Abstract
Background:
There is increasing evidence that menstrual changes (MC) should be considered as an adverse event resulting from COVID-19 vaccination. However, the contributing health factors are still poorly understood.
Objectives:
The aim was to analyze the characteristics and factors influencing MC after the administration of the severe acute respiratory syndrome coronavirus 2 vaccine.
Design & Methods:
A retrospective observational study of currently menstruating women (N = 14,550) in Spain was conducted during the month of December 2021 using an online survey. Among others, general characteristics of the menstrual cycle, medical history, and adverse events following vaccination—including MC—were recorded. Bivariate logistic regression analysis was performed to determine the influencing factors.
Results:
45.0%—50.9% of respondents reported experiencing mostly transient MC after both doses of COVID-19 vaccine, including different spotting (41.6%—49.0%), longer periods (26.5%—29.5%) and/or heavier flow (28.7%—31.6%). Binary logistic regression analysis showed that some of the inter-individual factors that may be involved in this unexpected event are age (dose 2: adjusted odds ratio (aOR): 1.02; 95% confidence intervals (CI): 1.02–1.03), heavy menstrual flow (dose 1: aOR: 1.12; 95% CI: 1.00–1.25), use of short- (dose 1: aOR: 1.38; 95% CI: 1.24–1.54) to medium-term contraception (dose 1: aOR: 1.31; 95% CI: 1.09–1.57), number of previous pregnancies (dose 1: aOR: 1.11; 95% CI: 1.03–1.19), pre-existing diagnoses of certain clinical conditions—including endometriosis (dose 1: aOR: 1.33; 95% CI: 1.11–1.59)—and suffering from other vaccine adverse events.
Conclusion:
Currently menstruating women may experience MC after COVID-19 vaccination. Further research is warranted to address the influencing factors, considering their heterogeneity according to the geographical background of the target population. This kind of evidence could prove instrumental in the context of future viral outbreaks, helping healthcare professionals to provide scientifically up-to-date information to patients for making informed decisions regarding their well-being, particularly in societies where menstruation remains a taboo subject.
Plain Language Summary
Health-related factors such as age, heavy menstrual flow, contraception, previous pregnancy, endometriosis, and other vaccine-related adverse events may underlie menstrual changes after COVID-19 vaccination.
Keywords
Background
The menstrual cycle is a sign of a woman’s health and fertility. It is controlled by the complex functioning of the hypothalamic-pituitary-gonadal axis, which can be affected by a variety of stressors.1,2 Nevertheless, any abnormal changes should not be ignored,3,4 even in the context of the coronavirus disease (COVID-19) pandemic, probably the most challenging public health emergency faced by modern society.
Over the past 4 years, a significant number of studies1,2,5–19 and reports from national public health surveillance agencies20–24 have claimed that menstrual changes (MC) should be considered as adverse events resulting from COVID-19 vaccination. While the vaccine against the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) was undoubtedly one of the most significant preventive strategies to halt the progression of the pandemic, it is crucial that we continue to analyze the factors influencing the occurrence of related side effects. This will allow us to understand how this, and other new vaccines, may affect the reproductive health of both sexes and, consequently, to provide scientifically based medical advice to those patients who seek help for this unexpected health event.
To date, several transient MC have been described in currently menstruating women after receiving the SARS-CoV-2 vaccine, including those related to menstrual cycle length5,6–9,12,14,15,17–19—including period length,5,10,16,17 menstrual flow,5,6,10–12,14–17 and other menstrual irregularities.2,5,6,12,13,15,16,18 In the context of limited understanding of menstrual physiology, it is conceivable that neuro-immune-endocrine interactions may underpin the observed phenomenon.5,25,26 However, little is known about the health factors that may be associated with its occurrence. The expected outcomes of this study include improving outbreak preparedness, strengthening evidence-based clinical practice, and fostering trust by validating and responding to patient-reported side effects. The aim of this study is to contribute to the existing body of scientific knowledge by analyzing the characteristics of this unexpected event in one of the most affected countries, Spain, and the factors that may have influenced it.
Materials and methods
Experimental design
An online survey (Microsoft Forms®; Microsoft Corporation, Washington, DC, USA) was used to conduct a retrospective, observational study of adult Spanish women. The study was conducted in accordance with the Declaration of Helsinki and approved by the Institutional Review Board of the University of Extremadura (ref. 180/2021). The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement. 27 Accordingly, a completed STROBE checklist is provided as a Supplemental Material to ensure comprehensive and transparent reporting of the study design, methods, results, and interpretation.
Recruitment, data collection, and participants
The online survey was launched in Spain in December 2021. Recruitment was done through social networks using the snowball method. Only participants who agreed to be contacted by the research group by email for additional data collection gave electronic informed consent. A total of 17,512 people were recruited within 15 days irrespective of their menstrual status. From this larger sample, the research team identified data from a subpopulation of currently menstruating women as being of interest and thus selected them for the present analysis (N = 14,550). Inclusion criteria were women: (1) over 18 years of age, (2) being currently menstruating women, and (3) who had received at least one dose of COVID-19 vaccine (Figure 1). Exclusion criteria included age <18 years, unvaccinated, formerly menstruating women and incomplete surveys.
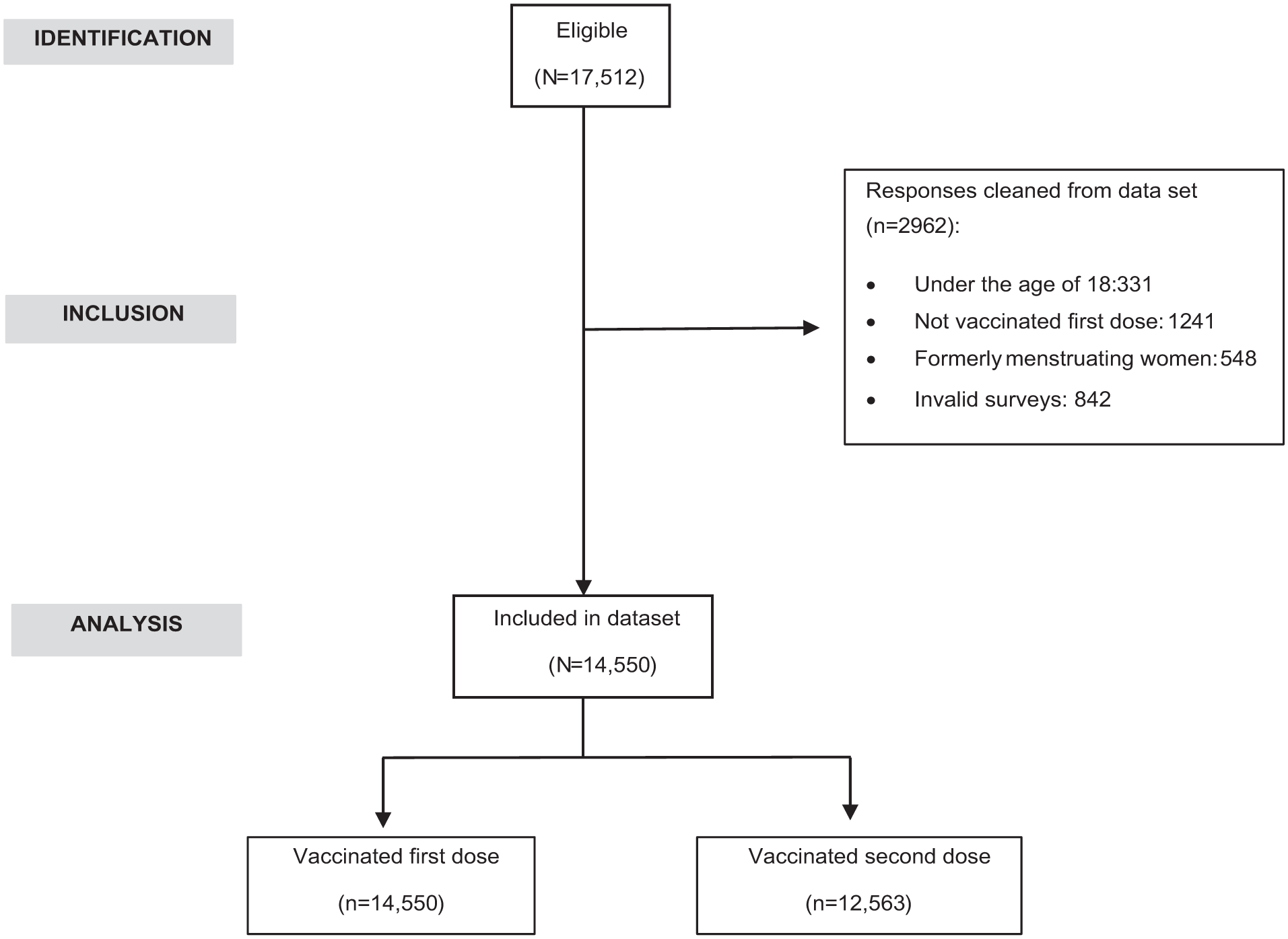
STROBE flow diagram.
Survey information
Based on the study by Lee et al., 10 the research team designed a customized questionnaire with 56 multiple-choice and text-entry questions divided into 6 sections regarding (1) the general characteristics of the menstrual cycle—or its absence and the cause, (2) SARS-CoV-2 infection, (3) COVID-19 vaccine, (4) menstrual experiences both after the SARS-CoV-2 infection and COVID-19 vaccination compared to the expected period symptoms—for example, shorter/longer/same, heavier/lighter/same. . ., (5) other MC—for example, spotting, breast pain, hot flashes, premenstrual syndrome, and other bleeding, (6) time between infection/vaccination and MC, (7) duration of MC, (8) adverse events of each vaccine dose, (9) reproductive history, (10) medical history, and (11) demographics. Some modifications were made to the original survey instrument to adapt it to the national context, including the two most used contraceptives in Spain: combined hormonal contraceptives and nonhormonal intrauterine device. The survey took 20–25 min to complete.
Statistical analysis
Participants were grouped according to the occurrence (MC subgroup) or non-occurrence (n-MC subgroup) of MC after COVID-19 vaccination. Quantitative variables were expressed as median and interquartile range, while qualitative variables were expressed as number of participants and frequency (%). Statistical analysis includes chi-square, Mann-Whitney U, and McNemar mid-p tests. Subsequently, bivariate logistic regression analysis was performed to investigate possible associations between the occurrence of MC following vaccination (dependent variable) and the independent variables that were significant (p ⩽ 0.05) in the previous analyses. Results were presented as adjusted odds ratios with 95% confidence intervals (CI). The above analyses were performed with the Statistical Package for Social Sciences (SPSS v.25; IBM, New York, NY, USA) for Windows. Statistical significance was set at p ⩽ 0.05.
Results
Anthropometric characteristics and medical history
65.2% of the study participants (N = 14,550, age: 33.0 (28.0–38.0)) had normal weight (body mass index, BMI: 22.8 (20.7–25.7)). Focusing on medical history, 23.0% had a diagnosis of non-autoimmune conditions and 8.9% of autoimmune diseases, both cases mainly related to thyroid. Regarding gynecological history, most women reported regular menstrual cycle (87.7%) and menstruation lasting 3–5 days (60.6%) with moderate flow (58.6%). Hormonal contraception (14.5%) was the most used method. 32.3% had a diagnosis of gynecological disease, the most common being polycystic ovary syndrome (43.6%) and endometriosis (12.4%; Table 1).
Anthropometric characteristics and medical history of the study population (currently menstruating women, N = 14,550).

BMI: body mass index; HPV: human papillomavirus; IUD: intrauterine device; PCOS: polycystic ovary syndrome.
Values are expressed as median (interquartile range).
Values are expressed as n (%).
Impact of vaccination on the menstrual health of currently menstruating women
On the other hand, most participants received the Pfizer-BioNTech Company® (New York, USA) vaccine. Moderna Spikevax® (Cambridge, USA) and Oxford-AstraZeneca® (Cambridge, UK) vaccines followed (Table 2).
COVID-19 vaccination of the study population (currently menstruating women, N = 14,550), according to the dose received.
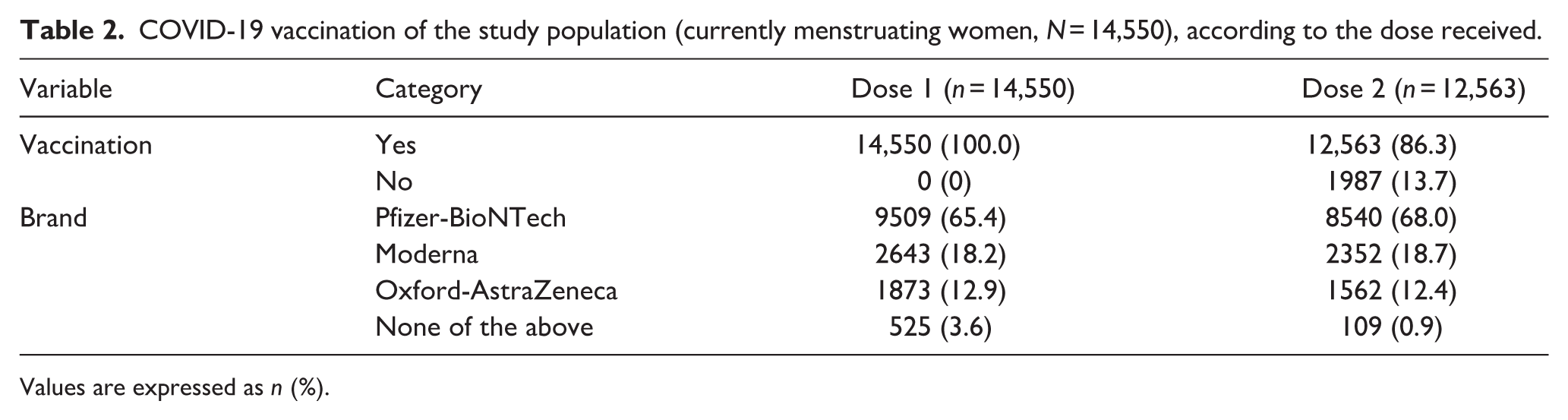
Values are expressed as n (%).
Comparative analysis of the occurrence of MC and other adverse events after COVID-19 vaccination
Compared to the first dose (45.0%), a significantly higher percentage (50.9%) of women experienced MC after the second dose (Figure 2(a)–(g)), as well as the simultaneous occurrence of two or more symptoms (Figure 2(b)), mainly spotting. Focusing on changes in menstrual bleeding, there were also significant differences between doses. For example, after the second dose, more women reported longer periods (Figure 3(a)), and heavier flow (Figure 3(b)). In addition, 30.6% of women reported that these changes had lasted “to date” (versus 17.1% for the first dose; Figure 3(d)).

Comparison between vaccine doses of frequencies (%) for variables related to MC (currently menstruating women, n = 12,563). (a) Occurrence of MC; (b) number of symptoms related to the MC; (c) spotting; (d) different spotting; (e) breast pain; (f) hot flushes; (g) premenstrual syndrome.

Comparison between vaccine doses of frequencies (%) for variables related to menstrual bleeding changes (currently menstruating women, n = 12,563). (a) Length; (b) flow; (c) time between vaccination and period; (d) duration of menstrual bleeding changes.
The occurrence of two or more adverse events was also significantly more frequent after the second dose (Figure 4(b)). The most common were fatigue (Figure 4(d)), fever (Figure 4(e)), and headache (Figure 4(f)).

Comparison between vaccine doses of frequencies (%) for variables related to other adverse events (currently menstruating women, n = 12,563). (a) Occurrence of other adverse events; (b) number of adverse events; (c) arm pain; (d) fatigue; (e) fever; (f) headache; (g) nausea, (h) breast lumps; (i) swollen glands; (j) others adverse events.
Factors associated with the occurrence or not of MC after COVID-19 vaccination
Table 3 shows the comparative analysis between the subgroups. Compared to the n-MC subgroup, women who experienced MC after receiving the two doses were heavier, with significant differences in the BMI category “pre-obesity/overweight.” These women were also significantly older than those who did not report MC after dose 2. The prevalence of some non-autoimmune clinical conditions was significantly higher in the MC subgroup according to the dose considered, such as rheumatic/articular and other conditions (dose 1), infectious (dose 2), and neurological/mental and gastrointestinal disorders (doses 1 & 2). In addition, allergies were observed to be more common in this subgroup compared to the n-MC subgroup (dose 1). For COVID-19, significant differences between subgroups were observed for both diagnosis (dose 2) and non-common vaccine brands (doses 1 & 2).
Differences in the study variables according to the dose received and the occurrence or not of MC after COVID-19 vaccination (currently menstruating women: n = 14,550 (dose 1); n = 12,563 (dose 2)).

BMI: body mass index; HPV: human papillomavirus; IUD: intrauterine device; MC: menstrual changes subgroup; n-MC: non-menstrual changes subgroup; PCOS: polycystic ovary syndrome.
Values are expressed as median (interquartile range).
Values are expressed as n (%).
p ⩽ 0.001. •p ⩽ 0.01. †p ⩽ 0.05 versus n-MC.
Focusing on the data on gynecological history (Table 3), menarcheal age was significantly lower in MC women compared to those in the n-MC subgroup (dose 2). Regarding menstrual cycle characteristics, a significantly higher percentage of women with irregular length (dose 1), period length of more than 5 days or other (doses 1 & 2), and heavy flow (doses 1 & 2) was observed in the MC subgroup compared to the n-MC subgroup. Moreover, significant differences between the subgroups were detected for both doses in terms of duration of use and type of contraceptive, as well as previous pregnancies, which were more common in the MC subgroup. Endometriosis and uterine bleeding were also more frequently observed in this subgroup. Finally, those who experienced MC after both doses were more likely to experience adverse events.
Binary logistic regression analysis for dose 1 (Table 4) showed that factors influencing MC after COVID-19 vaccination in currently menstruating women may include heavy menstrual flow, the phase of the menstrual cycle at the time of vaccination, heavy period flow, the presence of certain clinical conditions―rheumatic/articular, neurological/mental or gastrointestinal non-autoimmune conditions, and endometriosis―use of short- to medium-term contraception, having been pregnant more than two times, and experiencing vaccine adverse events such as fatigue, headache, nausea, swollen glands, breast lumps, and others.
Factors associated with the occurrence of MC after receiving the first dose of the COVID-19 vaccine: binary logistic regression.
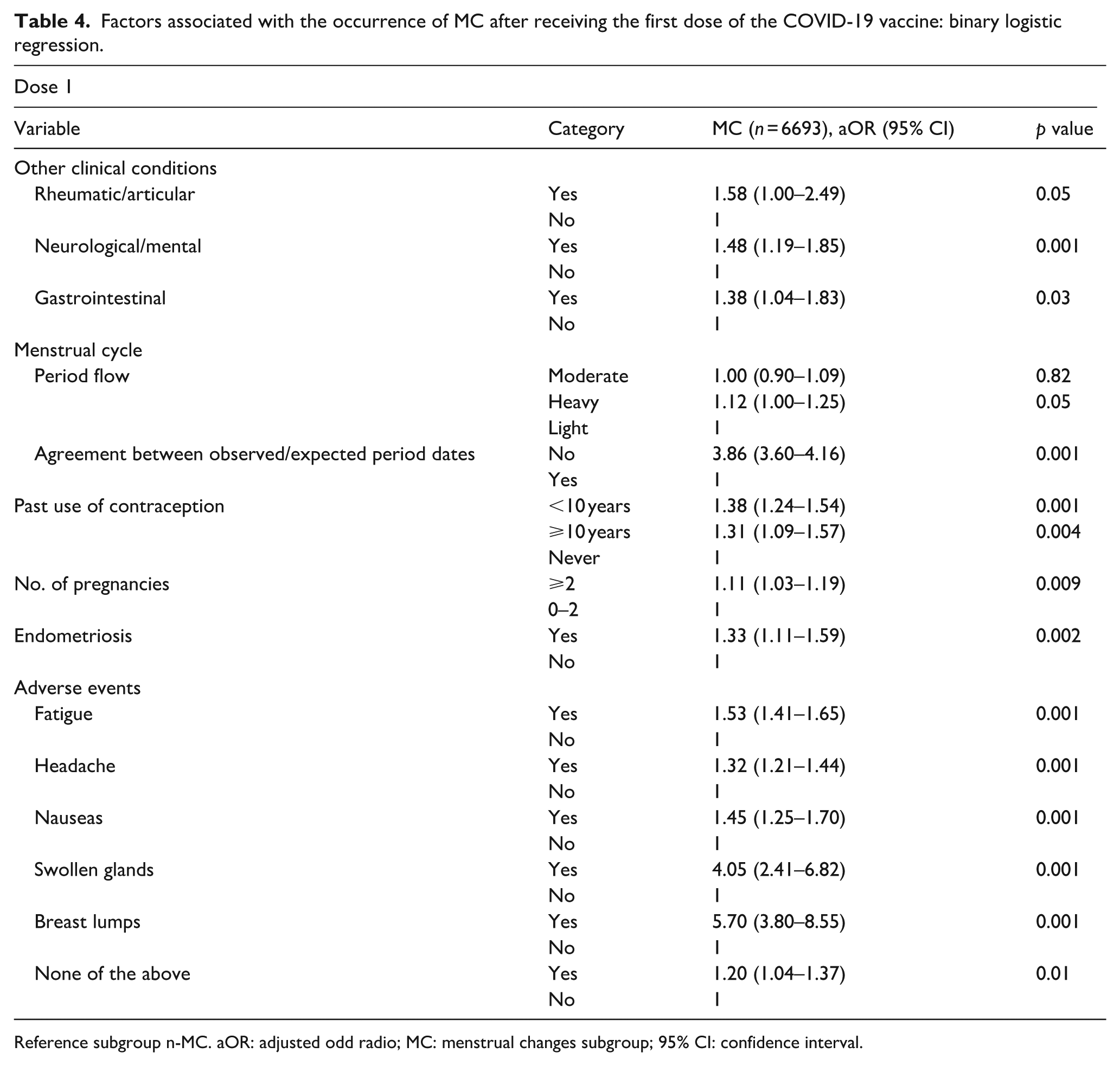
Reference subgroup n-MC. aOR: adjusted odd radio; MC: menstrual changes subgroup; 95% CI: confidence interval.
On the other hand, the age, phase of the menstrual cycle at the time of vaccination, previous diagnosis of an infectious disease, experiencing MC after the first dose, and suffering from some adverse events―for example, fatigue, fever, headache, nausea, swollen glands, breast lumps, and others―may be some of the factors associated with MC after receiving the second dose (Table 5).
Factors associated with the occurrence of MC after receiving the second dose of the COVID-19 vaccine: binary logistic regression.

Reference subgroup n-MC. aOR: adjusted odd radio; MC: menstrual changes subgroup; 95% CI: confidence interval.
Discussion
Currently menstruating women may experience MC, most commonly in the 2 weeks after receiving the COVID-19 vaccine. Inter-individual factors influencing this unexpected event may include age, heavy menstrual flow, use of short- to medium-term contraception, number of previous pregnancies, pre-existing diagnoses of certain clinical conditions―including endometriosis―and suffering from other vaccine adverse events.
Our findings add to a growing body of research showing that currently menstruating women may experience MC. As in our study, other authors have found changes in menstrual bleeding patterns, reporting longer periods5,20 and/or heavier flow5,6,10–12,14–17 than usual in vaccinated women. Although evidence suggests that these MC are mostly transient in nature,5,7–9,12,15,17,18,20 a non-negligible percentage of women reported suffering from them for more than 12 months.
Consistent with previous studies,5,12,14,16–18,20 the occurrence of MC seems to be significantly higher after the second dose of the vaccine. This was also observed in our study of formerly menstruating women. 25 For Male et al., 26 the interval between doses (3–4 weeks in the case of Spain) 28 may explain this additive effect, suggesting the importance of the phase of the menstrual cycle at the time of vaccination, later confirmed by binary logistic regression. Further research is needed, however, because of conflicting results.8,12 It should be remembered that the menstrual cycle is controlled by a complex hormonal process that varies at different stages and can be influenced by several internal and external factors.3–5 In the past, vaccinations against other viruses29–32 have also been associated with MC. Thus, human papilloma vaccine has been linked to premature ovarian insufficiency and increased number of hospital visits for abnormal amount of menstrual bleeding. 33 On the other hand, viral infections have also been reported to induce reproductive alterations.34–36 In the case of SARS-CoV-2 infection, similar percentages of vaccinated women with MC were observed regardless of prior infection history5–13; however, Muhaidat et al. 5 found a significant association between post-vaccination menstrual abnormalities and the severity of COVID-19. For their part, Li et al. 37 conclude that the average sex hormone concentrations and the ovarian reserve did not significantly change in those COVID-19 women of child-bearing age who experienced temporary MC. Our recent research indicate that SARS-CoV-2 infection may also result in menstrual-related disturbances in formerly menstruating women, including unexpected vaginal bleeding and spotting. Our analysis revealed that perimenopausal status and a history of menorrhagia are associated with an increased likelihood of experiencing these events. 38 Taken together, this evidence would support the theory proposed by other researchers5,25,26 about the role of unknown immunological mechanisms as triggers of MC resulting from both infection and vaccination. The interaction between the immune and neuroendocrine systems has been shown to control some of the processes of the menstrual cycle, including the cyclic build-up and breakdown of the uterine endometrium.6–8 In this sense, thrombocytopenia39,40 and changes in lymphocyte subpopulation patterns 34 may be involved. Furthermore, from our point of view, the possible involvement of oxidative events in this phenomenon should not be ignored. Unfortunately, our knowledge of the basic uterine and menstrual physiology is not enough to understand more complex processes of this kind, as demonstrated by the increasing evidence about an unknown internal circamonthly timing system. 41
Binary logistic regression analysis showed that other inter-individual factors may be involved in the occurrence of MC following COVID-19 vaccination in currently menstruating women, such as age, heavy menstrual flow, use of short- to medium-term contraception, number of previous pregnancies, pre-existing diagnoses of certain clinical conditions―including endometriosis―and suffering from other vaccine adverse events. Several of these factors were also reported by Lee et al. 10 A previous study in Spanish women 2 also points to age as one of the factors influencing this event. But again, the current scientific evidence is mixed. Other authors observed that the association between vaccination and MC did not differ according to age, use of contraceptives/hormones, or history of gynecological disease(s),5,16 but found a significant association with common vaccine side effects,5,11 in contrast to Alvergne et al. 8 From our perspective, these factors likely reflect distinct neuro-immune-endocrine contexts that may be involved in the occurrence of this vaccine side effect. Further research is warranted to clarify these complex interactions, considering their heterogeneity according to the geographical background of the target population. Moreover, sociodemographic factors may also underlie the difference in MC rates between countries. Thus, the country of residence could be an influencing factor, both for the predominant use of certain brands of vaccines—and the corresponding schedule—and for cultural reasons that may discourage young women from seeking help.5,42 That aside, most studies5,7–9,12,14,15 seem to agree that the occurrence of MC does not seem to be brand-specific and therefore independent of the technological approach (adenovirus-vectored or mRNA vaccines), suggesting shared immunological mechanisms.7,8
Given the current evidence, it is necessary to study in depth the impact of the vaccine, not only at reproductive age, but also throughout the different stages of a woman’s life, taking in account the potential influence of inter-individual factors (weight, genetic, medical history, smoking. . .).2,5,26 Further research must be also focused on determine the underlying physiological mechanisms of this unexpected event, including hemostatic—and inflammatory—changes to the endometrium after the acute immune response to the vaccine against SARS-CoV-2 virus. Furthermore, other unknown aspects such as the influence of the vaccine administration during the different menstrual phases and the potential effect of the vaccine adjuvants must be also dilucidated. From our point of view, the hypothesis of pandemic-related stress as an influencing factor on MC after vaccination could be discarded,1,7 but not on coronavirus disease. 43
Limitations
Certain limitations of the current study merit mention. While the sample analyzed was large enough to be statistically representative, a priori sample size calculation was not performed as the prevalence of this unexpected event was unknown. The experimental design may also have influenced the detection of significant differences, and causal relationships cannot be properly determined. The online questionnaire was not pre-validated due to the unexpected health crisis, but it gave us the opportunity to sample a target population in real time and investigate this unexpected event before it was reflected in official reports. In any case, although this type of data is potentially subject to errors such as recall bias or self-selection, self-reports are considered the gold standard for menstrual cycle data and are useful for quickly identifying potential signals or rare adverse events. Finally, we would like to point out that, as in similar studies, our results may not be applicable to countries other than Spain for some of the reasons mentioned above. In the view of foregoing, we agree that a longitudinal and multinational study could help to establish the cause-effect relationship and to clarify the factors influencing the occurrence of changes in the menstrual cycle after COVID-19 vaccination.
Conclusions
The findings of this study reinforce previous evidence and underscore the need for further research. Vaccine clinical trials should systematically account for potential menstrual side effects and the factors that may influence their occurrence, regardless of individuals’ reproductive status. Expanding research in this area would contribute directly to achieving the WHO Sustainable Development Goals, particularly goals 3 (“Good health and well-being”) and 5 (“Gender equality”). Moreover, generating robust evidence on this subject is vital for preparedness in the face of future viral outbreaks. Ensuring that healthcare professionals are equipped with up-to-date, evidence-based information is key to empowering patients to make informed decisions that support their quality of life. It also helps prevent the dismissal or gaslighting of individuals who report unexpected vaccine-related side effects.
Supplemental Material
sj-docx-2-whe-10.1177_17455057251406958 – Supplemental material for Characteristics and health factors influencing menstrual changes after COVID-19 vaccination: A Spanish retrospective observational study in currently menstruating women
Supplemental material, sj-docx-2-whe-10.1177_17455057251406958 for Characteristics and health factors influencing menstrual changes after COVID-19 vaccination: A Spanish retrospective observational study in currently menstruating women by Miriam Al-Adib, Ana B. Rodríguez and Cristina Carrasco in Women's Health
Supplemental Material
sj-pdf-1-whe-10.1177_17455057251406958 – Supplemental material for Characteristics and health factors influencing menstrual changes after COVID-19 vaccination: A Spanish retrospective observational study in currently menstruating women
Supplemental material, sj-pdf-1-whe-10.1177_17455057251406958 for Characteristics and health factors influencing menstrual changes after COVID-19 vaccination: A Spanish retrospective observational study in currently menstruating women by Miriam Al-Adib, Ana B. Rodríguez and Cristina Carrasco in Women's Health
Footnotes
Acknowledgements
The research team would like to thank all the participants who have collaborated in the study in an unselfish manner.
Ethics considerations
The study was conducted in accordance with the Declaration of Helsinki and approved by the Institutional Review Board of University of Extremadura (ref. 180/2021).
Consent to participate
An electronic informed consent was only obtained from those who agree to be contacted via email by the research group for additional data collection.
Consent for publication
Not applicable.
Author contributions
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Regional Government of Extremadura-ERDF funds (grant BBB021-GR21042); Cumlaude Lab under research agreement with the University of Extremadura (ref. 431/22). Neither the government agency nor the sponsoring company have played a role in the study design; in the collection, analysis, and interpretation of data; in the writing of the report; and in the decision to submit the article for publication.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
The data presented in this study are available on request from the corresponding author. The data are not publicly available due to ethical restrictions.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
