Abstract
Osteoporosis affects one in every five women over the age of 50 worldwide. With a rapidly ageing population, the prevalence of fragility fractures, considered a largely preventable consequence of osteoporosis, is expected to increase. Age is also a major risk for cardiovascular disease and mortality, thus highlighting the importance of cardiovascular profiling of osteoporosis interventions. Although calcium and vitamin D are essential for a healthy bone metabolism, excessive supplementation may be associated with increased risk. Conversely, early pre-clinical data have suggested a possible cardiovascular benefit from bisphosphonate therapy. This review evaluates the evidence behind the cardiovascular benefits and risks that may be associated with osteoporosis therapy.
Introduction
Age is the leading risk factor for several illnesses, including osteoporosis and cardiovascular (CV) disease. Mortality and morbidity increase dramatically following a fragility fracture and remain elevated for at least 5 years.1–3 The risk of disability after a fracture also seems to be a function of age, with a reduced risk of utility recovery following a fracture that increases for each decade of life. 4
Although the direct effect of calcium and vitamin D supplements on fracture risk and bone density (BMD) has been subjected to continued debate, adequate calcium and vitamin D metabolism remains essential for skeletal turnover, particularly in high-risk patients and/or those initiating osteoporosis pharmacotherapy. 5
Several pharmacotherapeutic interventions have shown a significant decrease in risk of fracture, particularly in postmenopausal women, with an even more pronounced absolute risk reduction in older adults.6,7
In the past decade, there has been increased concern over CV safety of osteoporosis interventions, including over-the-counter calcium and vitamin D supplements. On the other hand, animal studies and secondary/subgroup analyses of major osteoporosis clinical trials have raised the possibility of a cardioprotective effect of pharmacotherapeutic agents used for fracture risk reduction.
Many interpretations of the data have overlooked the quality of the evidence and provided blanket recommendations that may not apply to patients at high risk of fracture. We, thus, herein provide a review of the evidence behind the CV effects of osteoporosis interventions. We focused our review on calcium supplements, vitamin D supplements, and anti-resorptive therapy (bisphosphonates and denosumab). A separate review in this series addresses CV risk associated with osteoanabolic therapy.
Calcium supplementation
Early human observational studies looking at the association between calcium intake and CV outcomes had shown protective effect of calcium supplementation. The Iowa women’s health study, which included 34,486 postmenopausal women, 55–69 years of age, without a history of ischemic heart disease 8 found that women on calcium supplementation had a decreased risk of ischemic heart disease mortality over an 8-year follow-up compared to those who did not take calcium supplementation [relative risk (RR), 0.69 (95% confidence interval (CI) 0.52, 0.92) and RR, 0.64(95% CI 0.50, 0.82), for calcium supplementation > 500 mg/day and 1 to 500 mg/day, respectively] (Table 1). Similar protective effects were noted based on total calcium intake but not on dietary calcium intake. Similarly, the Nurse’s Health Study, which included 85,764 women, 34–59 years of age, without CV disease, found that those in the highest quantile of calcium intake, including dietary and supplemental calcium, with a median of 1145 mg/day had a lower risk of incident ischemic stroke with an adjusted relative risk of 0.69 (95% CI 0.50, 0.95) compared to those in the lowest quantile over a 14-year follow-up period. 9
Calcium supplementation and cardiovascular outcomes.

RCT: randomized controlled trial; RR: relative risk; MI: myocardial infarction; CHD: coronary heart disease; MACE: major adverse cardiovascular event; CVD: cardiovascular disease; HR: hazard ratio; OR: odd ratio.
More recently, however, reports of increased CV risk with calcium supplementations have emerged based on observational studies. The European Prospective Investigation into Cancer and Nutrition study (EPIC-Heidelberg) of 23,980 participants, 35–64 years of age, showed that in comparison with non-users of any supplements, those on calcium supplements had a high risk of myocardial infarction (MI) [hazard ratio (HR), 1.86 (95% CI 1.17, 2.96)], with a higher risk in those taking calcium only supplement [HR, 2.39 (95% CI 1.12, 5.12)]. 12 Similarly the Swedish mammogram cohort of 6433 women showed that, when compared to daily calcium intake of 600–1000 mg, intake in excess of 1400 mg of dietary and supplemental calcium was associated with an increased incidence of MI [RR, 2.14 (95% CI 1.48, 3.09)] CV death [RR, 1.49 (95% CI 1.09, 2.02)], and all-cause mortality [RR, 1.40 (95% CI 1.17, 1.67)]. 14
Evaluating CV-related mortality, one of the largest prospective cohort studies in the USA included 388,229 participants. 13 During a mean of 12 years of follow-up, 7904 CV deaths in men and 3874 CV deaths in women were noted. The result of the study suggested that a high intake of calcium supplements (>1000 mg/day) was associated with an excess risk of CV mortality in men [RR: 1.20 (95% CI 1.05, 1.36)] but not in women [RR 1.06 (95% CI 0.96, 1.18)]; cerebrovascular disease mortality was not significantly associated with calcium supplements in either women or men.
Other observational studies looked at surrogate markers of CV outcomes and showed an association between calcium supplementation and risk of incident coronary artery calcification [RR, 1.22 (95% CI 1.07, 1.39)], 15 and the prevalence of atrial fibrillation [OR, 3.89 (95% CI 1.28, 11.81)], 16 but an improvement in systolic blood pressure (Table 2).17–20
Calcium supplementation and surrogate markers of cardiovascular risks.
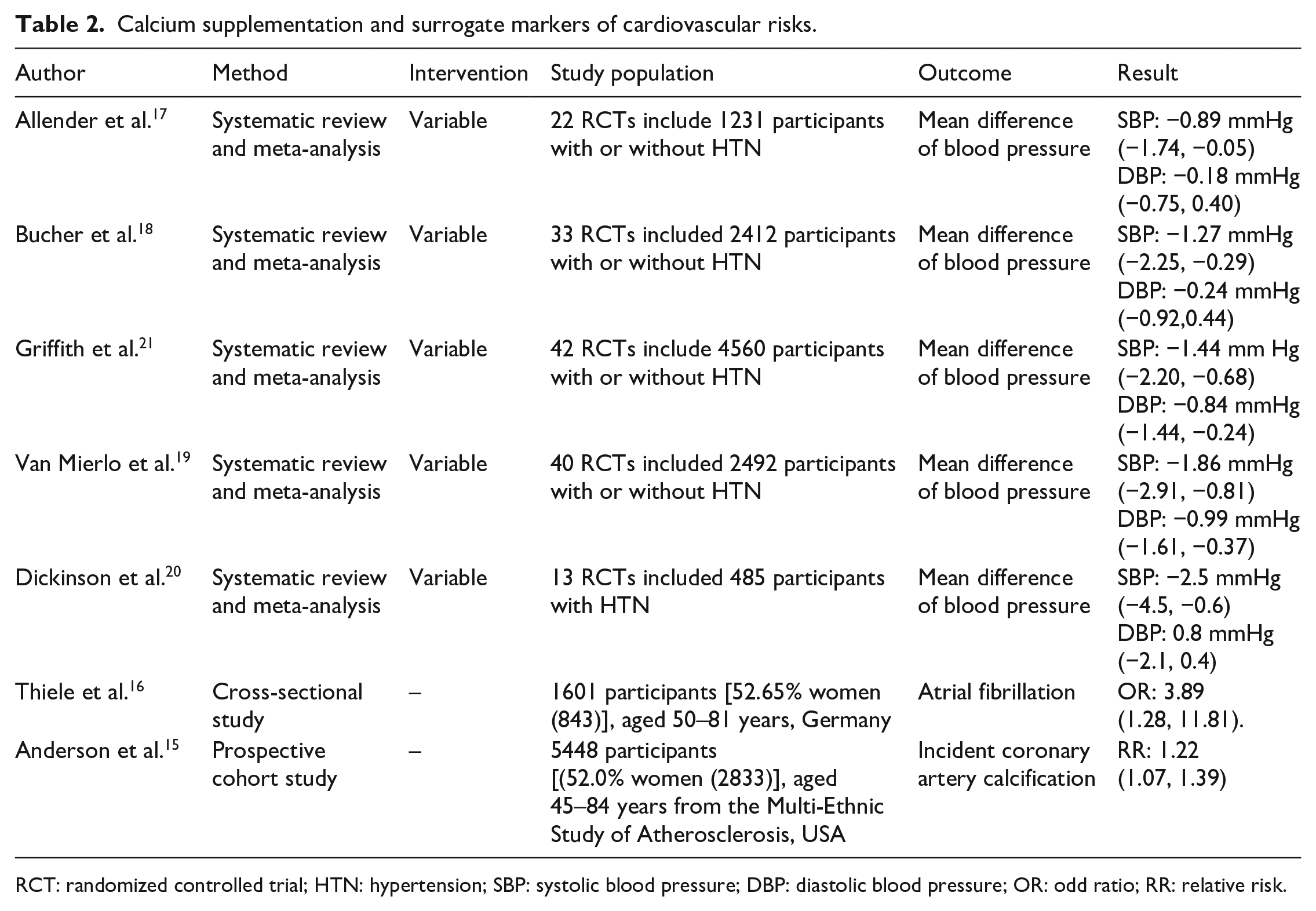
RCT: randomized controlled trial; HTN: hypertension; SBP: systolic blood pressure; DBP: diastolic blood pressure; OR: odd ratio; RR: relative risk.
Results from randomized control trials (RCTs) have also shown variable results of CV risk associated with calcium intake.10,11 Meta-analyses of published RCTs showed no significant effect of calcium supplement on CV events [pooled RR, 1.14 (95% CI 0.92, 1.41)], stroke [pooled RR, 1.12 (95% CI 0.92, 1.36)], all-cause mortality [pooled RR, 1.07 (95% CI 0.95, 1.19)], or major adverse cardiovascular event (MACE) [pooled RR, 1.21 (95% CI 0.85, 1.72)].22–24
An RCT of 1471 postmenopausal women in New Zealand, who had an average of about 800 mg of dietary calcium intake, showed that additional supplemental calcium citrate at 1000 mg/day was associated with a higher risk for MI [RR, 2.12 (95% CI 1.01, 14.47)] compared to placebo. 11 Similarly, a meta-analysis of 11 RCTs (11,921 participants, 76.5% women with median age 76.5 years) showed that the addition of calcium supplements (doses ranging between 500 mg/day and 2000 mg/day) to dietary calcium (baseline intake ranging between 400 mg/day and 1200 mg/day) showed an increased risk of MI [HR, 1.31 (95% CI 1.02, 1.67)], but no significant effect on stroke [HR, 1.20 (95% CI 0.96, 1.50)] or death [HR, 1.09 (95% CI 0.96, 1.23)]. 23 These results were replicated in a recent meta-analysis of 26 prospective cohorts and 16 RCTs (over 1,200,000 participants) that showed no CV risk associated with dietary calcium but an increase in risk of MI with additional calcium supplements (doses 1000–1500 mg/day).
A transient increase in serum calcium levels following calcium supplements ingestion may explain the impact of calcium supplements on the CV system. 25
Taken together, the evidence to date indicates that excessive doses of calcium supplements, especially when added to a diet that is sufficient with calcium, may be associated with increase in CV risk, particularly MI. On the other hand, when dietary intake of calcium does not provide the recommended daily calcium intake (Table 5), the addition of calcium supplements that do not exceed these daily goals is largely considered safe.
Vitamin D supplementation
Multiple population-based studies have associated vitamin D deficiency or vitamin D insufficiency (<15 ng/mL) with increased CV risk.26–29 Data from the U.S. Third National Health and Nutrition Examination Survey in adults aged 20 years or older showed that being in the lowest quartile of 25-hydroxyvitamin D levels (<17.8 ng/mL) was associated with a significant increase in all cause-mortality but non-significant increase in CV risk. 30 A Scottish population-based family study showed no association between vitamin D < 15 ng/mL and CV events or mortality. 31 Elevated 25-hydroxyvitamin D levels have also been associated with increased risk. A case–control study of Indian men showed a higher prevalence of elevated vitamin D levels (>89 ng/mL) in those with evidence of ischemic heart disease compared to controls. 32 In addition, genetic studies have noted an association between vitamin D receptor polymorphism and CV markers such as hypertension.33,34
A vast number of interventional studies have investigated the role of vitamin D supplementation, using various dosing regimens, on CV outcomes.
The Iowa women’s health study (see details in Calcium section) found that participants taking vitamin D supplements (1–400 IU/day and > 400 IU/day) had lower risk of ischemic heart disease mortality when compared to those without any vitamin D supplements. 8 Similarly, the Women’s Health Initiative that included 36,282 postmenopausal women in the USA demonstrated no increase in stroke or CV events in participants taking vitamin D 400 IU/day with calcium carbonate 1000 mg daily compared to placebo [HR, 0.95 (95% CI 0.82, 1.10) for stroke and 1.04 (95% CI 0.92, 1.18) for MI or CV death] (Table 3). 35 The RCT conducted on postmenopausal women in New Zealand evaluated monthly high doses of vitamin D (200,000 IU once, followed by 100,000 IU monthly) compared to placebo, and demonstrated no effect on MACE [HR, 1.02 (95% CI 0.87, 1.20)], incident MI [HR, 0.90 (95% CI 0.50, 1.50)], heart failure [HR, 1.19 (95% CI 0.84, 1.68)], stroke [HR, 0.95 (95% CI 0.55, 1.62)], and arrhythmias [HR, 0.93 (95% CI 0.62, 1.39)]. 36 The VITamin D and OmegA-3 TriaL (VITAL) randomized 25,871 participants to either vitamin D3 (2000 IU/day) and marine n−3 (omega-3) fatty acids (1g/day) or placebo and showed no increase in the incidence of CV events in the intervention group.37,38 Subgroup analysis similarly showed no effect of sex or race on the CV outcomes.
Vitamin D supplementation and cardiovascular outcomes.

RCT: randomized controlled trial; RR: relative risk; CVD: cardiovascular disease; MI: myocardial infarction; BP: blood pressure; WMD: weighted mean difference; MACE: major adverse cardiovascular event.
A meta-analysis of 23 RCTs that studied vitamin D supplementation in 89,251 participants similarly demonstrated no association with MACE [RR, 0.98 (95% CI 0.91, 1.05)], MI [RR 0.96 (95% CI 0.85, 1.09)], all-cause mortality [RR, 0.95 (95% CI 0.68, 1.32)] or CV mortality [RR, 1.00 (95% CI 0.85, 1.18)].(21, 23, 38, 39) These results support those of a prior meta-analysis of 51 RCTs that showed no increase in the incidence of stroke [RR, 1.05 (95% CI 0.88, 1.25)], MI [RR, 1.02 (95% CI 0.93, 1.13)], or death [RR, 0.96 (95% CI 0.93, 1.00)] with vitamin D supplementation. 40 Other meta-analyses looking at various dietary supplements similarly showed no effect of vitamin D supplementation (with or without calcium supplements) on CV outcomes.42,43
Numerous studies have examined the effect of vitamin D supplements on surrogate markers of CV disease (Table 4). An RCT from the UK that included 265 healthy postmenopausal women found that vitamin D3 at 400 or 1000 IU/day had no effect on lipid profiles, estimates of insulin resistance, inflammatory biomarkers, or blood pressure.44–46 In addition, vitamin D supplements had no effect on arterial stiffness or endothelial dysfunction.47–49 Finally, a number of studies, including RCTs and meta-analyses, showed no effect of vitamin D supplementation on metabolic changes such as the incidence of type 2 diabetes (T2DM) [HR, 0.88 (95% CI 0.75, 1.04)], 50 insulin resistance [MD −0.04 (95% CI −0.30, 0.2)], hemoglobin A1C (HbA1C) levels [MD −0.05% (95% CI −0.12, 0.03)], or blood glucose concentrations [SD −0.10 (95% CI −0.26, 0.07)]. 51
Vitamin D supplementation and surrogate cardiovascular markers.

RCT: randomized controlled trial; T2DM: type 2 diabetes mellitus; HR: hazard ratio; TC: total cholesterol; HDL-c: high-density lipoprotein cholesterol; LDL-c: low-density lipoprotein cholesterol; TG: triglyceride; HOMA- IR: Homeostatic Model Assessment for Insulin Resistance; hsCRP: high-sensitivity C-reactive protein; SBP: systolic blood pressure; DBP: diastolic blood pressure; MD: mean difference; HbA1C: hemoglobin A1C; OR: odd ratio; SMD: standardized mean difference; WMD: weighted mean differences.
Thus, the evidence to date suggests a reverse J-shaped curve associating circulating vitamin D levels with CV risk, as previously suggested by the CopD study, with very low 25-hydroxyvitamin D levels being associated with the highest risk. 54 On the other hand, vitamin D supplementation is considered safe. Table 5 summarizes the recommended daily intake for both calcium and vitamin D from The Bone Health and Osteoporosis Foundation (previously the National Osteoporosis Foundation) and the National Academy of Medicine (previously the Institute of Medicine).5,55
Recommended calcium and vitamin D intakes for women and men. 5

BHOF: The Bone Health & Osteoporosis Foundation; NAM: National Academy of Medicine.
Bisphosphonates
Bisphosphonates are the first line and most prescribed medications for management of skeletal fragility and other bone mineralization disorders. These agents suppress bone resorption through the inhibition of calcification and hydroxyapatite breakdown and have been found to limit osteoclast and osteocyte apoptosis. 56
Long-term studies on bisphosphonate use have demonstrated significant decrease in all-cause mortality in patients with osteoporosis regardless of previous history of fracture, and after accounting for potential confounders and competing risk [HR, 0.31 (95% CI 0.17, 0.59)]. 57 This mortality benefit has been consistent in different cohorts of frail older adults on oral alendronate, etidronate or risedronate [HR, 0.73; (95% CI 0.56, 0.94)]; 58 patients with osteoporosis after acute hip fracture on oral risedronate or alendronate [HR, 0.92; (95% CI 0.88, 0.97)]; 59 and patients with osteoporosis after acute hip fracture on intravenous zoledronate [HR, 0.72; (95% CI 0.56, 0.93)]. 60 Although the most intuitive mortality benefit would be the reduction in risk of incident fractures, it has been demonstrated that fracture prevention is only responsible for a small amount of the mortality reduction observed in patients on bisphosphonates. 61
There have been previous efforts to identify additional causes behind the mortality reduction in patients with bisphosphonates other than fracture risk reduction. 62 Given the well-established epidemiologic association between osteoporosis and CV disease, 63 there has been special interest on a potential CV mortality benefit from bisphosphonate use. Several studies have failed to demonstrate statistically significant differences in stroke, MI, or death from CV causes with the use of zoledronate.60,64 However, these studies have not been powered for the evaluation of CV mortality, and there has been a tendency towards reduced incidence of CV and cerebrovascular related deaths.57,62 In addition, risedronate has been previously reported to reduce the risk of MI in patients with rheumatoid arthritis [HR, 0.56; (95% CI 0.37, 0.86)], 65 has been shown to have a tendency towards decreasing CV mortality [HR, 0.77; (95% CI 0.57, 1.03)], and has been associated with reduction in stroke mortality in patients with osteoporosis [HR, 0.5; (95% CI 0.29, 0.88)]. 66
The mechanism behind the CV benefit of bisphosphonates has not been clearly elucidated but it is likely multifactorial. Historical studies on atherogenic diet-fed animals suggest that etidronate use inhibits arterial wall calcification, 67 leading to a decrease in size and lipid-rich necrotic content of existent atherosclerosis plaques,68,69 and preventing the development of new ones. 67 Comparative studies with other bisphosphonates have proposed several mechanisms behind their potential antiatherosclerotic and cardioprotective benefit including the inhibition of proliferation, migration, adhesion and osteogenic transformation of vascular smooth muscle cells,70,71 inactivation of tissue macrophages, 72 reduction of the arterial smooth muscle contraction, 73 and prevention of cardiac remodelling and dysfunction through the inhibition of the mevalonic pathway. 74 Noteworthy, these effects vary with the severity of the atherosclerosis lesions and stronger associations have been established with the use of high bisphosphonate doses and intravenous formulations.
In humans, the evidence on anti-atherosclerotic benefit from bisphosphonates is inconsistent. Previous studies have had short follow-up, and included small samples, subjects with other independent CV risk factors such as age, T2DM, menopause, dyslipidemia, and end stage kidney disease on dialysis. 75 Koshiyama and colleagues were the first to identify a significant decrease in the carotid artery intima-media thickness (−0.038 ± 0.011 mm, p < 0.005) despite no change in other risk factors (including lipid profile, HbA1C and BMD) in patients with T2DM who received etidronate for 12 months. 76 This agent was also found to have a statistically significant suppression on the progression of the coronary artery calcium score in patients on dialysis in whom the median annualized decrease in absolute calcified volume was −273 mm3 (p = 0.01). 77 By contrast, another study in patients on haemodialysis did not identify any significant impact on coronary artery calcium after 6 months of etidronate but demonstrated a 64.1% decrease in the aortic calcification score (p = 0.009) without associated changes in serum calcium, phosphorus, parathyroid hormone, activated cholecalciferol or serum lactate dehydrogenase levels. 78 Importantly, the follow-up period in these studies was 12 months or less, and there is only a single previous report of etidronate persistently inhibiting the progression of vascular calcification following 23 months of treatment in patients on haemodialysis. 79
Additional studies have evaluated the antiatherosclerotic effect of other bisphosphonate agents. A 12-month course of alendronate was found to significantly decrease the intima-media thickness in post-menopausal women. 80 However, these findings were not replicated in a separate study in post-menopausal women with osteoporosis. 81 More recently, a retrospective analysis of 34,940 individuals in Hong Kong that had sustained a hip fracture showed that alendronate reduced CV mortality [HR 0.33, (95% CI 0.17, 0.65)] and incident MI [HR 0.55, (95% CI 0.34, 0.89)] at 1 year when compared to propensity-matched individuals with hip fractures who were not exposed to bisphosphonates, with sustained benefits at 3, 5, and 10 years. 82 There was also a reduction in incident stroke, but it did not reach statistical significance. A similar pattern was seen in those exposed to any nitrogen-containing bisphosphonate after a hip fracture. Regarding intravenous zoledronate, it has been reported that a 12-month course could lead to reduction of the carotid artery intima-media thickness, increase in the high-density lipoprotein levels and decrease in the low-density lipoprotein levels, 83 and has been associated with a reduction in FGF23 which is directly and independently associated with the carotid artery intima-media thickness. 84 Furthermore, a 12-month course of risedronate therapy has been found to prevent the progression in plaque score and abdominal aorta coronary score in postmenopausal patients with osteoporosis T2DM, 85 but other agents (such as ibandronate) have not been found to have any impact on the progression of vascular calcification in post-menopausal women with osteoporosis following 3 years of treatment. 86
In addition to these observed changes in the CV system following bisphosphonate exposure, data have shown that zoledronate can have immunomodulatory effects including reduction in senescence-associated chemokines (such as CCL7, IL-1β, TNFRSF1A, and TGFβ1) and possible impact on immune cell subpopulations.87–89 The role of these changes in the CV system and their impact on CV outcomes remain unclear. In figure 1, we have summarized the cardioprotective benefits of bisphosphonate therapy with a hypothesized mechanism based on the available pre-clinical and clinical data.

Proposed mechanisms behind bisphosphonates cardiovascular benefits.
Aside from the potential CV risk reduction and antiatherosclerotic benefits, the association of bisphosphonate therapy with arrhythmogenic activity has been an area of investigation. The initial results from the largest prospective study for zoledronate [Health Outcomes and Reduced Incidence with Zoledronic Acid Once Yearly (HORIZON)-pivotal fracture trial] reported a significantly increased rate of arrhythmia and serious atrial fibrillation in patients who received this medication as compared to the control group (6.9% versus 5.3%, p = 0.003; and 1.3% versus 0.5%, p < 0.001, respectively). 64 However, no differences were observed in the incidence of arrythmia or atrial fibrillation in the subsequent HORIZON-recurrent fracture trial. 60 Moreover, while the alendronate’s Fracture Intervention Trial reported a non-significant tendency towards increased risk of atrial fibrillation in the intervention group [HR, 1.51; (95% CI: 0.97, 2.4; p = 0.07)], 90 a 2010 meta-analysis of RCTs failed to identify a statistically significant association between alendronate use and the risk of developing atrial fibrillation [RR, 1.16; (95% CI 0.82, 1.93; p = 0.33)]. 91 These findings were in agreement with the results of a large long-term epidemiologic study (>47,000 patients) in which the pooled use of any bisphosphonate agent was not associated with an increased risk of atrial fibrillation [HR, 0.82; (95% CI 0.66, 1.01; p = 0.63)]. 92
In summary, while bisphosphonate use has a well-reported all-cause mortality benefit and previous studies have shown a tendency towards decreased incidence of cerebrovascular and CV-related deaths, the current evidence is insufficient to demonstrate a statistically significant association with reduced CV mortality. Furthermore, whereas multiple comparative studies have demonstrated several anti-atherogenic mechanisms of bisphosphonates, results in human studies have been inconsistent and there is no robust data to support their potential cardioprotective effect in patients with osteoporosis or those with high risk of CV disease, MI, or stroke. Finally, although the initial pivot trials for zoledronate and alendronate suggested an association with increased risk of arrhythmia and atrial fibrillation, this risk has not been identified in meta-analysis and large epidemiologic studies.
Denosumab
Denosumab is a medication that is widely used for treatment of osteoporosis and bone mass augmentation in men on androgen deprivation therapy or in women on aromatase inhibitor therapy with high risk of fractures. This human monoclonal antibody exerts its antiresorptive effect through the inhibition of the receptor activator of nuclear factor-kB ligand (RANKL) which prevents osteoclasts’ activation, maturation, and survival. 93
Osteoprotegerin (OPG) is the natural counter-regulatory molecule of RANKL. As denosumab, OPG binds to RANKL preventing the activation of RANK which limits osteoclastogenesis. 94 Given the known pathophysiologic correlations between osteoporosis and atherosclerosis, the role of OPG, RANK, and RANKL on the development of CV disease has been an area of investigation. 95 OPG and RANKL have been found in aortic and carotid artery atherosclerotic plaques.96,97 Immunohistologic studies have demonstrated the presence of RANKL in early atherosclerotic plaques, 97 established an association between RANKL expression and atherosclerotic plaque destabilization, 98 and described RANKL mediated induction of calcification in vascular smooth muscle cells. 99 Conversely, induced OPG deficiency in mice has been correlated with increased calcification of the renal and aortic arteries, 100 and treatment with OPG has demonstrated potential inhibition of arterial calcification in rats. 101 Thus, it has been hypothesized that OPG could have a potential counterregulatory response aimed at limiting the vascular calcification process.96,102 However, these pre-clinical findings are discordant with recent epidemiologic data, which suggest that high OPG concentrations have a statistically significant association with increased risk of coronary artery disease, stroke and all-cause mortality that could be explained by pronounced upregulation of OPG leading to a paradoxical increase in vascular proinflammatory, pro fibrotic and pro apoptotic effect.103,104
Given that denosumab has the same mechanism of action as OPG, it is reasonable to question whether denosumab use has any impact on CV risks and mortality. While no prospective RCTs have been specifically designed to address this issue, retrospective analysis from denosumab’s pivotal trial provides some initial insight. The 3-year Fracture Reduction Evaluation of Denosumab in Osteoporosis Every 6 Months (FREEDOM) trial (n = 7808) identified a non-statistically significant trend towards decreased mortality in postmenopausal women with osteoporosis in the denosumab group (1.8% versus 2.3%, p = 0.08); In addition, compared to the placebo group, no statistically significant differences were identified in the rate of CV events (4.8% versus 4.6%, p = 0.74), stroke (1.4% versus 1.4%, p = 0.89), coronary artery disease (1.2% versus 1.0%, p = 0.41), peripheral vascular disease (0.8% versus 0.8%, p = 0.93), or atrial fibrillation (0.7% versus 0.7%, p = 0.98). 105 Furthermore, a subset analysis of the FREEDOM participants that were deemed to have high risk of CV disease (n = 2386; scoring ⩾ 4 points on Modified RUTH trial criteria) did not identify statistically significant differences in the mean score change in abdominal aorta calcification over 3 years (0.1 at 12 months, 0.2 at 24 months, and 0.4 at 36 months for both groups; all p values > 0.05) or the frequency of progression (5% versus 4% at 12 months, p = 0.37; 12% versus 10% at 24 months, p = 0.33; 22% versus 22%, p = 0.98) between the denosumab and placebo groups regardless of baseline abdominal aorta calcification status or progression of glomerular filtration rate. 106 However, these results need to be analyzed carefully given that the 3-year follow-up period might have been insufficient to establish statistically significant differences in mortality, incidence of CV events and atherosclerosis progression. In addition, three recent meta-analyses failed to identify differences in a composite of CV adverse events (including coronary artery disease, MI, stroke, heart failure, atrial fibrillation or CV death) and overall mortality in post-menopausal women with osteoporosis or low bone mass who received treatment with denosumab compared to placebo.107–109
In summary, while there are mechanistic and pathophysiologic reasons as well as pre-clinical data which suggests that denosumab could have an impact on CV disease, our current knowledge does not support any significant effect (neither positive nor negative) of denosumab on CV risk. Our review provides a descriptive summary of the literature on the CV effects of calcium, vitamin D, and antiresorptive agents in the management of skeletal fragility. Limitations of this review include the reliance on observational and retrospective studies when robust RCTs were not available. In addition, many studies looked at surrogate markers of CV disease rather than clinical outcomes. Finally, we have made every effort to review the most clinically relevant and up-to-date literature, but the unintentional omission of other studies may have been unavoidable.
Conclusion
Although CV risks have been linked to excessive supplementation, recommended daily calcium and vitamin D doses (Table 5) are not only safe, but essential for a healthy bone metabolism. Antiresorptive therapy, when added to sufficient calcium and vitamin D intake, provides a well-established reduction in fracture risk. Pre-clinical and early clinical data suggests potential CV benefits from antiresorptive therapy, independent of the fracture risk reduction. Additional longitudinal and adequately powered research studies are, however, needed to shed a better light on the long-term CV effects of osteoporosis therapy in humans, as well as their potential impact on patient-important outcomes.
