Abstract
Background:
Large-scale longitudinal studies with biological samples are needed to examine the associations between prenatal cannabis use and birth and developmental outcomes.
Objectives:
The aim of this study was to understand the feasibility and acceptability of collecting umbilical cord tissue for the purpose of cannabis use testing in a community sample.
Design:
This is a mixed methods research study consisted of a prospective cohort study and a qualitative descriptive study.
Methods:
This study was conducted in Vancouver, British Columbia between January 2021 and August 2022. Participants were recruited during pregnancy, and the umbilical cord tissues were collected at birth and tested for the presence of cannabinoids. After the completion of the study, participants completed an online open-ended questionnaire about their overall experience. Data were analyzed using descriptive and thematic analyses.
Results:
Among the 85 eligible individuals, 57 people (67%) consented to the study. The cord tissue was collected for 39 participants (68.4%). The collection rates for participants with vaginal, elective, and emergency cesarean delivery were 73.0%, 71.4%, and 53.8%, respectively, and for those with spontaneous and induced labor were 81.5% and 50%, respectively. Four (7.0%) and seven participants (12.3%) reported prenatal cannabis use in direct and probing self-report questions, respectively. The agreement between any self-report and cord tissue test was moderate (kappa 0.53, 95% confidence interval 0.06–0.99). Qualitative findings were classified into five themes.
Conclusion:
The collection of cord tissue was perceived acceptable by most participants. Implementation of collection protocols for complex labors, a central hospital unit to liaise direct communications and active participants’ involvement might increase the feasibility of future studies.
Introduction
Socio-legal perspectives are evolving around substance use including cannabis. Cannabis use during pregnancy is common with increasing trends in recent years.1,2 For instance, based on the US National Survey on Drug Use and Health (NSDUH), from 2002 to 2020, the prevalence of cannabis use in the first, second, and third trimesters increased from 6.3%, 1.9%, and 2.0% in 2002 to 16.0%, 4.2%, and 4.7% in 2020, respectively. 2
The accumulating evidence shows that cannabis use is associated with an increased risk of adverse events from the preconception to postpartum period. A recent study of those with a previous pregnancy loss found that preconception cannabis use was associated with 41% reduced fecundability after adjusting for confounders. 3 A 2022 systematic review and meta-analysis of 16 studies with 59,138 participants reported that cannabis exposure during pregnancy was associated with increased risk of adverse pregnancy (low birth weight, small for gestational age, and preterm birth) and neonatal outcomes (neonatal intensive care unit (NICU) admission and decreased Apgar score at 1 min and mean head circumference). 4
Evidence of the adverse developmental impacts of prenatal cannabis use is also emerging. A recent exploratory study showed that prenatal THC-COOH exposure was associated with statistically significant higher scores (worse attention) in the Conners’ Attention scale at 48 months of age; however, scores in both groups remained within the average range. 5 In 2023, Hiraoka et al. conducted a secondary analysis using data from the adolescent brain cognitive development (ABCD) study to determine the effect of prenatal cannabis exposure on cognitive abilities and brain volume development among over 11,000 children aged 9–11 years from 22 sites across the United States. The findings showed an adverse effect of cannabis exposure (retrospectively ascertained through parents’ self-report) on the development of visuo-spatial processing abilities of children. 6
While maternal self-report is a valuable source of information about cannabis exposure particularly during early pregnancy, 7 sole reliance on maternal self-report may introduce classification bias. 8 To examine the associations between prenatal cannabis use and birth and developmental outcomes, large-scale prospective studies with biological samples are needed. Limited data are available about the feasibility and acceptability of biological data collection during the perinatal period, particularly for the purpose of studying substance use. Recent evidence shows that umbilical cord tissue can be a convenient and appropriate substitute matrix to meconium for identifying in-utero exposure. 9 Previous research using umbilical cord tissues for ascertainment of prenatal cannabis use has used anonymous cord tissues 1 or secondary data where participants were not aware their cord tissues were tested for cannabis. 7 The aim of this study was to understand the feasibility and acceptability of collecting umbilical cord tissue for prenatal cannabis research in a community sample of pregnant persons.
Methods
Study design and setting
This feasibility study was conducted in Vancouver, British Columbia (BC) between January 2021 and August 2022. We used a mixed methods approach comprising a prospective cohort study and a qualitative descriptive study. The goal of the quantitative component was to determine the enrollment rate, retention rate, and the number of umbilical cord tissues successfully collected. We also collected information about cannabis use patterns and reasons to use/quit during pregnancy. The aim of the qualitative component was to understand the participants’ perspectives about the feasibility and acceptability of collecting umbilical cord tissue.
Participants
Pregnant people with a singleton pregnancy, 20 weeks gestation or above who were residents of BC, and planning to give birth in a tertiary hospital were eligible to participate. Street drug use and age ⩽ 18 years were exclusion criteria. Participants were enrolled in the study between January 15, 2021 and December 30, 2021. Participants were recruited through study posters distributed in various outpatient maternity care clinics, the hospital’s ultrasound clinic, and social media. Interested individuals were referred to the study website for more information and to complete an expression of interest (EOI) form. Once the potential participant submitted their EOI form, a PDF copy of the consent form became available to review prior to the initial phone call with the research assistant (RA). During the phone call, after confirming the eligibility criteria, the RA reviewed the consent form with the potential participant and addressed any questions. If a person decided to participate, the consent was obtained via an electronic consent form on a secure Canadian data collection platform, Research Electronic Data Capture (REDCap), and a copy of the completed consent form was emailed to the participant.
The consent form provided information about the purpose of the study and what participation involved and described measures of confidentiality and privacy. It also indicated that the participation was voluntary and the participant had the right to withdraw anytime without giving reasons. The consent form indicated that the collected cord tissue would be tested for cannabinoids and that the results would not be shared with anyone outside of the study team or become part of the participant’s or neonate’s medical records. The consent form also indicated that participation did not exclude the participant from any substance screens/tests ordered by their healthcare provider, the hospital, or the Ministry for Children and Family Development as part of standard care. Participants were informed that each cord tissue sample would receive a unique ID number as soon as it was collected and would remain anonymous while undergoing testing and that after the conclusion of the study, the samples would be retained in frozen storage for 1 year and then destroyed. The consent form also stated that participation in this study would not interfere with ability to donate or bank umbilical cord blood through the Canadian Blood Services (CBS).
Data collection
After consenting, participants completed an online questionnaire through REDCap that included questions about their pregnancy, physical and mental well-being, substance use, and demographic information. They then completed monthly surveys until the birth to report on cannabis and other substance use. Depending on the gestational age at the time of enrollment, participants completed between 0 and 4 monthly surveys. The estimated time commitment for completing surveys varied between 10 (monthly surveys) and 30 min (first survey). The cord tissue was collected by hospital staff after delivery. Participants completed two surveys at 2 and 4 months postpartum to report on labor information, postpartum cannabis and substance use, and information about their infant’s well-being and development. Participants received incentives in the form of gift cards for completion of each data collection point. Information about labor and birth characteristics and neonatal well-being up to discharge from hospital was also extracted from the medical charts. After the completion of the study, participants completed an online open-ended questionnaire to provide feedback about their overall experience with the study, including signing up/enrollment, facilitators and challenges of participation, collection of cord tissue, completion of surveys, and their suggestions about how to conduct the study on a larger scale.
Questionnaires
We used two categories of questions to determine self-reported cannabis use during pregnancy:
Self-report—direct question
Participants were directly asked about cannabis use during pregnancy using the Canadian Maternity Experiences Survey (MES) 10 (i.e. “after you realized you were pregnant, did you use cannabis in any form?”). The MES was designed by the Public Health Agency of Canada’s Maternity Experiences Study Group and was pilot-tested by the Statistics Canada in 2005. 11
Self-report—probing intention-to-quit question
Participants were asked “If you are still using cannabis during your pregnancy, do you plan to quit?,” with five possible response options: Yes, I plan to quit; Yes, I plan to cut back; No, I do not plan to quit or cut back; No, I cut back already; and Not applicable, I am currently not using cannabis. This question was inspired by the findings of Mark et al. 12 that showed the majority of pregnant persons who used cannabis report an intention to quit and worded by our research team.
Questions about cannabis use patterns and reasons to use were adapted from the Canadian Tobacco, Alcohol and Drugs Survey. 13 Participants’ motivations to quit cannabis during pregnancy were collected using questions developed by Mark et al. 12 Self-reported data for cigarette and alcohol use during preconception, that is, 3 months before pregnancy/prior to pregnancy recognition, during pregnancy, and at 2- and 4-month postpartum were also collected using questions from the Canadian MES. 10
Umbilical cord tissue
Each person registered for delivery at the hospital had registration paperwork filed with the admitting clerk. On admission to the hospital, the admitting clerk placed a study kit in participants’ hospital chart. The study kit included a biohazard bag, two specimen containers with the participants’ study ID (one for delivery unit to collect the tissue and one for the pathology unit to store the processed sample), and instructions for tissue collection. The cord tissues were collected immediately after birth. Hospital staff were instructed to cut a 7–10 cm section of the umbilical cord, place it in the specimen container, print date, time, and their initials on the container, place container back into the biohazard bag and seal, and place entire study kit in the designated area for the pathology laboratory pick up. After transferring to the pathology laboratory, the samples were cleaned, processed, and temporarily stored until transported to the BC Provincial Toxicology Center. The samples were stored at −20°C. Analysis was performed in batches of samples. In addition to the study samples, 15 blank anonymous cord samples were collected to develop the analytical methods and to prepare the cannabinoids calibration standards and quality controls in cord tissue. Calibration standards and quality controls were prepared as per methods described by Jensen and McMillin. 14 For sample extraction, 1.0 g of umbilical cord was weighed in a tube. The exact weight was recorded for final analyte concentration calculations and 50 µL of internal standard solution was added to all calibration standards, quality control (QC), and study samples, followed by 3 mL of water. All tubes were capped tightly and homogenized using Biotage Bead Ruptor. The homogenates were let to stand at room temperature for 30 min before being transferred to clean tubes and sodium hydroxide (2.5 mL) added. The samples were hydrolysed for 20 min. pH of the homogenates was adjusted to pH 6–7 using 1M hydrochloric acid. Then, 7 mL of hexane: ethyl acetate (9:1, v:v) was added to the samples and the samples were rocked for 10 min, before being centrifuged at 3000 r/min for 10 min. The organic layers were transferred into clean tubes and the solvent evaporated. The extracts were reconstituted in methanol (100 µL). Samples were analyzed on a liquid chromatography tandem mass spectrometry system comprising a Shimadzu LC and a Sciex 4000 QTRAP mass spectrometer and were tested for the presence of δ9-tetrahydrocannabinol (THC), 11-Hydroxy-δ9-tetrahydrocannabinol (OH-THC), 11-nor-9-Carboxy-δ9-THC (COOH-THC), cannabidiol (CBD), and cannabinol (CBN), with a limit of quantitation of 1 ng/g.
Statistical analysis
Descriptive analyses including proportions, means, and measures of dispersion were used to describe the sample demographic, obstetrics and substance use characteristics, and labor, birth and newborn information. Cannabis use rate during pregnancy was determined using both self-report and cord tissue test results. Cannabis use was initially defined based on self-report and then combined with the results of cord tissue testing to determine the exposure as: Negative Exposure
Results
Recruitment and retention
A total of 182 EOI submission forms were received. Of these, 83 entries were determined to be spam (i.e. identical forms submitted in large batches; use of same phone number in multiple different forms). Of the remaining 99 entries, 9 did not meet the eligibility criteria and 5 gave birth before signing up for the study. Of the remaining 85 entries, 26 persons were lost to follow-up after the initial contact and 2 opted out. Overall, 57 participants signed the consent form and were enrolled in the study. The mean age of participants was 33.7 years (3.8) and 23 participants (40.4%) were primigravida. Tables 1 and 2 demonstrate the characteristics of participants and labor and neonates’ information. All participants were planning to give birth at a tertiary hospital except one who was planning to give birth in another hospital and requested to join the study. After consultation with the ethics board and given that this was a feasibility study, we decided to enroll this participant. Of the total 57 enrolled participants, the retention rate was 100% at 2-month postpartum and 94.7% (54 out of 57) at 4-month postpartum. Overall, 48 participants (84.2%) completed the feedback questionnaire.
Characteristics of the participants, Vancouver, British Columbia, Canada (N = 57).

Labor and newborn characteristics.

Newborn’s length, head circumference, and Apgar scores were extracted from medical charts. NICU = neonatal intensive care unit.
Based on medical chart data—missing data for 39.6% of the sample.
Based on medical chart data—missing data for 37.8% of the sample.
Based on medical chart data—missing data for 19.3% of the sample.
Based on medical chart data—missing data for 19.3% of the sample.
Cord tissue collection
The cord tissue was collected for 39 participants (68.4%). The sample weights varied between 5.8 and 12.2 g. The sample weight used for analysis varied between 0.9 and 1.1 g. The reasons for not collecting samples that our team was able to identify included study kit not pulled at registration desk (n = 8), staff forgetting to collect/save sample after delivery (n = 5), and unknown reasons (n = 5). In qualitative data, two participants noted that the staff forgot to collect samples due to pregnancy complications or emergency cesarean. Quantitative data showed that the rates of cord tissue collection for participants with vaginal, elective cesarean, and emergency cesarean delivery were 73.0%, 71.4%, and 53.8%, respectively. The rates of collection for participants with spontaneous and induced labor were 81.5% and 50%, respectively. The rates of cord tissue collection for participants who received most of their prenatal care from a midwife, family physician, and obstetrician were 73.3%, 66.7%, and 61.5%, respectively. The rates of cord tissue collection (missing data for six participants) were similar for those who planned to donate cord blood (70.6%) and those who did not (73.3%).
Cannabis use
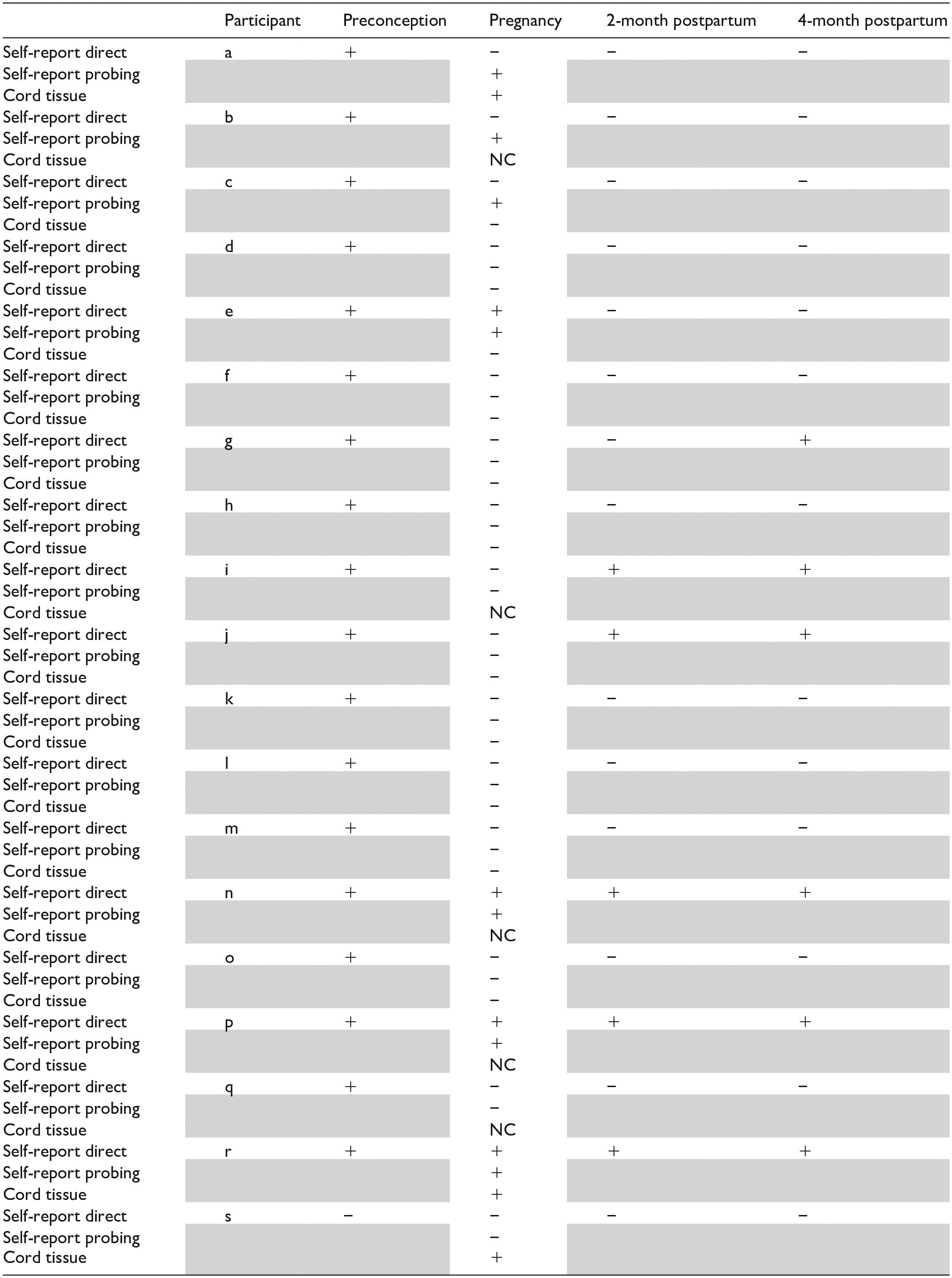
In total, 18 participants (31.6%) reported using cannabis during the preconception period. Four participants (7.0%) reported cannabis use during pregnancy in direct self-report questions. Three more participants reported cannabis use during pregnancy in probing questions with a total of seven participants (12.3%) reporting cannabis use during pregnancy in self-report probing questions. Three participants (5.3%) tested positive for cannabinoids in umbilical cord tissue testing. Based on either self-report or cord tissue test, the rate of cannabis use during pregnancy in this sample was 14.0% (n = 8). Figure 1 demonstrates the cannabis use during pregnancy based on self-report and cord tissue test results.

Cannabis exposure ascertainment (N = 57).
Five and six participants (8.7% and 11.11%) reported cannabis use at 2- and 4-month postpartum, respectively. Table 3 demonstrates cannabis use during the preconception/pregnancy/postpartum periods. The agreement between self-report direct questions and cord tissue test was fair (kappa 0.36, 95% CI −0.20 to 0.92). The agreement between any self-report (direct and probing) and cord tissue test was moderate (kappa 0.53, 95% CI 0.06–0.99).
Cannabis use from preconception to 4-month postpartum.
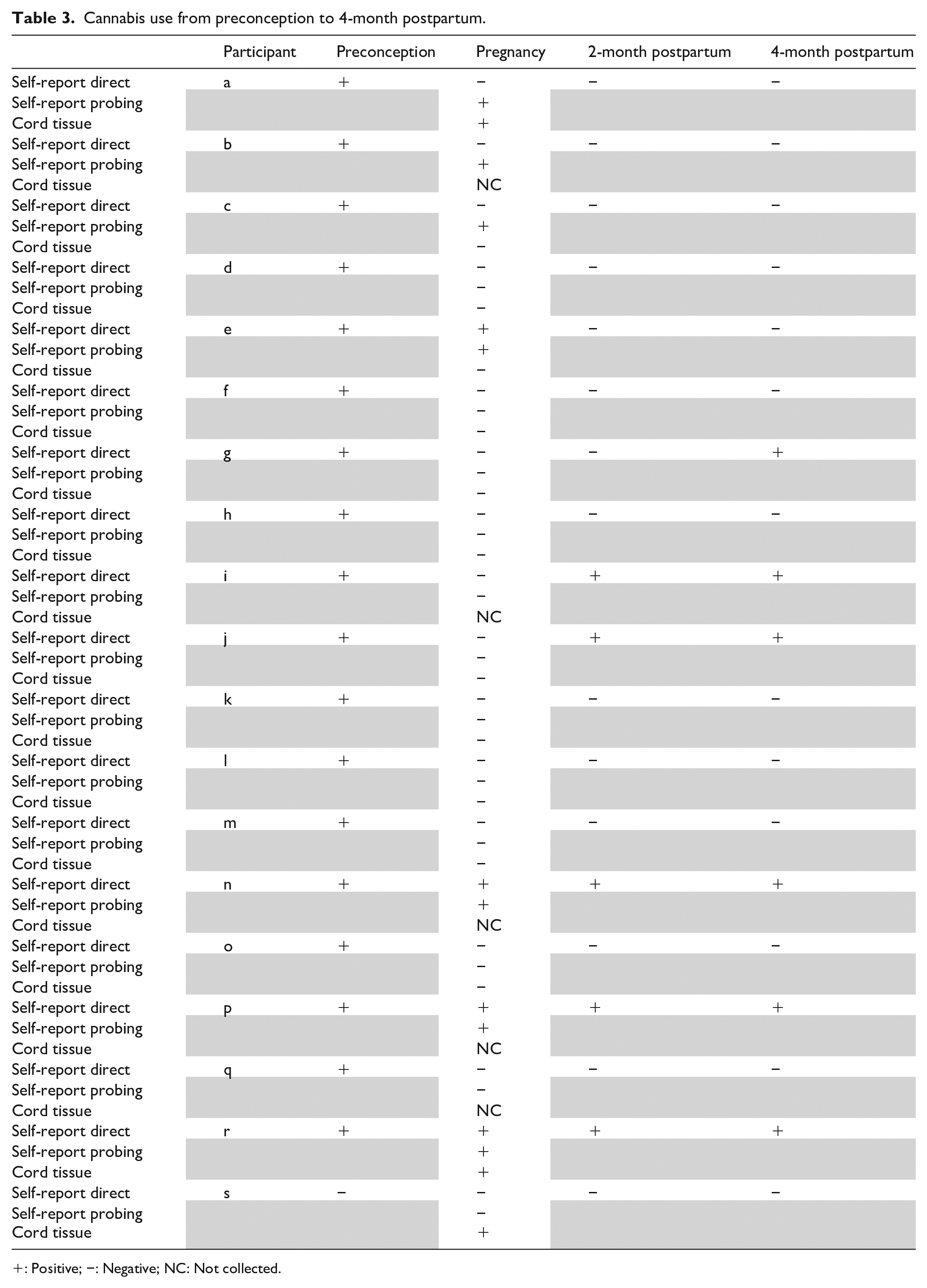
+: Positive; −: Negative; NC: Not collected.
Frequency of use
Table 4 summarized the frequency of cannabis use during preconception and pregnancy. All persons who used cannabis daily during the preconception period (n = 4) reported continuing to use during pregnancy, among whom two participants continued to use daily, one participant reported using a few times a week, and one participant reported occasional use.
Method and frequency of cannabis use during preconception (n = 18) and pregnancy (n = 4), (self-report; direct questions).

CBD: Cannabidiol; THC: δ9-tetrahydrocannabinol.
Co-use of cannabis, cigarettes, and alcohol
Of the seven participants who self-reported cannabis use during pregnancy, one reported using cigarettes and another one reported using alcohol. No participants in this sample self-reported using all three substances during pregnancy.
Methods of use
Most persons who used cannabis reported using cannabis in various methods. Table 4 summarized the methods of cannabis use during preconception and pregnancy. All persons who reported cannabis use during pregnancy reported smoking cannabis. Two participants reported other methods of use in addition to smoking.
THC/CBT concentration
Of the seven participants who self-reported cannabis use during pregnancy, four reported that the THC/CBT concentration of the cannabis products that they use varied each time. Three participants reported that they did not know the THC/CBT concentration of the cannabis products that they were using. Three participants reported obtaining the cannabis products from partner or friend. Three purchased and one reported growing it.
Medical use
Among persons who used cannabis during preconception, six participants reported using cannabis for medical purposes, such as insomnia, chronic pain, nausea/vomiting, depression, or anxiety. During pregnancy, two participants reported use for medical reasons, including anxiety and nausea/vomiting, of whom both reported daily use.
Planning to quit
Of the seven participants who self-reported cannabis use, one reported quitting at the time of completing the survey, two reported no intention to quit, and four reported an intention to quit. In response to the question about the reasons that the participant would want to quit or cut back cannabis use, participants who reported planning to quit/cut back stated the following reasons: not fitting with who they want to be, it can hurt the pregnancy/cause long-term harm for the baby, proving to themselves that they can quit, to avoid legal problems/ or involvement with child protection services after the birth and saving money. Participants who reported no intention to quit used cannabis daily and reported the following reasons as potential motives to quit: avoiding legal problems/child protection services involvement after the birth and saving money.
Participants’ perspectives
Overall, 48 participants provided qualitative data. Findings were classified into five themes: appreciating the value of research, feeling well-informed during the consent process, perception of study processes as efficient, perceptions and feedback about cord tissue collection, and feedback on questionnaires. Table 5 demonstrates themes and representative quotes.
Themes and representative quotes.

Appreciating the value of research
Most participants recognized the topic of the research to be important as there is limited knowledge in the area. They reported an aspiration to contribute to the new knowledge for two different reasons: to identify harms associated with cannabis or eliminate stigma around cannabis use during pregnancy.
Feeling well-informed during the consent process
Participants reported having “ample opportunity to read and understand” the consent form, as the consent form was available to them prior to the initial RA phone call. They also reported that they were provided with “all information needed to make an informed decision.” They described the enrollment and sign-up process to be easy and straightforward. Participants also reported that because they were providing biological samples, the initial review and debrief of the consent form, and answering questions helped them to understand what they were “getting into” and “what to expect” and to “feel comfortable” throughout the study.
Perception of study processes as efficient
Participants reported that the time commitment for the study was reasonable and “not too much of a burden.” They described study communications to be timely and concise and non-judgmental. They reported that the overall process was seamless and straightforward, all information was clearly conveyed and that they liked receiving gift cards as incentives.
Perceptions and feedback about cord tissue collection
Overall, participants reported that the collection of cord tissue for cannabis testing was acceptable. One participant noted that at the beginning of the study, they felt slightly uncomfortable about the idea of cord tissue collection but they reported feeling being comfortable when the time came. Another participant noted that because they understood that the cord tissue would only be used for the purpose of this study, they felt very comfortable about providing tissue. Participants had mixed reports about their experience with cord tissue collection at hospital. Several participants noted that the staff were aware of the study and the collection process went smoothly. However, several participants reported their samples were not collected for various reasons, such as pregnancy complications, emergency cesarean delivery, staff forgetting to collect, or being unaware about the study. Some participants noted that sometimes after the participant informed or reminded the staff, the cord tissue was still not collected. Participants’ suggestions indicated that they were interested in being actively involved in the collection process to ensure their samples were collected. For example, one participant suggested providing “printout on what need to be done so we can hand to our nurse/midwife on delivery day.” Another participant noted, “let patient know what the collection kit looks like so they can remind partners/doulas to remind hospital staff to collect it.”
For participants who were planning to donate their cord blood, the cord tissues were collected by our team after collection of cord blood by CBS staff. To coordinate with CBS, the research team sent the CBS team a biweekly list of participants who indicated participation in cord blood donation. The CBS team cross-referenced this list with each sample they received to determine which samples must be saved for the study. To ensure patient confidentiality, the CBS team followed a protocol to delete all identifiable patient information after the transfer of the cord to research team. Overall, the coordination with the CBS was smooth and cord tissues from most participants planning to donate were collected. Participants reported that their decision to participate in the study was facilitated by knowing that participation would not interfere with cord blood donation. Although, the study consent form indicated an ability to simultaneously participate in the study and donate cord blood, some participants recruited at the beginning of the study reported that they perceived by participating in the study they would not be eligible for cord blood donation. Later, we included an explicit conversation statement in the initial intake call to enquire about participants’ intention to donate cord blood and discuss the process.
Feedback on questionnaires
Given the longitudinal design of the study, several questions were repeated in each survey. Participants provided mixed feedback about the repetition of the questions. Some participants reported that they liked that the questions were consistent and that they felt “checked-in” with repeated questions. However, some participants disliked the repetitive questions and noted that information did not change between the surveys. Some participants suggested having an option to have access to their previous answers to the same question and an option to add additional comments if needed.
Participants noted that they would prefer shorter surveys for the postpartum period as with arrival of the new baby their time was more limited. Participants also reported a need for text reminders and phone-friendly surveys given the busy nature of the postpartum period. Participants also suggested including open-ended questions at the beginning of the survey and before multiple-choice questions.
Participants particularly provided several feedback comments about cannabis use questions. Some reported that these questions were straightforward and “asked in a non-judgmental and matter of fact way.” One participant who used cannabis described some questions to be vague and not applicable to the actual use of cannabis. This participant suggested providing options for various types of cannabis strains. Other feedback included adding two separate questions to determine postpartum cannabis resumption after the baby was born and after weaning the baby from breastfeeding.
Discussion
Overall, we found that the collection of cord tissue for cannabis research was acceptable for most pregnant people. Although it was difficult to estimate how many individuals viewed the study poster, of the 99 individuals who submitted the EOI form, 85 were eligible to participate. Among these, 57 individuals (67%) consented to the study. This rate is similar to that reported in a UK feasibility study (65%) for cord blood and stool sample collection. 18 Our qualitative data indicated that the use of cord tissue solely for research purposes, clear elaboration of study steps in the consent form, and an initial phone call to discuss details helped the participants to feel informed and comfortable with participation.
We were not able to locate a feasibility study regarding the collection of cord tissue for the purpose of measuring cannabinoid levels in the literature. The limited available data show that the collection rates for other cord products may vary based on the study setting. In a feasibility study conducted in Germany on the topic of childhood leukemia, Ernst et al. 19 reported that of the 41 participants who consented to the collection of umbilical cord blood, samples were collected for 54% participants. In a pilot study in the United Kingdom on the topic of microorganisms and immunological events, Bailey et al. 18 examined the feasibility of stool and cord blood sample collection and reported that 66% of participants provided at least one sample and 79% provided samples for cord blood. This rate is slightly higher than the rate of cord tissue collection in our study (68.4%). The samples in Bailey et al. 18 were collected by the research staff. Similar to our study, the samples in the Ernst et al. study were collected by hospital staff. The introduction of collection of additional biological samples from patients in hospitals can be challenging. A qualitative study that described the experience of frontline staff at the CBSs’ Cord Blood Bank found that the frontline staff may experience added emotional labor, which may include workload related to communication coordination for a positive donors’ experience and acquiring the acceptance and support of hospital staff and navigating organizational hierarchies. 20 In our study, not placing study kit in participant’s chart at admission was one of the main reasons for samples being missed. A Canadian pilot study that evaluated the feasibility of conducting a randomized controlled trial on the impact of a new technology reported that clerk unfamiliarity with the study can hinder the study progress. 21 While, we made several efforts to improve this clerical task through introducing our study to admitting clerks, due to large number of often part-time clerks working at various shifts, we found this challenging. Admitting clerks were not hospital employees and reported to managers outside the hospital, which made liaising with clerk supervisors for task clarification more challenging. These data suggest that a central unit at hospitals to facilitate resource allocation and direct communication between research groups and various hospital units can facilitate future studies.
Participants’ notes indicated that complicated labor and emergency cesarean delivery interfered with their cord tissue collection. We observed that the rates of cord collection were higher among those with vaginal delivery or planned cesarean than those with an emergency cesarean. The collection rate was also higher among those with spontaneous labor compared to those with labor induction. Other studies have also reported higher rates of cord product collection in planned cesarean deliveries 18 or samples not being collected as a result of emergency procedures and labor complications. 19 While some studies suggested that cord blood banking may be given priority over research studies, 18 in our study, the rates of cord collection were similar for those who were planning to donate and those who were not. This may be explained by the fact that our study was collecting a different cord product (tissue) than the CBS (blood). Indeed, we found liaison and coordination with the CBS more feasible given that there were fewer staff with whom to communicate and coordinate the collection process.
In 2020, using cross-sectional data from the 2016 Pregnancy Risk Assessment Monitoring System in the United States, Skelton et al. 22 reported a prevalence rate of 14.73% and 5.65% for cannabis use in the preconception and during pregnancy, respectively. The rate of preconception and pregnancy cannabis use in our sample was 31% and 14%, respectively, which is also higher than the rates reported in a previous BC study. 23 This might be partially related to the fact that study was conducted during the COVID-19 pandemic. A US-based study showed that compared to the cannabis use rates before the pandemic, the rates during the pandemic increased 25% among pregnant persons. 24
In our study, to improve maternal self-report during pregnancy, we used two types of questions during pregnancy to determine self-report: direct and probing intention-to-quit questions. In our sample, 43% of self-reported use were identified through the probing intention-to-quit questions. Given the stigma around substance use, it is possible that framing questions around intention to quit provides a more socially desirable context to initiate conversation about cannabis use during pregnancy. The agreement between self-reported cannabis use with biomarker detection in umbilical cord tissues in our study was moderate and was similar to that reported in a secondary linkage of 116 surveys data with umbilical cords in Colorado. 7 In a state-wide analysis of anonymous cord tissues in Utah, 2.5% of umbilical cords tested positive for cannabis. 1 Accumulating evidence suggests that the levels of cannabinoids in umbilical cord are mainly associated with the cannabis use in the last 4 weeks of pregnancy and may not be reliable indicators of earlier use. 7
Limitations
This study included a convenient sample and thus is prone to selection bias. A sample size between 24 and 50 participants has been recommended for feasibility studies.25 –27 While our sample size was 57, the small number of persons who used cannabis in our sample is a limitation of this study; therefore, findings cannot be generalized. Another limitation of the study is the potential recall bias for maternal self-report. An important consideration is that the study was conducted during the pandemic when the availability of prenatal care or classes might have been limited or altered. Another limitation is that the study did not include other forms of biological confirmatory testing in addition to cord tissue testing. We were not able to quantify the levels of cannabinoids in cord tissues. The study samples were between 8 and 20 months old at the time of analysis. Data on stability of cannabinoids in frozen umbilical cord are limited and range between 14 days and 1 year.28,29 In addition, the limit of quantitation used in our laboratory was 1 ng/g. Previous studies have used a limit of quantitation of 100 pg/g. 7 Despite these limitations, the detection agreement between self-report use with umbilical cords was similar to that reported previously. 7 The study sample was highly educated and over 80% of participants reported their ethnicity as White or Asian thus the findings may not be generalizable to other populations.
Implications
Our data suggest that rates of cord tissue collection may be increased through implementation of collection protocols for pregnancies with labor interventions or emergency procedures. Our findings also suggest that active involvement of participants in the collection process, having a central hospital unit to liaise direct communication and providing reminders for hospital staff to collect samples, may improve collection rates.
Prior evidence suggests that only people who use cannabis during the preconception period continue to use during pregnancy. 30 Given the comparable self-report rates among general population of females and preconception people, it is possible that individuals are more comfortable to disclose preconception use. To improve maternal self-report during pregnancy, our data suggest using two-step screening in which all pregnant people are asked whether they used cannabis during the preconception period. If they indicated use, then a question about an intention to quit should be followed rather than a direct question as to whether or not they used during pregnancy.
Conclusion
The collection of cord tissue was perceived to be acceptable by most participants. Use of detailed consenting procedures and sole use of tissue for study purposes supported participants’ decision to participate. Having a central hospital unit to liaise direct communication and active involvement of participants might facilitate biological sample collection. Our observation that collection rates were lower among participants with complicated labors suggests that logistical barriers might be overcome by implementation of collection protocols for persons requiring more complex intrapartum care. These approaches could ensure proportional representation of biological samples from all perinatal risk categories in larger studies and reduce the potential of selection bias.
Footnotes
Acknowledgements
The authors thank the Perinatal Wellbeing Research team for their contribution in conducting the study, Mary Bunka for developing cannabis-related questions, and the study participants who generously shared their experiences.