Abstract
Background:
Endometrial hyperplasia, a precursor to endometrial cancer, is increasing due to rising obesity and lifestyle risks. Scarce data exist on the molecular characterization of endometrial hyperplasia for prognostic and preventive applications,
Objectives:
This study investigates the expression of core pluripotency stem cell markers in endometrial hyperplasia, given their emerging role in cancer stem cell regulation in various cancers, including endometrial cancer,
Design:
This is a case–control study utilizing archival endometrial tissue blocks. In this study, we assessed the immunohistochemical expression of NANOG, OCT4, and SOX2 in hyperplastic endometrium compared to benign non-hyperplastic endometrium obtained from women with abnormal uterine bleeding,
Results:
The investigation included 165 patients (83 cases with endometrial hyperplasia and 82 controls). The prevalence of endometrial hyperplasia in our cohort was 20.8% (of 500 patients). NANOG showed moderate to high expression in 60.2% of endometrial hyperplasia cases, significantly higher than in controls (19.5%), p < 0.0005. OCT4 showed moderate/high expression in 12% of endometrial hyperplasia cases compared to 3.7% of controls (p = 0.045). In comparison, SOX2 was expressed in 18% of endometrial hyperplasia with no significant difference from controls (19.7%), p = 0.823. Logistic regression showed that age and NANOG expression were significant predictors of endometrial hyperplasia risk, with women exhibiting moderate/high NANOG expression having an 11-fold increased risk of endometrial hyperplasia (95% confidence interval of odds ratio: 3.484–35.864, p < 0.001). Using receiver operating characteristic curves, women’s age predicted SOX2 expression in endometrial hyperplasia cases (area under the curve (AUC) = 0.867, cutoff = 49 years, Youden’s index = 0.722, sensitivity = 100%, specificity = 72.2%, p < 0.001), and NANOG in controls (AUC = 0.734, cutoff = 42 years, Youden’s index = 0.37, sensitivity = 68.8%, specificity = 68.2%, p = 0.004).
Conclusion:
NANOG shows promise for a potential predictive role in patients with endometrial hyperplasia, with moderate/high expression indicating a higher risk. SOX2’s role was unclear but age-related. Further studies are needed to validate these findings and explore hormonal links.
Plain language summary
Findings suggest a potential role for these markers, particularly NANOG, in endometrial hyperplasia. NANOG may have prognostic value in stratifying risk for women with abnormal uterine bleeding, pending further clinical validation. Prospective studies are needed to confirm these findings and assess clinical translation. The pluripotency stem cell markers have been studied in several gynecologic and other cancers, but limited data exist on their expression in pre-cancerous lesions. This study examines the expression of core pluripotency markers (OCT4, SOX2, NANOG) in endometrial hyperplasia and non-hyperplastic endometrium from patients with abnormal uterine bleeding.
Introduction
Endometrial hyperplasia is a precursor of endometrial cancer, which has seen a steady global increase in incidence. In most industrialized countries, endometrial cancer is the most commonly diagnosed gynecologic cancer, with incidence rates rising significantly in recent decades across different regions.1–5 A similar trend is being observed in countries with recent rapid socioeconomic growth.6,7 Despite limited data available on the true incidence of endometrial hyperplasia, it is expected to increase in parallel with the rising rates of endometrial cancer. 8 Therefore, studying the basic cellular and molecular characterization of endometrial hyperplasia is imperative to develop novel preventive interventions for endometrial cancer.
The role of cancer stem cells in the initiation and progression of cancer, including endometrial cancer, has attracted significant research interest in recent years.9–13 OCT4, SOX2, and NANOG are transcription factors with significant interdependence and are established as core regulators of pluripotency in embryonic stem cells. Specifically, they regulate gene expression that promotes the remarkable plasticity of these cells and repress genes that promote differentiation. These properties are shared with cancer stem cells, with growing evidence supporting the role of these transcription factors as markers of cancer stem cells in various cancers.11,14
In endometrial cancer, a few studies characterized the expression of pluripotency markers, providing evidence of their higher expression in endometrial cancer cells compared to the normal endometrium, in addition to the association of the upregulation of these markers with an enhanced tumor formation capacity.13,15,16 However, sparse data exist on the expression of these markers and their role in endometrial hyperplasia.17,18 Both endometrial hyperplasia, as a clinical lesion, and the cancer stem cells, as a cellular anomaly, can be considered predispositions to endometrial cancer. Identifying associations between these two entities is crucial, as it could offer valuable insights into the molecular pathways of endometrial cancer pathogenesis. This understanding may facilitate the development of prognostic markers in the management of endometrial hyperplasia patients, especially for women at higher risk of endometrial cancer due to hereditary predisposition or acquired factors.
In this study, we investigated the tissue expression of the three master pluripotency stem cell markers (OCT4, SOX2, and NANOG) in hyperplastic endometrial tissue and compared it to control samples of non-hyperplastic endometrial tissue.
Materials and methods
Study design and sample selection
This is a retrospective case–control study that utilized archived, anonymized endometrial tissue specimens. The study was approved by the Research Ethics Committee (University of Sharjah REC-24-02-21-02-PG and the Ministry of Health and Prevention Research Ethics Committee MOHAP/DXB-REC/A.M.J/No. 79/2024). The tissue blocks used in the study were sourced from the institutional tissue bank at the Sharjah Institute of Medical Research, which had originally acquired the specimens from Al Baraha Hospital following the completion of their clinical retention period. The tissues were originally collected from patients who underwent diagnostic or therapeutic procedures for abnormal uterine bleeding (including dilatation and curettage/endometrial biopsies, and hysterectomies). The immunohistochemical study continued between May 2024 and February 2025. We included tissue blocks diagnosed as endometrial hyperplasia (typical/atypical) or common benign histologic findings (proliferative/secretory patterns, irregular shedding). Endometrial cancer and inconclusive diagnosis were excluded.
Immunohistochemistry studies
Primary antibodies included anti-OCT4 (ab109183, clone # EPR2054, 1:750 dilution; Abcam, Cambridge, UK), anti-SOX2 (ab92494, clone# EPR3131, 1:100 dilution; Abcam), and anti-NANOG (ab109250, clone# EPR2027, 1:200 dilution; Abcam) processed as per manufacturer instructions and our preliminary optimization studies. The specificity of the antibodies was validated using established positive tissue controls. Specifically, OCT4 and NANOG expression was confirmed in testicular germ cells, 19 while SOX2 was confirmed using low-grade breast cancer tissue. 20 Negative controls were prepared by omitting the primary antibodies during staining.
Tissue samples were obtained as blocks embedded in paraffin and sectioned at a thickness of 3 μm using a microtome. Tissue sections from selected blocks of study participants were deparaffinized in xylene, rehydrated through a graded series of ethanol, and subjected to heat-induced antigen retrieval. SOX2 and NANOG were retrieved using Tris–EDTA buffer (pH 9.0), while OCT4 was retrieved with citrate buffer (pH 6.0), all in a microwave. Endogenous peroxidase activity was blocked using 3% hydrogen peroxide. Sections were blocked with protein block and incubated with primary antibodies for 2 h at room temperature in a humidified chamber. After washing, sections were incubated with biotinylated secondary antibodies for 30 min at room temperature, followed by incubation with streptavidin-HRP and visualization using Diaminobenzidine (DAB) substrate. Hematoxylin was used for counterstaining. Stained tissue sections were imaged using an Olympus BX43 microscope (Olympus, Tokyo, Japan) equipped with a digital camera. Images were captured at 10×, 20×, and 40× magnification using Olympus cellSens software (Evident, Tokyo, Japan), which measured mean pixel intensity in five random fields per tissue section. Areas of interest were identified by focusing on regions with representative staining.
Immunohistochemical scoring
The expression of OCT4, SOX2, and NANOG in endometrial glandular epithelial cells was evaluated using a semi-quantitative method based on previously published protocols.21–23 For each marker, the extent of positively stained epithelial cells was classified on a 5-point scale: 0 (no staining, 0%), 1 (1%–10%), 2 (11%–25%), 3 (26%–50%), and 4 (51%–100%). The staining intensity was assessed as weak (+1), moderate (+2), or strong (+3). The final immunohistochemical score was obtained by multiplying the extent score by the staining intensity, resulting in a total score ranging from 0 to 12.
Based on previously reported scoring practices and the distribution of our data, the expression levels were categorized as low expression (0–2) and moderate/high expression (3–12). Representative images of each category are shown in Figure 1. All slides were independently evaluated by two blinded histopathologists, showing high concordance (κ = 0.86). In 10% of randomly selected samples, triplicate sections were re-examined for each marker to confirm consistency in scoring and interpretation.
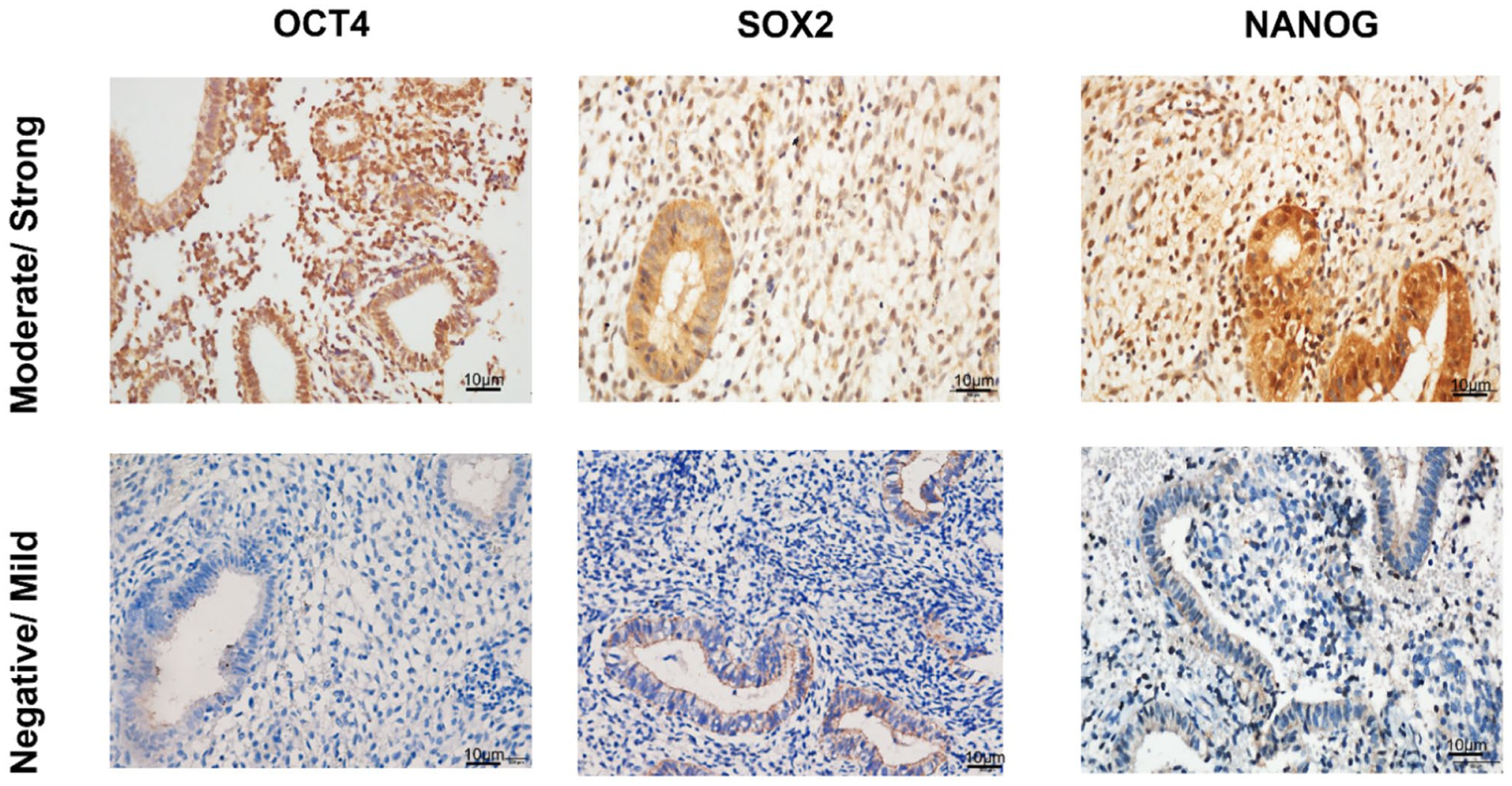
Representative immunohistochemical images (40× magnification) illustrating the expression of stem cell markers OCT4, SOX2, and NANOG in study subjects. The images depict the reference categories used in this study, distinguishing between negative/mild and moderate/high expression levels.
Statistical analysis
The study sample included all technically valid available endometrial hyperplasia tissue blocks from the institutional tissue bank. A prior study by Al-Kaabi et al. 17 reported NANOG expression in 88.4% of endometrial carcinoma cases compared to 15.0% in normal controls. Based on this 73.4% difference, a sample size calculation (with 80% power and a 5% significance level) indicated a minimum requirement of 6 participants per group, or 12 in total. The sample size included in our study exceeded this minimum. As there are no previous publications reporting differences in OCT4 and SOX2 expression between endometrial carcinoma cases and controls, a post hoc power analysis was performed to determine the study’s power to detect the observed effects for these two biomarkers.
Data were compiled into a Microsoft Excel sheet and then imported into the Statistical Package for Social Sciences (SPSS) program. 24 Descriptive statistics, including frequency and percentage, were calculated for categorical variables, while means and standard deviations and medians and interquartile ranges were reported for normally distributed and skewed scale data, respectively. The normality of scale data was first evaluated visually using histograms and Q–Q plots, and then statistically using the Kolmogorov–Smirnov test. 25 Age in years was categorized into two groups: 20–39 and 40 years and above. The expression level of each biomarker was grouped into “Low expression” (0–2) and “Moderate/High expression” (3–12). The Pearson chi-square test was then used to compare categorical variables between cases and controls. Bar charts were used to demonstrate variations in biomarker levels between cases and controls.
To identify the significant predictors of endometrial hyperplasia, a Binary Logistic Regression model was applied, including the input variables: age group, OCT4, SOX2, and NANOG. The assumptions of multicollinearity and multivariate outliers were checked before performing the regression model, using the variance inflation factor (<10) and the Mahalanobis distance, respectively. To verify the adequacy of the model, the Hosmer and Lemeshow test was used. An omnibus test was also performed to evaluate the significance of the model in predicting endometrial cancer cases. Cox and Snell R2 and Nagelkerke R2 were used to indicate the proportion of variation in the outcome variable explained by the regression model. The strengths of associations were presented using the odds ratio (OR) with its 95% confidence interval (CI), where a p-value of 0.05 or lower indicated statistical significance.
The receiver operating characteristic (ROC) curve and the area under the curve (AUC) were calculated separately among the cases, controls, and total study population to identify whether age, in years, is a significant predictor of the Moderate/High level of expression of any of the three biomarkers (OCT4, SOX2, and NANOG). ROC-AUC value above 0.70 indicates a moderate to high level of predictive accuracy. For each AUC value, statistical significance was assessed against chance by calculating its 95% CI and associated p-value. For each significant AUC value, a data-driven approach was used to determine, for age, the cut-off value that best predicts Moderate/High expression of the biomarker, yielding optimum values of sensitivity and specificity. Specifically, the age cut-off value was determined by maximizing Youden’s index (sensitivity + specificity − 1). Consequently, the sensitivity and specificity, along with their 95% confidence intervals, were calculated and reported.
Results
Our analysis included endometrial samples from 165 patients selected from an initial pool of 500 tissue blocks. The cohort comprised 2 matched groups: 83 patients with endometrial hyperplasia (including 11 atypical cases) and 82 controls with benign endometrial findings (proliferative/secretory patterns or irregular shedding). This selection reflected the overall prevalence in our tissue bank, where 20.8% of samples showed hyperplasia and only 0.4% were malignant. The groups were well-balanced by age, with similar proportions of patients ⩾40 years (53.8% hyperplasia versus 52.4% controls, p = 0.110).
To improve statistical power, effect size, and clinical interpretability, negative and mild expressions of the tissue markers were grouped into one category, whereas moderate and high expressions were grouped into another.
The percentage of moderate/high tissue expression compared to negative/low expression of the three markers was compared between endometrial hyperplasia cases and the control group (n = 165 for OCT4 or NANOG, n = 111 for SOX2), as shown in Figure 2 and Supplemental Material. Among the three markers, NANOG demonstrated the highest percentage of moderate/high expression in endometrial hyperplasia patients, reaching 60.2%, with a statistically significant difference between cases and controls. In contrast, the endometrial tissue expression of OCT4 and SOX2 was predominantly negative/low in over 80% of both cases and controls. However, OCT4 expression was marginally higher in endometrial hyperplasia (12%) compared to controls (3.7%, p = 0.045). Post hoc analysis revealed that the study had a power of 51% to detect the observed difference in OCT4 expression, 59% for SOX2, and 99% for NANOG between cases and controls.
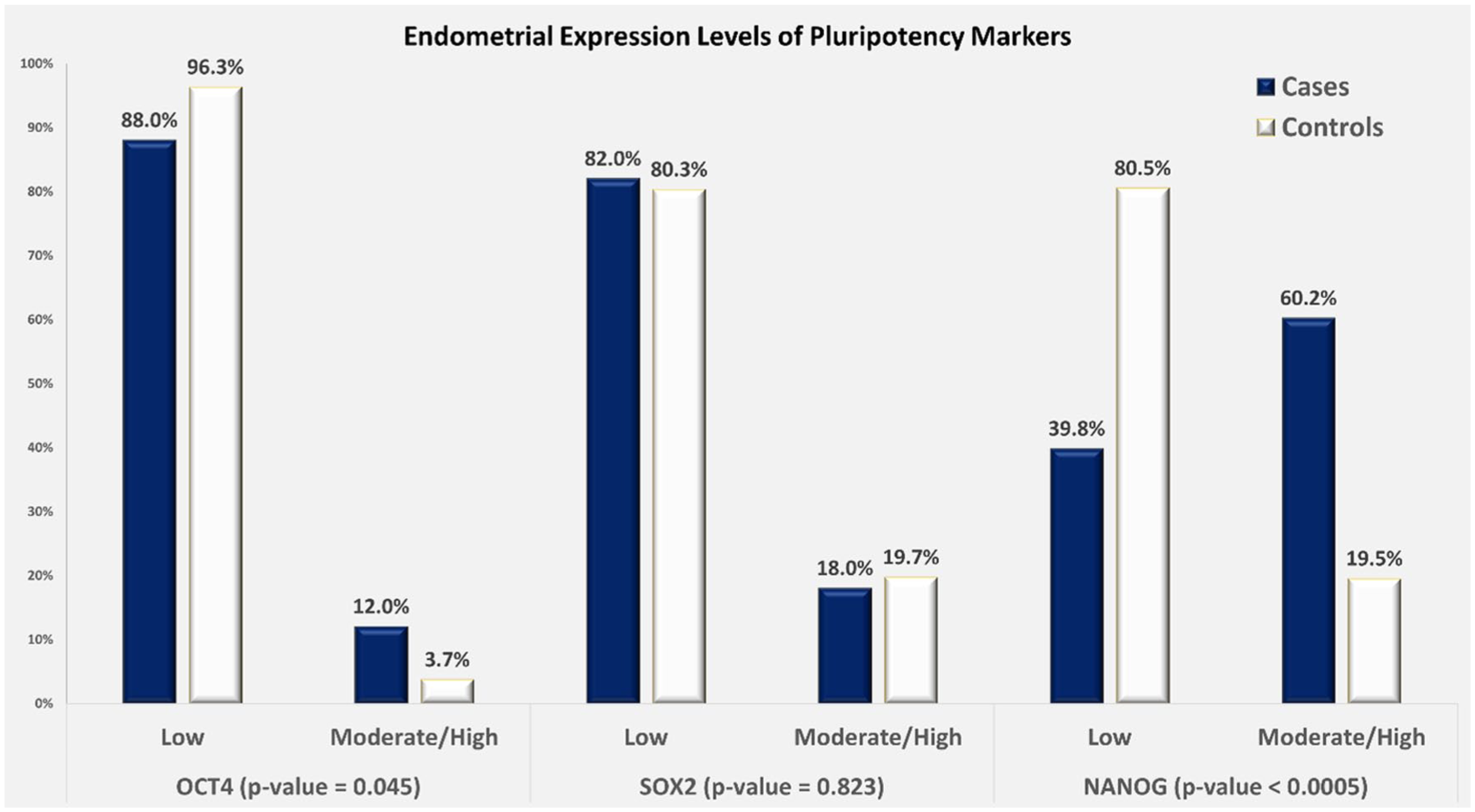
A bar graph demonstrating the level of expression of OCT4 (n = 165), SOX2 (n = 111), and NANOG (n = 165) in the endometrial tissue samples of patients with endometrial hyperplasia (cases) and without hyperplasia (controls). The association was calculated using the chi-square test. Statistically significant expression was identified for NANOG and OCT4 in endometrial hyperplasia compared to the control endometrium (p < 0.05). The number of cases and controls in each group for the three markers is included in Supplemental Figure 2.
Among the endometrial hyperplasia cases, we identified 11 cases with atypical endometrial hyperplasia. NANOG showed the highest percentage of moderate/high tissue expression in 63% (7 out of the 11 cases) compared to 59% in the endometrial hyperplasia without atypia group (43 out of 72 cases). OCT4 and SOX2 showed moderate/high expression in only 1 out of 11 and 1 out of 7 assessed atypical endometrial hyperplasia cases, respectively. There were no statistically significant differences between the moderate/high expression percentages between the atypical and non-atypical endometrial hyperplasia for any of the three markers (p = 1, using Fisher’s exact two-sided significance; Figures 3 and 4).
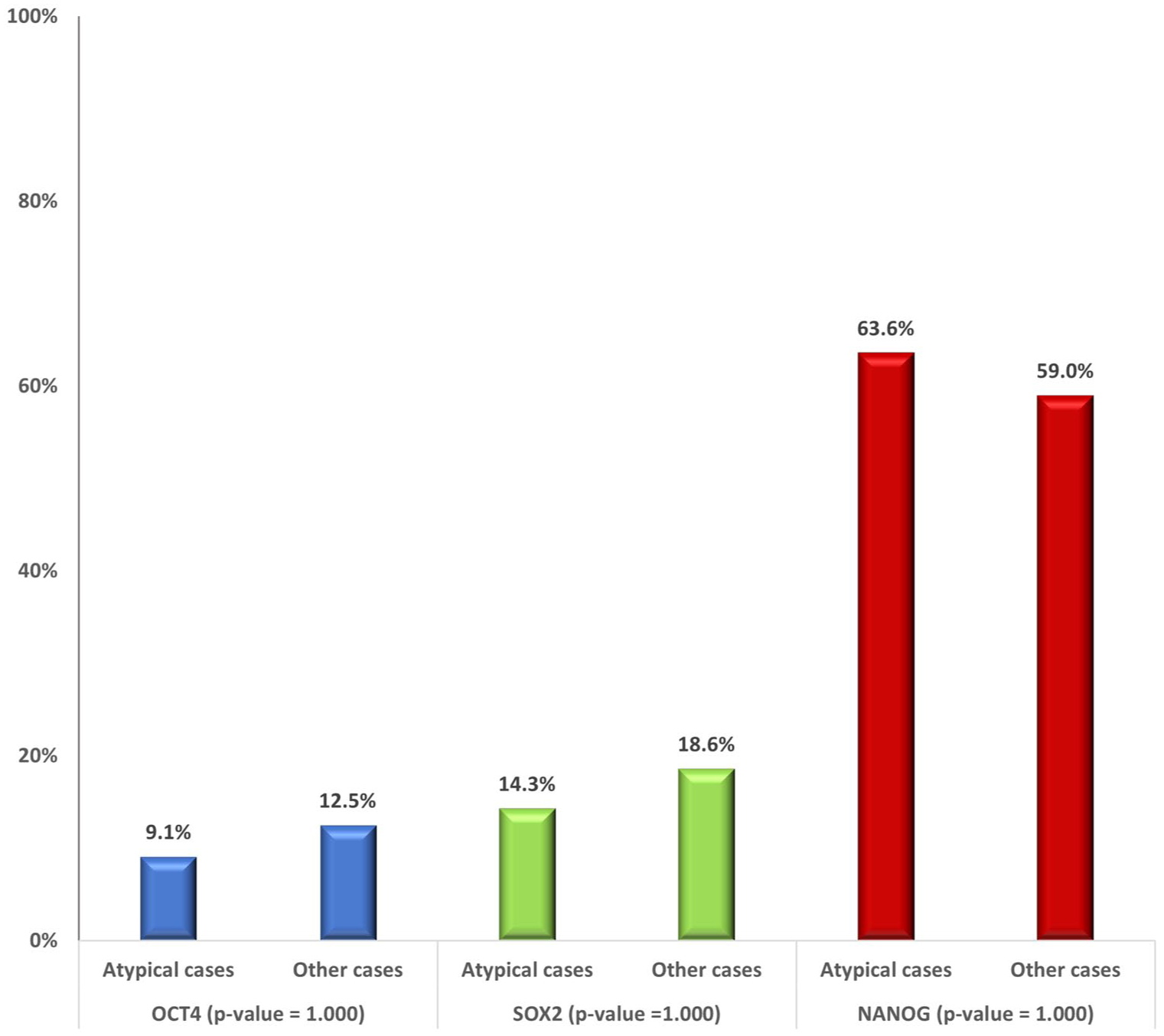
A bar graph comparing the moderate/high tissue expression of OCT4, SOX2, and NANOG within endometrial hyperplasia cases (atypical hyperplasia (n = 11 for SOX2 and NANOG, n = 7 for OCT4) versus hyperplasia without atypia). p-value was calculated by Fisher’s exact two-sided significance.
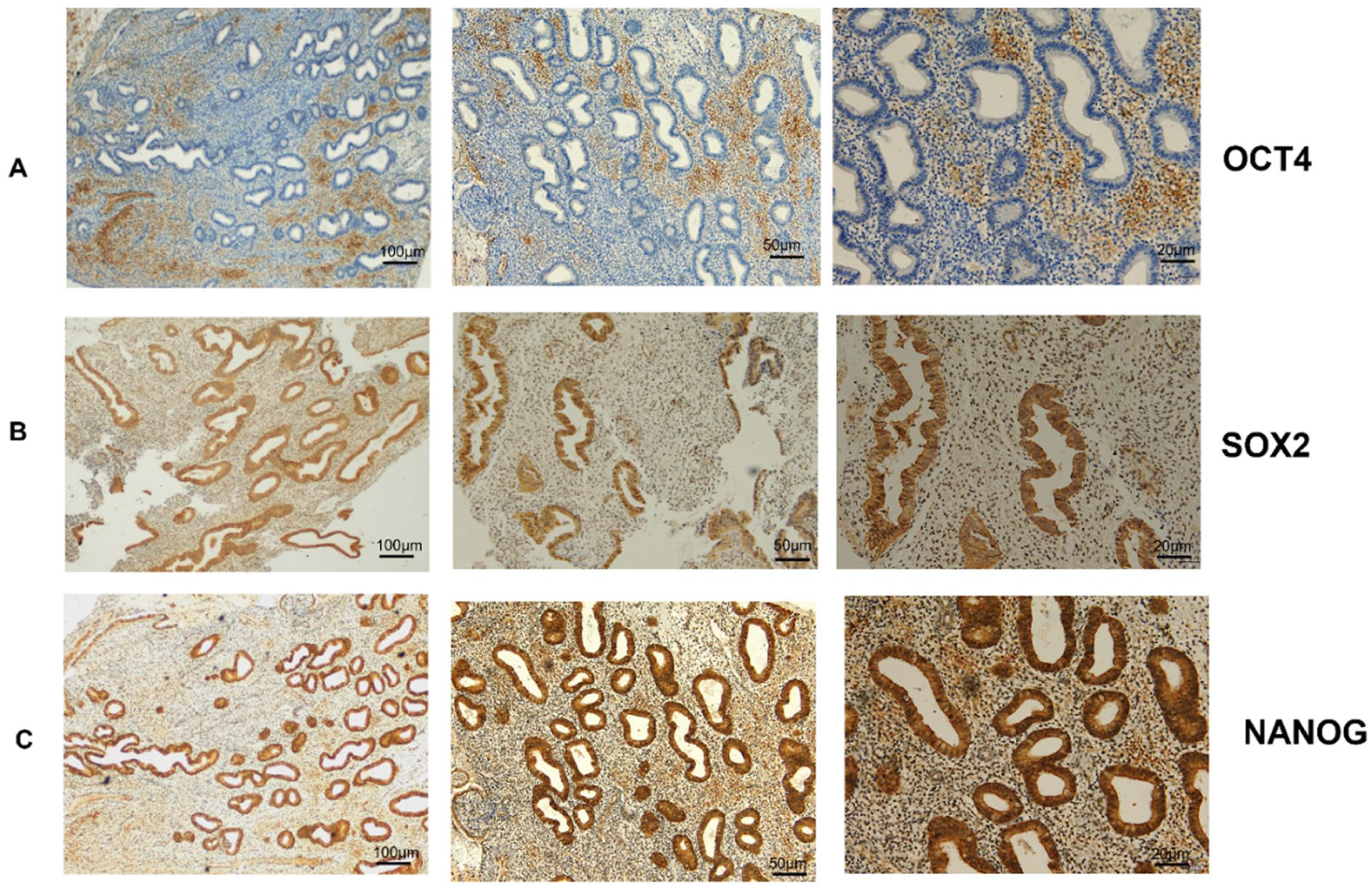
IHC staining for OCT4 (a), SOX2 (b), and NANOG (c) in a patient with atypical hyperplasia, visualized at three magnifications (4×, 10×, and 20×). Positive nuclear staining is indicated by brown chromogen (DAB (Diaminobenzidine)).
A binary logistic model was performed to predict the risk for endometrial hyperplasia using the input factors of age group (20–39 and 40+), OCT4 (low expression and moderate/high expression), NANOG (low expression and moderate/high expression), and SOX2 (low expression and moderate/high expression). The model was significant in predicting the outcome (Omnibus test chi-square = 30.900, df = 4, p < 0.001), and it explained between 25.3% and 34.0% of the outcome variation (Cox and Snell R2 = 0.253 and Nagelkerke R2 = 0.340). The model showed that age group and NANOG expression level were the only significant predictors of endometrial hyperplasia while keeping other variables constant. Women aged 40 years or older were 3.425 times more likely to have endometrial hyperplasia compared to those aged between 20 and 39 years (95% CI of OR: 1.328–8.835, p = 0.011). In addition, women with moderate/high levels of expression of NANOG had 11.178-fold increased odds of having endometrial hyperplasia as compared to those with low expression levels of NANOG (95% CI of OR: 3.484–35.864, p < 0.001; Table 1).
Binary logistic regression model for biomarkers predicting cases of endometrial hyperplasia.

Omnibus test (chi-square 30.900 (df = 4, p < 0.001)),
Cox and Snell R2 = 0.253 Nagelkerke R2 = 0.340. β: unstandardized coefficient; SE: standard error; OR: odds ratio; CI: confidence interval.
Reference group.
Findings from the ROC–AUC analysis indicated that age in years was a significant predictor of SOX2 expression in the endometrial hyperplasia group, and NANOG in the control group as well as in the total sample (combined group of cases and controls). The highest accuracy level was found in the prediction of moderate/high expression levels of SOX2 expression among the endometrial hyperplasia cases (AUC = 0.867, 95% CI 0.764–0.970, p = 0.001) where the optimal cut-off age value, as determined by the maximum Youden’s index of 0.722, was 49 years (sensitivity = 100% and specificity = 72.2%; Figure 5(a3)). Similarly, age predicted moderate/high expression levels of Nanog among the controls (AUC = 0.734, 95% CI 0.607–0.861, p = 0.004) with Youden’s index value of 0.37 and an optimal cut-off value of the age of 42 years (sensitivity = 68.8% and specificity = 68.2%; Figure 5(b2)). In the combined total sample, the cut-off age of 43 years predicted moderate/high expression levels of NANOG with an AUC = 0.644 (95% CI 0.555–0.732, p = 0.104); Youden’s index was 0.214, sensitivity was 54.1%, and specificity was 67.3% (Figure 5 (c2)).

The ROC curve illustrates the performance of age cut-offs in predicting the moderate-to-high expression of the three core pluripotency markers (OCT-4, Nanog, Sox-2) in endometrial hyperplasia patients (cases A1 to A3), in the controls (B1 to B3) and in the total study population (C1 to C3). The curve plots the true positive rate (sensitivity) against the false positive rate (1 − specificity) across various age thresholds.
Discussion
This study investigated the expression of pluripotency markers (OCT4, SOX2, NANOG) in endometrial tissue from women with abnormal uterine bleeding, comparing those with endometrial hyperplasia to controls with other benign diagnoses. NANOG exhibited the most pronounced expression difference: 60.2% of hyperplasia cases showed moderate/high expression, compared to only 19.5% in controls (p < 0.0005). In contrast, SOX2 was moderately/highly expressed in 18% of hyperplasia cases versus 19.7% of controls (not statistically significant). OCT4 expression was low overall but still significantly higher in hyperplasia (12%) than in controls (3.7%, p = 0.045). Logistic regression confirmed NANOG as the only independent predictor of hyperplasia, with moderate/high expression associated with an 11-fold increased risk compared to low expression.
In the present study, NANOG demonstrated the strongest association with endometrial hyperplasia among the evaluated markers, suggesting its potential predictive value. Following the establishment of NANOG’s role as a master transcription factor in embryonic stem cells and reprogramming, compelling evidence suggests that NANOG is one of the most critical cancer stem cell markers in various cancers.26,27 As thoroughly reviewed by Jeter et al., NANOG was shown to drive the core cancer stem cell phenotype by enforcing the undifferentiated cell state, enabling self-renewal and sphere formation in vitro, and conferring key in vivo properties such as tumor initiation, metastasis (through enhanced migration/invasion), and drug resistance via markers like MDR-1. 28 Conversely, knockdown of NANOG—either alone or in combination with OCT4—enhanced chemosensitivity, inhibited proliferation, induced cell cycle arrest, and suppressed invasion and tumor formation across different cancer types.29–31 NANOG was shown to play these biological roles through the modulation of several other key signaling pathways in cancer, such as P53 and STAT3 and other stemness and cell-cycle-related molecular pathways.26,32,33
Although OCT4, SOX2, and NANOG are well-established core pluripotency factors in embryonic stem cells, their individual roles in somatic tissues remain unclear. 34 Emerging evidence suggests these factors regulate distinct cell fate decisions rather than uniformly promoting self-renewal. 35 Notably, NANOG drives the transition to ground-state pluripotency and reactivates the silenced X chromosome in female embryonic stem cells, a role that may, hypothetically, translate to carcinogenesis by facilitating undifferentiated cancer stem cell phenotypes in female cancers. 36 These findings invite further investigation into NANOG’s contribution to cancer stem cell-like properties in endometrial hyperplasia.
Only one prior study (Al-Kaabi et al.) has examined NANOG role in endometrial hyperplasia, reporting expression in 33% of cases (10/30), predominantly in atypical hyperplasia (8/12). 17 Our study found moderate/high NANOG expression in 63% of atypical cases (11 in total), though this did not significantly differ from non-atypical hyperplasia. Small sample sizes of atypical endometrial hyperplasia cases in both studies limit definitive conclusions for this subgroup.
Likewise, a few previous studies investigated the role of pluripotency markers in endometrial cancer. Zhou et al. found NANOG expressed in 81% of endometrial cancers (55 samples) but absent in benign tissue, with OCT4 and SOX2 expressed in 76% and 70% of cancers, respectively. Moreover, NANOG-positive tumorospheres showed high tumorigenicity, suggesting its potential in cancer stem cells and as a therapeutic target. 16 Al-Kaabi et al. reported NANOG in 88% of cancers versus 15% in normal tissue, correlating with clinically aggressive disease. 18 However, other studies implicate OCT4 as pivotal—its overexpression in endometrial cancer cells upregulated pluripotency and stemness markers (ALDH1A1, CD133) while enhancing proliferation, colony formation, and chemoresistance. 13
The role of SOX2 in endometrial cancer remains unclear, with the few available studies reporting conflicting results. Some link high SOX2 expression to the advanced stage, aggressive behavior, and poor survival,37,38 while others show SOX2 downregulation and less mRNA expression in the cancer compared to normal tissue, attributed to epigenetic silencing through promoter hypermethylation. 39 In our study, SOX2 expression was slightly lower in hyperplasia than in normal tissue, though not significantly. Further research on SOX2 methylation in hyperplasia could clarify its role.
In the present study, age was a significant predictor of endometrial hyperplasia, consistent with established evidence. Women ⩾40 years had three times higher risk than those aged 20–39, aligning with the recognized association between older age (particularly peri-menopausal status) and increased endometrial cancer risk. No prior studies have examined age-related expression of pluripotency markers in the endometrium. We assessed marker expression across age in the entire cohort, as well as in hyperplasia and normal endometrium separately (Figure 4). Notably, age ⩾49 predicted moderate/high SOX2 expression in hyperplasia (100% sensitivity). NANOG expression in hyperplasia was not age-dependent, but age ⩾42 predicted moderate/high NANOG in controls (68.8% sensitivity). OCT4 showed no age-related expression patterns in any group. Establishing the baseline expression of these markers in normal endometrial tissue across age groups is critical. A gap in the literature makes it unclear whether age itself influences expression in healthy individuals, which is a significant question given that age is a recognized risk factor for endometrial cancer. Our analysis of controls provided an essential internal validation, revealing that the significant association between NANOG and endometrial hyperplasia is not merely a proxy for age but an independent factor. The weak predictive power of age for these markers in control patients, as demonstrated by the ROC analysis, strengthens this conclusion.
Consideration for clinical validation
Pluripotency markers have prognostic potential in cancer, but the endometrium’s unique regenerative capacity complicates their interpretation. Normal endometrial stem cells may express these markers cyclically, as one study found elevated NANOG, OCT4, and SOX2 levels in human endometrial cells, along with enhanced reprogramming efficiency. 40 Likewise, a side population with human endometrial stem cells properties was generated under hypoxic culture conditions, in which a gene profile characteristic of undifferentiated cells was identified. The profile included the key pluripotency markers NANOG and OCT-4, providing further evidence for the presence of stem cells in the normal endometrial tissue. 41 Thus, establishing baseline expression patterns across the menstrual cycle—in normal endometrium, hyperplasia, and cancer—is critical to ensure specificity and determine optimal assay timing.
Prolonged unopposed estrogen exposure—whether from exogenous sources, peripheral fat synthesis in obesity, or selective estrogen receptor modulators—significantly increases endometrial hyperplasia risk.42–44 Women with these risk factors represent ideal candidates for prognostic studies evaluating estrogen’s effects on endometrial pluripotency marker expression.
Current gynecologic practice lacks objective methods to predict whether endometrial hyperplasia will regress or progress to cancer, complicating patient counseling. A critical research priority is understanding how progesterone (first-line therapy for non-atypical hyperplasia) affects molecular markers and whether any molecular changes correlate with histologic resolution. For instance, the three pluripotency markers (NANOG, OCT4, and SOX2) were identified in one study as potential drivers of progestin resistance in endometrial cancer cell lines. Targeting these genes with embryonic sac fluid stimulated cell differentiation in a progestin-resistant endometrial cancer cell line and restored its therapeutic sensitivity to progestin. 45 Developing consistent research evidence in this direction could guide clinical decisions in the future, helping gynecologists determine when to discharge patients from prolonged follow-up/biopsies, continue progesterone, or escalate to hysterectomy (particularly for women on systemic progestins or those declining treatment). This unmet need is recognized in international clinical guidelines. 46
Study limitations
This study has several limitations. First, control tissues came from symptomatic women with benign uterine bleeding, which may introduce heterogeneity. While comparing against asymptomatic, normal endometrial tissue would be the ideal standard. However, obtaining endometrial tissue from completely healthy, asymptomatic women presents a significant practical and ethical challenge due to the invasiveness of the procedure. While controls from hysterectomy specimens for non-bleeding, non-pathological reasons (e.g., vaginal hysterectomy for uterine prolapse) represent a possible source of normal endometrial tissue, such cases are increasingly getting scarce and do not constitute a feasible source for a sufficiently sized control cohort. Therefore, our selection of controls from women with benign abnormal uterine bleeding could be considered a clinically justified compromise. Nevertheless, this approach may have had its advantage as it ensured that the differential expression we observed for NANOG is specific to hyperplasia within the standard clinical population presenting with this shared symptom of abnormal uterine bleeding. Second, the small sample size, particularly for atypical hyperplasia cases, has resulted in wide confidence intervals as well as low statistical power when comparing observed differences, especially for OCT4 and SOX2 biomarkers. Third, while we analyzed tissue expression, we lacked data on hormonal/reproductive risk factors that could influence these markers. Finally, the retrospective design precludes establishing causal relationships between marker expression and hyperplasia development. Despite these limitations, our findings remain valid as observed variations fall within the spectrum of benign endometrial changes. Larger prospective studies are needed to confirm these results.
Conclusion
NANOG shows promise as a marker of potential predictive value in endometrial hyperplasia, with moderate/high expression significantly increasing risk. While the role of SOX2 and OCT4 remains unclear, SOX2 exhibited age-related expression in hyperplasia. These preliminary findings warrant validation in prospective studies, particularly regarding hormonal influences and clinical utility.
Supplemental Material
sj-docx-1-whe-10.1177_17455057251408631 – Supplemental material for Expression of pluripotency stem cell markers in endometrial hyperplasia
Supplemental material, sj-docx-1-whe-10.1177_17455057251408631 for Expression of pluripotency stem cell markers in endometrial hyperplasia by Noha A. Mousa, Zeinab Al-Rawi, Amal Hussein, Abdalla Suliman, Ghada Mohammed, Rifat Hamoudi and Suha Al-Naimi in Women's Health
Footnotes
Acknowledgements
We would like to thank and acknowledge the contributions of Ms. Nival and Ms. Razan for providing initial technical help in immunohistochemistry and tissue sectioning.
Ethical considerations
Ethical approvals were obtained from the University of Sharjah Research Ethics Committee (REC-24-02-21-02-PG) and the Ministry of Health and Prevention Research Ethics Committee (MOHAP/DXB-REC/A.M.J/No. 79/2024). This study utilized entirely anonymized, archival tissue specimens obtained from the Sharjah Institute of Medical Research tissue bank. The specimens were irreversibly delinked by the source hospital from any patient identifiers or detailed clinical data before submission to the tissue bank. The ethics committees waived the requirement for obtaining consent for this research, as the samples were archival, fully anonymous, and the study nature was deemed minimal risk.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Author contributions
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The project was funded by the Competitive Grants #1901090266 and #22010902116
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
All original immunohistochemistry tissue sections, data collection sheets, and statistical analysis data are available from the corresponding author and will be provided upon reasonable request.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
