Abstract
Abdominal pregnancy is a rare form of ectopic pregnancy associated with increased maternal and perinatal risks. In low-resource countries, diagnostic challenges complicate management. Prompt diagnosis requires a high level of suspicion. We report a 34-year-old, G3P1A+1SA, referred as a suspected abdominal pregnancy. Despite several ultrasounds and clinical signs, the diagnosis was delayed until 29 weeks’ gestation with a live fetus. She was managed conservatively. A live infant was successfully extracted via laparotomy at 32 weeks of gestation. This case highlights diagnostic delays and difficulties in abdominal pregnancy, even as ultrasound use increases in resource-limited settings.
Introduction
Abdominal pregnancy is an uncommon obstetric complication with a high maternal and perinatal mortality.1–4 The majority of obstetrician-gynecologists are not likely to encounter an abdominal pregnancy during their professional careers. Pre-operatively, it is difficult to diagnose and poses a critical challenge to physicians. 5 Abdominal pregnancies are predominantly observed in developing countries, largely due to the high prevalence of genito-pelvic infections. 2 This emphasizes the importance of maintaining a high index of suspicion to improve early detection and reduce adverse complications.
This article presents a case of advanced abdominal pregnancy at 32 weeks’ gestation. The patient had a history of multiple uterine evacuations. Repeated obstetric ultrasounds showed an intrauterine pregnancy. The case was managed at the Obstetrics and Gynecology Department of Kwadaso SDA Hospital in Kumasi, Ghana. This is the first reported case of abdominal pregnancy managed in our facility. The objective is to report this rare case and provide a brief review of relevant literature.
Case summary
A 33-year-old woman, gravida 4 para 1, with 2 spontaneous miscarriages. She was referred to the Obstetrics and Gynecological department of the Kwadaso-SDA hospital, Kumasi, Ghana, from a sub-district hospital on account of an intra-abdominal pregnancy at 29 weeks and 3 days for further management. She had no complaints at presentation. There was no history of pain, no bleeding per vaginam, no loss of amniotic fluid, but she could perceive fetal movements. She booked for an antenatal visit at 7 weeks and 6 days and had attended six (6) antenatal visits at the time of referral. All antenatal laboratory tests done were essentially normal. She had two obstetric scans at 7 weeks and 3 days and at 12 weeks and 6 days (Figure 1). She admitted to an episode of postcoital bleed at 12weeks for which she was reassured and given hematinic. She had a live delivery four (4) years ago and two spontaneous miscarriages about four (4) and two (2) years ago, respectively. Her last miscarriage resulted in three evacuation procedures of the uterus with suction curettage. She had no other significant surgical or medical history. (a)Ultrasound at 7w+3d showing a gestational sac outside the uterus, (b) ultrasound at 12w+6d showing absent surrounding myometrial wall.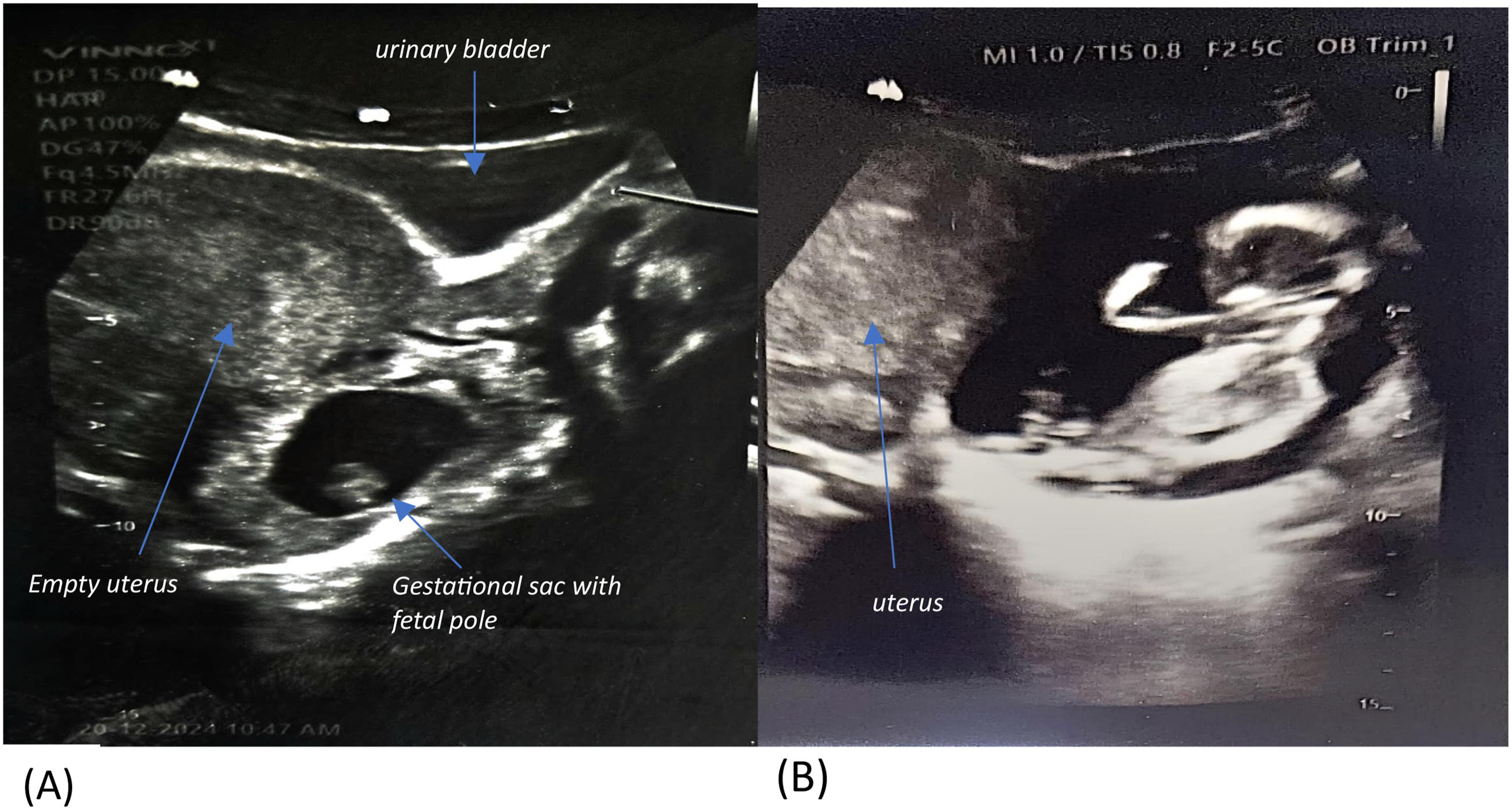
She was admitted to our facility on 29th May, 2025. Her vitals on admission were: blood pressure 105/58 mmHg, pulse rate 93 bpm, oxygen saturation 98%. Normal cardiovascular and respiratory system findings. Examination of the abdomen revealed a gravid uterus with symphysiofundal height of about 26cm, mild tenderness in the right iliac fossa, and easily palpable fetal parts. The fetal lie was transverse with the head to the maternal left. The fetal heart rate was 138bpm. An urgent ultrasound was requested, which confirmed an intra-abdominal pregnancy with fetal cardiac activity at 29 weeks and 4 days. Estimated fetal weight of 1397g. The placenta adhered to the posterior wall of the uterus. The uterus was bulky with normal echo-texture and an empty uterine cavity. There were also multiple well-defined anechoic hepatic cystic lesions distributed throughout the segments on the maternal liver, the largest measuring 4.4x4.2cm.
Patient was counseled on the diagnosis, its implications, and prognosis. She was admitted and managed expectantly, with a planned delivery between 32 and 34 weeks. Corticosteroids were given for fetal lung maturity. Antepartum fetal surveillance was assessed with daily fetal heart rate checks with a handheld Doppler, daily fetal kick counts to 10, twice-weekly biophysical profile, and Doppler velocity. Hemoglobin was optimized to 12.6 g/dL; 2 units of whole blood were secured. Her blood group was O positive. Hepatitis B and HIV screening were negative. Renal and liver function tests were normal. Planned cesarean delivery occurred due to reduced fetal movements and low amniotic fluid volume. A multidisciplinary team of an obstetrician, general surgeon, and pediatrician was involved.
Laparotomy was done under general anesthesia using a midline incision. Intra-operatively, we found a live fetus in a transverse lie, dorso-anterior, within an intact amniotic membrane, in the abdominal cavity (Figure 2(a)). The uterus was hyperemic at about 12-14 weeks, with a normal vesicouterine pouch, normal-looking fallopian tubes and ovaries bilaterally, and normal bowel loops and omentum (Figure 2(b)). A live neonate was extracted with an APGAR score of 2/10 and 5/10 in the 1st and 5th minutes, respectively (Figure 3(a)). Immediate and successful resuscitation was performed by the pediatrician, and the baby was sent to the Mother-and-Baby Unit for monitoring. Baby findings are as follows: sex-male; weight- 1.6kg; Fetal Length - 35cm; Head circumference - 30cm; pulse- 148bpm; Respiratory Rate- 64 bpm; temperature- 34.0 °C; RBS- 2.8mmol/l. The baby had no gross abnormalities (Figure 3(b)). The placenta was morbidly adherent to the posterior lower uterine wall, extending to the pouch of Douglas and the upper one-third of the vagina. There was no hemoperitoneum and no placenta infiltration into the surrounding structures. However, there were multiple cystic lesions on the liver capsule, measuring approximately 0.5-1cm (Figure 4(b)). (a)fetus in an intact amniotic sac in the abdominal cavity, (b) uterus with normal looking fallopian tubes and ovary. (a) Delivery of baby, (b) healthy baby. (a) posterior uterine wall. Placental bed showing uterine wall weakness, (b) liver with multiple cystic lesions. Normal looking bowel and omentum.


Placental removal was complicated by significant hemorrhage. The bleeding was controlled with suture ligation of the vessels. An estimated 800 mL of blood loss was recorded. Gross examination revealed a focal area of myometrial weakness and loss of structural integrity at the placental attachment site (Figure 4(a)). Intraoperative management included transfusion of one unit of whole blood. The patient’s immediate postoperative course was unremarkable, and she remained hemodynamically stable. A second unit of blood was transfused postoperatively, and broad-spectrum antibiotics were started.
On postoperative day 9, the patient had to undergo a relaparotomy for the drainage of a large volume of abscess in the pouch of Douglas. Wound swab culture isolated Klebsiella species. Therapy was transitioned to culture-directed antibiotics. The patient and the neonate were discharged after a 3-week hospital stay. Post-discharge follow-up was unremarkable.
The pediatrician examined a healthy baby weighing 3.6 kg with no abnormalities. The mother’s wound had healed completely and without complications (Figure 5). Abdominopelvic ultrasound revealed normal pelvic findings but showed hepatomegaly with multiple simple cysts, the largest measuring 5.3 x 4.6 cm. No free fluid or intra-abdominal masses were seen. Liver function test was unremarkable. (a) Intra-abdominal abscess with wound dehiscence, (b) a month after re-laparotomy and secondary wound closure.
Histology findings on the placenta revealed a placental weight of 300g with mature villi and focal areas of infarction. Maternal decidua showed areas of infarction (45% of placental volume). Fetal membranes and umbilical cord were essentially normal. (Figure 6(b)). (a) Placental fetal surface showing vessels, umbilical cord, membranes and chorion, (b) placental maternal surface showing disrupted cotyledons.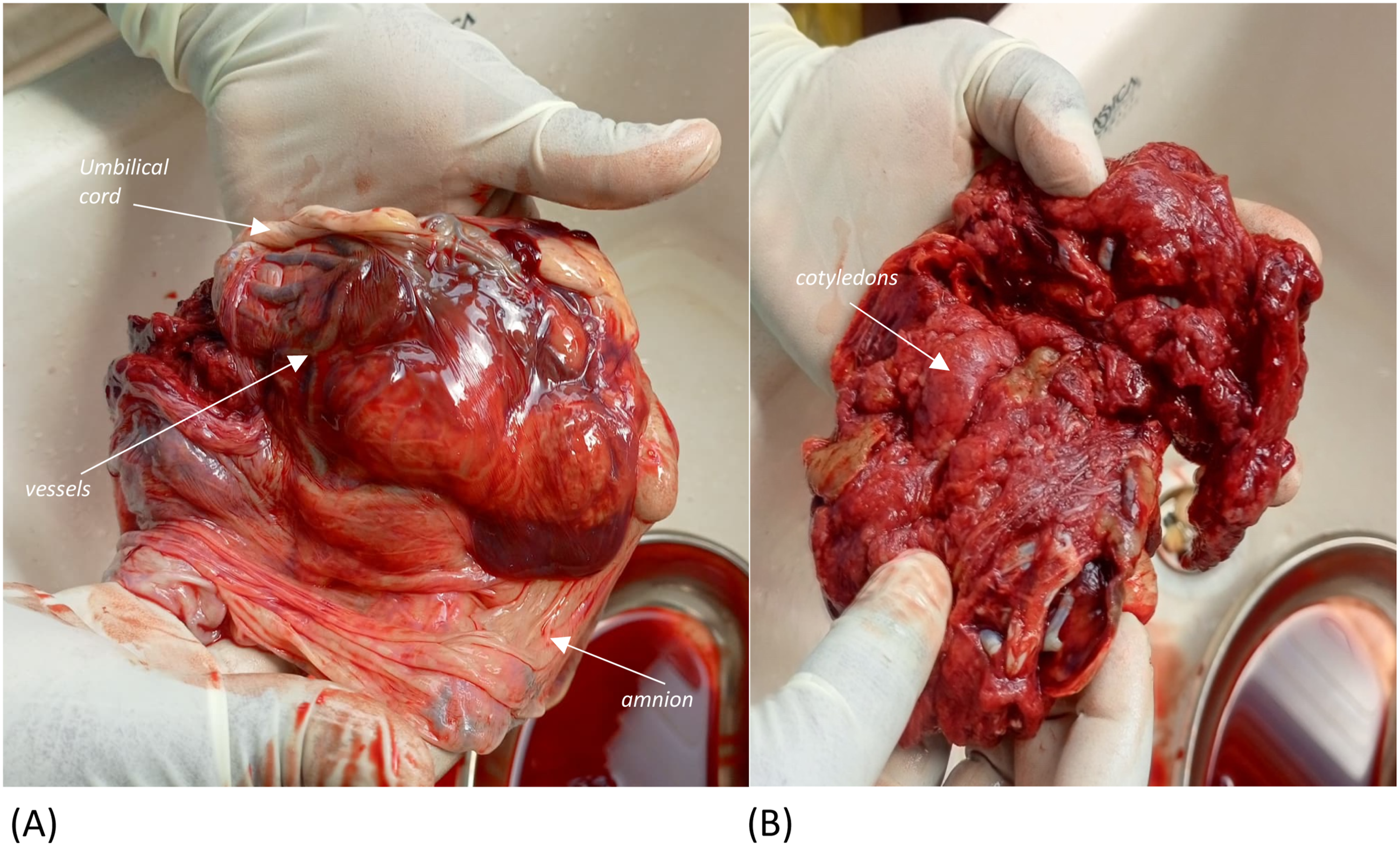
Discussion
Abdominal pregnancy is a rare condition. The earliest descriptions of abdominal pregnancies were recorded nearly 1000 years ago; however, it was first documented during an autopsy in 1701, and since then, several cases have been reported globally. 6 It is a type of ectopic pregnancy in which the fetus develops in the peritoneal cavity.1,4,5 It is a fatal condition that requires a high index of suspicion and swift intervention to prevent adverse obstetric outcomes.
It is classified as primary or secondary based on its pathophysiological mechanisms. 2 Primary abdominal pregnancy is rare, and it occurs when a fertilized ovum implants directly into the abdominal cavity.2,4,7 However, secondary abdominal pregnancy occurs when a fertilized ovum is reimplanted in the peritoneal cavity due to tubal rupture, fimbrial abortion, ruptured uterine or cornual pregnancy, artificial insemination, or the migration of an intrauterine pregnancy through a uterine perforation, hysterectomy breach, or a rudimentary horn.1,2,7 The majority of abdominal cases are secondary. We believe that the mechanism in our index case is a secondary abdominal pregnancy due to its inability to satisfy all of Studdiford’s criteria, notably intact fallopian tubes and ovaries, the absence of a uteroplacental fistula, and the pregnancy’s exclusive relation to the peritoneal surface. 8 Our patient’s prior history of uterine evacuation likely contributed to anatomical distortion and an inflammatory environment, creating conditions favorable for ectopic implantation and increasing the suspicion of secondary abdominal pregnancy.
An advanced abdominal pregnancy (AAP) is defined as a pregnancy that progresses beyond 20 weeks of gestation, with a fetus that is either alive or has shown evidence of having once lived and developed within the maternal abdominal cavity.9,10 These advanced forms are more commonly observed in regions with limited access to healthcare and few medical professionals. 2 The index case, therefore, is a secondary advanced abdominal pregnancy in which the patient was repeatedly misdiagnosed despite repeated obstetric ultrasound scans from a remote clinic.
Abdominal pregnancy represents about 1% of ectopic pregnancies.1,7 In Africa, the prevalence of abdominal pregnancy varies by country, with reported rates of 1 in 1134 in South Africa, 1 in 2583 in Dakar, and 1 in 3750 deliveries in Libreville, Gabon. 11 These figures are notably higher than those reported in developed countries, where the incidence ranges from 1 in 10,000 to 1 in 30,000 deliveries. 12 In a two-year study conducted at Komfo Anokye Teaching Hospital in Ghana, the incidence of advanced abdominal pregnancy was found to be 1 in 1,320 deliveries. Additionally, the ratio of advanced abdominal pregnancy to ectopic pregnancy was reported as 1 in 43.7. 13 The variability in the incidence of abdominal pregnancy is largely attributed to the delayed or missed diagnosis of early forms in developing countries, often resulting from low socioeconomic status and limited utilization of antenatal care services.1,2
It can remain undetected until an advanced gestational age, complicating subsequent management. 14 This was evident in our case, where the condition was not identified until 29 weeks of gestation.
Diagnosing abdominal pregnancy is often missed, with about 45% of cases diagnosed during the antenatal period. 9 This is because most patients have no or non-specific symptoms, making early detection difficult. 14 In certain cases, diagnosis is made intraoperatively. 5 Diagnostic error is about 50-60%.7,15 As a result, there are no standard protocols for diagnosis or treatment. This emphasizes the need for a high index of suspicion and the use of appropriate diagnostic tools. A thorough clinical and ultrasound examination is crucial for detecting abdominal pregnancy. 16
In literature, frequently reported clinical symptoms of uncomplicated abdominal pregnancy include abdominal or suprapubic pain (100%), gastrointestinal symptoms such as nausea and vomiting (70%), painful fetal movements (40%), general malaise (40%), altered bowel habits, bloody vaginal discharge, and no delay in menses. 17 Even though she reported an episode of bleeding per vaginam following sexual intercourse, our patient did not exhibit clear symptoms that could have led to a more focused and frequent evaluation and thus, early diagnosis. Also, the most common physical findings reported are abdominal tenderness (100%), easily palpable fetal parts, an abnormal fetal lie (70%), and a displaced uterine cervix (40%). 17 Other findings may include fetal growth restriction, absent fetal heart sound, failed medical termination or induction of labor, or absence of labor at term. 18 The index case on physical examination showed a transverse lie, minimal abdominal tenderness, and a firm, long, posteriorly displaced uterine cervix.
The first-line diagnostic modality is ultrasound. It is dependent on the quality of the ultrasound and the operator’s skills. 10 In using ultrasound, abdominal pregnancies can be missed in 50% of cases in the second and third trimesters. 19 Our patient had three obstetric ultrasound scans before referral to our facility, and none indicated the possibility of an abdominal pregnancy for prompt diagnosis and timely intervention. This may be due to the operator’s inexperience in poorly resourced centers.
Based on review of literature, the key sonographic features of an intrabdominal pregnancy may include visualizing a fetus in a gestational sac outside the uterus; identifying an abdominal or pelvic mass separate from the fetus; an absent myometrial wall between the fetus and the urinary bladder; recognizing a close proximity of the fetus to the maternal abdominal wall; and locating the placenta outside the uterine cavity. 20 This index case showed similar ultrasound findings with a fetus in a transverse lie associated with oligohydramnios, and a placenta implanted at the posterior uterine wall. Further imaging modalities, such as MRI or CT scan, can be used when the diagnosis is in doubt or suboptimal to determine the limits of the placental insertion and its relation to other viscera.2,8,21 But in our context, an MRI/CT scan was not requested due to financial constraints.
Maternal and perinatal outcomes are determined by early diagnosis, immediate laparotomy, surgical competence, and availability of adequate facility resources. 18 In our case, the team included an obstetrician-gynecologist, a pediatrician, a general surgeon, an anesthetist, and transfusion services to improve the maternal and fetal outcomes. In advanced centers, the team may also include a radiologist, intensivist, pathologist, specialized nursing staff, and counselors. In general principle, laparotomy is recommended once the diagnosis of abdominal pregnancy is made, given the unpredictable and severe maternal adverse outcomes.2,8
Conservative management is seldom appropriate because it carries a very high risk of life-threatening intraperitoneal bleeding. However, in certain cases, a conservative approach may be recommended up to 34 weeks for fetal lung maturation, after which laparotomy is performed. 22 Other studies propose planned delivery between 32 and 35 weeks.1,4,8 Conservative management is considered when there are no congenital anomalies, the fetus is alive, adequate feto-maternal monitoring, well-equipped and adequate staffing, and the placenta is implanted away from the liver or spleen. 5
We managed our case expectantly, promoting fetal lung maturation with steroids and improving survival in the NICU after delivery in a low-resource setting. A successful caesarian delivery was done at 32 weeks’ gestation due to reduced fetal movements and reduced amniotic fluid volume.
Management of the placenta is the most contentious issue of abdominal pregnancy. The placenta may infiltrate several organs, such as the fallopian tubes and ovaries. Other reported sites are the omentum, liver, spleen, and the diaphragm.9,23–25 The optimal management of the placenta remains uncertain, as various methods have been described in the literature. The choice should take into account the risk of hemorrhage and injury to invaded structures.
Often, it is preferable to leave the placenta in situ for natural regression if it is not easily removable. 26 This choice is best in cases of extensive adherence to the bowels and with a high risk of hemorrhage. Placenta may remain functional for fifty (50) days until total regression is completed in four (4) months4,5 and monitored by checking human chorionic gonadotropin levels. The use of methotrexate to accelerate placental regression is still controversial, as this can increase the risk of peritonitis, abscess formation, intestinal obstruction, the need for surgical re-intervention, and prolonged hospitalization. 22 Other adjuvant therapies are arterial embolization and mifepristone administration. 22 In our case, laparotomy revealed placental attachment to the lower half of the posterior uterine wall extending into the pouch of Douglas without bowel or major vessel involvement. The placenta was successfully removed; however, there was excessive hemorrhage that necessitated intraoperative blood transfusion. Fortunately, the bleeding vessels were identified and ligated. The patient’s immediate postoperative course was satisfactory. Nevertheless, our patient developed an intra-abdominal abscess accompanied by wound dehiscence, necessitating surgical re-intervention. We believe this complication may be attributable to either remnants of placental tissues or the presence of gestational material within the peritoneal cavity as a foreign entity. In addition, the hepatomegaly diagnosed on ultrasound might be a postoperative complication from the intrabdominal abscess. However, the liver function test was unremarkable.
Recent studies have reported 12% maternal mortality, 80% maternal haemorrhage requiring blood transfusion, 72% fetal mortality, and 20 to 40% fetal morbidity and anomalies. 1 Other reported causes of maternal death include toxemia, pulmonary embolism, coagulopathy, and infection. 27 There are reports of fetal malformations as high as 40% associated with abdominal pregnancies, and only 50% of these babies survive up to one week postpartum.26,28 Most frequent neonatal complications reported are torticollis, flattening of the head, facial or cranial asymmetry, thoracic malformations, limb defects, joint abnormalities, or central nervous system malformations related to pressure effects and oligohydramnios.17,18,29 Our newborn baby was evaluated by the pediatrician at birth, who confirmed the absence of any gross congenital anomalies.
Conclusion
An advanced abdominal pregnancy resulting in a healthy newborn is very uncommon. Accurate diagnosis remains difficult despite advances in imaging technologies. This emphasizes the need to train personnel to improve diagnostic techniques. This case highlights the importance of thorough early clinical assessment and comprehensive ultrasound, particularly in settings with limited resources. A high index of suspicion and timely surgical intervention are essential for improving maternal and fetal outcomes in Sub-Saharan Africa, where the burden of advanced abdominal pregnancy is heightened by a lack of adequate healthcare infrastructure.
Supplemental material
Supplemental material - Expectant management of advanced abdominal pregnancy with successful outcome: A case report and literature review
Supplemental material for Expectant management of advanced abdominal pregnancy with successful outcome: A case report and literature review by Opoku-Achampong Emmanuel, Randolph Baah Adu, Rdeini Mohammed Walid, Agyemang Charles Osei Tutu, Asiedu Amissah Albert, Abdul-Majeed Fadila, Gyedu Linda in Women’s Health.
Footnotes
Author note
The reporting of this study conforms to the CARE statement (CARE checklist 2013).
Acknowledgements
The authors wish to thank all the staff of the Departments of Obstetrics and Gynaecology, Child Health, and Surgery, as well as the theatre team, for their immense support in the care of this patient.
Consent to participate
Patient approved and provided written informed consent for participation in the manuscript.
Consent for publication
Written informed consent and associated images were obtained from the patient for publication of manuscript.
Author contributions
OAE, RBA, AMF, and AAA were responsible for clinical management. Surgery was performed by OAE, RBA, RMN, and GL. Neonatal care was provided by COTA. Conceptualization, manuscript preparation, and revisions were carried out by OAE, RBA, and GL. All authors have read and approved the final manuscript submitted.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
Available upon request to the corresponding author.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
