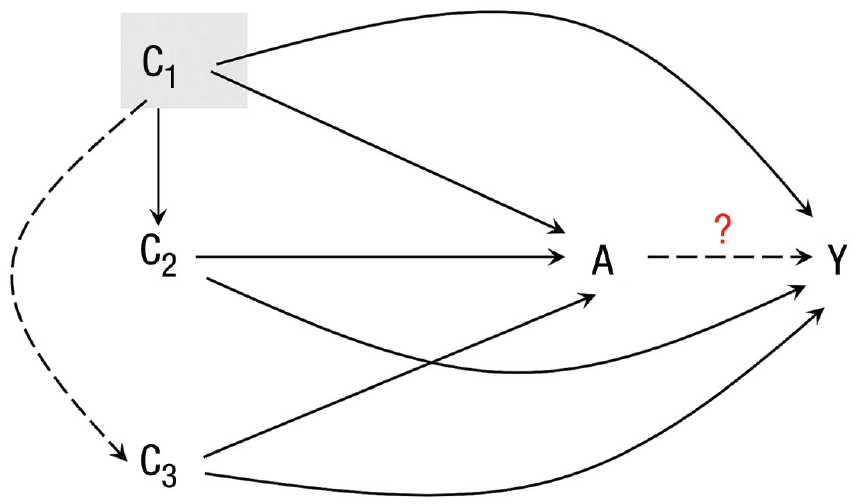
Causal diagram with intervention/exposure A (ARHL), outcome Y (CI), and confounding variables C1 (age), C2 (lifestyle and cardiovascular risk factors in Box 1), and C3 (noise exposure in Box 2). Full lines represent confirmed casual relationships; dashed lines represent potential casual relationships that require further verification. C1 is highlighted to represent that age might be a final confounder. ARHL = age-related hearing loss; CI = cognitive impairment.
Theoretically, one methodological approach to avoid the confounding bias is the stratification of subjects according to the potential confounding factors. However, this is difficult to achieve because of the requirement for a much larger sample size. Another simple method is the direct intervention of ARHL using the HA equipment. However, performing a rigorous verification of the efficacy of HA is inherently challenging. Some authors have suggested that the development of novel equipment that can concomitantly measure sensor-neurocognitive function may help avoid the potential confounding bias (Fischer et al., 2016).
Methodological Flaws During the Assessments of CI and ARHL
From the perspective of clinical epidemiology, when the causal relationship between a certain exposure factor (such as ARHL) and a clinical outcome (such as CI) is measured, an unbiased estimation can be obtained only when the assessments for these two issues are independent without mutual interference (Asakawa, Fang, Sugiyama, Nozaki, Hong, et al., 2016). Conversely, for example, if the assessments of the clinical outcome are also affected by the exposure factor, the relationship is likely to be exaggerated or underestimated. Our previous study discussed the cases using behavioral indices such as “speed” and “initiation time” to evaluate the cognitive state in subjects with PD. Because these indices were affected by PD, we could not distinguish whether the slowness of speed was a result of PD per se or caused by cognitive decline (Asakawa, Fang, Sugiyama, Nozaki, Hong, et al., 2016). Such issues can be regarded as a sort of confounding bias. In the subsequent section, we discuss such methodological flaws in the assessments of CI and ARHL.
Flaws in the identification, classification, and assessment of CI
The Mini-Mental State Examination (MMSE) is the most commonly used tool for assessing cognitive function (Asakawa, Fang, Sugiyama, Nozaki, Kobayashi, et al., 2016). The MMSE is also used for the identification, classification, and assessment of CI. However, hearing loss may notably influence performance in neuropsychological assessments (Hill-Briggs et al., 2007). In a study by Jorgensen et al. (2016), hearing loss was found to significantly affect MMSE performance in young adults (Jorgensen et al., 2016). Another study found that a noticeable reduction in MMSE scores could be attributed to hearing loss, particularly in older populations with a low education level (Gaeta et al., 2019). The use of neuropsychological assessments (Table 1) that are potentially affected by hearing loss to explore the association between ARHL and CI will lead to a biased conclusion. Some authors have even summarized this bias as an “overdiagnosis hypothesis,” which refers to a disadvantage during assessments in which subjects cannot clearly hear testers’ voices because of hearing impairment (Uchida et al., 2019).
Commonly Used Cognitive Assessments Involved in ARHL Studies
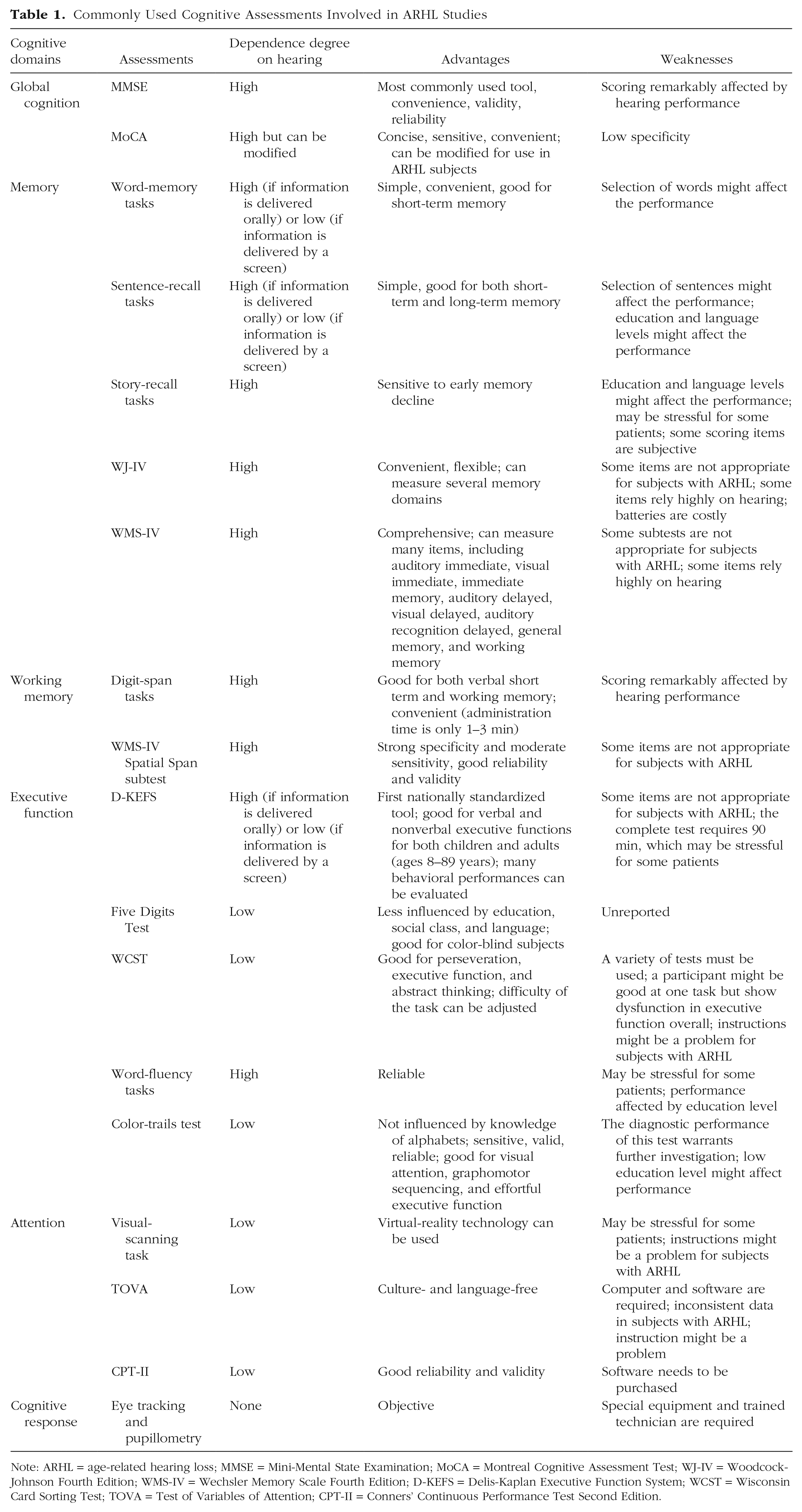
Note: ARHL = age-related hearing loss; MMSE = Mini-Mental State Examination; MoCA = Montreal Cognitive Assessment Test; WJ-IV = Woodcock-Johnson Fourth Edition; WMS-IV = Wechsler Memory Scale Fourth Edition; D-KEFS = Delis-Kaplan Executive Function System; WCST = Wisconsin Card Sorting Test; TOVA = Test of Variables of Attention; CPT-II = Conners’ Continuous Performance Test Second Edition.
Another important issue is the classification of CI. CI can be classified into different stages, such as preclinical stage, cognitive-decline stage, CI stage, and dementia stage. CI may also involve many domains, such as global cognition, executive function, episodic memory, semantic memory, and visuospatial ability. Moreover, the relationship between ARHL and CI may vary in different stages and in different domains. Loughrey et al. (2018) found a small but significant association of ARHL with accelerated cognitive decline, CI, and dementia. However, no significant association was found between ARHL and dementia. With respect to cognitive domains, global cognition, executive function, episodic memory, semantic memory, and visuospatial ability were significantly associated with ARHL. Only fluency exhibited no significant association. Although the authors attributed the lack of statistical significance to the small sample size, the difference of a relationship relevant to different stages and domains requires serious attention (Loughrey et al., 2018). Nevertheless, the classification of CI based on the assessments that are affected by hearing loss (such as MMSE scores) increases the risk of a biased association.
As for the solutions, the simplest method is to select the appropriate scoring system that is less reliant on the auditory function. Dawes et al. (2014) modified the Montreal Cognitive Assessment (MoCA) to evaluate cognitive function in individuals with hearing loss (Dawes et al., 2014). Their results are awaited. However, owing to the complexity of most neuropsychological tasks, even with the use of an appropriate test, the instructions for subjects with ARHL may pose a challenge. Another important solution is to select physiological measurements. Approaches such as EEG (Ohlenforst et al., 2017), eye tracking, and pupillometry (Van Engen & McLaughlin, 2018) have been used to evaluate cognitive function. However, these methods require special equipment and experienced researchers. The development of novel CI assessments, particularly for individuals who have hearing impairment, is challenging.
Flaws in the measurement of hearing loss
Pure-tone audiometry (PTA) is the current gold-standard method for the identification of ARHL. It is a critical tool for the diagnosis, assessment, and classification of hearing loss. PTA is commonly used to measure the hearing thresholds that represent the lowest detectable levels of pure tones at certain frequencies. However, PTA is far from a faultless method for evaluating ARHL. There are several known limitations regarding the applications of PTA. First, technologically, a normal examination of PTA requires a silent examining room, an experienced audiologist, and a lot of time. This makes it unsuitable for use in a large-scale investigation. Second, overall, hearing loss is commonly classified into four categories: sensory, neural, metabolic, and mechanical ARHL. PTA can provide information only regarding hearing sensitivity. It provides no information regarding the central auditory processing and auditory processing of real-world signals (Musiek et al., 2017). Hence, PTA can provide limited insights into functional hearing, especially in patients with central deafness. For example, some older patients with changes in the central auditory nervous system and dysfunction in temporal processing ability may present no changes in PTA (He et al., 2008; Hopkins & Moore, 2011). Patients with damage of the central auditory nervous system (e.g., difficulty in understanding meanings of speech in a noisy background) may show normal pure-tone thresholds measured with PTA (Anderson et al., 2011). Conversely, PTA sometimes may provide misleading results during the identification of central hearing loss. A series of studies by Musiek et al. (2017) showed that in patients with brain damage (such as cerebral ischemia) who do not have hearing loss, PTA may show severe to profound hearing loss (Musiek et al., 2017). Some researchers may disregard the classification of hearing loss in ARHL studies. However, regardless of classification, the evidence presented above seems to indicate a limited role for PTA in the investigation of hearing loss caused by central nervous system disorders. Finally, PTA cannot detect subtle hearing loss in patients with tinnitus and those who are exposed to noise (Ishak et al., 2013; Schaette & McAlpine, 2011).
Central deafness is affected not only by “peripheral” hearing and sound transduction but also by the “central” processing and comprehension of the auditory input. These findings indicate that PTA may not be adequate for evaluating auditory function, particularly in patients who have central deafness or deafness resulting from mixed mechanisms (central and peripheral). More behavioral assessments and objective electrophysiological tests should be included to enable a comprehensive evaluation of the auditory function in individual patients. C. Wang et al. (2021) used a binaural-integration paradigm, which can measure the ability to detect and store binaural temporal fine-structure cues of wideband noise. They used this paradigm to evaluate the binaural processing of speech comprehension in a noisy environment. They found that the neural adaptation for binaural processing can reflect the cognitive stages. The authors claimed that the binaural-integration paradigm may be a promising tool for screening for CI (C. Wang et al., 2021). Moreover, some objective electrophysiological tests, such as the auditory brainstem response (ABR), acoustic reflexes, frequency-following response, otoacoustic emissions, and endocochlear potential (EP) can be considered during the evaluation of ARHL.
Problems Involving the HA Studies
Theoretically, the appropriate evaluation of the effect of treatment of hearing loss on cognitive function in older people can help clarify the causal relationship between ARHL and CI. Wearing HA equipment is the most simple and common treatment for ARHL. Therefore, the pertinent question is whether using HA can improve cognitive function. However, because of numerous methodological difficulties, there is limited evidence of the value of HA in ameliorating cognitive function. Mamo et al. (2018) conducted a systematic review of studies that investigated the value of hearing-loss treatment in the amelioration of cognitive function in older people. The available evidence showed that treating auditory dysfunction in patients with CI may ameliorate difficulties in communication and dementia-related symptoms; however, because of the limitations of the available studies, there was no definitive evidence of the beneficial effect of HA use on cognitive function (Mamo et al., 2018). The problems in the current HA studies pertain to several aspects. First, the rate of using HAs among people with CI is quite low. It was estimated that only approximately 30% people with ARHL used HA equipment. Second, it is difficult to set up a control group because of the ethical problems associated with denying treatment for ARHL. However, the lack of a control group reduces the strength of evidence. Third, it is difficult to blind the researchers to the cognitive situation and treatment group. Last, the technological limitations of the currently used HA equipment may enhance the experimental difficulty. Haque et al. (2012). reported a typical scenario in which patients with CI experience problems using HA equipment. A patient with CI had worn dysfunctional HA equipment for many years because she could neither manage the battery nor report that the HA was not working. Such cases may not be rare, which may increase the difficulties in HA–CI investigations (Haque et al., 2012). Routine maintenance of the HA equipment may be a solution; however, it will impose a financial burden on the patients. Hence, the development of novel HA equipment, particularly for patients with hearing loss and CI, that can be easily managed and reported by the caregiver (such as management by smart phone) is a key imperative. Solutions of these problems includes the popularization of HA use; development of better smart HA machines with lower cost, lower risk, and cheaper maintenance; and improvement of the experimental design (see later section).
Other Difficulties in Studies Concerning ARHL and CI
Difficulties in functional imaging
Functional-imaging modalities such as functional MRI (fMRI) and PET are attractive tools for investigating the central nervous system mechanisms associated with ARHL and CI. These imaging technologies enable the observation of changes in regional cerebral blood flow, volume, metabolism, and oxygen extraction, which can be used to investigate neural activation. In addition, resting-state fMRI is also used to explore the functional connectivity between the auditory cortex and other brain structures in subjects with ARHL and CI. fMRI using the blood oxygen-level dependent signal is useful for investigating activation over a relatively long period of time. In contrast, conventional approaches for the assessment of “potential,” “response” (such as ABR, EP), and motor-evoked potential are useful for the investigation of momentary neural activity (Slade et al., 2020). However, because of the methodological differences, the results of previous fMRI studies have been largely inconsistent. Panouillères and Mottonen (2018) observed the activation of the tongue motor cortex during speech listening in young subjects and older subjects with normal hearing, whereas such activation was weaker in older subjects with ARHL. These findings suggest a motor-network-related mechanism. However, Du et al. (2016) found enhanced activation of the frontal speech motor areas in older subjects (compared with young adults) during a listening task, suggesting a compensation-related mechanism (Du et al., 2016). Hence, the functional-imaging approaches are highly dependent on the methodology, including experimental design, data-acquisition methods, and analysis methods. However, the fMRI data-processing methods are complex and continuously developing because the available data-processing methods have various limitations (Cole et al., 2010). Hence, the methodological differences between studies make their results incomparable. Another difficulty lies in the investigation of age-related CI and hearing loss. If age is considered a factor influencing CI and hearing loss, the investigation of CI and hearing loss in different age groups, depiction of the variation tendency of CI and hearing loss with aging, or comparison of the difference of CI and hearing loss between young and older populations is crucial. Unfortunately, the prevalence of hearing loss in young subjects (< 40 years) is quite low (< 2%; Wilson et al., 2017). Such experimental design is indeed quite difficult to conduct. Moreover, such approaches involving fMRI or PET are costly and require expensive equipment and highly trained technicians. These approaches cannot be used in a large-scale investigation. These methodological difficulties restrain further application of the functional-imaging approaches on this topic.
Animal studies might be invaluable but challenging
Experiments entailing exposure of subjects to a high-level noisy environment and investigating the hearing loss are unethical and cannot be conducted in humans. Therefore, many animal models have been used in studies exploring the casual relationship between ARHL and CI, such as chinchilla, gerbils, rat, and mouse. Although the animals have a short life span, which is helpful for the researcher to observe the changes caused by natural aging, no animal model can perfectly mimic all aspects of human ARHL and CI.
Chinchillas
The chinchilla animal model is the most commonly used model for noise-induced hearing loss. It has many advantages for studying hearing loss (Table 2); however, it has many limitations for an ARHL-based study. First, chinchillas have a longer gestation time (112 days vs. 21 days in rats and mice) and longer life span (8–20 years vs. 2 years in rats and mice; Trevino et al., 2019). Although some authors believe that the chinchilla model is good because it enables the researcher to gain enough time to explore the hearing loss more accurately (Trevino et al., 2019), this would increase the cost and reduce the experimental efficiency for tracking the longer aging time in chinchillas. Second, compared with mice, chinchillas lack the genetic-related ARHL; therefore, it is not appropriate for studies evaluating attributes or manipulations of a certain gene to induce ARHL. Last, the behavioral assessments and applications for the evaluation of CI in chinchillas are limited. These limitations restrain the use of chinchillas as the preferred animal model for ARHL/CI studies.
Commonly Used Animal Models in Studies Concerning the Relationship Between ARHL and CI

Note: ARHL = age-related hearing loss; CI = cognitive impairment.
Rats
The most commonly used rat model involved in ARHL research is the Fischer 344 rat model. One advantage of this model is the rapid development of progressive ARHL (starting from the age of 12 months and low-frequency damage from the age of 18 months). Cheaper Wistar albino rats have also reportedly been developed as an ARHL model. The advantages and weaknesses of the rat model are listed in Table 2.
Mice
The mouse model has been the mainstay model for investigating ARHL. Because of their genetic diversity, mice are used to investigate the genetic basis of ARHL. Moreover, because of their short lifespan, well-developed behavioral assessments for cognitive functions, and relatively easier genetic manipulations (such as knock-in and knockdown), mice are invaluable in ARHL/CI studies. The most commonly used mice are C57BL/6 strains, which begin to display progressive loss of sensory hair cells (Bowl & Dawson, 2019). Nonetheless, the mouse model also has many limitations. The main disadvantage of the mouse model is that mice have a different hearing range from humans, which makes it difficult to determine which cochlear regions in mice and humans are appropriate for comparison. The small size of mice and hearing-related structures also limit some experiments and surgeries. Moreover, mice are very sensitive to noise, which might be a confounding factor in ARHL research (Table 2).
Gerbils
Gerbils are the most preferred animal for ARHL research. With regard to their biological characteristics, gerbils looks like a “compromised” animal between chinchillas and mice. They are smaller than rats but have the same hearing range as humans. Their life span is intermediate (3–4 years) and between that of chinchillas and rats. They can be trained to complete complicated behavioral tests. They shares some advantages/disadvantages with chinchillas and rats (Table 2).
To summarize, these four animal models have their respective advantages and disadvantages (Table 2). Researchers should select the best model in terms of the aims of their study.
Concluding Remarks
There is no robust evidence to date pertaining to the casual relationship between ARHL and CI. Because of the methodological and systematical flaws/challenges, very few clinical trials can be satisfactorily conducted regarding the causal relationship between ARHL and CI and whether the prevention or treatment of ARHL can ameliorate CI. These stumbling blocks motivated us to undertake this review. Other than these methodological flaws, exploration of the robust therapeutic targets for ARHL is also crucial. Several potential suggestions might be helpful for future investigations.
Regarding the experimental design
Several studies have shown that the investigation of a relationship between ARHL and CI requires a large sample size (Bowl & Dawson, 2019) because of the numerous confounding factors (Fig. 1). Subgroup analysis based on these factors necessitates enrollment of more participants. In addition, long-term observation is indispensable for studying hearing loss and CI. Moreover, examinations such as PTA are expensive. These problems increase the cost of the study beyond that covered by most research grants. Hence, the organization of a large transnational research group might be a solution. A large transnational research initiative such as the Lancet Commission on Dementia Prevention, Intervention, and Care (Livingston et al., 2017) may overcome the insufficiency of research grants and manpower and should be the future direction of this sort of study.
Objectivity might be the key to resolve some methodological flaws
“Objectivity” here refers to the use of objective evaluation criteria and the development or application of objective behavioral assessments for both hearing and cognitive function. The use of more objective tools can resolve some methodological flaws, such as “lack of blinding.” We believe our insights into behavioral assessment in PD may also inspire the development of assessments for ARHL, namely the principles of objectification, multipurpose, and simplification (Asakawa, Fang, Sugiyama, Nozaki, Hong, et al., 2016; Asakawa, Fang, Sugiyama, Nozaki, Kobayashi, et al., 2016). Moreover, with the development of computerized technology, new technologies such as wearable sensors, virtual reality, artificial intelligence, and big data have been used for the development of behavioral assessment (Asakawa et al., 2019). These technological advances may provide unprecedented leverage for exploring the relation between ARHL and CI.
Footnotes
Acknowledgements
We thank Enago for the English translation.
Transparency
Action Editor: Tina M. Lowrey
Editor: Interim Editorial Panel
Author Contributions
T. Asakawa: Conceptualization, Investigation, Writing – original draft, Writing – review & editing, Visualization, Supervision, Project administration. Y. Yang: Investigation, Writing – original draft. Z. Xiao: Investigation, Writing – original draft. Y. Shi: Investigation, Writing – original draft. W. Qin: Investigation, Writing – original draft. Z. Hong: Investigation, Writing – original draft, Supervision, Project administration. D. Ding: Conceptualization, Writing – original draft, Writing – review & editing, Supervision, Project administration, Funding acquisition. All of the authors approved the final manuscript for submission.
