Abstract
Background:
Sickle cell disease in pregnancy is associated with high maternal and fetal mortality. However, studies reporting pregnancy, fetal, and neonatal outcomes in women with sickle cell disease are extremely limited.
Objectives:
The objectives of the study are to determine whether women with sickle cell disease have a greater risk of adverse pregnancy, fetal, and neonatal outcomes than women without sickle cell disease and identify the predictors of adverse pregnancy, fetal, and neonatal outcomes in women with sickle cell disease.
Design:
A retrospective pair-matched case-control study was conducted to compare 171 pregnant women with sickle cell disease to 171 pregnant women without sickle cell disease in Muscat, Sultanate of Oman.
Methods:
All pregnant Omani women with sickle cell disease who delivered between January 2015 and August 2021 at Sultan Qaboos University Hospital and Royal Hospital, who were either primipara or multipara and who had a gestational age of 24–42 weeks, were included as patients, whereas women who had no sickle cell disease or any comorbidity during pregnancy, who delivered within the same timeframe and at the same hospitals, were recruited as controls. The data were retrieved from electronic medical records and delivery registry books between January 2015 and August 2021.
Results:
Women with sickle cell disease who had severe anemia had increased odds of (χ2 = 58.56, p < 0.001) having adverse pregnancy outcomes. Women with sickle cell disease had 21.97% higher odds of delivering a baby with intrauterine growth retardation (χ2 = 17.80, unadjusted odds ratio = 2.91–166.13, p < 0.001). Newborns born to women with sickle cell disease had 3.93% greater odds of being admitted to the neonatal intensive care unit (χ2 = 16.80, unadjusted odds ratio = 1.97–7.84, p < 0.001). In addition, the children born to women with sickle cell disease had 10.90% higher odds of being born with low birth weight (χ2 = 56.92, unadjusted odds ratio = 5.36–22.16, p < 0.001). Hemoglobin level (odds ratio = 0.17, p < 0.001, 95% confidence interval = 0.10–3.0), past medical history (odds ratio = 7.95, p < 0.001, 95% confidence interval = 2.39–26.43), past surgical history (odds ratio = 17.69, p < 0.001, 95% confidence interval = 3.41–91.76), and preterm delivery (odds ratio = 9.48, p = 0.005, 95% confidence interval = 1.95–46.23) were identified as predictors of adverse pregnancy, fetal, and neonatal outcomes in women with sickle cell disease.
Conclusion:
As pregnant women with sickle cell disease are at increased risk for pregnancy, fetal, and neonatal adverse outcomes; improved antenatal surveillance and management may improve the outcomes.
Keywords
Introduction
Sickle cell disease (SCD) is an inherited autosomal recessive disorder that affects the structure of hemoglobin (sickle hemoglobin) and is a frequently occurring hemoglobinopathy. In individuals with SCD, sickle cells die early, causing a constant shortage of red blood cells (RBCs). Globally, approximately 3,000,000 neonates are born with SCD yearly. 1 Owing to migration and inter-racial relationships, SCD has become the most common monogenic disorder, affecting 30,000,000 people across all continents worldwide. 2 Sub-Saharan Africa, South America, the Caribbean, Saudi Arabia, India, and the Mediterranean are regions with the highest prevalence of SCD. 3 Approximately 2000 babies are born with SCD in the United States annually. 4 In Middle Eastern Arab countries, an elevated prevalence of SCD is observed, and the clinical presentation of SCD manifests in both benign and severe forms. 5 SCD is more prevalent in this region owing to increasing rates of consanguineous marriages between first cousins.5,6 For example, Saudi Arabia has a relatively high prevalence of SCD, with an estimated number of 145/10,000 cases, 7 and the Saudi premarital screening program has estimated the prevalence of SCD at 3.8 cases per 1000 people. 8
The prevalence of hemoglobin disorders in Oman is 3.5–4.7/1000 infants, and approximately 3000 patients with SCD live in the country. 9 Furthermore, the prevalence of SCD ranges from 0.2% to 0.3%. 10 Approximately 6% of Omanis are carriers of the gene for sickle cell anemia (SCA). 11 SCA is a disease in which the body produces crescent or sickle-shaped RBCs, which have a shorter lifespan than normal, round RBCs, leading to anemia. The sickling of cells reduces their flexibility, resulting in various complications. 12 More than two-thirds of SCD cases have a mild disease history, and most patients (61.2%) have homozygous SCA. 11 Accordingly, it is estimated that one of every 323 newborns in Oman has SCA. 13 Despite the availability of advanced treatment options to manage SCD, the quality of life of people living with SCD is substantially limited. 10 Therefore, early diagnosis and advances in comprehensive care are essential for people living with SCD. 14
SCD in pregnancy is considered a high-risk pregnancy. Pregnant women with SCD and their newborns develop more complications than children born to healthy women. 15 The reported pregnancy outcomes include maternal death, 16 - 18 perinatal mortality, 19 anemia, 19 - 23 sickle cell crisis, 19 bacterial infections,19,20,24,25 preterm delivery,20,23,26 pre-eclampsia,8,16,20,23 and eclampsia. 20 Furthermore, outcomes such as blood transfusion during pregnancy,16,20,22 abortion, 27 cesarean delivery,8,19,20,22,24 uterine rupture, 20 postpartum hemorrhage (PPH), 20 acute chest syndrome,19,23 cardiomyopathy, venous thromboembolism, 8 antepartum hospital admission, 25 bone and joint pain, urinary tract infection, hepatosplenomegaly, 23 preterm rupture of membrane, 26 meconium-stained liquor, 23 and postpartum infection 25 have been reported. The fetal outcomes identified include intrauterine growth retardation (IUGR),8,19-21,23,25 fetal distress,20,23 and stillbirth. 17 -19,21-23,25 Meanwhile, the neonatal outcomes reported are low birth weight (LBW),17,18,20-23,24,26 neonatal death,20,21,24,27 prematurity,24,26 neonatal intensive care unit (NICU) admission,21,24 neonatal asphyxia, 21 meconium aspiration syndrome, 23 and low Appearance, Pulse, Grimace, Activity, and Respiration (APGAR) score.17,18
SCD causes significant morbidity throughout Arab and Middle Eastern countries, including Oman. 28 This underscores the need for a multidisciplinary approach to managing patients during pregnancy, delivery, and postpartum. 19 In Oman, only one study has reported the fetal outcomes in women with SCD. Examining the pregnancy, fetal, and neonatal outcomes in women with SCD can provide baseline data and direct the initiation of evidence-based interventions to manage women with SCD during pregnancy and prevent complications in the growing fetus. Therefore, this study aimed to evaluate the adverse pregnancy, fetal, and neonatal outcomes in Omani women with SCD and identify the predictors of adverse pregnancy, fetal, and neonatal outcomes in women with SCD.
Methods
This study was reported in accordance with the Strengthening the Reporting of Observational Studies in Epidemiology guidelines.
Design
A retrospective pair-matched case-control research design was used, with cases and controls matched for age, gravidity, and parity. The study was conducted to assess whether women with SCD have a greater risk of adverse pregnancy, fetal, and neonatal outcomes than women without SCD. In addition, the predictors of adverse pregnancy, fetal, and neonatal outcomes in women with SCD were identified.
Setting
This research was conducted in two tertiary care hospitals in Muscat, Oman: Sultan Qaboos University Hospital (SQUH) and Royal Hospital (RH). These hospitals were selected as they both offer tertiary care services to the general population and women with SCD. Moreover, these hospitals typically serve the population across Oman. At SQUH, approximately 30,000 patients were admitted in 2019. Around 120,000 patients sought treatment in the outpatient department, and approximately 65,000 patients were admitted to the emergency department. Moreover, approximately 4000–5000 deliveries are conducted yearly. At SQUH, the rate of Cesarean Section has been steady in the last few years (12%–15%). Oman’s LBW rate has been increasing since the 1980s. It was approximately 4% in 1980 and had nearly doubled (8.1%) by 2000. Since then, a slow but steady rise has been observed, reaching 10% recently. Notably, SQUH received platinum-level accreditation from a Canadian accreditation body for maintaining the quality of international standards. RH is equipped with specialties including Medicine, Surgery, Obstetrics, Gynecology, Pediatrics, and Oncology. Furthermore, this hospital adheres to international quality standards and has obtained the Canadian Qmentum International Accreditation, an organization that approves standards of excellence in quality care and services of Canadian health institutions and hospitals. Moreover, these hospitals cover the population across Oman.
Population
The target study population included all pregnant mothers who were diagnosed with SCD who delivered. Particularly, the patient group comprised all pregnant Omani women with SCD who delivered at SQUH and RH during the study period. The control group included healthy pregnant women who delivered in the same hospitals during the same period.
Sample
Pregnant Omani women with SCD who delivered between January 2015 and August 2021 at SQUH and RH and met the inclusion criteria were selected as patients. Pregnant women without SCD who delivered during the same period at SQUH and RH and met the inclusion criteria were selected as controls. Chi-square homogeneity testing was conducted to ensure that age, gravidity, and parity matched between both groups.
Inclusion criteria (patients)
All pregnant Omani women with SCD (diagnosed in accordance with ICD-10 criteria) of any age who delivered between January 2015 and August 2021 at SQUH and RH and who were either primipara or multipara, with a gestational age of 24–42 weeks, were included as patients.
Inclusion criteria (controls)
Women without SCD or any comorbidity during pregnancy, who delivered between January 2015 and August 2021 at SQUH and RH, and who were either primipara or multipara, with a gestational age of 24–42 weeks, were included as controls.
Exclusion criteria (patients)
All pregnant women with other hemoglobinopathies such as congenital dyserythropoietic anemia and various types of thalassemia (beta-thalassemia or alpha-thalassemia); women with hypertension, diabetes mellitus, myasthenia gravis, and systemic lupus erythematosus; and women with SCD who had been infected with coronavirus disease 2019 (COVID-19) were excluded, as these conditions might affect the outcome variables. In addition, women with HIV were excluded from the study because of the possible effect of HIV on pregnancy outcomes.
Sample size
Following the rule of thumb with the number of predictors (n = 17) in the study, the investigators recruited 170 participants for each group. For each variable, 10 samples were calculated, considering the need for a regression analysis. The predictors included in the study were age, gravidity, parity, number of antenatal visits, type of delivery, gestational age, history of infertility, previous abortion, medical history, obstetric history, surgical history, family history of SCD, height, weight, level of hemoglobin, type of SCD, and history of blood transfusion. The ratio for the patient and control groups was 1:1.
Sampling technique
Through convenient sampling, data specific to the mothers with and without SCD were collected, considering the inclusion and exclusion criteria.
Description and interpretation of the study tool
The investigators developed a data collection sheet with three sections by reviewing the published literature.
Section 1: maternal demographic variables
The maternal demographic variables included maternal age, parity, number of antenatal visits, mode of delivery, nature of pregnancy, gestational age, history of infertility, pregnancy loss, medical history, surgical history, presence of medical illness, and family history of SCD.
Section 2: pregnancy outcomes
The pregnancy outcomes included preterm labor, pregnancy-induced hypertension, pre-eclampsia, eclampsia, and severe anemia during pregnancy. These outcomes were determined based on yes/no-type questions.
Section 3: fetal and neonatal outcomes
The fetal outcomes included IUGR and fetal distress, while the neonatal outcomes included NICU admission, low APGAR score, and LBW. These outcomes were determined based on yes/no-type questions.
Data accuracy
The accuracy of the data was evaluated through quality checks. The collected data were counter-checked and verified by the principal investigator (PI) and the research assistant (RA). The investigators also verified whether there were missing data in the data collection sheet. The missing data were verified manually, and the missing data sheets were discarded and replaced by collecting additional cases.
Ethical considerations
Ethical approval was obtained from the Research and Ethics Committee of the College of Nursing, SQU (CON/TRC-GR/2021/01). The research proposal was submitted to the Medical Research and Ethics Committee of the College of Medicine and Health Sciences, SQU (SQU-EC/410/2021). In addition, ethical approval was obtained from the ethics committee of the Ministry of Health (MoH/DGPS/CSR/PROPOSAL_APPROVED/28/2021). As the data were retrospectively collected from the delivery register book and electronic medical records, no direct contact with the participants was required; therefore, informed consent was not required. No identifying information of the participants was collected. The collected data were treated confidentially, stored in a password-protected file, and exclusively handled by the research team.
Pilot study
A pilot study was performed for 1 week in June 2021, involving approximately 20% of the samples (n = 40), which were divided into 20 patients and 20 controls. A few minor modifications were made to the tool after the pilot study. Blood transfusion and anemia during pregnancy were added to the tool, while puerperal sepsis was deleted, as these data were difficult to obtain. The pilot study samples were excluded from the main study.
Data collection
The PI and RA obtained permission from the medical records department of the hospital and the obstetric unit to access the records. Thereafter, the data were retrieved manually using the tool prepared by the investigators from the participants’ electronic medical records (Track care + Al Shifaa) and delivery register book. Both the PI and RA verified the accuracy of the data collected through crosschecks. The data were collected and entered manually in the data collection sheets. Thereafter, the data were transferred electronically into the Statistical Package for the Social Sciences software (IBM, SPSS Inc).
Data processing
The data were entered and cleaned. Data cleaning was conducted to ensure consistency, eliminate errors, and check the values of the variables. In addition, frequency tables were used to analyze missing data and monitor outliers.
Statistical analysis
Descriptive statistics, including frequencies, percentages, means, and standard deviations (SDs), were used to describe the sample characteristics. Unadjusted odds ratios were utilized to estimate the potential risk factors. The chi-square test and t test were used to evaluate the differences between the patient and control groups. An unpaired t test was conducted to determine any significant differences in age, body mass index (BMI), and hemoglobin level between the women with and without SCD. A bivariate analysis was performed to test for any significant difference in the risk factors between the women with and without SCD. The predictors of adverse pregnancy, fetal, and neonatal outcomes among the women with SCD were evaluated using a logistic regression analysis.
Results
Table 1 shows the characteristics of women with and without SCD who visited the selected hospitals in Oman from January 2015 to August 2021. The majority of women in both the case and control groups were aged between 20 and 34 years. In addition, both groups had a similar distribution in gravida and parity. Both case and control groups were statistically matched for age, gravida, and parity. Women with SCD had a significantly lower BMI (mean ± SD = 26.22 ± 4.29) than those without SCD (mean ± SD = 28.42 ± 4.95) (t = 4.25, df = 316, p < 0.001). Similarly, women with SCD had a lower hemoglobin level (mean ± SD = 8.91 ± 1.19) than those without SCD (mean ± SD = 11.42 ± 1.22) (t = 19.05, df = 334, p < 0.001). Newborns born to mothers with SCD had 3.93% greater odds of being admitted to the NICU than those born to mothers without SCD (χ2 = 16.80, unadjusted odds ratio (UOR) = 1.97–7.84, p < 0.001). In addition, the newborns born to mothers with SCD had 10.90% higher odds of having an LBW than those born to mothers without SCD (χ2 = 56.92, UOR = 5.36–22.16, p < 0.001).
Sample characteristics of women with and without SCD in Oman from January 2015 to August 2021.
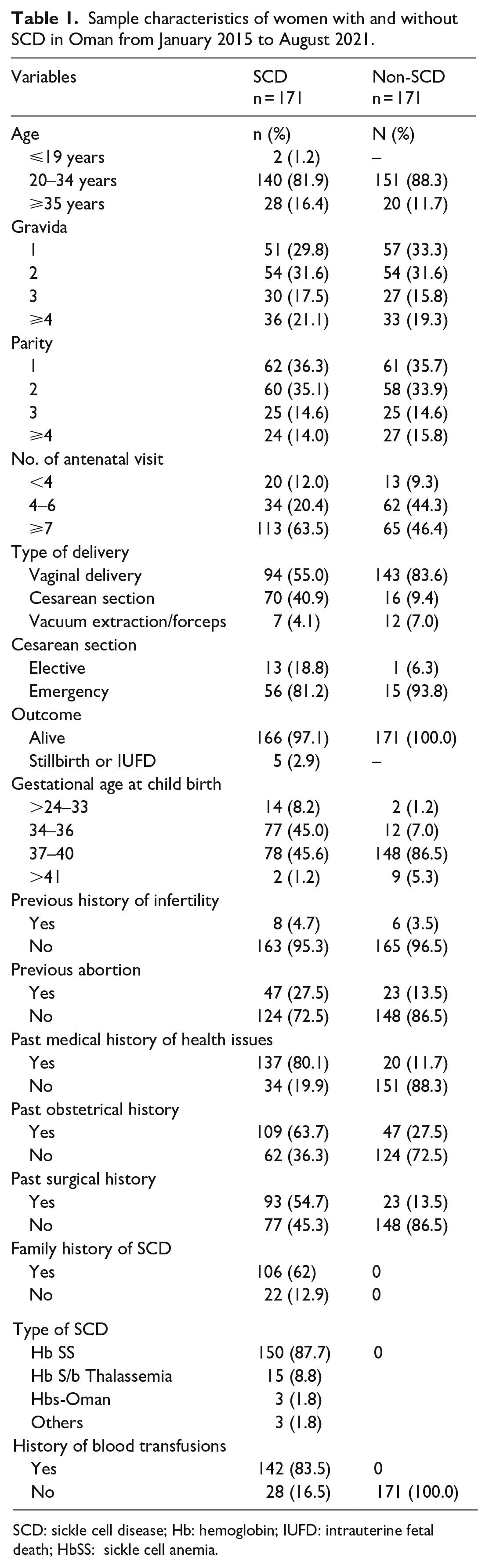
SCD: sickle cell disease; Hb: hemoglobin; IUFD: intrauterine fetal death; HbSS: sickle cell anemia.
There was a significant difference in the risk factors between the women with and without SCD. Fewer than four antenatal visits (χ2 = 20.38, p < 0.001), cesarean section (χ2 = 49.07, p < 0.001), and gestational age (χ2 = 82.61, p < 0.001) were found to be the significant risk factors among the women with SCD. There was a significant difference in the medical history (χ2 = 161.19, p < 0.001), obstetric history (χ2 = 45.31, p < 0.001), surgical history (χ2 = 64.64, p < 0.001), and blood transfusion history (χ2 = 244.76, p < 0.001) between the women with and without SCD.
Table 2 displays the estimated odds of the occurrence of adverse pregnancy outcomes in the women with and without SCD. The women with SCD and severe anemia had increased odds of having adverse pregnancy outcomes compared with the women without anemia (χ2 = 58.56, p < 0.001). Table 3 demonstrates the estimated odds of the occurrence of adverse fetal outcomes in the women with and without SCD. The mothers with SCD had 21.97% higher odds of delivering a baby with IUGR than those without SCD (χ2 = 17.80, UOR = 2.91–166.13, p < 0.001). Table 4 shows the estimated odds of the occurrence of adverse neonatal outcomes in the women with and without SCD. The newborns born to the mothers with SCD had a 3.93% greater odds of being admitted to the NICU than those born to the mothers without SCD (χ2 = 16.80, UOR = 1.97–7.84, p < 0.001). In addition, the newborns born to mothers with SCD had 10.90% higher odds of having an LBW than those born to mothers without SCD (χ2 = 56.92, UOR = 5.36–22.16, p < 0.001).
Estimated odds of occurrence of adverse pregnancy outcomes in women with and without SCD in Oman from January 2015 to August 2021.

Severe anemia in pregnancy is if Hb < 6.5 g/100 mL.
SCD: sickle cell disease; PIH: pregnancy-induced hypertension.
p < 0.001 level of significance.
Estimated odds of occurrence adverse fetal outcomes in women with and without SCD in Oman from January 2015 to August 2021.

SCD: sickle cell disease; IUGR: intrauterine growth retardation.
p < 0.001 level of significance.
Estimated odds of occurrence of adverse neonatal outcomes in women with and without SCD in Oman from January 2015 to August 2021.

SCD: sickle cell disease; NICU: neonatal intensive care unit; LBW: low birth weight; APGAR: appearance, pulse, grimace, activity, respiration.
Low APGAR score is if APGAR is < 7.
p < 0.001 level of significance.
The multivariate logistic regression analysis was conducted using the enter method, considering the significant factors from the bivariate analysis (up to the 0.2 level). The following factors were entered into the model: maternal age, BMI, hemoglobin level, number of antenatal visits, mode of delivery, history of abortion, medical history, obstetric history, preterm labor, IUGR, NICU admission, LBW, and gestational age. The probability of the model for obtaining a χ2 value is 294.098. The model was significant at p < 0.001 (Table 5).
Predictors of adverse pregnancy, fetal, and newborn outcomes in women with SCD in Oman from January 2015 to August 2021.
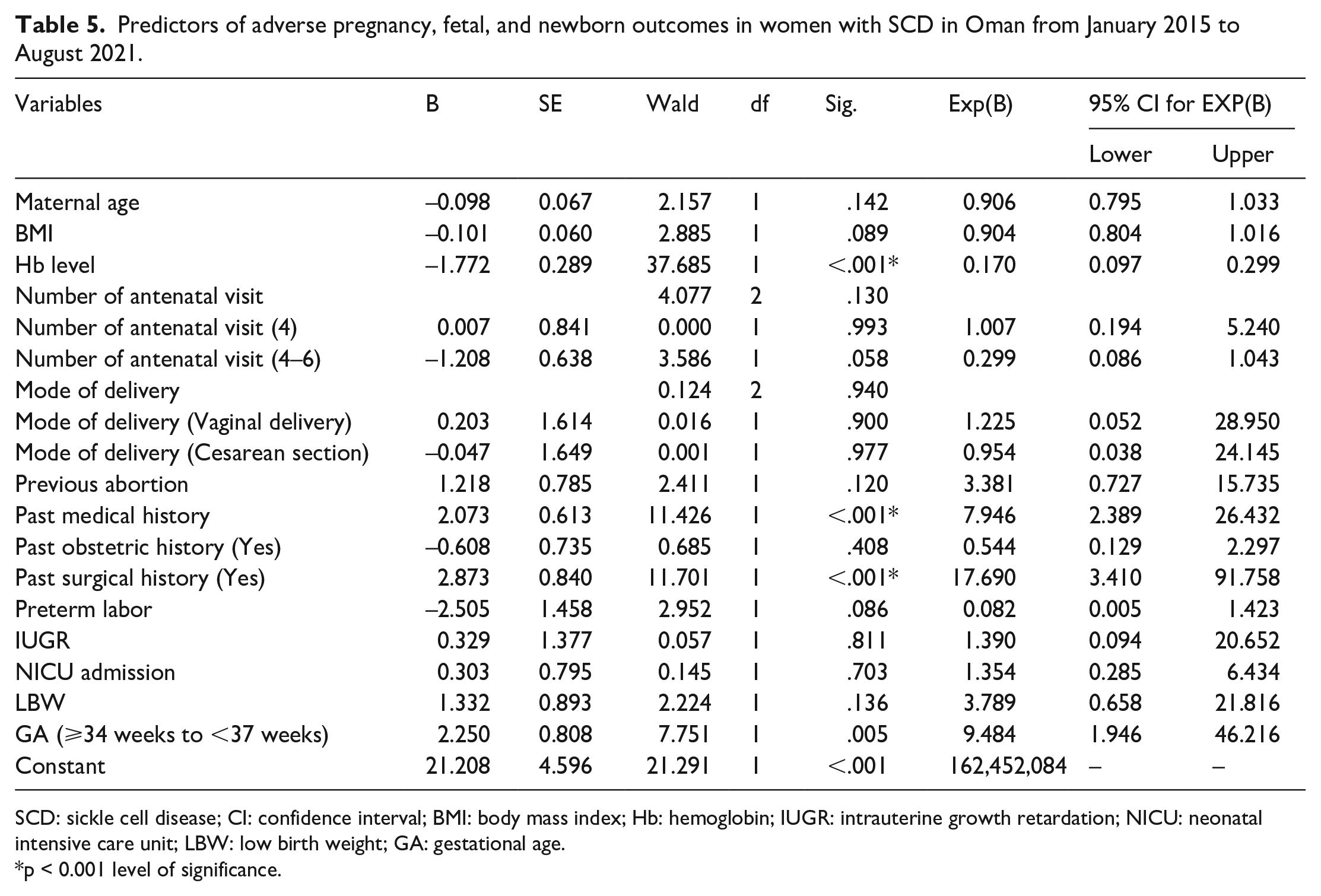
SCD: sickle cell disease; CI: confidence interval; BMI: body mass index; Hb: hemoglobin; IUGR: intrauterine growth retardation; NICU: neonatal intensive care unit; LBW: low birth weight; GA: gestational age.
p < 0.001 level of significance.
The model had a good fit (χ2 = 17.97, p = 0.021) with an adjusted Cox and Snell R2 value of 0.642. It also had a high sensitivity, specificity, and overall predictive power (94.8%). Collinearity was tested using tolerance and variance inflation factor (VIF) statistics. No collinearity issues were observed; the minimum tolerance value was 0.505; and the maximum VIF value was 1.981. After the factors were adjusted for in the model, the following factors remained as the independent predictors of SCD: hemoglobin level (OR = 0.17, p < 0.001, 95% CI = 0.10–3.0), medical history (OR = 7.95, p < 0.001, 95% CI = 2.39–26.43), surgical history (OR = 17.69, p < 0.001, 95% CI = 3.41–91.76), and preterm delivery (OR = 9.48, p = 0.005, 95% CI = 1.95–46.23). The other factors failed to achieve statistical significance.
Discussion
This study revealed that the women with SCD who had severe anemia had increased odds of having adverse pregnancy outcomes; mothers with SCD had higher odds of delivering a baby with IUGR; the newborns born to the women with SCD had greater odds of being admitted to the NICU; and the newborns born to the women with SCD had increased risk of having an LBW baby. The hemoglobin level, medical history, surgical history, and preterm delivery were identified as predictors of adverse pregnancy, fetal, and neonatal outcomes in women with SCD.
Herein, the proportion of women with pregnancy-induced hypertension was higher in the women with SCD than in those without. It has been postulated that women with SCD have a lower prostacyclin-to-thromboxane ratio, potentially indicating a greater proclivity to vasoconstriction, leading to pregnancy-induced hypertension. 29 Similar to the present findings, Haseeb and Al Qahtani 30 found hypertensive disorders in 13.3% of pregnant women with SCD. Babah et al. 31 also reported the occurrence of hypertensive disorders in pregnant women with SCD. In addition, Sousa et al. 32 recognized the occurrence of hypertensive disorders in pregnant women with SCD. Another study showed that pregnancy-related hypertensive disorders were more common among pregnant women with SCD (30.4%) than among those without. 33 Kuo and Caughey 34 also found the presence of pregnancy-induced hypertension in women with SCD. These findings strongly show the relationship between SCD and pregnancy-induced hypertension. Thus, special attention must be given to pregnant women with SCD to prevent and manage hypertensive disorders during pregnancy.
Generally, increased prostaglandin production is believed to be the cause of preterm labor. 35 Similarly, SCD increases the risk of preterm labor, as it causes additional physiological adaptations in the hematologic, renal, circulatory, and pulmonary systems during pregnancy, which can overburden the organs. 32 In this study, it was discovered that preterm labor was frequent among mothers with SCD. Consistent with this finding, preterm labor is reported in many other studies on women with SCD.15,24,36- 39 Therefore, mothers with SCD should deliver in a healthcare facility that is well-equipped to manage high-risk pregnancies.
Herein, mothers with SCD experienced pre-eclampsia and eclampsia. Similarly, Boafor et al. 40 reported that SCD was associated with an increased risk of pre-eclampsia and eclampsia in their systematic review and meta-analysis of 16 studies. Numerous studies worldwide have indicated an elevated incidence of pre-eclampsia in women with SCD, with reported percentages such as 3%, 56%, 20%, 6.1%, 33.3%, and 25% in India22,41- 44 ; 24% in Tanzania 45 ; 6% in West Indies 46 ; and 11% in French Guiana. 34 In addition to these studies, Lari et al. 47 recognized in their systematic review that pregnancy in women with SCD is associated with high maternal morbidity and mortality secondary to hypertensive disorders during pregnancy, as these may worsen the outcomes. Oteng-Ntim et al. 25 also demonstrated that SCD poses an increased risk of pre-eclampsia and eclampsia. Moreover, O’Hara et al. 33 recently identified that the presence of SCD in women increases the risk of pre-eclampsia and eclampsia and leads to adverse pregnancy outcomes. Collectively, previous studies predominantly show that pre-eclampsia and eclampsia are associated with SCD in pregnancy, emphasizing the need for providing meticulous care to pregnant women with SCD. Moreover, these women can be treated with low doses of aspirin from the beginning of the second trimester until 5 to 10 days before the due date of delivery. 48 Aspirin is currently the only medication recommended for the prevention of pre-eclampsia. 49 In addition, low- and high-dose calcium supplementation is effective for pre-eclampsia prevention in women with low calcium intake. 50 The survival of these women can be improved by developing and testing multidisciplinary evidence-based strategies. 51
Severe anemia was more frequently seen in the patient group than in the control group in this study. Generally, anemia has a negative impact on fetal growth. Similarly, in pregnant women with SCD, anemia poses an additional risk to the fetus. Vasculopathy and vaso-occlusion also affect placental health, further limiting fetal growth. 34 In their systematic review, Aroke et al. 52 identified the overall prevalence of anemia in pregnant women with SCD as 6.67%–83.33%. Elenga et al. 36 also noted anemia in 36.5% of pregnant women with SCD. Limited literature has explored the occurrence of severe anemia in pregnant women with SCD. Therefore, further studies to explore this aspect are recommended. As severe anemia escalates the complications and yields unforeseen outcomes in pregnant women with SCD, managing these pregnancies can be best accomplished by utilizing individualized care plans and a multidisciplinary team approach. 32
PPH was found more frequently in the women with SCD than in those without in this study. Similarly, Boulet et al. 53 reported the occurrence of PPH in women with SCD. Emezienna et al. 54 also reported that PPH occurred more frequently in women with SCD than in those without. Therefore, close monitoring and multidisciplinary care are recommended to improve pregnancy outcomes in women with SCD. Moreover, exceptional care is warranted for mothers with SCD during the antenatal, intranatal, and postnatal periods. Further studies are also needed to explore this outcome, as only a few studies have been conducted.
In the present study, IUGR and fetal distress were more commonly found in the patient group than in the control group. A few other studies have also reported that IUGR was associated with SCD in pregnancy, leading to perinatal mortality.16,38,42 An earlier study conducted in Oman also has shown the occurrence of IUGR and fetal distress in children born to mothers with SCD. 20 Similarly, Turner et al., 55 Sousa et al., 32 Kuo and Caughey, 34 and Oteng-Ntim et al. 25 recently reported the occurrence of IUGR in pregnancies complicated by SCD. It is believed that chronic fetal hypoxia, occurring because of impaired placental blood flow, causes a high incidence of perinatal complications, including IUGR and fetal distress. 56 Moreover, anemia in mothers with SCD causes impaired placental perfusion, which further limits the transport of nutrients and oxygen to the fetus. This circumstance causes an increased incidence of IUGR in pregnancies associated with SCD. 37 As few studies have identified the prevalence of IUGR and fetal distress in pregnancies complicated by SCD, more extensive and multicentric studies are suggested.
Herein, the women with SCD delivered newborns who had to be admitted to the NICU. Similarly, Elenga et al. 36 reported that newborns born to women with SCD had abnormal fetal heart rates, which necessitated admission to the NICU. Therefore, it is highly recommended that pregnancies complicated by SCD be managed by a multidisciplinary team comprising an obstetrician, a physician, and an integrated center for SCD for the comprehensive management of the mother and the child. More studies are also required to understand further the reasons for admitting children who are born to mothers with SCD to the NICU. In this study, the newborns born to the mothers with SCD had a higher incidence of LBW and low APGAR scores than those born to the mothers without SCD. In other studies, a more significant incidence of LBW39,40-43,46,47 and low APGAR scores41,46,47 has been noted. These findings warrant the need for comprehensive and meticulous care for laboring mothers with SCD and their children.
In this study, the women with SCD and severe anemia had a significant risk of having adverse outcomes compared with the women without anemia. Similar to these findings, maternal complications of SCD, including anemia, sickle cell crisis, and the need for blood transfusion during pregnancy, were found in another study. 16 Adverse pregnancy outcomes, including anemia in 89.4% of women with SCD and sickle cell crisis in 39.2%, were also reported in another study. 32 Numerous other studies worldwide have reported an increased incidence of anemia in women with SCD, including 23.3%, 64%, 24%, 22.1%, 33.3%, and 75% in India22,43- 46 and 16% in Tanzania. 45 Anemia during pregnancy compromises placental perfusion, reducing the transport of nutrients and oxygen to the fetus, consequently leading to an increased incidence of IUGR. Therefore, strategies must be designed to improve the pregnancy outcomes in women with SCD.
In this study, mothers with SCD had a higher risk of delivering babies with IUGR than the mothers without SCD, which aligns with the results reported by Haseeb and Al Qahtani, 32 where IUGR occurred in 19.2% of mothers with SCD. Several studies have also estimated the risk of IUGR in mothers with SCD, revealing percentages such as 3.3%, 33.3%, 2.8%, and 50% in India22,43,45 and 19% in French Guiana. 38 In Nigeria, IUGR was associated with SCD in pregnancy. 33 Close monitoring and advanced perinatal care are thus essential for managing pregnant women with SCD. Herein, the newborns born to the mothers with SCD were more frequently admitted to the NICU and more commonly had an LBW than the newborns born to the mothers with SCD. A previous study reported that 1.6% of neonates born to mothers with SCD were admitted to the NICU. 32 LBW has been reported in numerous studies worldwide, with percentages such as 53.3%, 77.7%, 84.6%, 56%, and 100% in India22,41- 44 and 33.5% in Tanzania. 47
As reported in the previous literature, SCD in pregnancy is the cause of many adverse maternal and fetal outcomes. Therefore, pre-conceptual counseling, early booking, watchful monitoring during pregnancy and multidisciplinary care for pregnant women with SCD can prevent adverse outcomes. The present study showed significant differences in the pregnancy, fetal, and neonatal outcomes between the women with and without SCD. Thus, the following null hypothesis of the study is rejected: “There are no significant differences in the pregnancy, fetal, and neonatal outcomes between women with and without SCD.”
In this study, the hemoglobin level, medical history, surgical history, and preterm delivery were the independent predictors of adverse pregnancy, fetal, and neonatal outcomes. Similarly, lower hemoglobin levels during the first trimester, numerous blood transfusions before pregnancy, admissions in the year before pregnancy for vaso-occlusive events, maternal cardiac complications before pregnancy, and SCD genotype predicted the maternal risk in another study. In addition, younger age and SCD genotype allowed for early prediction of the fetal risk. 57 These findings show that the following null hypothesis of this study is rejected: “There is no significant association between adverse pregnancy, fetal, and neonatal outcomes and demographic variables of women with SCD.”
Limitations
First, the findings of this study are limited only to the Omani population. Moreover, as the data were collected from two public hospitals in Oman and as the record-keeping differs between these hospitals, the investigators faced challenges while collecting the data. Some data could not be retrieved, as they were not documented; thus, these data were omitted from the analysis. In addition, our study excluded women with SCD who had co-morbid illnesses such as hypertension, diabetes mellitus, myasthenia gravis, systemic lupus erythematosus, women with HIV, and women who had been infected with COVID-19, as these conditions could potentially impact the outcome variables, which could have affected the study results significantly.
Strengths
This study included women with SCD who can adequately represent the Omani population. As the study utilized a matched case-control design, bias was limited. Moreover, to the investigators’ knowledge, this study is the first to assess the pregnancy, fetal, and neonatal outcomes among women with SCD in Oman.
Implications
This research contributes valuable data regarding adverse pregnancy, fetal, and neonatal outcomes in pregnant women with SCD in Oman. The findings offer baseline data that can assist healthcare workers in designing evidence-based interventions for pregnant women with SCD. Furthermore, curriculum designers in nursing education can use the findings to enlighten nursing students in providing better nursing care considering the outcomes. The findings can also guide healthcare system planning for future studies to achieve the best standards of care in clinical practice.
Recommendations
A multidisciplinary approach is recommended for the comprehensive management of pregnant women with SCD. Further studies are required to investigate additional predictors of adverse outcomes in pregnant women with SCD in Oman. In addition, the relationship between SCD and fetal anomalies, as well as the optimal delivery timing and outcome in women with SCD, should be studied.
Conclusion
Our study demonstrated that women with SCD who experienced severe anemia had increased odds of getting adverse pregnancy outcomes. Notably, women with SCD had higher odds of getting a baby with intrauterine growth retardation, and newborns born to women with SCD had greater odds of being admitted to NICU. In addition, the children born to women with SCD had higher odds of being born with low birth weight. Hemoglobin level, past medical history, past surgical history, and preterm delivery were identified as predictors of adverse pregnancy, fetal, and neonatal outcomes in women with SCD. SCD during pregnancy poses significant risks and adverse outcomes to both the mother and the newborn. Various factors predict the risk of adverse pregnancy, fetal, and neonatal outcomes in women with SCD. Therefore, it is crucial that a multidisciplinary team of hematologists, obstetricians, and pediatricians closely monitor these pregnancies. To mitigate these risks, women with SCD must obtain adequate information, receive education, and undergo communication sessions. In addition, identifying subgroups of women with SCD who are at a high risk of adverse events will allow early intervention to improve pregnancy outcomes.
Supplemental Material
sj-doc-1-whe-10.1177_17455057231220188 – Supplemental material for Adverse pregnancy, fetal and neonatal outcomes in women with sickle cell disease in a Middle Eastern country
Supplemental material, sj-doc-1-whe-10.1177_17455057231220188 for Adverse pregnancy, fetal and neonatal outcomes in women with sickle cell disease in a Middle Eastern country by Salwa Saif Said AL Harthi, Judie Arulappan, Basma Al Yazeedi and Asma Hassan Salmeen Al Zaabi in Women’s Health
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
