Abstract
Background:
Intimate partner violence in its various forms increases HIV exposure in female victims and potentially jeopardizes the HIV treatment cascade, for instance, by impeding engagement in and adherence to care. Elevated rates of HIV and intimate partner violence are reported in Central Africa. Evidence on the effect of intimate partner violence on antiviral therapy interruption is lacking in Cameroon, where only 330,000 women live with HIV and only 19% of HIV-positive people are virally suppressed. This study aimed to assess the prevalence and factors of intimate partner violence against HIV-positive women and its relationship with antiretroviral therapy interruption ⩾1 month.
Methods:
The EVOLCam cross-sectional survey was conducted in 19 hospitals in the Center and Littoral regions. The study sample comprised antiviral therapy–treated women declaring at least one sexual partner in the previous year. Scores of recent emotional, physical, extreme physical and sexual intimate partner violence were built using principal component analysis and categorized under no, occasional or frequent intimate partner violence. Multivariate logistic analyses were performed to investigate the relationship between intimate partner violence and recent antiretroviral therapy interruption ⩾1 month, and associated factors.
Results:
Among the 894 analyzed women, the prevalence of intimate partner violence was 29% (emotional), 22% (physical), 13% (extreme physical) and 18% (sexual). Frequent physical intimate partner violence was a significant risk factor of antiretroviral therapy interruption ⩾1 month (adjusted odds ratio = 2.42 (95% confidence interval = 1.00; 5.87)). It was also associated with HIV-related stigma (2.53 (1.58; 4.02)), living with a main partner (2.03 (1.20; 3.44) and non-defensive violence against this partner (5.75 (3.53; 9.36)).
Conclusion:
Intimate partner violence is a potential barrier to antiviral therapy continuity and aggravates vulnerability of Cameroonian HIV-positive women. The prevention and detection of intimate partner violence by HIV services might help to reach the last “90” of the 90-90-90 targets.
Introduction
Violence toward women is a worldwide public health issue and affects one woman in three. Perpetrators are mostly their intimate partners. Indeed, the latter are responsible for one-third of murders of women. Women physically or sexually abused by their partners have poorer physical, reproductive and mental health. Aside from mortality and injuries resulting from physical violence, they are also more likely to experience depression, to acquire sexually transmitted diseases and to have induced abortions or babies with low birth weight.1,2 Intimate partner violence (IPV) also has an intergenerational effect on IPV itself 3 and on poor health outcomes.
IPV increases the risk of HIV acquisition, may delay or prevent HIV testing and HIV status disclosure to partners4,5 and exacerbates the vulnerability of women living with HIV (WLHIV). WLHIV are at greater risk for domestic and family exclusion than other women. 4 IPV among people living with HIV (PLHIV) is associated with decreased access or use of healthcare services,6–9 poorer engagement in HIV care, sub-optimal antiviral therapy (ART) adherence10,11 and lower health-related quality of life, which is a strong predictor of death in PLHIV.11,12 Suicidal ideation and attempts, depression, anxiety or post-traumatic disorders are more frequent in HIV-positive persons suffering from IPV than in other PLHIV, all of which affect ART adherence, immune functions and viral suppression.12,13 To summarize, IPV is greatly interconnected with HIV and health outcomes of PLHIV and may jeopardize the implementation and success of various elements of the HIV treatment cascade.
IPV and HIV are both endemic in Central Africa.4,14 In 2014, Cameroon was the 15th country most affected by HIV worldwide. Only 37% of PLHIV there received ART and only 19% were virally suppressed. 14 The lack of real progress toward achieving the UNAIDS 90-90-90 targets is a concern for the control of the epidemic in the country. In 2016, of the 32,000 new HIV infections, 4000 were due to mother-to-child transmission. 14 The 330,000 WLHIV in Cameroon account for 65% of all PLHIV in the country, a prevalence of 5.1% in women compared with 2.5% in men. In addition, Cameroon has the fifth highest prevalence of IPV among Sub-Saharan African countries. According to the 2011 Demographic Health Survey, nearly half of all Cameroonian women had recently suffered emotional, physical and/or sexual IPV. 15 In this setting, where the prevalence of both HIV infection and IPV is particularly high, no study has yet investigated the relationship between IPV and ART discontinuity in women. Antiretroviral therapy interruption (ATI) compromises the control of the HIV epidemic 16 and is a stronger risk factor than sub-optimal ART adherence for virological failure and HIV drug resistance17,18 which are both very prevalent in Cameroon. 19
The ANRS-12288 EVOLCam cross-sectional survey was conducted in 2014 in Cameroon to study the living conditions of PLWH followed up in 19 HIV services in the Center and Littoral regions, established as part of the national ART program. A previously published analysis from that survey reported that 21% of PLWH reported ATI. 20 The two objectives of this study were first to investigate whether IPV was associated with ATI among WLHIV participating in this survey and second to describe the prevalence of the different forms of IPV in this sub-population and identify associated risk factors.
Methods
Study design, participants and data collection
The ANRS-12288 EVOLCam study was a cross-sectional survey conducted in the Center and Littoral regions of Cameroon between April and December 2014 which recruited 2357 eligible PLHIV (⩾21 years old, HIV-diagnosed >3 months) attending one of the study’s 19 participating HIV services. Details about recruitment can be found elsewhere. 20 Demographic, socioeconomic, domestic and psychosocial data were collected during face-to-face interviews performed by independent, trained interviewers. The questionnaire included a section dealing with IPV previously developed by the World Health Organization (WHO) for a multi-country study on women’s health and domestic violence against women. 21 Participating women were asked 12 behavior-specific questions about acts of violence (physical and mental) they might have had suffered from their most recent intimate partner in the 12 months prior to the survey and the frequency of these acts (never occurred, occurred occasionally or occurred frequently). Clinical data were obtained from medical files. A blood sample was collected from patients treated >6 months to measure HIV viral load (VL) and was analyzed by a reference HIV laboratory in Yaounde accredited by the WHO (quantification threshold 100 copies/mL). All participants provided written informed consent. ANRS-12288 EVOLCam was approved by the Ministry of Public Health in Cameroon and the Cameroonian National Ethics Committee. The study population of the present analysis included women declaring at least one intimate partner in the 12 months prior to the survey and having no missing data in the IPV section of the questionnaire.
IPV scores
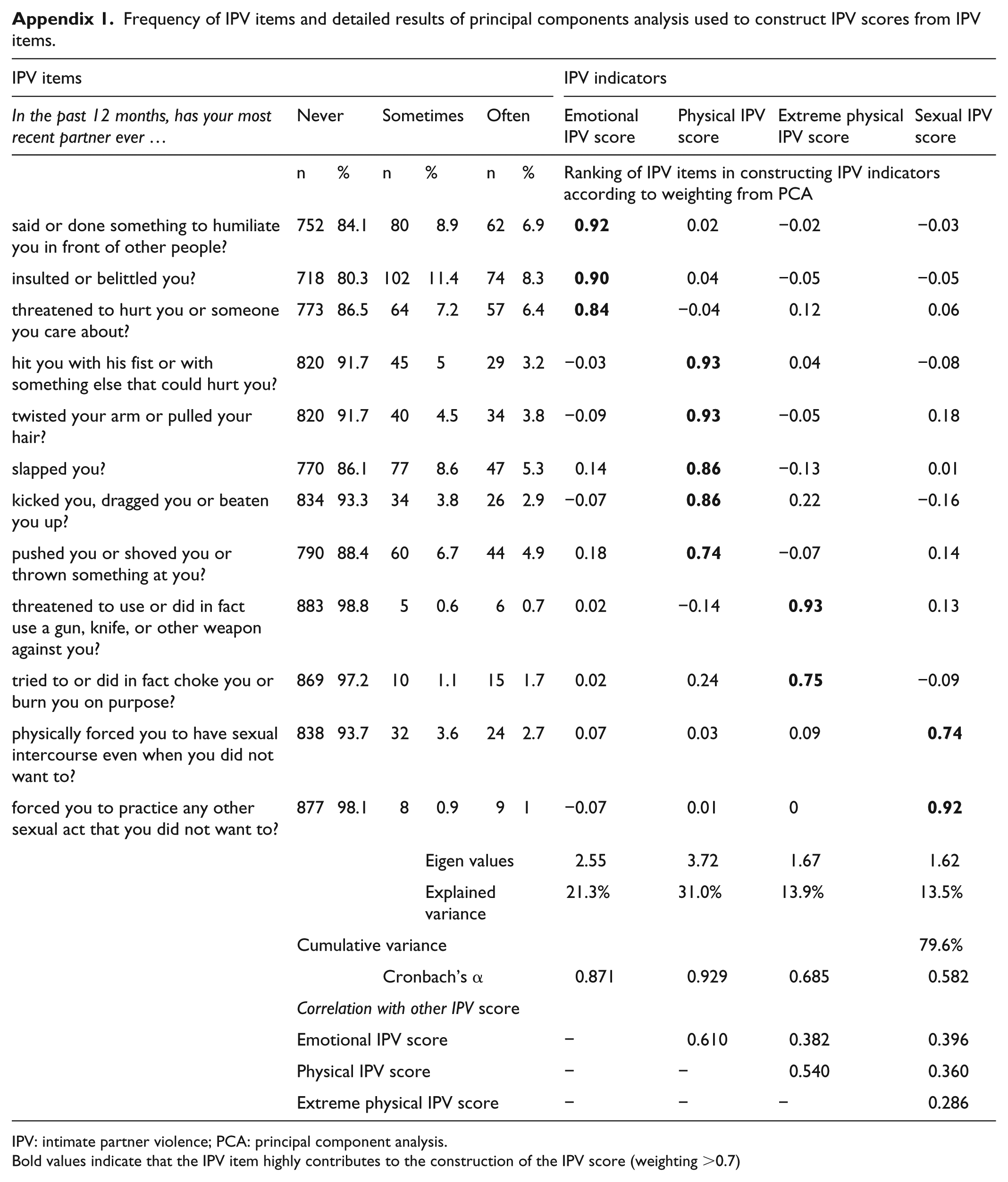
We performed a principal component analysis (PCA) on the 12 items of the IPV questionnaire section to build IPV scores which, by construction, were standardized and computed using values ranging from 0 (no violence) to 1 (high level of violence). The Promax rotation technique, which enables correlation between factors, was implemented to improve the fit of the data. 22 IPV was broken down into four factors allowing the construction of the following scores (Appendix 1): emotional IPV (based on three items, eigenvalue = 2.55, Cronbach’s α = 0.871), physical IPV (five items, eigenvalue = 3.72, Cronbach’s α = 0.929), extreme physical IPV (two items, eigenvalue = 1.67, Cronbach’s α = 0.685) and sexual IPV (two items, eigenvalue = 1.62, Cronbach’s α = 0.582). The previous scores were considered highly reliable when Cronbach’s α value was ⩾0.7 and moderately reliable when the value was [0.5; 0.7[.23,24 The IPV scores explained cumulatively 80% of the total variance. A three-class IPV variable (no, occasional and frequent violence) was built using the following IPV score cut-offs: score = 0 (no IPV), score <median among non-zero values (occasional IPV) and score ⩾median among non-zero values (frequent IPV).
Outcomes
ATI was an intermediary outcome in order to verify whether it was associated with one or more IPV scores. ATI ⩾1 month (ATI ⩾ 1m) was classified into three types: recent, that is, ATI ⩾ 1m in the 6 months prior to the survey; former, that is, ATI ⩾ 1m older than 6 months; and none, that is, no ATI > 1m since the beginning of ART.
IPV scores were defined as binary variables (frequent vs occasional or none). Those associated with recent ATI ⩾ 1m were then used as the main outcomes of this study in order to investigate related factors.
Explanatory variables
The following variables were common to both the intermediary and main analyses: age (in four categories defined using quartiles), living setting (rural, urban), educational level (primary, secondary, post-secondary), professionally active (yes, no), monthly household income (in four categories defined using quartiles), having children (yes, no), relationship with the household head (being the household head, spouse or ascending line, descending line or other family relationship or no family relationship), having a main partner (yes, no), experiencing ART stock-outs in the previous 3 months (yes, no), HIV clinical stage at ART initiation (stages 1–4). 25
In addition, the following factors were used in the analysis investigating factors associated with ATI-related IPV: duration of relationship with the main partner (in four categories defined using quartiles), living together (living with the main partner, not living together, no main partner), marital status (legal or customary marriage, common-law couple, no main partner), kind of union (polygamous, monogamous, no main partner), educational level of the main partner (primary, secondary, post-secondary, unknown, no main partner), decision making about how to spend the respondent’s income (the main partner alone, other responses), decision making about the respondent’s health care (the main partner alone, other responses), HIV status disclosure to the most recent intimate partner (yes, no), HIV status of the most recent intimate partner (positive, negative, unknown), age difference between the respondent and her most recent intimate partner (>5 years, ⩽5 years, unknown), having perpetrated non-self-defensive violence against her intimate partner (yes, no) and having experienced stigma (stigma score >0, stigma score = 0 as measured by the HIV Stigma Scale). 26
Statistical analysis
Relationship between IPV and ATI ⩾ 1m
Univariate logistic regressions were performed to evaluate the relationships between ATI ⩾ 1m and (a) the binary variables of emotional, physical, extreme physical and sexual IPV; (b) all the previously listed possible explanatory variables.
Multivariate logistic regression was performed including the binary variables of emotional, physical, extreme physical and sexual IPV, age and variables with a p value of <0.2 in the univariate analysis. Causal mediational analysis was further performed to investigate the influence of mental and physical quality of life (scores calculated by using the SF-12 scale) 27 on the association between recent ATI ⩾ 1m and IPV.
Factors associated with ATI-related IPV
Univariate logistic regression was then performed to investigate the associations of IPV scores previously identified as related to recent ATI ⩾ 1m and the previously cited explanatory variables. Multivariate logistic regression was performed including variables with a p value of <0.2 in the univariate analysis, using the backward stepwise technique. All the statistical analyses were performed using R 3.5.0. 28
Results
Characteristics of participants
Of the 2138 participants in the ANRS-12288 EVOLCam survey, 1387 (65%) were female. Among them, 934 women declared at least one intimate partner in the previous 12 months. Of the latter, 894 (64% of all women included in the survey) had no missing values in the IPV questionnaire section and were therefore included in this study. Median (interquartile range (IQR)) age was 37 (32–42) years (Table 1). A large proportion of women were living in urban areas (80%), were professionally active (63%), and had a secondary educational level (60%). Seventy three percent of participants had experienced HIV-related stigma. Sixteen percent of those included experienced ART stock-out(s) in the 3 months prior to the survey and 28% percent had a detectable VL.
Characteristics of HIV-positive ART-treated Cameroonian women who declared at least one intimate partner in the previous 12 months a (ANRS-122988 EVOLCam survey, n = 894).

ART: antiviral therapy; ATI: antiretroviral therapy interruption; IQR: interquartile range; IPV: intimate partner violence.
With no missing data in the IPV questionnaire.
Alcohol consumption: moderate, ⩽3 big or 6 small bottles of beer (i.e. ⩽195 cL); elevated, ⩾4 big or 7 small bottles of beer (i.e. ⩾230 cL).
In the 12 +months prior to the survey.
ATI ⩾ 1 month: recent, ATI ⩾ 1m = in the 6 months prior to the survey and former, ATI ⩾ 1m = more than 6 months prior to the survey.
Data on women with ART ⩾6 months (n = 852); undetectable VL ⩽100 copies/mL.
A large proportion of women had disclosed their HIV status to their most recent intimate partner (70%), but 39% did not know this partner’s HIV status, while 37% and 24%, respectively, declared that their most recent intimate partner was HIV-negative and HIV-positive.
Description of IPV
The proportions of women reporting occasional or frequent IPV was 29% for emotional IPV, 22% for physical IPV, 13% for extreme physical IPV and 18% for sexual IPV (Table 2). Median scores of emotional IPV, physical IPV, extreme physical IPV and sexual IPV were, respectively, 0.297, 0.288, 0.013 and 0.026 among women who declared IPV. Thirteen percent of participants declared they had perpetrated non-self-defensive physical violence against their most recent intimate partner.
Description of intimate partner violence in HIV-positive ART-treated Cameroonian women who declared at least one intimate partner in the previous 12 months a (ANRS-122988 EVOLCam survey, n = 894).

ART: antiviral therapy; IPV: intimate partner violence; IQR: interquartile range.
With no missing data in the IPV questionnaire.
Median score and IQR calculated for all non-zero scores.
IPV score >0.
IPV score < median IPV scoreb.
IPV score ⩾median IPV scoreb.
IPV score equal to zero.
“Have you ever beaten up, slapped, hit, kicked or physically assaulted your (most recent) intimate partner when he was not assaulting you?”
Association of IPV with recent ATI ⩾1m
For this study, we focused only on IPV association with recent ATI ⩾ 1m, due to the cross-sectional design of the survey. Accordingly, results for previous ATI ⩾ 1m are not shown. The analysis of IPV association with ATI ⩾ 1m was performed on 815 of the 894 WLHIV included in this study, as 5 had missing data on treatment interruption and 74 women were not receiving ART at the time of the study. In the final model, the risk of recent ATI ⩾ 1m was higher in women suffering from frequent physical IPV than in those reporting no or occasional IPV (Table 3; adjusted odds ratio (aOR) = 2.42 (95% confidence interval (CI) = 1.00; 5.87), p = 0.051). Recent ATI ⩾ 1m was reported, by 8%, 6% and 5% of women who reported, respectively, frequent, occasional or no physical violence. Other IPV scores were not associated with recent ATI ⩾ 1m. The other factors also independently associated with recent ATI ⩾ 1m as follows: experience of ART stock-outs in the previous 3 months (2.14 (1.04; 4.41), p = 0.039), women aged 32–37 years (0.19 (0.04; 0.87), p = 0.032), monthly household income per consumption unit >75th percentile, that is, ⩾US$26.8 (4.67 (1.64; 13.29) p = 0.004) and ]50th–75th] percentile, that is, ]US$14.9–US$26.8] (2.77 (0.95; 8.15), p = 0.063). In the causal mediational analysis, no mediation effect of mental and physical quality of life scores was found in the association between frequent physical IPV and recent ATI ⩾ 1m (average causal mediation effects: mental life quality index, coefficient = –0.00175, p = 0.616; physical life quality index, coefficient = –0.000581, p = 0.870; data not shown).
ART: antiviral therapy; ATI: antiretroviral therapy interruption; CI: confidence interval; cu: consumption unit; OR: odds-ratio; aOR: adjusted odds ratio; IPV: intimate partner violence
Recent ATI ⩾ 1 month = ATI ⩾ 1 month in the 6 months prior the survey
Due to five missing values on ATI ⩾1 month and 74 women not receiving ART
Except IPV variables, only associations with p value of <0.2 are presented (variables included in the multivariate analysis)
Only associations with p value of <0.1 are presented
<25th percentile
]25th–50th] percentile
]50th–75th] percentile percentile’ is correct as given in the footnote of Table 3.]
⩾75th percentile
Frequent IPV score above median score (calculated for all non-zero scores)
Frequent IPV score equal to zero
Frequent IPV score below median score (calculated for all non-zero scores)
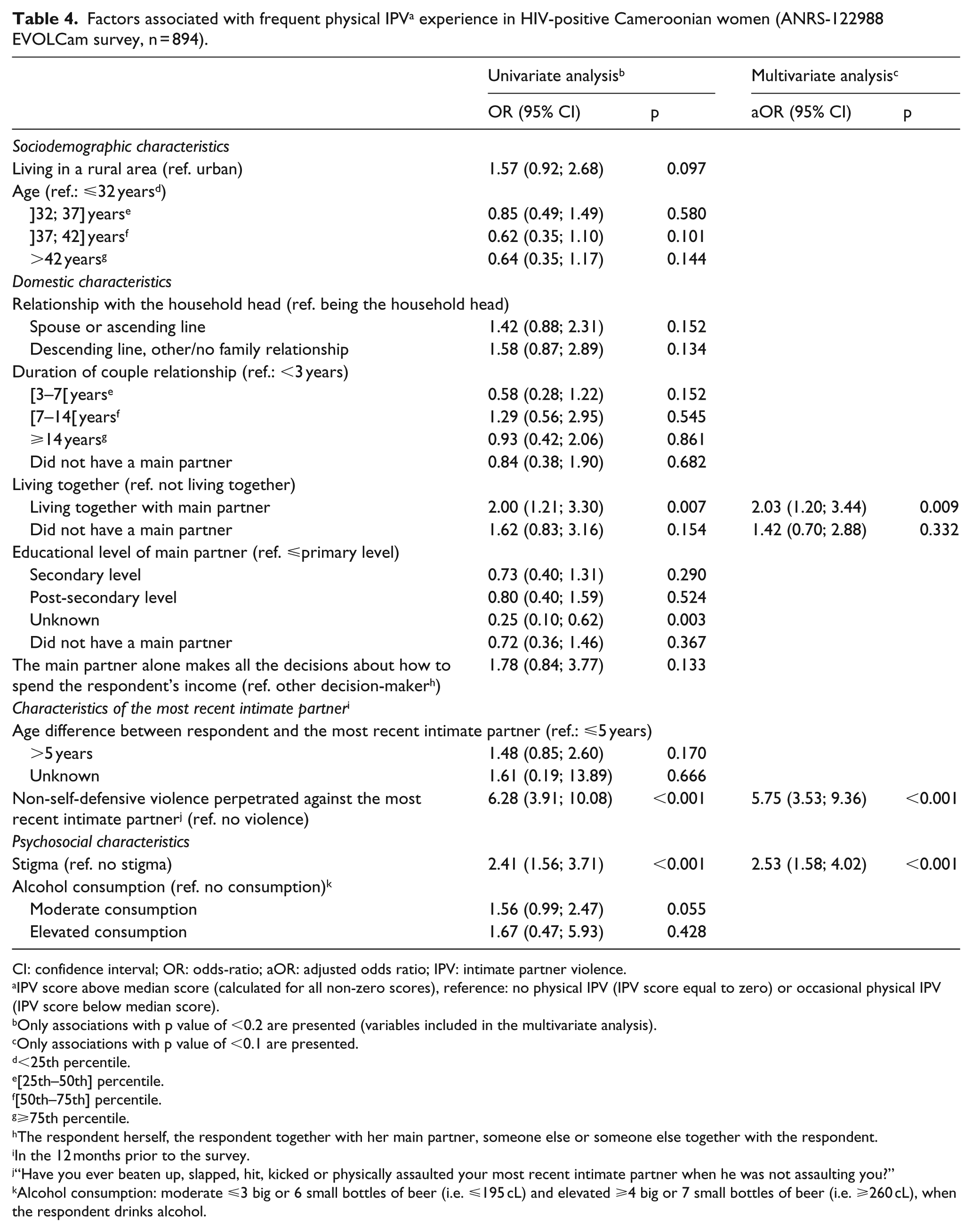
Factors associated with frequent physical IPV
Eight hundred and nighty-four women were included in the analysis of factors associated with frequent physical IPV. In multivariate analysis (Table 4), women living with their main partner (2.03 (1.20; 3.44), p = 0.009), those who experienced HIV-related stigma (2.53 (1.58; 4.02), p < 0.001) and those who reported non-self-defensive violence against their most recent intimate partner (5.75 (3.53; 9.36), p < 0.001) were all significantly more likely to experience frequent physical IPV.
Factors associated with frequent physical IPV a experience in HIV-positive Cameroonian women (ANRS-122988 EVOLCam survey, n = 894).
CI: confidence interval; OR: odds-ratio; aOR: adjusted odds ratio; IPV: intimate partner violence.
IPV score above median score (calculated for all non-zero scores), reference: no physical IPV (IPV score equal to zero) or occasional physical IPV (IPV score below median score).
Only associations with p value of <0.2 are presented (variables included in the multivariate analysis).
Only associations with p value of <0.1 are presented.
<25th percentile.
[25th–50th] percentile.
[50th–75th] percentile.
⩾75th percentile.
The respondent herself, the respondent together with her main partner, someone else or someone else together with the respondent.
In the 12 months prior to the survey.
“Have you ever beaten up, slapped, hit, kicked or physically assaulted your most recent intimate partner when he was not assaulting you?”
Alcohol consumption: moderate ⩽3 big or 6 small bottles of beer (i.e. ⩽195 cL) and elevated ⩾4 big or 7 small bottles of beer (i.e. ⩾260 cL), when the respondent drinks alcohol.
Discussion
High IPV prevalence in Cameroonian WLHIV
IPV is an important concern in HIV-positive women in Cameroon, with one-third of our participants suffering emotional IPV, one-fifth suffering sexual and/or physical IPV, and one-eighth suffering extreme physical IPV. These findings are consistent with results which reported higher exposure to IPV in PLWH than in other related HIV-negative sub-populations, in Sub-Saharan Africa,29,65 and in various settings. 5 However, the prevalence of IPV, especially sexual and emotional IPV, observed in this study, was particularly high compared with other research conducted among WLHIV in Sub-Saharan Africa.29–31 The higher prevalence of recent IPV might reflect the higher prevalence of IPV in the Cameroonian general population than in other countries. 14
In our study population, the prevalence of emotional IPV was similar to that of women in the general population of Cameroon. However, sexual IPV was higher and physical IPV was lower, as indicated by the 2010 demographic and health national survey (respectively, 29%, 22% and 18% in the general population vs 33%, 29% and 11% in our study population (15)). The higher exposure of HIV-positive women to sexual IPV than in the general population of women in Cameroonian might partly explain their HIV acquisition. Indeed, there is growing evidence that IPV, especially sexual IPV, increases HIV exposure and incidence in women.4,32–35 Moreover, abusive partners are more likely to have HIV risky behaviors and sexually transmitted infections (STIs) than non-violent partners, which increases women’s risk of exposure to HIV. 1 However, it is also possible that HIV diagnosis and status disclosure to their intimate partner in our population may have increased their exposure to physical violence.4,12 In this study, two-thirds of women had disclosed their HIV status to their most recent intimate partner, which was their main partner in most cases. However, no statistical association was found between serodisclosure and frequent physical IPV, perhaps because IPV might occur as a result of disclosure, 6 or indeed prevent disclosure, 36 due to a fear of violence.
IPV and ATI
This study highlights an association between frequent physical IPV and prolonged ATI. One study among American WLHIV showed more frequent prolonged healthcare interruptions leading to viral rebound among victims of IPV. 37 Unstructured prolonged ATI has been found to compromise immunological recovery and viral suppression and increase comorbidity, mortality and resistance to antiretroviral treatment.38–40 Increased VL in HIV-positive people who interrupt their ART is a major predictor of the risk of vertical 41 and horizontal transmission of HIV. 42 Because women who are victims of IPV are more likely to interrupt ART for a long period, both their new-born children and HIV-negative intimate partners are at greater risk of HIV infection, as are their abusive partners’ concurrent partners. IPV jeopardizes HIV control at both the individual and collective levels.
Furthermore, the experience of emotional, physical or sexual violence is associated with poor psychosocial outcomes in HIV-positive women,37,43 which are known to be predictors for sub-optimal ART continuity or adherence. 44 Therefore, the effect of violence on linkage to and retention in health care is either direct, because of freedom deprivation, or indirect, by degrading the quality of life or mental health, 45 which in turn affects healthcare use. However, in this study, quality of life was shown to have no mediation effect on the association between frequent physical IPV and recent ATI ⩾ 1m. As suggested in a previous national survey conducted in Cameroon, ATI is more related to exogenous events than to individual decisions. 46 Therefore, in this specific setting, violent partners might directly affect women’s use of health care by controlling them or by limiting how much money they have and how they use it, or by limiting their use of transportation rather than by degrading their mental quality of life.
Factors associated with physical IPV among Cameroonian WLVIH
Stigma was associated with frequent physical IPV in HIV-positive women in this study. Women who suffer HIV-related stigma are more likely to be socially isolated and helpless toward their partners’ violence. Non-defensive violence perpetrated by the women on their partners, as well as cohabitation with their partners, were identified as risk factors for frequent physical IPV in this study, which is consistent with findings of a multi-country study conducted by the WHO. 47 Qualitative research investigated how cohabitation with partners is linked to IPV and poor-related health outcomes among HIV-positive women, through sexual slavery, confinement and healthcare access control and dissuasion. 45 Aside from these factors, no educational or socioeconomic characteristic of the women or their partners was associated with recent physical IPV in this study, which is consistent with observations made in the general population in Cameroon. 15
Further research in Cameroon should investigate the health effect of domestic violence in HIV-positive women and men in greater detail. Domestic violence is not limited to violence perpetrated on women by their partners, but includes emotional and physical violence inflicted by co-wives in polygamous unions (which accounted for 13% of our study sample), or by in-laws. 48 Violence perpetrated by women on their partners, reported by 13% of our study sample, is related to violence they suffer and may also have an adverse impact on their male partner’s health and HIV-related outcomes. 48
Addressing IPV in HIV-positive women
When we consider the large proportion of women who are victims of IPV, the absence of demographic or socioeconomic criteria to identify those at risk of IPV, and the fact that in Cameroon, most physical and sexual IPV starts in the early years of a relationship,15,16 we see that a major proportion of women followed up in HIV services are at risk of IPV. This fact justifies the need for systematic screening for IPV in these patients.
The most promising interventions to concomitantly address HIV and IPV in low-income countries are those which combine economic strengthening and gender-transformative interventions 49 and involve men. 50 However, these are mostly designed to prevent HIV acquisition by women in contexts where IPV is widespread51–53 as opposed to help women already infected. Data on interventions to reduce IPV endured by HIV-positive African women and related poor outcomes are scarce. Skills and practices of workers in HIV services, as well as their regular contact and lifelong relationship with patients should be used to address IPV-related effects. Peers strongly recommend integrating universal screening for IPV in mother-to-child HIV transmission prevention programs, and in outreach for testing and ART programs, in order to achieve viral suppression in WLHIV. 54 IPV appears to be less spontaneously discussed by women in health services than other intimate issues such as condom and contraceptive use. 31 This suggests that healthcare providers need to raise the IPV subject instead of waiting for their patient to do so, and to screen for IPV by searching for identifying features (e.g. wounds and traces of punches). 12 A previous study investigated how acceptable it was to women to be asked about their experiences of IPV in counseling and testing services. 55 A few interventions to facilitate safe HIV serostatus disclosure options and sex negotiation in HIV-positive women who experience or fear IPV have been implemented, but with limited evidence for their success. 56
Consequently, we recommend implementing universal counseling and screening for IPV (endured or mutual) and other forms of domestic violence, in both male and female patients, in healthcare services dedicated to testing, post-testing and ART provision, in accordance with the guidelines from the WHO 2006 report 57 and 2013 programming tool. 58 We also recommend that same-gender counselors be used where possible. 59 Implementing and evaluating the efficiency of such healthcare provider-led care interventions to improve ART continuity, viral suppression, HIV transmission and other health outcomes in the context of IPV is a currently unexplored opportunity for research. 60
Strengths and limitations of the study
The study did not include women attending HIV services in areas other than Center and Littoral regions, and more importantly, it did not include HIV-positive women not attending HIV services, a sub-population believed to be at greater vulnerability. Accordingly, our results are not representative of the overall situation of WLHIV in Cameroon. However, the study is representative of HIV-positive women receiving ART within the Cameroonian ART program in the two regions cited. Furthermore, the study’s cross-sectional design did not allow us to establish causal mechanisms. In addition, our study relied on self-reported IPV, which is a sensitive issue, possibly inducing declarative bias. However, the WHO questionnaire used to collect IPV data has been validated and is considered highly reliable in discriminating different forms of violence in different social contexts. 59 PCA has already been used by other researchers to construct scores of IPV using a large number of items reporting IPV in face-to-face questionnaires. 60 To our knowledge, this is the first study to investigate IPV suffered by ART-treated HIV-positive Cameroonian women and its associated factors, as well as the association between IPV and prolonged ATIs among HIV-positive women in Cameroon.
Conclusion
The high proportion of women attending HIV services in the Center and Littoral regions of Cameroon who suffer from various forms of IPV is a great cause for concern. While sexual IPV may have contributed to their HIV acquisition, physical IPV increases the risk of prolonged ATIs, which may degrade health outcomes, increase vulnerability and negatively affect both the control of HIV and the HIV treatment cascade. In the context of the interconnection between the IPV and HIV epidemics in Cameroon, screening for IPV and providing counseling in HIV services offer the opportunity to identify patients at risk of poor health outcomes and to mitigate the effects of IPV not only on themselves but on their partners, their children and on public health. Combined with measures to address supply-related barriers to ART continuity, identified in the ANRS-12288 EVOLCam survey, 20 these efforts could both consolidate the important progress which Cameroon has already made in engagement in HIV care since the adoption in 2007 of a national policy of free access to ART63,64 and improve the treatment cascade.
Footnotes
Appendix
Frequency of IPV items and detailed results of principal components analysis used to construct IPV scores from IPV items.
| IPV items |
IPV indicators |
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| In the past 12 months, has your most recent partner ever … | Never | Sometimes | Often | Emotional IPV score | Physical IPV score | Extreme physical IPV score | Sexual IPV score | |||
| n | % | n | % | n | % | Ranking of IPV items in constructing IPV indicators according to weighting from PCA | ||||
| said or done something to humiliate you in front of other people? | 752 | 84.1 | 80 | 8.9 | 62 | 6.9 |
|
0.02 | −0.02 | −0.03 |
| insulted or belittled you? | 718 | 80.3 | 102 | 11.4 | 74 | 8.3 |
|
0.04 | −0.05 | −0.05 |
| threatened to hurt you or someone you care about? | 773 | 86.5 | 64 | 7.2 | 57 | 6.4 |
|
−0.04 | 0.12 | 0.06 |
| hit you with his fist or with something else that could hurt you? | 820 | 91.7 | 45 | 5 | 29 | 3.2 | −0.03 |
|
0.04 | −0.08 |
| twisted your arm or pulled your hair? | 820 | 91.7 | 40 | 4.5 | 34 | 3.8 | −0.09 |
|
−0.05 | 0.18 |
| slapped you? | 770 | 86.1 | 77 | 8.6 | 47 | 5.3 | 0.14 |
|
−0.13 | 0.01 |
| kicked you, dragged you or beaten you up? | 834 | 93.3 | 34 | 3.8 | 26 | 2.9 | −0.07 |
|
0.22 | −0.16 |
| pushed you or shoved you or thrown something at you? | 790 | 88.4 | 60 | 6.7 | 44 | 4.9 | 0.18 |
|
−0.07 | 0.14 |
| threatened to use or did in fact use a gun, knife, or other weapon against you? | 883 | 98.8 | 5 | 0.6 | 6 | 0.7 | 0.02 | −0.14 |
|
0.13 |
| tried to or did in fact choke you or burn you on purpose? | 869 | 97.2 | 10 | 1.1 | 15 | 1.7 | 0.02 | 0.24 |
|
−0.09 |
| physically forced you to have sexual intercourse even when you did not want to? | 838 | 93.7 | 32 | 3.6 | 24 | 2.7 | 0.07 | 0.03 | 0.09 |
|
| forced you to practice any other sexual act that you did not want to? | 877 | 98.1 | 8 | 0.9 | 9 | 1 | −0.07 | 0.01 | 0 |
|
| Eigen values | 2.55 | 3.72 | 1.67 | 1.62 | ||||||
| Explained variance | 21.3% | 31.0% | 13.9% | 13.5% | ||||||
| Cumulative variance | 79.6% | |||||||||
| Cronbach’s α | 0.871 | 0.929 | 0.685 | 0.582 | ||||||
| Correlation with other IPV score | ||||||||||
| Emotional IPV score | − | 0.610 | 0.382 | 0.396 | ||||||
| Physical IPV score | − | − | 0.540 | 0.360 | ||||||
| Extreme physical IPV score | − | − | − | 0.286 | ||||||
IPV: intimate partner violence; PCA: principal component analysis.
Bold values indicate that the IPV item highly contributes to the construction of the IPV score (weighting >0.7)
Acknowledgements
We thank all the female participants and all the staff from the participating HIV centers who agreed to take part in the survey. We also thank Jude Sweeney for revising and editing the English version of the manuscript. The members of the EVOLCam study group are as follows: C. Kuaban, L. Vidal (principal investigators); G. Maradan, A. Ambani, O. Ndalle, P. Momo, C. Tong (field coordination team); S. Boyer, V. Boyer, L. March, M. Mora, L. Sagaon-Teyssier, M. de Sèze, B. Spire, M. Suzan-Monti (UMR912—SESTIM); C. Laurent, F. Liégeois, E. Delaporte, V. Boyer, S. Eymard Duvernay (TransVIHMI); F. Chabrol, E. Kouakam, O. Ossanga, H. Essama Owona, C. Biloa, M.T. Mengue (UCAC); E. MpoudiNgolé (CREMER); P.J. Fouda, C. Kouanfack, H. Abessolo, N. Noumssi, M. Defo, H. Meli (Hôpital Central, Yaoundé); Z. Nanga, Y. Perfura, M. Ngo Tonye, O. Kouambo, U. Olinga, E Soh (Hôpital Jamot, Yaoundé); C. Ejangue, E. Njom Nlend, A. Simo Ndongo (Hôpital de la Caisse, Yaoundé); E Abeng Mbozo’o, M. Mpoudi Ngole, N. Manga, C. Danwe, L. Ayangma, B. Taman (Hôpital Militaire, Yaoundé); E.C. Njitoyap Ndam, B. Fangam Molu, J. Meli, H. Hadja, J. Lindou (Hôpital Général, Yaoundé); J.M. Bob Oyono, S. Beke (Hôpital Djoungolo, Yaoundé); D. Eloundou, G. Touko, (District Hospital, Sa’a); J.J. Ze, M. Fokoua, L. Ngum, C. Ewolo, C. Bondze (District Hospital, Obala); J.D. Ngan Bilong, D. S. Maninzou, A. Nono Toche (Hôpital St Luc, Mbalmayo); M. Tsoungi Akoa, P. Ateba, S. Abia (District Hospital, Mbalmayo); A. Guterrez, R. Garcia, P. Thumerel (Catholic Health Center, Bikop); E. Belley Priso, Y Mapoure, A. Malongue, A.P. Meledie Ndjong, B. Mbatchou, J. Hachu, S. Ngwane (Hôpital Général, Douala); J. Dissongo, M. Mbangue, Ida Penda, H. Mossi, G. Tchatchoua, Yoyo Ngongang, C. Nouboue, I. Wandji, L. Ndalle, J. Djene (Hôpital Laquintinie, Douala); M.J. Gomez, A. Mafuta, M. Mgantcha (Catholic Hospital St Albert Legrand, Douala); E.H. Moby, M.C. Kuitcheu, A.L. Mawe, Ngam Engonwei (District Hospital, Bonassama); L.J. Bitang, M. Ndam, R.B. Pallawo, Issiakou Adamou, G. Temgoua (District Hospital, Deido); C. Ndjie Essaga, C. Tchimou, A. Yeffou, I. Ngo, H. Fokam, H. Nyemb (District Hospital, Nylon); L.R. Njock, S. Omgnesseck, E. Kamto, B. Takou (District Hospital, Edea); L.JG Buffeteau, F. Ndoumbe, JD Noah, I. Seyep (Hôpital St Jean de Malte, Njombe).
Author contributions
M.S.-M., M.-T.M., L.V., C.K., L.M., C.L., B.S., S.B. and M.F. contributed to the concept and design of the work and acquisition of data. K.N., M.F. and L.S.-T. performed the analysis of data. M.F. drafted the paper. M.S.-M., B.S., S.B. and L.S.-T. contributed to revision of the paper.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by the French National Agency for Research on AIDS and viral hepatitis (ANRS).

