Abstract
Background:
Studies had compared single-embryo transfer to double-embryo transfer with cleavage stage embryos and found that while single-embryo transfer was less costly, it was also associated with a lower live birth rate than double-embryo transfer. A single blastocyst transfer has been shown to improve the live birth rate per cycle compared to single-embryo transfer at cleavage stage.
Objectives:
To compare live birth rates and real costs of elective single-embryo transfer to double-embryo transfer and to determine the incremental cost-effectiveness ratio of these two strategies in an unselected pool of women in a single center.
Design:
Retrospective study.
Methods:
We analyzed data of 4232 women who underwent their first fresh in vitro fertilization/intra-cytoplasmic sperm injection cycles with at least two embryos available for transfer in KK Women’s and Children’s Hospital from 2010 to 2017.
Results:
Five hundred and sixty-four women underwent elective single-embryo transfer and 3668 women underwent double-embryo transfer. One hundred and fifty-six women who failed to achieve a live birth in their fresh elective single-embryo transfer cycle underwent a sequential thaw single-embryo transfer cycle. Live birth rate of fresh elective single-embryo transfer was significantly higher at 41.3% than that of double-embryo transfer at 32.6%. Cumulative live birth rate for sequential elective single-embryo transfer (fresh elective single-embryo transfer + thaw single-embryo transfer) was 47.9%. After accounting for variables which may affect live birth rates such as age and stage of embryo transfer, the odds of achieving a live birth from double-embryo transfer was 24% lower than that from sequential single-embryo transfer, although not statistically significant. For every live birth gained from an elective single-embryo transfer compared to double-embryo transfer, cost savings were S$20,172 per woman. If a woman had to have a sequential single-embryo transfer after a failed single-embryo transfer in her fresh cycle, cost savings were reduced to S$1476 per woman.
Conclusion:
Single-embryo transfer is a dominant strategy in an unselected population and adopting it in assisted reproductive treatments (ART) can produce cost savings without compromising on live birth rates.
Introduction
Transferring two or more embryos in order to increase live birth rate (LBR) have resulted in a significant rise in multiple pregnancies. 1 Compared to singleton pregnancies, twins and higher order multiple pregnancies are associated with increased obstetric and perinatal morbidity and mortality and increased healthcare costs. Intuitively, single-embryo transfer (SET) appears to be the way to reduce multiple pregnancies with its attendant risks. As such, many committees such as the Human Fertilization and Embryology Authority in 2008 2 and the American Society for Reproductive Medicine (ASRM) in 2012 3 have recommended utilizing elective single-embryo transfer (eSET) to reduce multiple birth rates.
A systematic review in 2020 on the number of embryos for transfer following in vitro fertilization (IVF) or intra-cytoplasmic sperm injection (ICSI) evaluated the approach of single- versus double-embryo transfer (DET) in the first cycle, and DET in the first cycle versus sequential SET. 4 Compared to DET, SET was found to reduce twinning rate by sevenfold but was also associated with a lower LBR. However, the use of sequential SET was found to have a comparable cumulative LBR to DET with a significantly lower risk of multiple pregnancies, albeit with a longer time frame. An increase in treatment costs is theoretically expected in sequential SET as the number of cycles taken to achieve a live birth is increased. However, the reduction in multiple pregnancies and the costs associated with complications of multiple pregnancies may offset the higher treatment costs of sequential SET.
A systematic review in 2007 on the cost-effectiveness of embryo transfer strategies (one fresh eSET versus one fresh DET, two fresh eSET versus one fresh DET, one fresh followed by one frozen SET versus one fresh DET) found that DET in a single fresh cycle produced the highest LBR but was also the most expensive due to the high costs of multiple pregnancies. 5 The LBR in eSET was only comparable if performed in younger patients with favorable prognoses and when a sequential frozen ET was included. A more recent systematic review on cost-effectiveness in 2016 showed similar results, with an eSET strategy showing a relative reduction of 12.5% in the LBR and a relative reduction of 16.9% in costs compared to DET. 6 When DET was compared with the sequential eSET, a smaller difference between the cumulative LBR was reported. Previously mentioned systematic reviews included studies which utilized cleavage staged embryo transfers or where the embryo stages at transfer were not stated. With the advent of extended embryo culture techniques, single blastocyst transfer (SBT) has been shown to improve the LBR per cycle compared to SET at cleavage stage. 7 A non-statistically significant increase in the cumulative LBR was also shown with a cycle of fresh SBT followed by a frozen SBT compared to a single fresh double blastocyst transfer (DBT). 8
Cost-effectiveness studies of SET strategies using blastocyst transfers have been limited. Crawford et al. 9 found that sequential SBT in the majority of cycles for women below 35 years of age reported lower medical costs and multiple births without compromising on the LBR as compared to DET. With the revision of ASRM guidance in 2017 to recommend SET even for women older than 38 years if good quality embryos are available, 10 there is also a need to evaluate the costs for eSET in older women. Hence, the objectives of this study are to compare the LBR and real costs of sequential eSET/SBT compared to DET and to determine the incremental cost-effectiveness ratio (ICER) of these two strategies in patients of unselected age groups who underwent IVF/ICSI in a single Academic Medical Centre in Singapore.
Materials and methods
This retrospective study was approved by the Centralized Institutional Review Board Singapore and informed consent had been waived. Data collection was carried out in accordance with relevant guidelines and regulations. Women who underwent their first IVF/ICSI cycle at KK Women’s and Children’s Hospital (KKH) between 2010 and 2017 were included. A comparison analysis of a sequential eSET versus DET was undertaken. Sequential eSET was defined as the elective transfer of a single embryo in the first fresh cycle and if a live birth did not occur, to perform another SET in the subsequent frozen cycle. DET was defined as the transfer of two embryos in the first fresh cycle. The inclusion criteria for the sequential eSET arm included a minimum of two embryos available for transfer during the first fresh cycle and a minimum of one frozen embryo available after fresh embryo transfer. For the DET arm, the inclusion criteria were having at least two embryos available for transfer. Exclusion criteria included having only one embryo available for transfer during the first fresh cycle, use of donor oocytes, and cycles with pre-implantation genetic screening. For the sequential eSET arm, women who did not achieve live births after fresh eSET were tracked using anonymized patient numbers and included for analysis if they subsequently underwent frozen cycles of SET.
Characteristics
Patient demographics and cycle characteristics of sequential eSET were compared to DET. Demographic data collected included age, race, paternity, and primary cause of infertility. Cycle characteristics collected included number of oocytes retrieved, number of embryos frozen, and stage of embryo transfer. Cycle outcomes included the LBR, mode of delivery, and gestation at delivery. A live birth is defined as a baby born at or after 24 weeks of gestation. Cumulative LBR was defined as the sum of live births from both fresh and frozen transfers over the number of women who underwent fresh cycles.
Estimated costs
All estimated costs in this study are based on rates prior to any government subsidies and were obtained from hospital averages in KKH. The total costs of the respective sequential eSET and DET arms include the costs of fresh IVF/ICSI cycles and medical costs for live births, which included both fees for delivery and neonatal care if delivery occurred before 37 weeks of gestation. This study assumed no additional cost for neonatal care for a baby born at 37 weeks and beyond. Total cumulative costs for sequential eSET also included costs for thaw cycles and medical costs for additional live births. Average costs per woman were then derived from the total costs divided by the number of women in each arm. The average cost of a fresh IVF/ICSI cycle in Singapore dollars with and without blastocyst culture was estimated to be $16,277 and S$15,742, respectively. This included the cost of ovarian stimulation, cycle monitoring, oocyte retrieval, embryo transfer, and embryology laboratory cost. A thaw cycle was estimated to cost S$3500, which included the cost of medications (if required), cycle monitoring, embryology, and transfer. For delivery, the average costs of a vaginal birth and a cesarean section were S$5220 and S$8390, respectively. Additional costs of neonatal care for a baby born before 37 weeks were estimated based on the gestation at which the baby was born. Neonatal care costs for pre-term babies ranged from S$1681 per baby born at 36 weeks to $323,219 per baby born at 24 weeks.
Statistical analysis
Analysis was performed with Stata 11.0. Continuous and categorical variables between the eSET and DET group were, respectively, compared using the Student t-test and the chi-square test. Univariate analyses were used to assess relationship between predictive variables and outcome of live births. Significant predictive variables from univariate analyses were then included in multivariate logistic regression analysis to evaluate their effect on live birth. An ICER was obtained from the formula: ICER = (mean SET cost − mean DET cost)/(percentage of live births in SET − percentage of live births in DET). 11 The ICER expresses the additional cost incurred by SET to gain an extra live birth over DET. The lower the ICER, the more cost effective the strategy. A strategy is considered “dominant” if it is less costly and more effective than an alternative strategy and a negative ICER will be obtained. If the strategy is superior but more costly (positive ICER), then the cost-effectiveness of the strategy will depend on the threshold which the society is willing to pay per extra live birth.
Results
A total of 4232 women were included in this study, with 564 women who underwent eSET and 3668 who underwent DET in their fresh cycles. Demographic data and cycle characteristics are shown in Table 1. The mean age of women in the eSET arm was 34 years compared to 34.5 years in the DET arm. The racial composition of the women in both groups had the majority as Chinese, which is reflective of Singapore’s racial composition. Both groups shared the top two causes of infertility, which were male factor and unexplained. The eSET group had higher percentage of women with ovulatory disorders (15.8% versus 8.8%) and other factors (0.9% versus 0.3%) and lower male factor infertility (35.8% versus 49.9%) compared to the DET group. The majority of women in both groups were nulliparous.
Demographic, cycle characteristics and outcomes of fresh cycles.

eSET: elective single-embryo transfer; DET: double-embryo transfer; ICSI: intra-cytoplasmic sperm injection; IVF: in vitro fertilization.
In our center, a combination of IVF and ICSI is performed if at least 15 oocytes were retrieved.
When women were stratified into age groups, the differences in live birth rates among the three age groups did not differ significantly in the eSET group but were significantly different in the DET group.
Statistically significant.
ICSI was performed in 78.7% and 88.7% of the women in the eSET and DET groups, respectively. The remaining underwent a combination of IVF and ICSI, which is the protocol in our center for women with at least 15 oocytes retrieved. The mean number of oocytes retrieved was 14.2 for the eSET group and 13.9 for the DET group. The eSET group had a higher percentage of blastocyst transfer (84.8% versus 1.7%) compared to the DET group. The mean number of embryos for cryopreservation was also higher in the eSET group compared to the DET group (4.6 versus 3.3).
The LBR for the fresh eSET group was 41.3% (233/564), of which 39.7% (224/564) were singletons, 1.6% (9/564) were twins with no triplets (Figure 1). The LBR for the DET group was 32.6% (1195/3668), of which 24.5% (898/3668) were singletons, 8.1% (296/3668) were twins, and 0.03% (1/3668) were triplets. The eSET group had a significantly higher LBR than the DET group (p < 0.0001). When the women were stratified into age groups and LBR was compared among the age groups separately in each arm, LBR was not significantly different among the different age groups in the eSET group. Whereas within the DET group, younger women had significantly higher LBR than older women (Table 1).

Outcomes and ICER.
Among the 331 women who failed to achieve a live birth in their fresh eSET cycles, 171 women (51.7%) embarked on a thaw cycle using embryos derived from the same fresh cycle (Figure 1). The interval between the fresh and thaw cycle ranged from 0.8 to 16.4 months, with the median being 3.5 months. In total, 156 women (47.2%) went on to have sequential eSET as 15 women (4.5%) had canceled cycles due to embryo attrition after thawing. Of the 156 thaw transfers, 90.4% (n = 141) had a SBT while 9.6% (n = 15) had cleavage stage SET (Table 1). There were 37 singleton live births (LBR of 23.7%) and all were born at term except for one baby born at 36 weeks (Table 2, Figure 1). There were no multiple pregnancy deliveries in the thaw transfers (Figure 1). Cumulative LBR for sequential eSET was 47.9% (Figure 1).
Unit and average costs of IVF/ICSI procedures, deliveries, and NICU admissions.
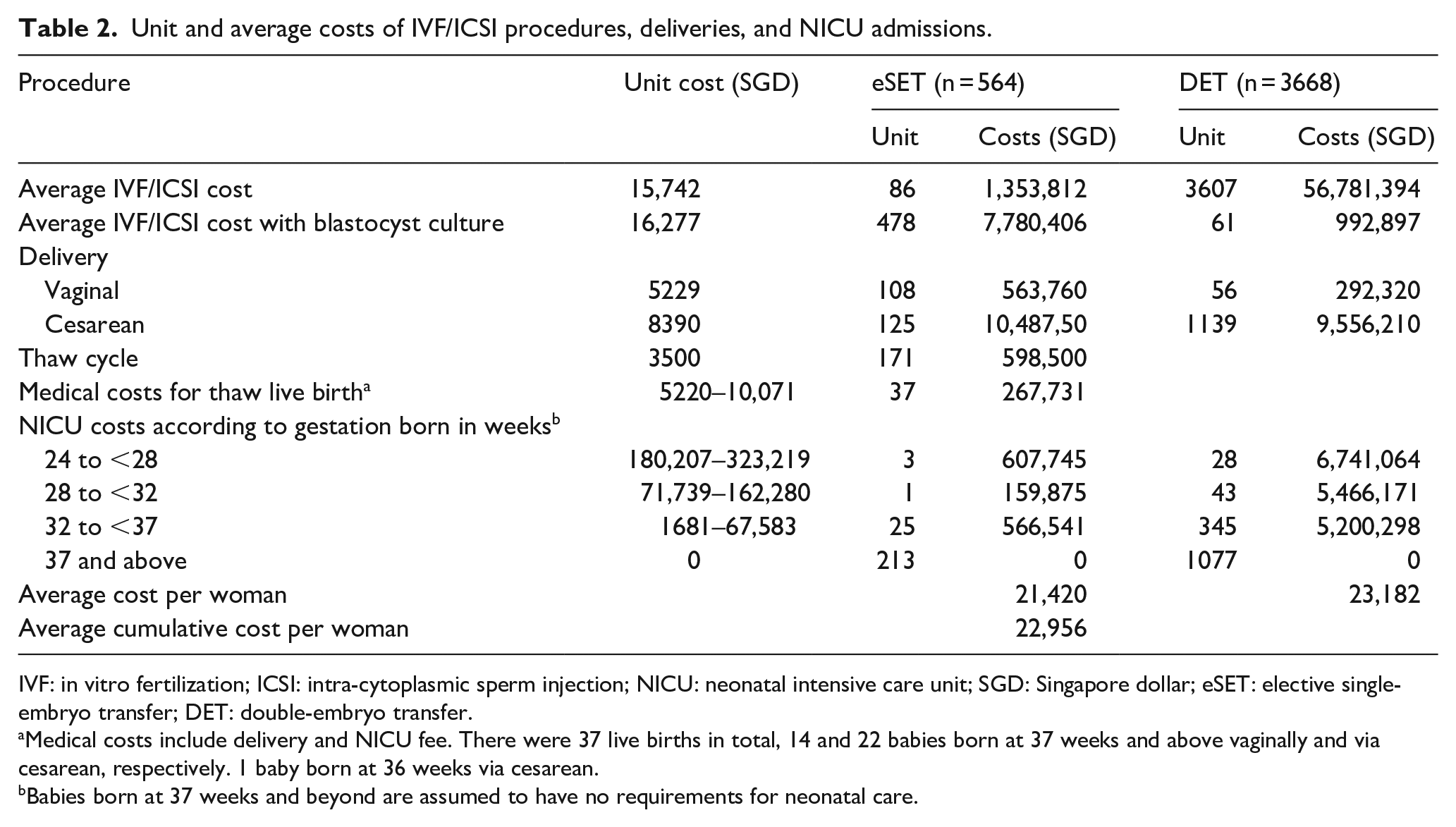
IVF: in vitro fertilization; ICSI: intra-cytoplasmic sperm injection; NICU: neonatal intensive care unit; SGD: Singapore dollar; eSET: elective single-embryo transfer; DET: double-embryo transfer.
Medical costs include delivery and NICU fee. There were 37 live births in total, 14 and 22 babies born at 37 weeks and above vaginally and via cesarean, respectively. 1 baby born at 36 weeks via cesarean.
Babies born at 37 weeks and beyond are assumed to have no requirements for neonatal care.
Using logistic regression, the odds of achieving a live birth from a fresh cycle of DET was 1% lower than that for eSET but was not statistically significant (Table 3). Using cleavage staged embryos and increasing age were significant independent factors associated with reduced chances of achieving a live birth. Comparing cumulative LBR from sequential SET and DET (Table 3), the odds of achieving a live birth from DET was 24% lower than that from sequential eSET. However, the reduced odds were not statistically significant. Table 2 lists the unit costs used in the cost analysis. The average cost per woman to have a fresh eSET cycle for a 41.3% LBR was S$21,420, and this cost increased to S$22,956 if she went on to have a sequential thaw SET with a corresponding increase in the LBR of 47.9% (Table 2, Figure 1). The average cost per woman to undergo a fresh DET was higher at S$23,182 for a lower LBR of 32.6%. The ICER over DET with a single SET and a sequential SET was −S$20,172 and −S$1476, respectively (Figure 1).
Cycle comparison of eSET versus DET and sequential SET versus DET.

eSET: elective single-embryo transfer; DET: double-embryo transfer; SET: single-embryo transfers; CI: confidence interval.
Statistically significant.
Discussion
Our center has been progressing moving toward blastocyst culture and eSET since 2014. In our study, 84.8% of the eSET cohort had fresh blastocyst transfers and 90.4% had thawed blastocyst transfers. A higher LBR was achieved in the eSET group (41.3% for SET and 47.9% for cumulative SET) compared to the DET group (32.6%). The higher live birth rate in the SET group was most likely due to the higher percentage of blastocyst transfers compared to DET group which had mostly cleavage staged embryo transfers. Two studies8,12 compared women undergoing SBT to women undergoing DBT and had different conclusions. One study 8 showed similar LBR between the SBT and the DBT arm (39% versus 44.7%) while the other 12 showed significantly lower LBR in the SBT arm compared to the DBT arm (49% versus 70%). In both studies,8,12 the DBT arm yielded significantly higher twinning rate (29.2%–45%) compared to the SBT arm (0%–4.2%). While DBT may or may not improve LBR, DBT significantly increases twinning rate compared to SBT. The potential benefits of DBT will have been negated by the risks of multiple live births which should be considered and relayed to the couple.
In our study, the multiple LBR was 1.6% and 8.1%, respectively, in the eSET and the DET group. When comparing single blastocyst versus single cleavage stage embryo transfer, the risk of twins appears higher with blastocyst embryos.13,14 A retrospective study of 937,848 SET cycles reported an increased odds of twinning by 1.79 times from blastocyst compared to cleavage stage transfer. 15 However, the increased risk of twinning from SBT is still relatively low compared to a fivefold increase in higher order pregnancies from DET as shown in this study.
While the magnitude of difference was not statistically significant, women who underwent DET were 24% less likely (odds ratio (OR) = 0.76, p = 0.12) to achieve a live birth than those who underwent sequential SET. The embryo stage at transfer was found to be a significant independent factor in determining LBR, and transferring a cleavage stage embryo was associated with a 32% reduction in odds of achieving a live birth compared to a blastocyst transfer. Increasing maternal age was also an independent factor in reducing the odds of achieving a live birth. The magnitude of reduction at 8%, however, was lower than that for using cleavage staged embryos. In the univariate analysis, women in the older age groups had lower LBRs than women in the younger age groups in both SET and DET groups. The differences were only significant in the DET group in which the majority of transfers used cleavage staged embryos. This suggests that if blastocysts are available for transfer, eSET is an option even for older women without compromising on LBR.
The analysis in this study showed eSET and sequential eSET to be more effective than DET as well as less costly. A study utilizing a decision tree model to evaluate cost-effectiveness of sequential SET versus DET with cleavage stage embryos also showed that sequential SET was more effective and less costly, and was the preferred strategy in younger women under 32. 16 In another retrospective cohort study, 9 sequential SET in patients below 35 years old was found to have lower total IVF/ICSI and pregnancy-associated medical costs than DET by reducing multiple births and preterm deliveries while maintaining the LBR. For every live birth gained from an eSET compared to DET, cost savings were S$20,172 per woman. If a woman had to have a sequential SET after a failed SET in her fresh cycle, cost savings were reduced to S$1476 per woman.
The rates of SET uptake depend on a myriad of clinical, social, and economic factors, including patients’ perception of SET success, their preferences, as well as the public funding model. 17 In Singapore, the national IVF/ICSI funding policy for restructured government hospitals provides subsidies of up to 75% for a maximum of three fresh and three thaw cycles. 18 The number of assisted reproduction cycles performed in Singapore has risen from below 5000 in 2012 to above 6000 in 2015. 19 A society which heavily subsidizes healthcare will benefit from cost savings using strategies such as eSET and sequential eSET to reduce multiple pregnancies while maintaining LBRs.
There are several limitations in this study. First, stimulation and thaw programs were not standardized due to the retrospective nature of this study. Second, cycle and neonatal intensive care costs were based on average estimates and were not patient specific, potentially resulting in over- or under-estimation of costs. The database also did not capture the treatments for ectopic pregnancies or miscarriages resulting from IVF/ICSI and hence these costs were not included. Third, our eSET group comprises largely of blastocysts (~85%) while the comparator group of DET were largely cleavage stage embryos (~98%). Performing a DBT in the first fresh cycle is uncommon in our center due to the risks of multiple births and its associated complications. Consequently, a comparison between SBT and DBT may not be clinically relevant. Finally, power analysis for sample size calculation was not performed in this study.
Conclusion
This study demonstrates that SET is a dominant strategy and adopting it in assisted reproduction can produce cost savings without compromising on LBRs. With blastocyst transfers, age is less of a deciding factor as to which strategy to choose. In addition, perinatal outcomes are improved with SET by reducing multiple pregnancies and preterm deliveries.
