Abstract
Background:
Hepatitis B infection has been associated with the development of gestational diabetes but the underlying mechanism is not known.
Objective:
To examine associations between viral activity, gestational diabetes mellitus (GDM), and insulin resistance in pregnant people with chronic hepatitis B infection (HBV).
Design:
Prospective cohort study across three tertiary maternity centres in Melbourne, Australia, between May 2021 and April 2023.
Methods:
Participants were followed prospectively through pregnancy to evaluate subsequent GDM diagnosis. Demographics, pregnancy outcomes, and markers of viral activity were compared between those with GDM versus those without. Logistic regression analysis was performed pre- and post-adjustment for known confounders. Sub-group analysis of participants from South East Asia (SEA) was performed. Outcome measures included GDM diagnosis, insulin resistance (Homeostatic Model Assessment Insulin Resistance score (HOMA-IR) score), HBV activity as measured by liver function tests, HBV viral load, hepatitis B e antigen, and quantitative hepatitis B surface antigen (quantHBsAg).
Results:
A total of 113 women were recruited. One third (38/112, 33.9%) developed GDM, mostly diagnosed on isolated postprandial hyperglycaemia (25/38, 65.8%). Over half were born in SEA (66/113, 58.4%). Mean quantHBsAg was significantly lower in those with GDM (p = 0.044). No other associations were identified between GDM or HOMA-IR and markers of hepatic activity on multivariate logistic regression analysis and on sub-group analysis of those born in SEA.
Conclusions:
QuantHBsAg was significantly lower in those with GDM; otherwise, no association between GDM and measures of HBV viral activity was found. QuantHBsAg may be useful as an early pregnancy marker for GDM risk and warrants further research.
Introduction
The prevalence of chronic hepatitis B (HBV) infection is estimated at over 240 million people worldwide, 1 with a significant number of these being of reproductive age. 2 HBV infection during pregnancy has been reported as associated with the development of gestational diabetes mellitus (GDM). 2 GDM is characterized by insulin resistance and glucose intolerance first manifesting during pregnancy, and has a global prevalence of approximately 15%. 3 It is associated with poorer short and long-term health for both mother and child, including obstetric complications of fetal macrosomia, pre-eclampsia, shoulder dystocia and requirement for operative delivery, as well as longer-term increased risk of overt diabetes mellitus in both mother and infant. 4 Many risk factors for GDM are not modifiable, such as maternal age and family history of diabetes; therefore, identifying and developing strategies to mitigate modifiable risk factors is important.
Most studies reporting an association between HBV and GDM have been conducted in Asia, where women of Asian ethnicity have both a higher baseline incidence of GDM 5 and HBV. 6 Outside of Asia, a study based in the United States of America found no correlation between HBV and GDM; 7 however, a recent study of over 670,000 women with HBV in Victoria, Australia identified an increased risk of GDM which persisted after adjusting for factors of age, parity, body mass index (BMI), smoking and region of birth. 2
The underlying mechanism by which HBV infection increases the risk of GDM is not well characterized; however, evidence suggests that women with HBV experience a chronic inflammatory state that predisposes them to developing GDM. 8 The association between various inflammatory markers and GDM have been explored. Serum alanine transferase (ALT) as a marker of hepatic inflammation has not been associated with GDM 9 whereas ferritin has been demonstrated to be higher in women with HBV infection and GDM. 10 Viral factors such as hepatitis B envelope antigen (HBeAg) and HBV DNA viral load 11 have been inconsistently found to be associated with the development of GDM. 9
The aim of this study was to explore the relationship between HBV activity, measured by liver function tests, hepatitis B viral load, quantitative hepatitis B surface antigen (quantHBsAg), HBeAg and the development of GDM.
Methods
This prospective cohort study recruited women receiving antenatal care at three tertiary maternity centres in Melbourne from May 2021 until April 2023. The three services provide care for approximately 27,000 pregnant women per year. Each of the maternity services have a perinatal infectious diseases clinic where women with HBV during pregnancy can be referred.
Women were eligible for the study if they were: HBsAg positive, aged 18 years or older, planning to give birth at one of the participating hospitals, available for the entire study period, and willing and able to comply with the scheduled visits and study procedures. Opportunistic sampling was used to recruit women who fulfilled eligibility criteria from outpatient clinics. Written informed consent was obtained by participants. Non-English-speaking participants were also eligible, with interpreting services engaged to obtain informed consent. Participants were excluded if they had pre-existing diabetes or were unable to provide informed consent.
A target sample size of 260 women was calculated based on an estimated eligible cohort of 480 women over 12 months, required to detect a difference between groups with 80% power and an alpha cut-off of 5%.
Data collection
Baseline demographic and clinical data were collected. Country of birth was grouped into close geographic areas except for South East (SE) Asia as this was expected to account for a high proportion of our population. If HBsAg positive, then hepatitis B viral load, HBeAg and hepatitis B e antibody (HBeAb) were also collected. Viral load was stratified as low if below 2000 IU/mL, and high if over 2000 IU/mL. Women at high risk of GDM are routinely recommended to have early screening for gestational diabetes with either fasting glucose and HbA1c or oral glucose tolerance test (OGTT); where performed, this data was collected. If an early screening test for GDM was not performed then routine GDM screening was performed at 24–28 weeks’ gestation as per local protocols, either with oral glucose tolerance testing, or with fasting glucose. Variation in practice occurred due to temporary changes in testing as part of the COVID-19 pandemic restrictions. Participants had additional blood tests for study purposes including fasting insulin and quantHBsAg. Fasting glucose and insulin, where performed, were used to calculate the Homeostatic Model Assessment Insulin Resistance score (HOMA-IR), 12 a validated score used to demonstrate insulin sensitivity or resistance. There is no universally defined threshold for HOMA-IR to delineate insulin sensitivity or resistance: this study used a cut-off of >2.0 to indicate insulin resistance based on a review of previous studies. 13 QuantHBsAg was further characterized as high (>100 IU/mL) or low (<100 IU/mL). 14
Prospective outcomes were collected at two further study points: antenatal and postnatal. Use of antivirals and pregnancy outcomes (date of delivery, gestation, mode of delivery, birth weight, APGAR score) were collected by clinical record review. Antivirals were prescribed in accordance with recommended clinical guidelines. Data were stored and managed using REDCap electronic data capture tools. 15
The STROBE guidelines were followed when preparing this manuscript. There was no patient or public involvement in this study design.
Statistical methods
Descriptive statistics were performed for all variables and data are presented as either means and standard deviation or counts and percentages as appropriate. Differences in demographics, HBV activity and birth outcomes between those with and without GDM were compared using independent t tests or chi-square tests unless otherwise specified. Multivariate logistic regression was used to compare the proportion of women with GDM (model 1) and insulin resistance (model 2) according to level of hepatitis B activity, adjusted for known risk factors of maternal age, BMI, history of GDM in a previous pregnancy and country of birth. Method of GDM diagnosis (OGTT vs fasting glucose) was also included as a confounding variable. Unadjusted and adjusted odds ratios, 95% confidence intervals and two-sided p-value are reported. Data were analysed using IBM SPSS Statistics (Version 29).
Ethics
This study was approved by the Monash Health Ethics Research Committee (RES-21-0000-190A).
Results
A total of 113 participants were recruited between May 2021 and April 2023. Of these, 30 (26.5%) had a high viral load and 38 (33.6%) were diagnosed with GDM. The demographic and clinical characteristics of the participants are presented in Table 1.
Gestational diabetes mellitus (GDM) incidence by maternal characteristics.

Data are expressed as n (%) unless otherwise specified. Abbreviations: SD, standard deviation; BMI, body mass index; GDM, gestational diabetes. aOne patient had no testing performed to analyse for GDM and was excluded from analysis;. bDenotes statistically significant p-value; cFisher’s exact test used due to small cell value; dComparison of those from South East Asia compared to those not from South East Asia. eNulliparous participants were included within the ‘no past history of GDM’ group for the purposes of analysis; fDefined as previous infant with birthweight > 4500 g or >90th centile at birth; gAll patients prescribed antivirals received tenofovir, apart from one patient who had tenofovir allergy and received lamivudine.
Patients who were diagnosed with GDM were older than those without (p = 0.004), had a higher pre-pregnancy body mass index (BMI) (p = 0.050), and were more likely to have a history of GDM in a previous pregnancy (p = 0.001). Most women were born overseas (N = 110, 97.3%) and the majority were born in SE Asia (N = 66, 58.4%) (Supplementary Table S1). This group made up most of those who developed GDM (N = 29, 76.3%), compared with almost half of those who did not (N = 36, 48.6%); this difference was significant (p = 0.005). Most (111/113, 98.2%) were singleton pregnancies and no patients had co-existing hepatitis C infection.
GDM status and HOMA-IR scores were compared with markers of viral activity and inflammation (Table 2). No association was found between these two outcomes and viral load, ALT, alkaline phosphatase (ALP), gamma-glutamyltransferase (GGT), total bilirubin, albumin, ferritin levels and HBeAg status. Mean quantHBsAg was significantly lower in those with GDM than those without (1948 IU/mL, compared to 4449 IU/mL, p = 0.044). However, mean quantHBsAg was higher, but not significantly so, in those with insulin resistance. QuantHBsAg expressed as high (>100 IU/mL) or low (<100 IU/mL) was not significantly different between groups with GDM or insulin resistance.
Comparison of measures of HBV viral activity with incidence of GDM and insulin resistance.
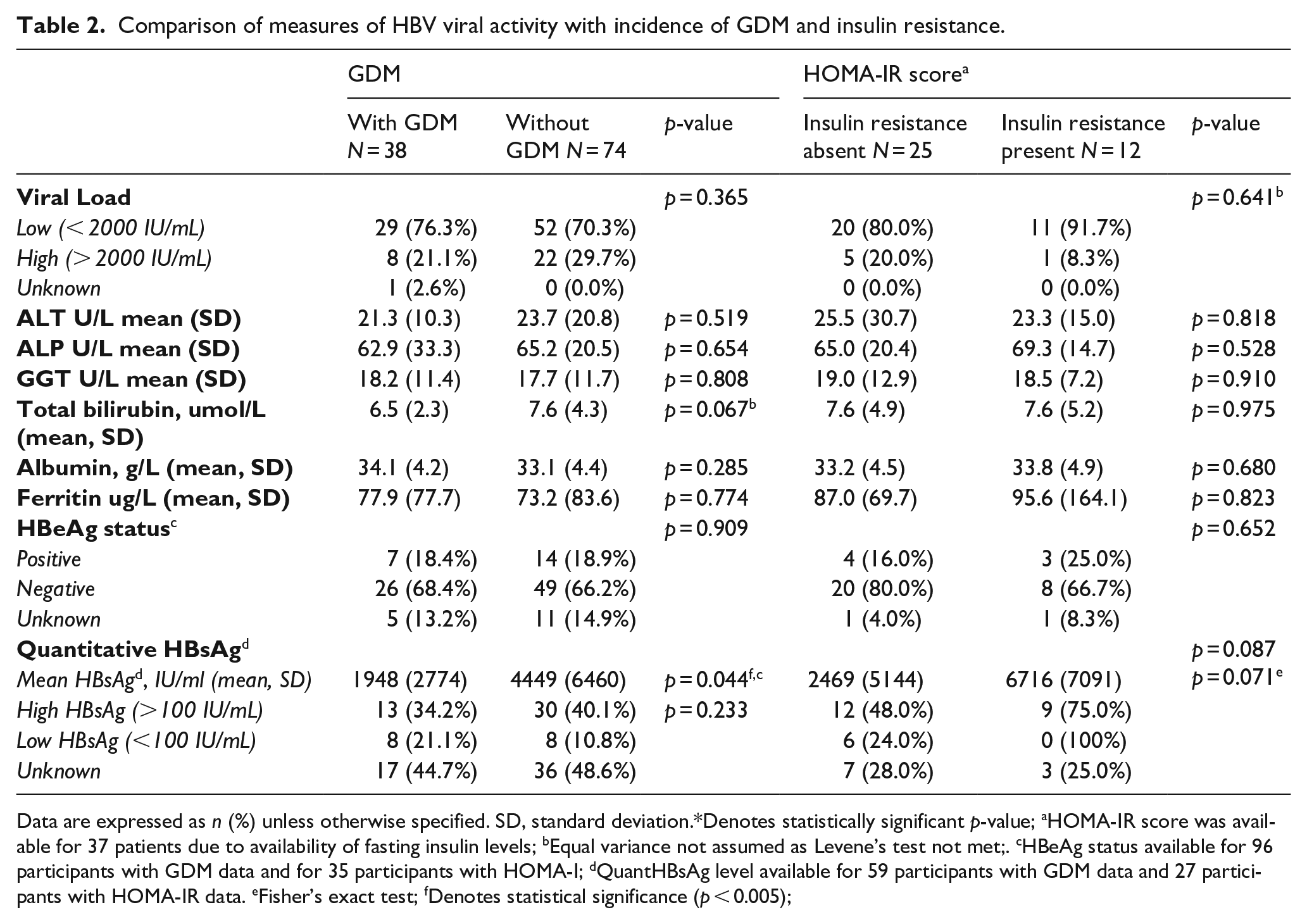
Data are expressed as n (%) unless otherwise specified. SD, standard deviation.*Denotes statistically significant p-value; aHOMA-IR score was available for 37 patients due to availability of fasting insulin levels; bEqual variance not assumed as Levene’s test not met;. cHBeAg status available for 96 participants with GDM data and for 35 participants with HOMA-I; dQuantHBsAg level available for 59 participants with GDM data and 27 participants with HOMA-IR data. eFisher’s exact test; fDenotes statistical significance (p < 0.005);
There were no significant differences in gestation at birth or average birthweight between patients with and without GDM (p = 0.163 and p = 0.545 respectively) (Table 3). Patients with GDM, but not insulin resistance, were significantly more likely to undergo induction of labour (p = 0.024, p = 0.718, respectively). Those with GDM were significantly more likely to have instrumental delivery or Caesarean section than unassisted vaginal delivery (p = 0.021). Insulin resistance was associated with increased birthweight; this trended towards significance (p = 0.059). Viral load was not associated with specific birth outcomes.
Birth outcomes in the study cohort compared with presence of GDM, viral load and insulin resistance.

Data are expressed as n (%) unless otherwise specified. SD, standard deviation; N/A, not available. aHigh viral load defined as HBV DNA viral load >2000 IU/mL and low viral load <2000 IU/mL, viral load was unavailable for one woman; bFisher’s exact test used due to small cell value; cNo women delivered after 42 weeks gestation; dDenotes statistically significant p-value; eExcluding five unknown and two sets of twins.
Most patients were screened for GDM with the recommended OGTT (N = 90, 80.4%) with a further 22 patients (19.6%) screened with fasting glucose alone. One patient had no testing performed and was managed as not having GDM. An analysis of the fasting glucose levels of the 34 patients diagnosed on OGTT identified that 10 patients (26.3%) would have been diagnosed if they had been screened using fasting glucose only; with most diagnosed with postprandial hyperglycaemia at 1 or 2 hours post-OGTT (63.2% and 67.6%, respectively).
Logistic regression analysis was performed to analyse GDM and HOMA-IR against markers of HBV activity when adjusting for the variables age, BMI, history of previous GDM, country of birth SE Asia and method of GDM diagnosis (OGTT versus fasting glucose) (Table 4). No significant associations were identified; multivariate analysis was not performed due to this and due to small sample size.
Logistic regression analysis: results for markers of viral activity related to presence of gestational diabetes and insulin resistance (HOMA-IR).

OR: odds ratio. CI: confidence intervals. Each marker of viral activity was assessed by logistic regression model with covariables of age, BMI, SE Asia country of birth, method of GDM diagnosis and past history of GDM. Where value is denoted with subscript10 this indicates that statistical values relate to changes per 10 units. – Unable to be calculated accurately within model due to low cell values. aOne record excluded as no record of whether GDM diagnosis occurred or not. bHOMA-IR able to be calculated for 37 of total participants.
A sub-group analysis was performed on participants of SE Asian ethnicity, as this group was highly represented in our sample and had significantly higher rates of GDM (Supplementary Tables S2 and S3). Previous GDM and higher BMI were significantly associated with GDM (p = 0.002, p = 0.044, respectively). A higher birthweight was significantly associated with the presence of insulin resistance as measured by HOMA-IR (p = 0.021) which persisted on logistic regression analysis (adjusted odds ratio (AOR) 0.0, confidence interval (CI) 0.000–0.381, p = 0.027). There were no significant associations with markers of viral activity identified on correlation analysis or regression analysis.
Discussion
Main findings
In our prospective study, participants with HBV were diagnosed with GDM at almost twice the rate (33.9%) of women undergoing antenatal care in Australia (17.9%). 3 This rate is reported among women of SE Asian ethnicity who made up the majority of our cohort. 2 Most diagnoses were made based on isolated postprandial hyperglycaemia (25/38, 65.8%).
We found that increased maternal age, pre-pregnancy BMI and history of GDM in a previous pregnancy were independently associated with developing GDM, in keeping with known risk factors for GDM. 4 Birth region of SE Asia was also over-represented in those with GDM which trended towards but did not reach significance, and has been well-documented in other studies.5,16 There was no difference in antiviral use between groups. Markers of viral activity were not associated with the development of GDM, apart from quantHBsAg, which was significantly lower among participants who developed GDM. The presence of insulin resistance, as measured by the HOMA-IR score calculated from fasting insulin and fasting was not associated with particular participant demographics nor markers of viral activity.
Interpretation
Previous studies have postulated several markers of inflammation as potential markers heralding an increased risk of GDM in women with HBV infection. These include serum ALT which has been found to be higher in pregnancy among those with HBV 9 but without a correlation with incidence of GDM, 17 a finding which our study replicated. We did not identify a difference in ferritin levels in women with and without GDM, although this has been reported elsewhere. 10 We found no association between GDM and markers of insulin resistance for other markers of hepatic inflammation including bilirubin, GGT and ALP. Elevated GGT has been found previously in women with GDM suggesting a role in pathogenesis 18 and raised ALP has been associated with adverse metabolic markers in women with and without GDM. 19 Higher levels of bilirubin appear to confer a lower risk of GDM; 20 however, we found no correlation in our cohort.
Other inflammatory markers not examined in our study include aspartate transaminase (AST), C-reactive protein (a marker of inflammation that has been shown to correlated with developing GDM 21 ) and tumour necrosis factor alpha (an important factor in immune-mediated control of HBV 22 ). Tumour necrosis factor alpha has been found to be elevated among women with GDM 23 although this has not been studied in women with dual pathologies of GDM and HBV. AST was not found to be associated with GDM in one recent study; 19 however, hepatic steatosis index was found to be associated with metabolic disturbance 19 and may represent a marker to identify women with HBV at increased risk of GDM.
Markers of viral activity include HBV DNA viral load and HBeAg, which are also used to assist with decision-making in relation to antiviral therapy to reduce perinatal transmission of hepatitis B. High viral load and HBeAg positivity have been suggested to be associated with a higher risk of GDM, but studies have shown varying results, with the overall relationship remaining unclear. 10 In our study, we found no correlation between HBV DNA viral load or HBeAg positive status and incidence of GDM.
Our study is the first to measure quantitative HBsAg in pregnancy and examine the relationship with measures of metabolic dysfunction. We found that mean quantHBsAg levels were significantly lower in women with GDM than those without (p = 0.044); although the presence of insulin resistance measured by HOMA-IR score was not significantly associated with HBsAg levels. Previous studies exploring the link between serum quantHBsAg levels and hepatic inflammation have shown a negative correlation, 24 with high levels of quantHBsAg commonly seen in the high replicative non-inflammatory state of HBV infection. 25 Patients with steatohepatitis were found to have lower quantHBsAg levels than those with steatosis alone, and those without fatty liver disease. 26 Our findings suggest that lower quantHBsAg levels may represent an increased risk of GDM as a marker for hepatic inflammation, and warrants further review particularly as other common scores did not identify a relationship in our study.
Diagnosis of GDM is recommended to be made using the oral glucose tolerance test; however, during COVID-19 pandemic restrictions, one of the maternity centres in our study used a two-step approach with the first step using fasting glucose with a diagnostic threshold of >5.1 mmol/L to diagnose GDM, and a second step with an OGTT performed if fasting glucose was between 4.7 and 5.0 mmol/L. A review of OGTT results of women in our study diagnosed via this method identified that 10 of 34 would have been identified if fasting glucose of >5.1 mmol/L was used, increasing to 14 of 34 if fasting glucose was between 4.7 to 5.0 mmol/L which would have prompted the testing with OGTT. This suggests that we may have underestimated the number of women with GDM in our study; potentially close to 60% of the participants screened with fasting glucose. This is similar to previously reported findings that HbA1c and/or fasting glucose levels have low sensitivities to detect hyperglycaemia in pregnancy 27 and that the use of fasting glucose of >5.1 mmol/L alone to diagnose GDM may miss the majority of GDM cases. 28 Previous studies have identified that between 48.4% 29 and 67% 28 of study participants with GDM only had abnormal post-load hyperglycaemia, compared with 71% in our cohort diagnosed with GDM on OGTT. Use of a lower fasting glucose cut-off of 4.7 mmol/L to exclude a diagnosis of GDM has been found to have similar perinatal outcomes compared to use of OGTT. 30
This observation is particularly interesting as this highlights a common pattern of insulin resistance in women in our population, notably that postprandial hyperglycaemia was more common than elevated fasting glucose. This pattern may be a clue to the specific metabolic disruption which occurs with HBV. Patients with both cirrhosis and diabetes have been found to have lower mean HbA1c than those without cirrhosis, with insulin more marked within the liver and skeletal muscle which is associated with higher postprandial glucose levels. 31 In addition to providing insights into the mechanism linking GDM and HBV, there is clinical importance as postprandial hyperglycaemia and elevated fasting glucose are individually associated with specific obstetric complications. Patients with isolated fasting glucose were more likely to develop macrosomia and large for gestational age infants, and undergo Caesarean delivery; while those with postprandial hyperglycaemia were more likely to have preterm labour and a trend towards small for gestational age infants.32 –34
Future research may include examining the role for use of insulin sensitizing agents such as metformin, which has been found to improve outcomes in both HBV 35 and GDM 36 separately, but has not been reviewed as a treatment strategy to improve obstetric outcomes in women with dual pathologies to our knowledge.
Strengths and limitations
Our study has several strengths including its prospective nature and the use of interpreters in order to recruit migrant women who make up the majority of referrals to our health services for antenatal management of HBV. Limitations included challenges to recruitment which were exacerbated by the concurrent COVID-19 pandemic, meaning that our study was under-powered. We suspect an underestimate of true GDM case numbers due to suboptimal GDM testing in the pandemic setting. The gold standard test for insulin resistance is the euglycaemic hyperinsulinaemic clamp; 37 however, due to practicalities we utilized paired fasting glucose and fasting insulin levels to produce HOMA-IR scores. These are not well validated across populations such as ours; with variable distributions known to exist depending on patient demographics including age, sex, and race, making the distinction between presence and absence of insulin resistance challenging. 38 We also limited our tests to those who had not undergone an OGTT in order to reduce additional testing burden to women during the pandemic; only 37 women (32.7%) had HOMA-IR scores calculated for evaluation.
Conclusion
The mechanism behind the relationship between HBV and GDM remains elusive; our study supports previous literature identifying no correlation between commonly recognized markers of viral activity including viral load, HBeAg positive, ALT, ferritin, GGT, and ALP. Our study is the first to examine the link between quantitative HBsAg and risk of GDM, finding that lower quantitative HBsAg is associated with increased insulin resistance. This may be useful as a surrogate marker to predict GDM. We also highlight that postprandial hyperglycaemia may be more likely to be present than elevated fasting glucose in those with GDM, suggesting a specific pathway of metabolic dysfunction in HBV deserving of future attention.
Supplemental Material
sj-docx-1-whe-10.1177_17455057241265083 – Supplemental material for Evaluating the relationship between hepatitis B viral activity and gestational diabetes mellitus: A prospective cohort study
Supplemental material, sj-docx-1-whe-10.1177_17455057241265083 for Evaluating the relationship between hepatitis B viral activity and gestational diabetes mellitus: A prospective cohort study by Naomi Whyler, Anwyn Pyle, Sushena Krishnaswamy, Joanne M Said and Michelle L Giles in Women’s Health
Supplemental Material
sj-docx-2-whe-10.1177_17455057241265083 – Supplemental material for Evaluating the relationship between hepatitis B viral activity and gestational diabetes mellitus: A prospective cohort study
Supplemental material, sj-docx-2-whe-10.1177_17455057241265083 for Evaluating the relationship between hepatitis B viral activity and gestational diabetes mellitus: A prospective cohort study by Naomi Whyler, Anwyn Pyle, Sushena Krishnaswamy, Joanne M Said and Michelle L Giles in Women’s Health
Supplemental Material
sj-docx-3-whe-10.1177_17455057241265083 – Supplemental material for Evaluating the relationship between hepatitis B viral activity and gestational diabetes mellitus: A prospective cohort study
Supplemental material, sj-docx-3-whe-10.1177_17455057241265083 for Evaluating the relationship between hepatitis B viral activity and gestational diabetes mellitus: A prospective cohort study by Naomi Whyler, Anwyn Pyle, Sushena Krishnaswamy, Joanne M Said and Michelle L Giles in Women’s Health
Supplemental Material
sj-docx-4-whe-10.1177_17455057241265083 – Supplemental material for Evaluating the relationship between hepatitis B viral activity and gestational diabetes mellitus: A prospective cohort study
Supplemental material, sj-docx-4-whe-10.1177_17455057241265083 for Evaluating the relationship between hepatitis B viral activity and gestational diabetes mellitus: A prospective cohort study by Naomi Whyler, Anwyn Pyle, Sushena Krishnaswamy, Joanne M Said and Michelle L Giles in Women’s Health
Footnotes
Acknowledgements
Prof. Allen Cheng, Professor/Director of Infectious Diseases, Monash Health, Melbourne and School of Clinical Sciences, Monash University, Victoria, Australia, for assistance with statistical analysis.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
