Abstract
Background:
Robotics-assisted surgery (RAS) offers several advantages over traditional laparoscopic surgery, such as enhanced precision, dexterity, and ergonomics, and allows stable movements with tremor filtering and motion scaling. The new multi-modular Hugo™ RAS system is commercially available in certain countries and is used in urological, gynecological, and general surgical procedures.
Objectives:
To document the experience with the use of the Hugo™ RAS system in 20 patients who underwent surgery for various gynecological conditions at a hospital in Hyderabad, India.
Design:
Real-world single-center study.
Methods:
Twenty patients (45.3 ± 6.5 years) who were admitted to a tertiary care center (September 2022–2023) underwent hysterectomy (n = 18), accessory and cavitated uterine mass excision (n = 1), and myomectomy (n = 1) using the Hugo™ RAS system. The evaluated outcome parameters included docking time, console time, blood loss (intraoperative and postoperative), length of hospital stay, postoperative complications, and postoperative pain perception measured as per visual analog scale (VAS) scores at multiple time points.
Results:
The mean docking time was 6.3 ± 2.0 min, and the mean console time was 86.9 ± 20.3 min. Blood loss (intraoperative and postoperative drain) was 103.5 ± 62.4 mL, not requiring blood transfusion for any patient. Per hospital norms, each patient had a hospital stay lasting for 2 days. None of the patients experienced immediate postoperative complications. Minor late complications were observed in two patients. Postoperative pain perception decreased with time. VAS scores were 3.2 ± 0.4 (1 h), 2.2 ± 0.4 (6 h), and 1.0 ± 0.0 (12 h), and no pain was perceived 1 week after surgery.
Conclusion:
Based on initial experience, the Hugo™ RAS system provides favorable results for gynecological patients, with benefits including efficient docking time, short surgery duration, minimal blood loss, short hospital stay, few postoperative complications, and low pain perception. Future studies comparing the Hugo™ RAS system with other robotic surgical platforms in gynecological procedures are essential.
Introduction
Minimally invasive surgical methods in gynecology are alternatives to traditional open surgery and offer numerous benefits compared to traditional laparotomy, including decreased estimated blood loss, a lower likelihood of complications, and shorter hospitalization and recovery periods. 1 These methods mainly include laparoscopy, employed for managing a variety of benign gynecological conditions. 2
Conventional laparoscopic surgery presents some limitations. 3 The surgeon’s limited field of view requires him/her to rely on a camera assistant, who maneuvers the laparoscope based on verbal instructions from the surgeon. Moreover, owing to the 2D perspective provided, depth perception and spatial awareness are restricted. Although laparoscopes have been modified to provide high-definition 3D vision to address this issue, the need for a camera assistant remains, and the “camera shake” due to fatigue of the person holding the camera could disorient the surgeon. 4 Laparoscope holders were among the first robots in laparoscopic surgery, with the AESOP® 3000 robot introduced in 1994. 5 The fixed positions of trocars used in conventional laparoscopy hinder the surgeon’s ability to maneuver with flexibility. 3 The master–slave robots enhance the surgeon’s flexibility by providing six degrees of freedom of hand motion at the console. 4 The da Vinci surgical system, introduced in the year 2000, was the first Food and Drug Administration-approved robotics-assisted surgery (RAS) system for general laparoscopic surgery. Since then, RAS systems have advanced, offering improved precision, dexterity, and ergonomics over conventional laparoscopic surgery, along with stabilized movements, tremor filtration, and motion scaling.6,7
In RAS systems, the 3D high-definition magnified images enhance depth perception, the robotic arm carts offer improved dexterity with increased degrees of freedom, tremors are mitigated through vibration filtration, wristed instruments counter fulcrum effects, robot-held endoscopes with direct control reduce dependence on assistants, and adjustable seats and user-friendly interfaces afford surgeons greater comfort during procedures compared to conventional laparoscopic systems. 8 Robotic-assisted techniques have found applications in a wide range of surgical fields, including hepatobiliary, pancreatic, endocrine, bariatric, anti-reflux, colorectal, pediatric, and gastric oncology surgeries, as well as hernia, prostatectomy, or abdominal wall reconstruction procedures.9,10 In the field of gynecology, robotic surgery is not only used for benign conditions such as endometriosis, pelvic organ prolapse, pelvic pain, sacropexy, tubal recanalization, uterine fibroids, and abnormal uterine bleeding but also for treating malignant conditions such as cancers of the ovary, endometrium, and cervix and performing lymphadenectomy.11,12 Patients undergoing gynecological procedures with RAS systems have been reported to exhibit early recoveries, shorter hospital stays, and fewer postoperative complications, including primary or secondary hemorrhage. 13
The Hugo™ RAS system (Medtronic Inc, USA) was first used at Clínica Santa María (Chile) for robotic prostatectomy in June 2021, and since then, several hospitals worldwide have adopted this RAS system. 14 The system comprises arm carts, a surgeon console, and a system tower (Figure 1) and is compatible with wristed articulating instruments of the same company, a 3D endoscope, the VITOM® 3D visualization system (Karl Storz SE & Co. KG, Germany), the Covidien ValleyLab™ FT10 high-frequency electrosurgical generator (Medtronic Inc, USA), and the VersaOne™ reusable positioning trocar system (User guide). The system provides high-resolution 3D images. The surgeon’s console has an interactive high-definition display with a highly adjustable ergonomic set-up. The surgeon’s wrist motion is translated into the robotic instrument’s actions with two hand controllers, facilitating fluid and precise movements. The sensors on the console track the surgeon’s 3D glasses, pausing instrument movement when the surgeon looks away from the display screen (User guide). The system’s tilt angle capability allows customization of the surgical approach according to the patient’s characteristics, especially in complex clinical scenarios. 15
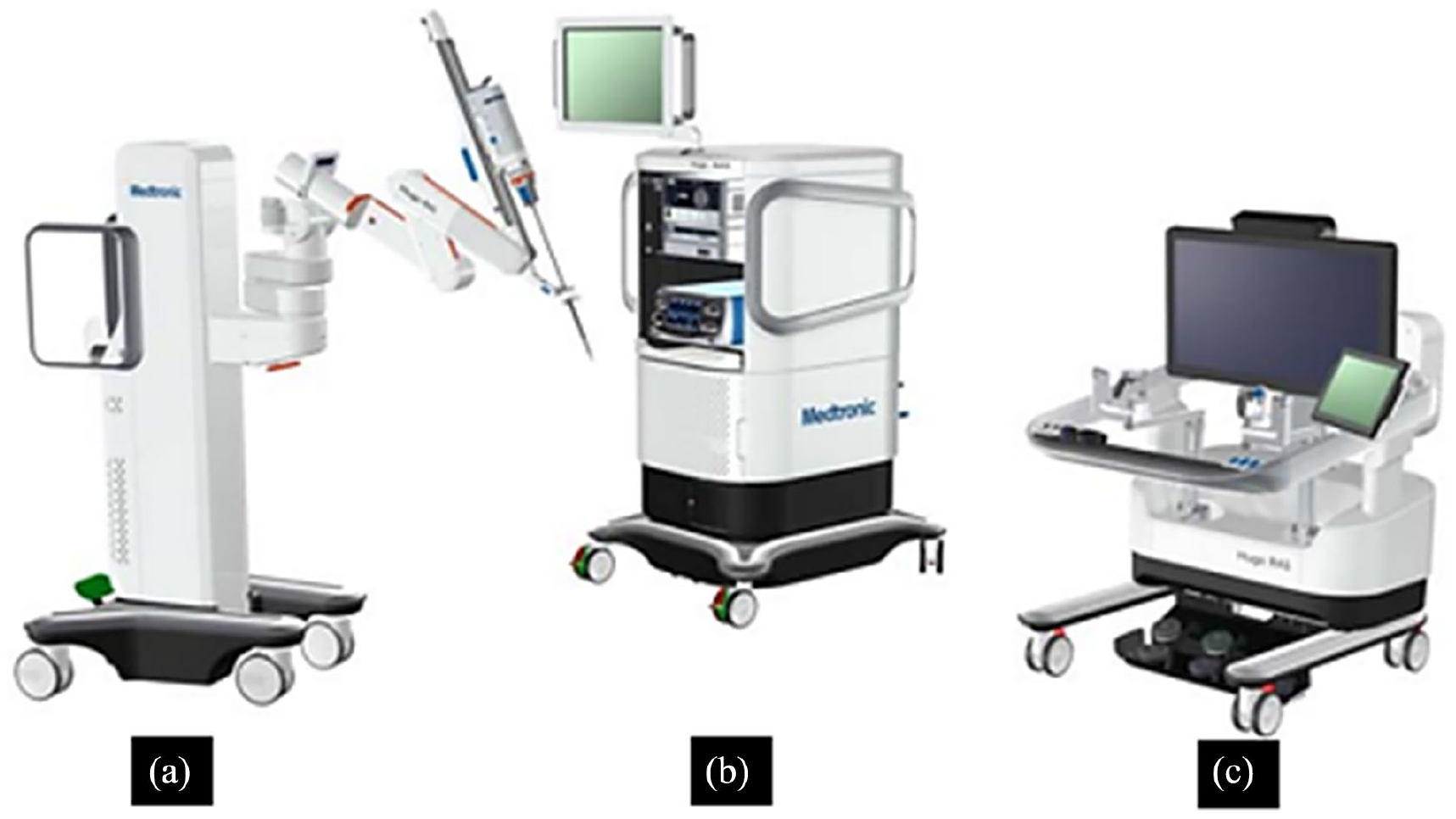
Components of the Hugo™ RAS system: (a) arm carts, (b) system tower, and (c) surgeon console.
Previous reports on the Hugo™ RAS system have demonstrated favorable results with no technical failures or intraoperative complications in urological procedures16,17; however, data on its use in gynecological surgeries is limited. This underscores the importance of surgeons sharing their experiences and outcomes to further solidify the evidence base for the application of robotic-assisted surgery in the field of gynecology.
In India, the first surgical robot was installed in 2006. By 2019, there were 66 robotic centers with 71 systems installed and more than 500 surgeons skilled in robotic surgery. 18 This article describes the initial experience of using the Hugo™ RAS system in gynecological surgeries at a single tertiary care center in India.
Methodology
Study design and patient population
This report presents a real-world single-center experience of using the Hugo™ RAS system on 20 patients who underwent surgeries for a range of gynecological conditions at the CARE Hospital in Banjara Hills, Hyderabad, from September 2022 to 2023. Since the study was aimed at reporting on the observations and experiences from a single center and was not intended to assess a specific hypothesis, no sample size calculation was performed. The STrengthening the Reporting of OBservational studies in Epidemiology (STROBE) reporting guideline was employed for this study. 19
Preprocedural evaluation
The patients underwent preoperative assessment for eligibility and were included in the study if they were fit for general anesthesia, required surgery for gynecological conditions, were undergoing surgery with Hugo™ RAS system, and provided written informed consent per institution and geographic requirement. Patients were excluded from the study if they were, or were expected to be, unavailable for follow-up if local regulations prohibited their participation, or if they were already enrolled in, or had intentions to enroll in, concurrent investigations involving drugs or devices that could potentially introduce bias to the results of this study. Written informed consent was obtained from all patients, and the study was conducted in accordance with the principles of the Declaration of Helsinki.
Patient position and port placements
Patients were laid in a supine steep Trendelenburg position of >25° with their legs in the low-mid lithotomy position (Figure 2(a)). There was no roll (0°), and the height of the bed was above 70 on the scale of the cart column. The endoscopic ports were placed near the patients’ umbilicus with a caudal offset of 5 cm between the right-hand surgeon port and the endoscopic port. The left-hand surgeon port was kept lateral to the endoscopic port at a distance of 14 cm. The reverse port was also placed lateral to the endoscopic port and the right-hand surgeon port at a distance of 8 cm from both. The assist port was placed 5 cm away from the endoscopic and left-hand surgeon ports. A distance of 2 cm was also maintained between any bony prominences and all the ports (Figure 2(b)). For the procedure, the tilt was adjusted for the arms, the carts were placed in the range of 18–24 inches, and the brakes were set. The laser was set parallel to the bed, and the position button was used to move the arms to the desired positions while docking to the port at the angles prescribed in the Medtronic user guide (Figure 3). Once the placements of the arms were finalized, they were confirmed on the tower screen. Since the cases reported in this paper were the initial cases operated with the system, all surgeries were performed using three arms. However, as per the advanced practice, the technique is now modified and the surgeries currently performed at the center are by using both, three arms and two arms. In case of the two-arm technique, one arm is for unipolar and other for bipolar.

Patient position and port placements. (a) Patient position during surgery and (b) port positions for gynecological surgeries.

Docking positions of the arm carts and position of the assistant.
Surgeries were performed by a single operator who had clinical experience of minimal access surgeries for about 28 years. The surgeries were assisted by a single assistant having clinical experience of minimal access surgeries for about 7 years.
Outcome measures
The outcome parameters evaluated were docking time (time from the first order given to position the robot to start at the console) and console time (time from the start of the procedure to suturing of the surgical incision), blood loss (intraoperative and postoperative drainage), and length of hospital stay. Postoperative complications were graded according to the Clavien-Dindo classification. 20 Postoperative pain perception was evaluated using a visual analog scale (VAS) at 1, 6, and 12 h as well as 1 week after surgery.
Data analysis
Descriptive statistics were performed using R software version 4.3.2. Continuous variables were presented as mean ± standard deviation (SD).
Results
Patient characteristics
All 20 patients (mean age 45.3 ± 6.5 (range 25–58) years) successfully underwent robotic surgery using the Hugo™ RAS system. Hysterectomy was performed on 18 patients, accessory and cavitated uterine mass excision on one patient, and myomectomy on one patient. Patient characteristics are shown in Table 1. All patients were followed up to 1-week post-surgery.
Patient characteristics.

ACUM: accessory and cavitated uterine mass; AUB: abdominal uterine bleeding; PID: pelvic inflammatory disease.
Procedural outcomes
The mean docking time was 6.3 ± 2.0 min, and the mean duration of the surgery was 86.9 ± 20.3 (range: 55.5–119.0) min. The mean blood loss (intraoperative and postoperative drainage) was 103.5 ± 62.4 (range: 50–250) mL; it was minimal in all cases with no requirement for blood transfusions. The duration of hospital stay was 2 days as per hospital norms.
Postoperative complications
All surgical procedures were uneventful, and no immediate complications were observed. Grade 1 complications according to the Clavien-Dindo classification were observed in two patients. These patients experienced minor late complications during the third week following surgery, characterized by vaginal spotting secondary to granulation tissue formation at the vaginal vault. Complications were addressed through a conservative approach on an outpatient department basis.
Pain perception
Postoperative pain perception declined over time; the mean VAS scores were 3.2 ± 0.4, 2.2 ± 0.4, and 1.0 ± 0.0 at 1, 6, and 12 h, respectively. No pain was perceived at 1 week after surgery.
Discussion
Robotic surgery is often seen as an extension or alternative to laparoscopic surgery rather than a definitively superior technology, despite having the benefits of being minimally invasive compared to open surgery, and there exists conflicting research on the comparative advantages and drawbacks of both robotic-assisted and laparoscopic approaches. 21 This report documents the experience of using the Hugo™ RAS system for performing gynecological surgical procedures on 20 patients at a single center in India.
Docking and operative times
Compared with conventional laparoscopic methods, RAS is generally associated with a longer operative time, which includes the time required for placement of ports, docking time, console time, and closing time. 22 The docking phase encompasses activities such as moving the robotic arms into the surgical field, setting the arms into their respective port sites, and inserting the robotic instruments in the abdomen, which account for approximately 3.5% of the operative time. 22 A systematic review of reports on hysterectomies performed using RAS systems (2016–2021) presented a wide variation in the operative time (75.4–306.0 min). 23 The docking time and console time for the Hugo™ RAS system in the present study were 6.3 ± 2.0 min and 86.9 ± 20.3 min, respectively. In a prospective observational study that used the Hugo™ RAS system for 192 gynecological surgical procedures performed at a single center in Italy, the docking time was reported to be 5.1 ± 2.0 min and the console time was 111.5 ± 40.2 min. 22 The data indicate that robotic set-up and docking with the new Hugo™ RAS system can be performed efficiently and that the specific robotic docking learning curve is comparable to preexisting data for other platforms. The Hugo™ RAS system has four independent arm carts, which makes it easier to park the device away from the patient while enabling precise docking of the surgical arm at the desired location on the surgical field. This convenience stands in contrast to the da Vinci® Surgical System, which features four arms attached to a single base. 24 In a prospective observational study in the Netherlands, 65 robot-assisted operations, including 14 gynecological procedures, were performed using the da Vinci Xi system (Intuitive Surgical Inc., USA), and the docking time was reported to be 7.8 ± 2.7 min. 25 The time required for docking and the surgery is largely influenced by the experience of the surgeon, the complexity of the surgery, and the techniques used for the surgery.11,23 Nevertheless, the presence of a learning curve underscores the importance of surgeons receiving comprehensive training to proficiently handle the docking process and carry out successful procedures with the Hugo™ RAS system. Preclinical laboratory training is crucial for ensuring optimal performance by surgeons. A structured and certified training program beforehand, focusing on trocar positioning and familiarizing with operating room protocols through dry simulations with the new system, is important for achieving significant outcomes. This training also prepares surgeons to effectively manage any technical challenges that may arise during surgery.26,27
Blood loss
The patients enrolled in this study had minimal blood loss (103.5 ± 62.43 mL), and none of them required blood transfusion. There is a general trend of reduced blood loss with hysterectomy performed using a RAS system compared with conventional laparoscopic hysterectomy,28,29 but there are also reports of no significant advantage of using a RAS system over conventional laparoscopy, with the estimated blood loss reported to be in the range of 50–237 mL with robotic hysterectomy and 50–230.5 mL in patients who underwent conventional laparoscopic hysterectomy. 23
Length of hospital stay
In the present study, the hospital stay after surgical procedures performed using the Hugo™ RAS system was 2 days per hospital norms, and no patient reported a longer duration. A systematic chart review of 152 women who underwent robotic hysterectomy for benign indications suggested that robotic assistance contributes to a short hospital stay even in patients with complex pathology. 30 However, there is a lack of evidence suggesting a statistically significant difference in the length of hospital stay when comparing gynecological procedures conducted with and without the use of a RAS system. For example, in a retrospective analysis (2018–2019) of surgical outcomes for patients who underwent hysterectomy for early endometrial cancer or benign indications using the da Vinci Xi system, the length of hospital stay was 3.74 ± 1.92 days, while it was 3.71 ± 2.29 days for patients who underwent conventional laparoscopic hysterectomy with no significant difference (p = 0.150). 31 It is important to note that the length of hospital stay in the present study was as per the hospital norm for this particular study. The patient was hospitalized 1 day prior to the surgery for preprocedural tests and to decide on their inclusion/exclusion in the study. Post-surgery, the pain scores were evaluated, the bowel movement was assessed the following day, and the patient was discharged, resulting in a 36-h stay. However, there is a possibility that the patient could have been discharged on the same day of the surgery in the absence of such a norm. Nevertheless, consideration was also given to the insurance practices followed in India. As per insurance protocols, medical expenses can only be claimed if the patient is hospitalized for a minimum of 24 h.
Postoperative complications
Evidence from a retrospective cohort study on patients who underwent robotic-assisted laparoscopic hysterectomy (n = 745) and conventional laparoscopic hysterectomy (n = 688) suggested that while rates of major postoperative complications were similar between robotic and laparoscopic surgery, the robotic approach exhibited notable advantages with lower rates of minor postoperative complications and conversion to laparotomy. 32 In this study, only two patients experienced postoperative complications when the Hugo™ RAS system was employed, and both cases involved late complications successfully addressed through a conservative approach. The variations in the incidences of postoperative complications could be due to confounding factors such as differences in patient characteristics, complexity of the surgery, presence of comorbidities, and the experience of the surgeon.33 –35
Obesity is a major risk factor for postoperative complications in patients undergoing hysterectomy, but robot-assisted laparoscopic hysterectomy is associated with lower morbidity and reduced length of hospital stay compared with conventional laparoscopic hysterectomy in obese patients.30,35 Robot-assisted laparoscopic surgery is reported to be more feasible than conventional laparoscopic surgery for obese patients and those with poor respiratory compliance. 36 A retrospective chart review of 1032 obese patients (Body Mass Index > 30 kg/m2) at two academic institutions in the United States showed that most obese patients could well tolerate gynecological surgeries performed using a RAS system, and only 3% of the study population developed pulmonary complications. 37
Pain
Compared with laparoscopic surgery, robotic surgery is considered to be associated with less postoperative pain and reduced need for analgesia, which could be possibly due to the instrument’s wrist-like movement occurring within the abdominal cavity and the minimized trauma to the abdominal wall. 33 In the present study, the VAS score for postoperative pain at 6 h was 2.2 with the Hugo™ RAS system. In a retrospective analysis of patients who underwent robotic-assisted radical prostatectomy during 2019–2021 using the da Vinci Xi system (n = 100), the postoperative pain score was reported to be 2.5 at 6 h. 38
Navigating robotic surgery challenges with the Hugo™ RAS system despite the advantages associated with robotic surgery, the challenges cannot be overlooked. The absence of tactile feedback, the surgeon’s distant positioning from the patient, and increased expenses relative to conventional laparoscopic surgery are some of the prominent disadvantages of robot-assisted laparoscopic surgery. 36
Lack of haptic feedback has been a concern with the da Vinci systems. 39 In contrast, the Hugo™ RAS system offers haptic feedback to surgeons, allowing them to identify and prevent tissue damage during the surgical procedure. 17
Various safety mechanisms incorporated in the Hugo™ RAS system enhance surgical precision and minimize risks, thereby ensuring patient safety. 17 The six hinges on the arm carts greatly increase the range of motion, creating a configured arrangement that minimizes the risk of collisions. 40 The higher expenses associated with the da Vinci systems primarily arise from the acquisition and maintenance of the system, increased instrument costs, the use of semi-disposable tools, and prolonged operative times. 39 The Hugo™ RAS system is more sustainable because the tower could be adapted for use with pure laparoscopy and one of the arms could function independently as a stationary assistant in traditional laparoscopic procedures, thereby reducing the need for trained personnel. 39 Analysis of the cost of robotic-assisted hysterectomies at a community teaching hospital in the United States demonstrated a 15.5% reduction in the total cost and 14.3% in the operative cost of hysterectomy procedures after 5 years of experience with the RAS system. 41 Thus, costs associated with robotic surgery are expected to diminish over time with improved surgical expertise, reduced operative duration and length of hospital stay, increased popularity of high-volume robotic centers, and adoption of minimally invasive surgery. 36
Comparing the Hugo™ RAS system with other robotic surgical platforms is essential as it will help in identifying the advantages of Hugo™ RAS over other systems in terms of efficiency, precision, outcomes, and potential areas of improvement. 42
Limitations of this study
This report is limited in its scope as it only records the early experiences of the usage of the Hugo™ RAS system from a single center and has a small sample size of 20 patients. Given the limited case volume, surgeons utilizing the Hugo™ RAS system may still be in the initial stages of their learning process.
Conclusion
Drawing from the initial experience, the use of the Hugo™ RAS system provides favorable outcomes for patients with gynecological conditions, demonstrating advantages in terms of short docking time and surgery duration, minimal blood loss, short length of hospital stay, few postoperative complications, and low pain perception. Further studies with more extensive sample sizes comparing the Hugo™ RAS system with other RAS systems and traditional laparoscopic procedures in the gynecological domain are needed.
Supplemental Material
sj-doc-1-whe-10.1177_17455057241302581 – Supplemental material for Robotics-assisted surgery in gynecology: A single-center experience with the Hugo™ RAS system in India
Supplemental material, sj-doc-1-whe-10.1177_17455057241302581 for Robotics-assisted surgery in gynecology: A single-center experience with the Hugo™ RAS system in India by Manjula Anagani, Ravula Sindura Ganga and Snehalatha Paritala in Women’s Health
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
