Abstract
Background:
After breast cancer (BC), women may face other severe symptoms such as sleep problems. The use of simple, fast, and reliable scales is necessary in the clinic to improve patient benefits, and sleep is an important aspect to be addressed.
Objective:
This study was conducted to adapt and validate the Spanish version of the satisfaction, alertness, timing, efficiency, and duration (SATED) scale for measuring sleep health in women who have completed treatment for BC in Spain (SATED-BC).
Design:
Cross-sectional study.
Methods:
The adaptation process involved adding a sixth item to the SATED-BC scale: “the impact of symptoms experienced after completing breast cancer treatment on sleep” item was not considered for scoring. The SATED-BC score ranged from 0 (poorest sleep health) to 10 (best sleep health). A validation analysis was performed using the Pittsburgh Sleep Quality Index, the Consensus Sleep Diary, and actigraphy, and the results were compared with those obtained using the SATED-BC scale.
Results:
The SATED-BC scale was reliable in terms of its internal consistency (Cronbach’s α = 0.70; McDonald’s ω = 0.72), showed high intrasubject reliability (r = 0.90), and was shown to be valid for use in women who have completed treatment for breast cancer.
Conclusion:
The SATED-BC scale is a reliable and valid tool for comprehensively evaluating sleep health in women who have completed treatment for breast cancer.
Introduction
The acute and long-term effects of breast cancer (BC) and its treatment can have important negative impacts on quality of life.1,2 However, these adverse effects may not always be clearly visible, silently impairing the health of women with BC.3,4 Epidemiological studies5,6 have found that sleep disturbances, for example, affect up to 90% of women who have completed treatment for BC, 7 and they may be exacerbated by the combination of medical treatments. 8 Women are more at risk of suffering from sleep disturbances than men and have poorer sleep health due to multiple reasons (age and hormonal changes across the life cycle 9 ); therefore, women are more susceptible to health problems associated with sleep disturbances, such as osteoporosis, depression, and anxiety. 10 These reasons, coupled with the high risk of sleep impairment due to cancer and treatments, make this population a high-risk group. 11 Indeed, sleep disturbances are among the top five most burdensome long-term effects of cancer treatment 12 and can be occur for more than 10 years after the completion of treatment. 13 Certainly, the physical and psychological impacts of sleep disturbances can have a strong impact on quality of life. 14 In addition, poor sleep health has even been linked to a greater risk of cancer recurrence 15 and higher mortality rates in BC survivors.16,17 Symptoms such as depression, pain, fatigue, and hot flashes, which are prevalent problems in women after treatment for BC, have been shown to be directly related to the subjective perception of sleep. 7 Therefore, several studies have emphasized that sleep should be a priority in the continuing care of individuals who have completed treatment for BC, 12 sleep should be comprehensively and continually assessed, and patients should be monitored for comorbidities. 18
Buysse 18 described sleep health as a “multidimensional pattern of sleep-wakefulness, adapted to individual, social and environmental demands, that promotes physical and mental well-being. Good sleep is characterized by subjective satisfaction, appropriate timing, adequate duration, high efficiency, and sustained alertness during waking hours.” Sleep health can therefore be examined via the latter five indicators. In practice, sleep has been examined using objective and subjective tools. The gold standard for the objective measurement of sleep is laboratory-based polysomnography, 19 followed by actigraphy. 20 However, these are quite expensive techniques; in particular, polysomnography requires a minimum of an overnight stay in a clinic or sleep laboratory. Moreover, while polysomnography may be the best method for diagnosing certain sleep disorders and for measuring sleep electrophysiology, its laboratory-based requirements mean that it cannot be used to measure the normal sleep characteristics of patients in their habitual sleep environments. Actigraphy also provides an objective measure of sleep–wake behavior and has an agreement rate of 90% with polysomnography. 20 Subjective tools are the most commonly used tools to evaluate sleep health in patients treated for cancer.21,22 These tools assess what people experience and consider important, but they can also measure habitual sleep patterns, are inexpensive, and are easy to administer. One of the most widely used tools to assess overall sleep quality is the Pittsburgh Sleep Quality Index (PSQI), 23 which has already been validated for use in patients with BC. 24 Unfortunately, the PSQI is not specific enough to detect factors influencing sleep in cancer patients. Additionally, the PSQI has the drawback of having a complicated scoring algorithm. Another option is the use of a sleep diary, 25 which is useful for prospectively measuring sleep patterns, can collect information on bedtime, sleep latency, sleep quality, etc., and can be complemented by actigraphy. 26 However, sleep diaries require a daily commitment if they are to be of any use. A short, simple assessment tool that specifically addresses sleep health-related issues is therefore needed.
The satisfaction, alertness, timing, efficiency, and duration (SATED) scale was developed to cover the five key dimensions of sleep health defined by Buysse 18 and is composed of five items assessing each of the dimensions, which are scored from 0 to 2. The sum of the scores of the five dimensions gives a total score (from 0 to 10) with higher values indicating better sleep health. The SATED scale has been validated for use in the general population in Portuguese, 27 English, 28 and Spanish, 29 providing reliable results. However, no version of the SATED scale has previously been validated in any cancer population, including BC patients. The sleep health dimensions of satisfaction, alertness, efficiency, and duration have been considered together in some tools, such as the PSQI, 24 which did not consider the timing dimension. From this idea, the present study emerged, which involved the adaptation and validation of the SATED scale for women who have completed treatment for BC in Spain (SATED-BC scale). For the adaptation of the scale, we decided to add an informative item that was not included in the final scoring; this item considered related symptoms such as psychological problems, fatigue, pain, and hot flashes, as these are likely present in this population and related to sleep health.
Methods
Study design
A cross-sectional study was conducted from June 2020 to March 2021 following the Strengthening the reporting of observational studies in epidemiology (STROBE) checklist, 30 as well as the recommendations of the standards for the development and adaptation of assessment instruments. 31
Subjects and setting
This study is part of the ATOPE PRO project (PI23/01646). Participants of this study were women after BC treatment who had participated in the preliminary validation of the ATOPE+ mobile app 32 (PI18/01840) for cancer patients, whose implementation will be improved in the mentioned project.
Study subjects were enrolled from the University Hospital Clínico San Cecilio and the University Hospital Virgen de las Nieves, Granada, Spain. All subjects were required to be over 18 years of age and to have completed medical-surgical treatment for BC (with the exception of hormone therapy) within the last 15 years. Subjects treated for other types of cancer, those who had a mental or psychological condition that impeded their ability to complete the assessment or that might affect the results (e.g., schizophrenia, anxiety, or depression), and those who had a diagnosed sleep disorder (e.g., obstructive sleep apnea), were excluded. For sample size calculation, we followed the recommendations of 5–10 participants per item.33,34 This yielded a minimum of 50 participants. In support of this sample size, a study of simulations reported that a sample size of 60 yielded convergent and admissible solutions using exploratory factor analysis in at least 95% of the cases where communality was wide. 35 Therefore, our sample size of 55 was likely to yield an acceptable solution.
Adaptation of the SATED scale to create the SATED-BC scale
The original developer of the SATED scale provided permission for the present adaptation to be made. 18 The SATED scale consists of five questions, each measuring a key item of sleep that influences health outcomes: satisfaction with sleep, alertness during waking hours, timing of sleep, sleep efficiency, and sleep duration. Each item is scored from 0 to 2, with 0 for “never” or “rarely,” 1 for “sometimes,” and 2 for “usually” or “always”. Respondents obtain a total score that ranges from 0 (the worst sleep health) to 10 (the best sleep health) 18 (no cutoff points have been established; Supplemental file 1). The Spanish version of the SATED scale was shown to have adequate internal consistency and reliability for use with the general population over 18 years of age. 29 For the adaptation, we added an additional informative item (not considered for the scoring and analysis) about the impact of symptoms experienced after completing treatment for BC on sleep, for which the following question was introduced: “6. Do any symptoms negatively influence your sleep?” This question had the same answer choices. The following question was also included in the scale: “6.1 Mark any symptom that has influenced your sleep with an X: fatigue, hot flashes, pain, depression/anxiety, worries, others” (Supplemental file 2). Since many subjects found Item four (sleep efficiency) difficult to understand in pilot testing, it was reworded to make it clearer. Specifically, the phrase “less than 30 min” was changed to “more than 30 min,” and the score for this item was inverted. Although it was not considered for the scoring, the new item added to complete the SATED-BC scale would also have an inverted score.
Data collection for reliability and validity analyses
The study subjects were contacted by telephone to confirm their participation. All subjects were asked to come to the PAIDI BIO-277 (CUIDATE) group’s facilities at the University of Granada for an explanation of the procedure, to sign an informed consent form, and to start the assessments. Data were collected by researchers with experience in the evaluation of patients with cancer and psychological variables. At baseline (t0), sociodemographic data were collected using a structured questionnaire, and clinical records were reviewed for cancer stage at diagnosis and the treatment received. All subjects completed the SATED-BC scale, and for its validation, they also completed the PSQI and completed the Consensus Sleep Diary for seven consecutive nights. All subjects wore a wrist accelerometer for these same consecutive nights and the corresponding days. At 1 week (t1), 20 subjects were contacted and invited to answer the items on the SATED-BC scale again in a test–retest reliability procedure. 36 The collected results were then compared. Details of the reference instruments used for this study are provided below.
Pittsburgh sleep quality index
The PSQI was used to measure the quality of sleep over the previous month through 18 questions that collected information for seven sleep domains: sleep quality, sleep latency, sleep duration, habitual sleep efficiency, sleep disturbances, the use of sleep medication, and daytime dysfunction. 23 Each domain score ranged from 0 to 3, and these were summed to provide a total score of 0–21. The PSQI has been shown to have adequate internal consistency and test–retest reliability in patients with BC. 24 Total scores higher than five indicated poor sleep quality; however, a cutoff score of eight has been suggested for populations susceptible to sleep issues, such as women with BC. 37
Consensus sleep diary
The consensus sleep diary was used to self-monitor sleep for seven consecutive nights 25 (a reliable tool for measuring sleep duration in adults when used over this length of time 38 ). Subjects were asked to complete the diary within an hour of getting out of bed in the morning. The diary collected information on bedtime and wake-up times, sleep quality, restful sleep, the number of awakenings and their duration, the time to fall asleep, the use of medication, and beverage consumption during the day, collecting information for four domains: sleep efficiency, total sleep time, the number of awakenings, and sleep latency.
Actigraphy
An Actigraph GT3X triaxial accelerometer was used to objectively assess sleep over the same seven consecutive nights as mentioned above. This device has been validated for measuring sleep, 39 including in patients with BC. 21 Subjects were asked to wear the accelerometer on their nondominant wrist (except in the presence of lymphoedema or when there was a risk of lymphoedema) for 24 h per day over eight consecutive days (thus covering the above seven nights). Epochs of 6-s light exposure and movement were transferred to a computer and analyzed using the ActiLife Sleep Analysis routine software (version 6.13.1), collecting information for five domains: latency, efficiency, total bedtime, total sleep time, and waking after sleep onset (WASO).
Statistical analysis
Normality was tested using the Kolmogorov‒Smirnov test. Demographic and clinical data, as well as the SATED-BC score, are presented as the means ± standard deviations (SDs) for continuous variables and as percentages for categorical variables. Missing data were not considered for statistical analysis. Missing data only occurred in secondary variables. Specifically, five data points were missed for the PSQI, seven were missed for the sleep diary, and six were missed for actigraphy. In these cases, analyses were conducted excluding cases per dependent variable. Significance was set at p < 0.05. All analyses were performed using STATA v.17 software (StataCorp LLC, College Station, TX, USA).
Factor analysis
Exploratory and confirmatory factor analyses were conducted to ensure that the proposed questionnaire focused on the single factor “sleep health in women treated for BC” using two independent subgroups randomly selected from the total sample (n = 30 for both analyses). A Kaiser‒Meyer‒Olkin (KMO) test and Bartlett’s test of sphericity were performed in both cases to determine the strength of the partial correlations and the adequacy of the factorial model obtained, respectively. A polychoric correlation matrix was used in the factorial analyses given the ordinal nature of the variables, the generalized least squares method was employed since the distribution of the scores for each SATED-BC item was not normal and the sample was relatively small, and Promax rotation (a type of oblique rotation that allows the factors to be correlated) was used given that high correlations between the items were expected. We assessed the fit of the one-factor model in the confirmatory factor analysis using recommended standard criteria and measures such as the root mean square error of approximation (RMSEA) (⩽0.06), the comparative fit index (CFI) (⩾0.95) and the standardized root mean residual (SRMR) (⩽0.08). 40
Reliability
The internal consistency of the proposed scale was analyzed using Cronbach’s coefficient α, McDonald’s omega, and item score-total score correlations. The scale was assumed to be ordinal, and Cronbach’s α and McDonald’s ω values >0.70 were deemed to indicate acceptable reliability. 41
The reproducibility of the scale results was tested using the test–retest method. Pearson correlation coefficients and intraclass correlation coefficients were used to verify the relationship between two measurements over time and to detect systematic bias. Intraclass correlations were performed for the subgroup of the 20 subjects who answered the items in the SATED-BC scale twice (with 1 week in between assessments); values >0.7 were deemed to indicate adequate test–retest reliability. Limits of agreement (LoA) were also calculated as the mean difference between the test and retest score ± 1.96 * SD of this mean; 95% of the differences were located between the upper and lower LoA.
Validation
Concurrent criterion validation of the SATED-BC satisfaction and alertness items was undertaken by determining the Spearman rank correlations with the different PSQI domains, and validation of the SATED-BC timing, efficiency, and duration items was performed by their Spearman rank correlation with the Consensus Sleep Diary and actigraphy domains.
Results
Subject characteristics
Out of the 68 potential subjects, 5 did not meet the inclusion criteria (3 women had recurrent cancer and 2 had a diagnosed sleep disorder), and 8 declined to participate mainly because of the COVID-19 pandemic (refusal of physical contact for the delivery of materials and emotional discomfort); consequently, the final sample consisted of 55 subjects.
Table 1 shows the subjects’ sociodemographic and clinical characteristics. At t0, the mean age was 54.41 ± 9.45 years, the mean BMI was 25.69 ± 3.92 kg/m2, and the majority (76.9%) of the sample was postmenopausal. The majority of subjects had a university level education (55.4%) and were married (70%). Most had stage II BC at the time of diagnosis (55.3%), followed by stage III (29.8%), stage I (12.8%), and IV (2.1%). A total of 73.3% of the sample received a combination of surgery, radiotherapy and chemotherapy, 8.3% received surgery and radiotherapy, 10% received surgery and chemotherapy, and one participant received surgery alone.
Main demographical and clinical characteristics.
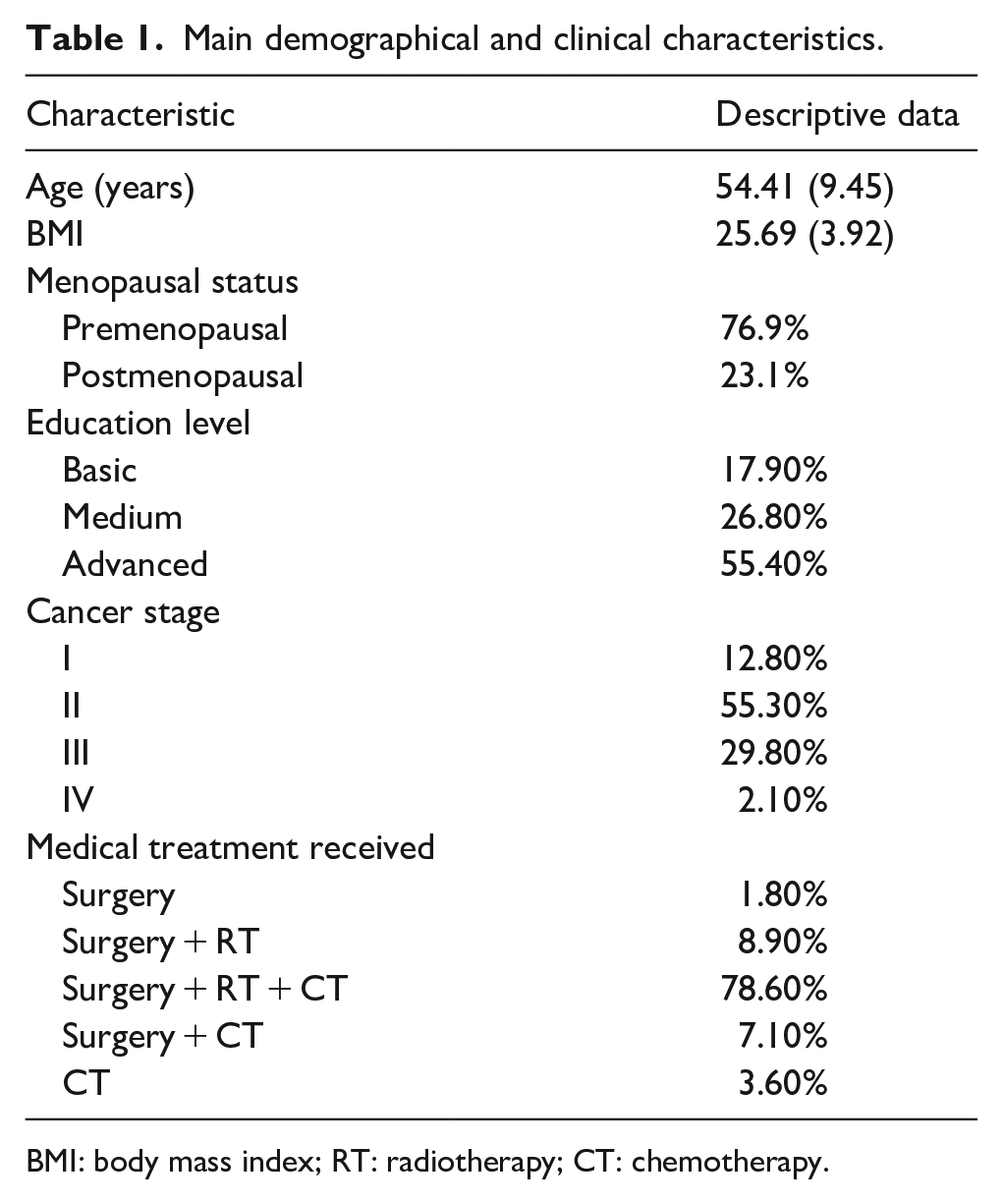
BMI: body mass index; RT: radiotherapy; CT: chemotherapy.
The SATED-BC scores were normally distributed (p < 0.05), and the mean summed score was 6.48 ± 3.09.
Factor analysis
The adequacy of the sample for exploratory factor analysis was verified (KMO = 0.532, Bartlett: χ2 = 34.809, df = 10, p < 0.001), and for confirmatory factor analysis, Mardia’s test showed that the multivariate normality assumption was met. The optimal implementation of parallel analysis showed a unifactorial solution. Therefore, exploratory and confirmatory analyses were performed considering only one dimension. The factor loadings of the items were above .40 in all cases (Table 2). Furthermore, the goodness-of-fit indices of the model showed adequate values (RMSEA = 0.000; CFI = 1.000; SRMR = 0.038) after confirmatory analysis.
Saturations of the items in the rotated factor of the exploratory factor analysis.

EFA: exploratory factor analysis; CFA: confirmatory factor analysis; h2: communality.
Reliability
The internal consistency of the SATED-BC scale was adequate (Cronbach’s α = 0.70; McDonald’s ω = 0.72). Table 3 shows the individual correlation analyses for each of the scores with the total score; significant correlations were found in all cases, ranging from r = 0.414 for the alertness item to r = 0.767 for the duration item.
Correlation between individual score of the SATED items and the total score.
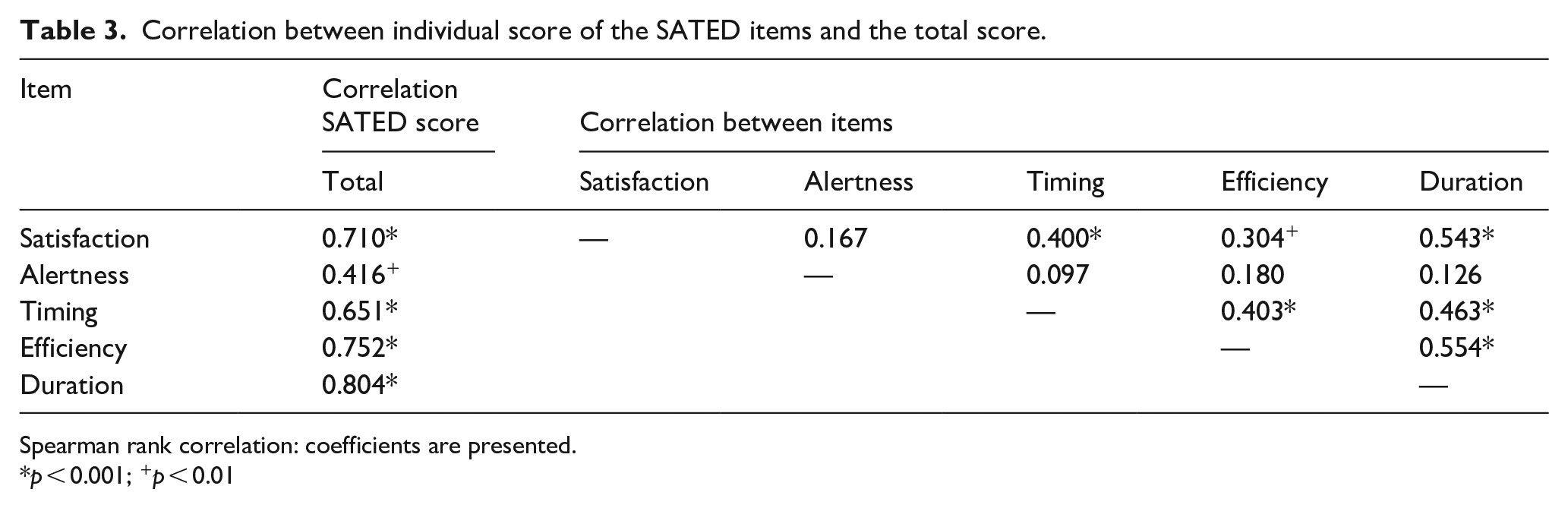
Spearman rank correlation: coefficients are presented.
p < 0.001; +p < 0.01
The test–retest reliability score, assessed via Pearson correlation and corroborated by intraclass correlation testing for 20 subjects, was 0.91 in both cases (p < 0.001; see Table 4). The Bland‒Altman plot (see Figure 1) showed a mean difference of 0.100 points, with LoAs between −2.436 and 2.636.
Test-retest reliability (n = 20).

Intraclass correlation coefficient and Pearson correlation coefficient (r) between baseline SATED-BC and one-week SATED-BC measurements. ICC: intraclass correlation coefficient; CI: confidence interval.

Bland–Altman scatterplots for SATED-BC.
Validity
Table 5 shows the Spearman rank correlation coefficients for the individual scores of the different items of the SATED-BC scale with the reference instruments of the consensus sleep diary, PSQI, and actigraphy domains. Satisfaction and alertness correlated significantly (in the expected directions) with the PSQI domains of sleep quality and daytime dysfunction, respectively. However, the SATED-BC timing item correlated only with the PSQI sleep latency domain. The SATED-BC item efficiency correlated (in the expected direction) significantly with the Consensus Sleep Diary total sleep time domain, the PSQI habitual sleep efficiency domain, and the actigraphy sleep efficiency domain. The SATED-BC duration item also correlated significantly with the Consensus Sleep Diary total sleep time domain and the PSQI sleep duration domain but not with the actigraphy total sleep time domain. Finally, the overall SATED-BC score showed a significant correlation with all PSQI domains, as well as with the consensus sleep diary domains.
Correlation between individual SATED-BC scale items, and sleep diary, PSQI, and actigraphy domains.
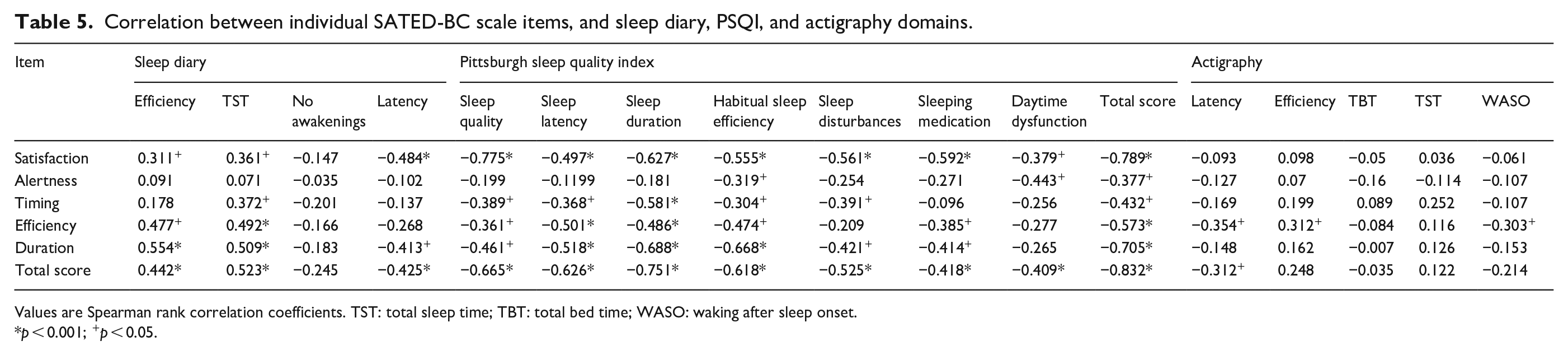
Values are Spearman rank correlation coefficients. TST: total sleep time; TBT: total bed time; WASO: waking after sleep onset.
p < 0.001; +p < 0.05.
Discussion
This study presents the adaptation and validation of the SATED-BC scale in Spanish. Our results confirm that this scale has adequate reliability in terms of internal consistency and intrasubject reliability, as well as good validity values.
The present exploratory and confirmatory factor analyses show that the SATED-BC scale adequately focuses on “sleep health in women treated for BC.” The scale showed adequate internal consistency (Cronbach’s α = 0.70) and intrasubject reliability (r = 0.90) and was found to be valid for use in subjects who have completed treatment for BC.
In the present work, one factor loading was found to be slightly low in the exploratory factor analysis (alertness item at <0.040). However, it was not omitted from the confirmatory analysis since it is generally considered important for measuring sleep health. Similar results regarding the factor loading for alertness were reported in a previous study. 29 As expected, the obtained internal consistency of the SATED-BC scale agreed with the Spanish version of the SATED scale developed for use in the general population (Cronbach’s α = 0.77). 29 The validation of the English version 28 showed a lower internal consistency value (Cronbach’s α = 0.64), but in that work, the RU-SATED version was used, which includes the “regularity of sleep” item. A higher internal consistency value was obtained in the validation of the Portuguese version (Cronbach’s α = 0.85). 27 The test–retest reliability for the SATED-BC scale was r = 0.91, which was very similar to the r = 0.93 reported in the Spanish validation study 29 (both were performed over the same time period of 7 days).
As reported in earlier studies,27,29 significant correlations were observed between SATED-BC satisfaction item and the PSQI sleep quality domain (r = −0.775). The SATED-BC alertness item correlated with the PSQI daytime dysfunction domain (r = −0.443) (the Spanish validation study for the SATED scale 29 found a correlation between the alertness item and the score returned by the Epworth Somnolence Scale). In addition, the present work detected a significant correlation between the SATED-BC timing item and the PSQI sleep latency domain (r = −0.368), whereas in the Spanish SATED validation study, the authors observed an association between timing and the Consensus Sleep Diary latency domain. 29 No previous validation of the Spanish SATED scale has involved actigraphy. The present results showed a significant correlation between the SATED-BC efficiency item and the actigraphy domains of efficiency, latency, and WASO. The nonsignificant correlations detected might be explained by the disturbed or fragmented sleep experienced by women who have completed treatment for BC, which could make actigraphy readings less accurate. 38
Limitations
This study has a number of limitations. Polysomnography (the gold standard for assessing sleep health) was not used, but rather, self-reporting and actigraphy were relied upon. Indeed, self-reporting is better for the present kind of study. 18 The new version of the SATED scale 28 was published after the recruitment of participants for this study; therefore, the regularity item was not included in the SATED-BC version of the scale. The sample was also small in relation to those usually used in factor analysis and the power analysis for sample size calculation was not done.
Strengths
This work addresses an important clinical problem: the adaptation and validation of a rapid, simple tool for use in patients who have completed treatment for BC. The adaptation of this scale allows for the comprehensive sleep assessment of such persons and takes the informative new items for factors associated with sleep onset and maintenance, such as fatigue, pain, depression, anxiety, and hot flashes, into consideration. Another strength of this work is that the validated scale can be extrapolated not only to the survival phase but also to other disease processes (i.e., the active treatment phase) where the assessment of sleep health is important and where the considered symptom cluster is present. In addition, it could be applicable to patients with other types of cancer, such as lung and head and neck tumors, 42 where sleep health remains an aspect for consideration to try to improve the quality of life of these patients.
Conclusion
The SATED-BC scale has been found to have good reliability and validity for comprehensively evaluating sleep health in women who have completed treatment for BC. These results provide evidence in favor of the usefulness of the SATED-BC scale to assess sleep satisfaction, alertness, timing, efficiency, and duration of sleep in a quick and easy way. Understanding sleep health in this population is crucial to plan interventions aimed not only at this problem but also at improving the general well-being and health-related quality of life of these women. Further studies would be needed to corroborate its usefulness at other times of the disease, as well as in other groups of cancer patients.
Supplemental Material
sj-doc-3-whe-10.1177_17455057241309779 – Supplemental material for Adapting and validating the satisfaction, alertness, timing, efficiency, and duration-breast cancer (SATED-BC) scale for measuring sleep health in Spanish women treated for breast cancer
Supplemental material, sj-doc-3-whe-10.1177_17455057241309779 for Adapting and validating the satisfaction, alertness, timing, efficiency, and duration-breast cancer (SATED-BC) scale for measuring sleep health in Spanish women treated for breast cancer by Ángela González-Santos, Mario Lozano-Lozano, Irene Cantarero-Villanueva, Paula Postigo-Martín, Lydia Martín-Martín, Rocío Gil-Gutiérrez, Roberto Muelas-Lobato and Maria Lopez-Garzon in Women’s Health
Supplemental Material
sj-docx-1-whe-10.1177_17455057241309779 – Supplemental material for Adapting and validating the satisfaction, alertness, timing, efficiency, and duration-breast cancer (SATED-BC) scale for measuring sleep health in Spanish women treated for breast cancer
Supplemental material, sj-docx-1-whe-10.1177_17455057241309779 for Adapting and validating the satisfaction, alertness, timing, efficiency, and duration-breast cancer (SATED-BC) scale for measuring sleep health in Spanish women treated for breast cancer by Ángela González-Santos, Mario Lozano-Lozano, Irene Cantarero-Villanueva, Paula Postigo-Martín, Lydia Martín-Martín, Rocío Gil-Gutiérrez, Roberto Muelas-Lobato and Maria Lopez-Garzon in Women’s Health
Supplemental Material
sj-docx-2-whe-10.1177_17455057241309779 – Supplemental material for Adapting and validating the satisfaction, alertness, timing, efficiency, and duration-breast cancer (SATED-BC) scale for measuring sleep health in Spanish women treated for breast cancer
Supplemental material, sj-docx-2-whe-10.1177_17455057241309779 for Adapting and validating the satisfaction, alertness, timing, efficiency, and duration-breast cancer (SATED-BC) scale for measuring sleep health in Spanish women treated for breast cancer by Ángela González-Santos, Mario Lozano-Lozano, Irene Cantarero-Villanueva, Paula Postigo-Martín, Lydia Martín-Martín, Rocío Gil-Gutiérrez, Roberto Muelas-Lobato and Maria Lopez-Garzon in Women’s Health
Footnotes
Acknowledgements
The authors thank Dr. Daniel Buysse for the methodological support and reviewing the manuscript, as well as all the women who participated in the study.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
