Abstract
Rheumatic diseases cover a wide spectrum of conditions, including primary and secondary degenerative joint diseases and autoimmune inflammatory rheumatic diseases. The risks of cardiovascular disease and osteoporosis and resultant fractures in aging female rheumatic disease populations, especially those with autoimmune rheumatic diseases, are increased. Changes in the immune system in aging populations need to be considered especially among patients with autoimmune rheumatic diseases. Immunosenescence is closely aligned to reduced adaptive immunity and increased non-specific innate immunity leading to chronic inflammation of inflammaging. The effective use of disease-modifying antirheumatic drugs to control autoimmune rheumatic diseases may also mitigate factors leading to cardiovascular disease and osteoporosis. Rheumatic diseases, which largely manifest as arthritis, predispose patients to premature joint degeneration and poor bone health and therefore have a higher risk of developing end-stage arthritis requiring joint arthroplasties sooner or more often than other patients without rheumatic disease.
Keywords
Introduction
Most developed nations are seeing a graying of their populations. In the United States, adults over the age of 65 will outnumber children for the first time, making up 21% of the population by the year 2030. In addition, the number of older adults is expected to nearly double to 95 million in 2060. 1 Recent medical advances have led to improved life expectancy from 68 years in 1950 to 77 in 2020. 2 However with an aging population, we now see a higher prevalence of infections and chronic and degenerative diseases, including cardiovascular disease (CVD), Alzheimer’s disease, arthritis, and malignancy.
Rheumatic diseases cover a wide spectrum of conditions, including primary and secondary degenerative joint diseases, and autoimmune inflammatory rheumatic diseases. We report herein the current practice in rheumatology and review selected relevant literature exploring the changes that occur in the immune system with aging, their potential impact on aging female rheumatic disease populations, and the inherent risks for the development of CVD and osteoporosis in this population. We also wish to briefly highlight the prevalence and impact of joint arthroplasties on aging female rheumatic disease populations.
Immunosenescence and inflammaging
The aging of the immune system, called “immunosenescence,” leads to a progressive decline in the body’s response to stressors and pathogens. 3 One of the main drivers of this process is the remodeling of lymphoid organs, specifically the involution of the thymus as one ages. This leads to decreased T lymphocyte function and ineffective central tolerance where there is a decrease in regulatory and naïve T cells.4,5 This immune system reorganization contributes to a chronic low-grade sterile inflammation seen in older adults called “inflammaging.” 6 With this process, there is an increase in acute phase reactants such as the C-reactive protein (CRP), and cytokines including interleukins 6 (IL-6) and 1 (IL-1) and tumor necrosis factor alpha (TNF-α) in the elderly. 7 This pro-inflammatory state is attributed to the somatic cellular senescence-associated secretory phenotype (SASP) and chronic innate immune activation. However, self-reactive T lymphocytes seem to be a primary contributor to inflammaging as well, making the process autoimmune or self-reactive in origin.5,8 Simply put, immunosenescence is linked to reduced adaptive immunity while there is an increase in non-specific innate immunity, leading to inflammaging. This complex interplay predisposes the aging individual to development of autoimmune disease, malignancies, infections, and decreased responsiveness to vaccinations,4,8,9 translating to an overall increased morbidity and mortality in the elderly. 10
Inflammaging and immunosenescence are implicated in the development of autoimmune rheumatic diseases (ARD) such as rheumatoid arthritis (RA). In general, with aging, there is a decline in the generation of naive and regulatory T cells which results in a compensatory proliferation of mature T cells in the periphery. Over time, the continuous replication of mature T cells leads to exhaustion and eventual production of its senescent phenotype. This process is accelerated in patients with RA, where senescent T cells are found much higher compared to the normal population (Figure 1). 11 Coupled with a decreased central tolerance, this likely contributes to the higher incidence of RA in adults >60 years old. The prevalence of RA is also higher at 2.03%–2.34%, compared to 0.5%–1% in younger adults. 12

Immunosenescence in rheumatoid arthritis. As the immune system ages, T-cell production from the thymus decreases and this process is accelerated in patients with rheumatoid arthritis. With the decrease of thymic T-cell population, a compensatory and excessive proliferation of peripheral T cells ensues causing them to eventually be senescent. Senescent T cells have an autoreactive and pro-inflammatory phenotype which contribute to the inflammatory features of the disease. Adapted from Weyand and Goronzy 2002. Image created BioRender.com (Weyand and Goronzy 11 ).
It should be cautioned, however, that while autoimmunity increases in the elderly this does not necessarily translate to an increase in autoimmune disease in this population. 13 Various autoimmune diseases present differently in older adults than in their younger counterparts. For example, in RA a subset of patients would develop elderly onset RA (EORA) at age >60. The phenotype of EORA contrasts from younger-onset RA with more balanced gender distribution, and an increased involvement of larger joints especially the shoulder girdle, resembling polymyalgia rheumatica (PMR). Also, there is a tendency for EORA to present more acutely with higher disease activity and increased systemic symptoms compared to younger counterparts. 14
In systemic lupus erythematosus (SLE), the majority of patients are young, reproductive age females. However, about 12%–18% will have a delayed onset of SLE after age 50–65. 15 Females are still predominantly affected but the female to male ratio in late-onset SLE is decreased from 9:1 to 5-7:1. 16 Late-onset SLE is considered to be more indolent compared to younger onset, especially with lower prevalence of anti-dsDNA and hypocomplementemia.15,16 There is also less involvement of skin, mucus membranes, joints, kidneys, and hematological abnormalities, but more frequent incidence of serositis, depression and thrombotic events when compared to their younger counterparts. 17
As one ages, there is an accumulation of comorbidities making it common for patients to have multiple disease processes at the same time, a condition called “multimorbidity” (Figure 2). In RA, the average patient has 1.6 additional conditions and this is expected to increase with age and disease activity. 18 These competing and interrelated diseases increase the complexity of how we care for older patients. Along with RA-related comorbidities, older adults are already at risk for cognitive impairment, depression, falls, urinary incontinence, and malnutrition. These are found to be more prevalent in patients with rheumatic diseases. These lead to increased adverse outcomes including accumulation of added morbidity, nursing home admission and mortality. 19 It is thus imperative for the clinician to recognize the changes and unique challenges that the older adult patients face in order to adequately care for them.
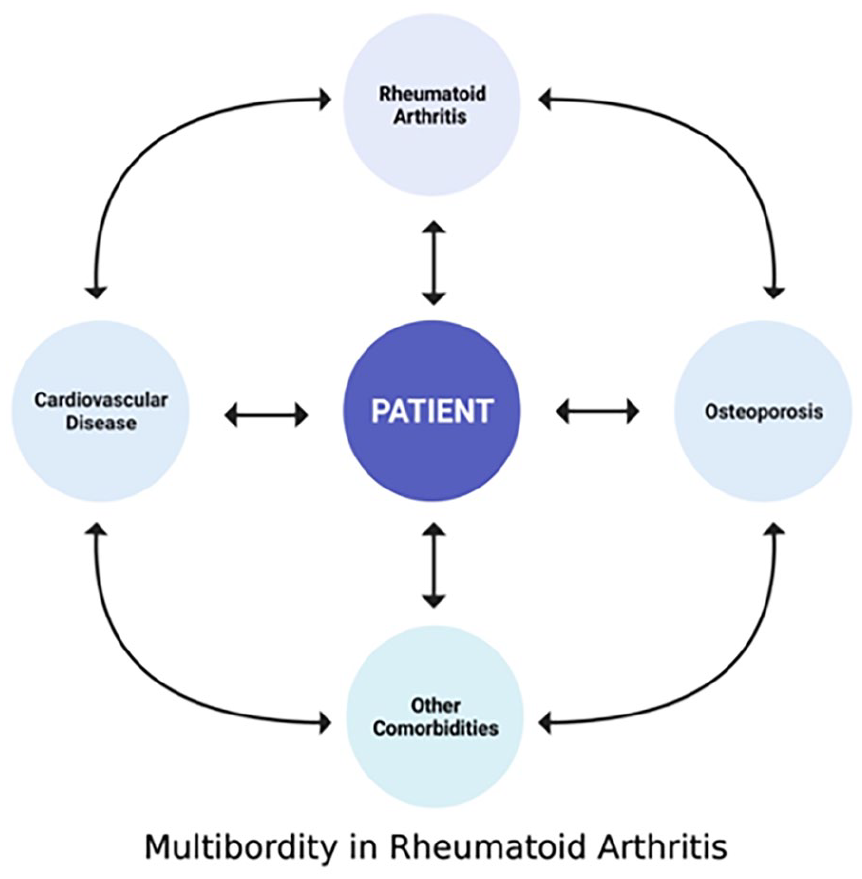
Rheumatoid arthritis (RA) is a systemic disease, and most patients have multiple other comorbid illnesses. Using the concept of multimorbidity, the patient is the center of the treatment paradigm where there is no index disease. Each comorbidity is given adequate importance, taking note of the interaction of each coexisting condition, and treating with the goal of patient improvement and quality of life. Adapted from Radner et al. 18 Created with BioRender.com.
CVD in aging rheumatic disease populations
In 2019, 35% of total deaths in women were due to CVD. 20 In countries with a low socio-demographic index (measured by a composite of income per capita, educational attainment, and fertility rates), CVD mortality is greater in women than men. 21 Even in developed countries like the United States and Canada; however, the decline in cardiovascular (CV) mortality has slowed with a more recent uptick in acute myocardial infarctions in younger women.22,23 Overall, CVD in women is underdiagnosed with an under-representation in clinical trials thereby limiting progress in diagnosis and management in this population. 24 The disparity of CVD treatment utilization in women compared to men is likely related to differences in clinical manifestations of CVD and comorbidities.25–27 Such disparities are further compounded by an aging population of individuals with rheumatic disease, which largely impact women greater than men. 28
Menopause, the heart, and autoimmunity
The risk for CVD increases with interrupted ovarian function, as in menopause, whereby the cardioprotective benefits of estrogen are reduced or lost. 29 Specifically, the declining estrogen levels trigger an increase in low-density lipoprotein (LDL) and total cholesterol in addition to the impact on receptors in the vasculature. 30 An earlier age at menopause is linked to a greater risk of CVD and all-cause mortality. 31 One hypothesis for this is that CVD risk is increased by mechanisms beyond estrogen depletion alone, and that menopause before age 45 years may be a red herring for CVD-related events. 32
In ARDs, CVD is a major cause for morbidity and mortality and occurs due to underlying disease activity, medication use, and additional risk factors. 33 When evaluating the burden of mortality associated with autoimmune diseases grouped together, they rank among the top 10 causes of death in women under 75 years of age.34,35 Furthermore, in evaluating menopause, autoimmunity, and heart disease, one must consider the impact of rheumatic disease treatments on the reproductive system. In SLE, for example, premature ovarian failure is described in more than 60% of patients with a history of treatment with cyclophosphamide, 36 a medication known for its adverse effects of teratogenicity and ovarian failure.
Pathogenesis
Atherosclerosis is a chronic disease that presents itself clinically as a myocardial infarction. What starts as endothelial dysfunction results in an atheromatous plaque formed by foamy macrophages in the arterial intima, where collagen, elastin, and proteoglycan are deposited. 37 Premature atherosclerosis is a result of chronic inflammation as seen in individuals with ARD. The impact of ARDs on traditional CV risk factors (e.g. smoking, hypertension) as well as the use of potentially cardio-toxic medications (e.g. non-steroidal anti-inflammatory drugs (NSAIDs), glucocorticoids (GCs), certain disease-modifying antirheumatic drugs (DMARDs) adds layers of complexity to the pathophysiology of CVD in ARDs. 38 Autoantibody formation in ARDs like SLE have also been implicated in premature atherosclerosis as demonstrated by the presence of IgG-continuing immune complex deposits in premature atherosclerotic lesions. 39
While obstructive coronary artery disease (CAD) is conventionally a disease of older males, cardiac syndrome X has been ascribed to myocardial ischemia occurring because of microvascular dysfunction (MVD), typically presenting in women with chest pain and myocardial ischemia without angiographic evidence of coronary stenosis. 40 MVD is largely underdiagnosed, impacts women aged 45–65 years, and is associated with major adverse CV events.41,42
Cardiovascular manifestations
It is important to remember that vascular disease in ARDs is typically described as premature atherosclerosis not otherwise related to traditional CV risk factors.
Valvular disease, typically of the mitral valve, is common in patients with ARD, seen in at least 30% of patients with RA and SLE, though largely asymptomatic.43,44 Such valvular abnormalities noted in ARD are to be considered in combination with underlying risks for valvular disease, including senescence.
Manifesting as sudden cardiac death (SCD), atrial or ventricular arrythmias, or atrioventricular block, electrical abnormalities are not infrequent in ARD. The main cause of SCD in RA is related to acute coronary syndrome and resulting ventricular arrhythmias. 45 In SLE, sinus tachycardia, atrial fibrillation, and ectopic atrial beats are associated with flares and myocarditis. 46 Antibodies to Ro/SSA are associated with sinus bradycardia and prolonged QT interval. 45 From small vessel vasculitis to fibrosis of the conduction nodes and bundle branches, ARDs can impact the electrophysiology of the heart in ways that can otherwise be improperly categorized as “age-related” or related to other comorbidities.
While pulmonary hypertension (PH) can complicate many ARDs, systemic sclerosis (SSc) is the foremost ARD that presents with PH. 47 Additional etiologies include thromboembolic PH seen in antiphospholipid syndrome or PH from veno-occlusive disease in SSc. PH is only second to interstitial lung disease in being the leading cause of death in SSc, thereby emphasizing the importance of prompt diagnosis and management. 48 Early diagnosis is limited by coexisting symptoms of ARD that may limit an optimal clinical evaluation. For example, fatigue and reduced exercise tolerance may be discounted as a manifestation of one’s age or underlying rheumatic disease. 49 Recognizing risk factors for PH and being proactive in evaluating for PH can mitigate some of these misinterpretations.
Myocardial disease due to ARDs typically presents in the form of congestive heart failure and is believed to occur from a chronic pro-inflammatory state. In SLE and SSc, most cases are asymptomatic.43,50
Pericardial disease is a frequent manifestation in SLE, SSc, and RA. Clinically, this may present as pericarditis with fever and pleuritic chest pain, or with more clinically significant pericardial effusion with shortness of breath and hypotension. 43
Cardiovascular risks
Senescence of various components of the CV system implicates biological age as a critical determinant of onset and progression of CVD. 51 Hypertension, which affects more than 60% of adults aged 45–75 years in the United States, is another key player in the development of CVD. 52
The 2018 AHA Guidelines on cholesterol management describe CVD risk-enhancing factors to include a family history of premature atherosclerotic CVD, persistently elevated LDL levels, metabolic syndrome, chronic kidney disease, a history of premature menopause (<40 years) or preeclampsia, high-risk ethnic groups like South Asians, persistently elevated triglycerides, elevated apolipoprotein B, and chronic inflammatory diseases like RA, psoriasis, and other ARDs (Table 1). 53 The Framingham Offspring Study demonstrated that women with SLE <45 years of age are 50 times more likely to have a myocardial infarction compared to non-SLE patients of the same age. 54 In RA, the risk of CVD mortality is 50% greater than the general population. 55 Traditional risk factors such as hyperlipidemia and hypertension are generally more prevalent in SLE patients, related either to disease state such as lupus nephritis or medications such as GCs. In addition, antiphospholipid antibodies, seen in 30%–50% of patients with SLE, may affect the lipid profile through lipid peroxidation, which furthers endothelial dysfunction.56,57
Cardiovascular disease risk factors in females with autoimmune rheumatic diseases.

Source: Adapted from Grundy et al. 53
CVD: cardiovascular disease; RA: rheumatoid arthritis; SLE: systemic lupus erythematosus.
Historically, GCs were used universally to treat ARD. More recently, however, many studies have demonstrated complications from long-term use and high doses of GCs, including but not limited to an increase in blood glucose, triglycerides, and blood pressure, all independent risk factors for CVD. 58 Similar associations of NSAIDs and other immune modulatory therapy with hypertension and CVD have been noted.
Lipid levels, though implicated in CVD, can be normal in half of all myocardial infarctions. 59 While lipid deposition within arterial vasculature is typical of atherogenesis, inflammation is an independent risk factor for CVD. 60 Serum high-sensitivity CRP (hs-CRP) and IL-6, both markers for inflammation, bear serologic risk for CV events.61,62 An elevated CRP is also associated with a greater likelihood of myocardial infarction in patients with angina in addition to death from CVD in smokers with other atherosclerotic risk factors.63,64 Cigarette smoking is independently associated with a higher CRP. 65
In SLE, older age at disease onset, longer duration of SLE, higher damage score, elevated serum LDL and homocysteine, and greater duration of treatment with GCs are associated with an increased risk for CVD. 66 The risk for PH in SSc includes the limited form of SSc, the presence of centromere antibody, telangiectasias, increased duration of illness, older age, and elevated uric acid levels. The risk for cardiopulmonary hospitalization in scleroderma is associated with dyspnea, pericardial effusion, and a diffusing capacity of carbon monoxide (DLCO) of less than 50%. 67 In RA, where the risk for a myocardial infarction is two-fold greater than controls, a reduced risk of CV disease is noted in those individuals treated with DMARDs such as TNF-α inhibitors and methotrexate.66,68
CVD risk models, including the Framingham risk score, Systematic Coronary Risk Evaluation (SCORE), and the Reynolds Risk Score underestimate CVD risk in RA and other rheumatic diseases. 69 Because of this, non-invasive cardiovascular risk stratification can be considered through coronary artery calcification score and carotid ultrasonography to identify those patients considered “moderate risk” concerning the SCORE or modified EULAR score.70–72 The European guidelines on CV disease prevention in clinical practice recommend multiplying the risk SCORE by 1.5 to ascertain CV risk in ARD. 73 In SLE, Petri et al. 74 established that the risk for CVD-related events varied based on SLE-related risk factors like SELENA-SLEDAI score, the presence of low C3 and lupus anticoagulant in addition to traditional risk factors. Urowitz et al. 75 proposed a CVD risk score by multiplying the Framingham risk score by two.
Diagnosis and management of CVD
The diagnosis of CVD in ARD can be made through traditional imaging and interventional modalities. Carotid ultrasonography may uncover subclinical atherosclerosis through carotid intima-media thickening. 76 Similarly, echocardiography, coronary computed tomography, and positron emission tomography are valuable tools aiding in the diagnosis and risk stratification of patients with ARD and suspected CVD. 77 Cardiac magnetic resonance imaging can also be used as a non-invasive modality to identify coronary MVD in patients with unremarkable conventional CVD evaluation. 78
PH evaluation is largely clinical on initial screening. When PH is suspected, or in the case of all patients with SSc and SSc-related diseases, annual screening echocardiography is warranted. 79 The DETECT algorithm incorporates the use of lung volume studies (forced vital capacity/diffusion capacity of the lung for carbon monoxide, FVC/DLCO ratio), positive centromere antibody, current or prior telangiectasias, serum uric acid and N-terminal-pro hormone B-type natriuretic peptide levels, and a right axis deviation on electrocardiography to assess risk of PH in patients with SSc. 80
The subclinical nature of CVD in women with ARD, combined with increased morbidity and mortality, calls for early diagnosis and prompt treatment. The association of CVD onset and progression with disease activity stresses the importance of concomitantly controlling signs and symptoms of ARD. Longitudinal registry data of patients with RA in the United States has demonstrated that reduced disease activity is associated with fewer CV events. 81 In more experimental phases, treatment with anti-inflammatory agents such as the IL-1β receptor antagonist canakinumab, and colchicine improves CVD outcomes in various cohorts.66–68
The presence of ARD plays a significant role in accelerated senescence, including CVD. Comprehensive risk stratification, assessment, and management of these additional CVD risk factors should be standard of care in women with ARD. Collaborative care efforts between cardiology, rheumatology, and primary care, are critical to closing the gap in CV care in women with ARD.
Bone health in aging rheumatic disease populations
Inflammation related to ARDs largely predisposes to the development of osteoporosis (OSP). When compounded with female sex and elderly age, the risk of OSP in aging females with ARDs becomes much higher (Table 2).
Assessment of osteoporosis in patients with autoimmune rheumatic diseases.

ARD: autoimmune rheumatic diseases.
Rheumatoid arthritis
RA is highly associated with OSP, affecting as many as 50% of all patients, with the risk for men and premenopausal women being double that of those without RA in age matched controls. 82 Periarticular osteopenia, a decrease in both cortical and trabecular bone, increased cortical porosity and lower volumetric bone mineral density (BMD) are all hallmarks of RA (Figure 3). 83 The mechanisms for this are many such as contributions by TNF-α and IL-6 release indirectly leading to receptor activation of nuclear factor kappa-B ligand (RANKL) activation which in turn activates osteoclast formation and activation while simultaneously decreasing osteoblastogenesis. The Wnt signaling pathway is also inhibited in RA patients via increased levels of Dickkopf-related protein 1 (Dkk-1). 84 This explains why higher RA disease activity is associated with higher degree of pathologic bone loss. Further, RA patients with anti-cyclic citrullinated peptide antibodies have an even higher incidence of OSP, which is thought to be mediated by IL-6-dependent osteoclast activation. 83
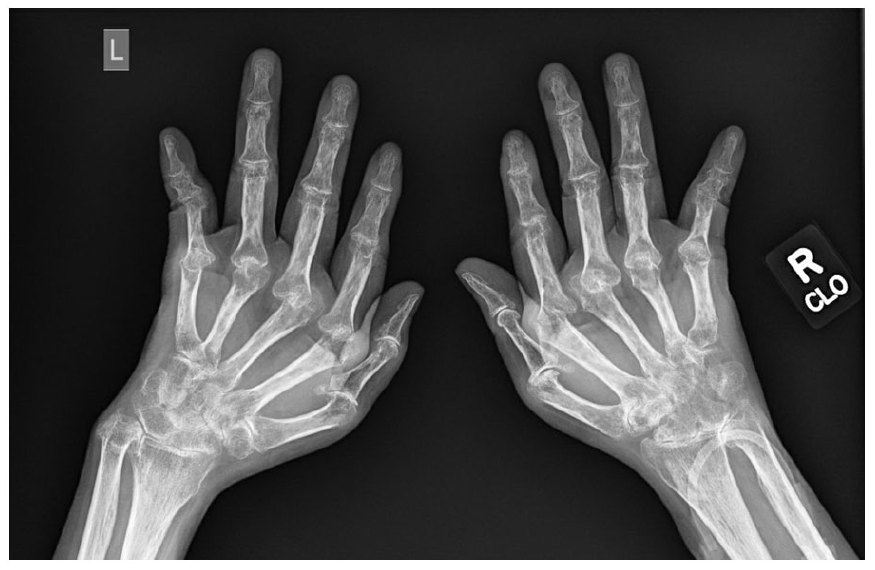
Plain radiograph of a patient with long-standing rheumatoid arthritis (RA). The image shows extensive erosive and destructive changes on all the metacarpophalangeal (MCP) joints on both hands. These resulted in ulnar deviation of the digits. Also note the significantly decreased bone density around the MCPs and carpal/wrist joints (periarticular osteopenia), which indicates chronic inflammatory activity from this patient’s years of untreated disease. RA is an independent risk factor for osteoporosis and affects as much as 50% of RA patients.
While uncontrolled RA is one contributor to poor bone health in this disease, decrease in physical activity and resultant bone loading and development of sarcopenia further exacerbates the risk of OSP and subsequent fractures. Further, patients with periarticular erosions are also at risk for trabecular bone loss which is another reason poor disease control is associated with an increased risk for OSP. 85
The role of GC-associated bone loss cannot be divorced from a discussion about OSP in RA patients. GCs directly encourage apoptosis of osteoblasts and an increase in Dkk-1 and sclerostin via the Wnt pathway in a time and dose dependent manner leading to loss of trabecular bone strength. Further, high doses of GCs shift production of bone stem cells from osteoblasts to adipocytes. GCs increase the osteoclast life cycle via the RANKL pathway. Finally, GCs inhibit absorption of calcium in the intestine and renal tubules. 86 Both RA and GCs cause decrease in muscle mass (rheumatoid cachexia and sarcopenia) which leads to a decrease in bone loading, further worsening the strength of the bones in the RA patient. Poor strength is a risk factor for poor balance and increased risk of falling, leading to an increased risk for fragility fractures.
On the other hand, GCs are a mainstay of terminating acute flares of RA, which decrease disease activity leading to an improvement in bone health. The interplay between RA disease activity, GC use, and bone health is complicated. Nevertheless, short courses of low dose GCs in patients with early and active RA appear to be safe and not have a significant negative effect on BMD. This is likely due to the established advantage of GCs in controlling disease activity. 87 It then makes sense that treating the underlying inflammation will improve or at least stabilize the BMD of patients with RA. Indeed, several studies have shown positive evidence supporting this. The use of TNF-α inhibitors was found to be associated with decreased vertebral fractures in a U.S.-wide observational cohort. 88 Similarly, favorable outcomes in BMD were found for other biologics such as abatacept and tocilizumab, with no significant difference in the risk of fractures between these drugs and TNF-α inhibitors. 89 Overall, treating the underlying RA is beneficial in preventing or even improvement bone loss in this population.
Systemic lupus erythematosus
A relationship between low BMD, resultant fractures, and SLE has been established in many populations worldwide. 90 An estimated prevalence of 20%–26% of vertebral fractures exists in SLE patients, with a 4.7-fold increase in risk of fracture in female SLE patients versus healthy controls. 91 In male patients with SLE, the incidence rate ratio for developing OSP is 5.35. 92
Risk factors unique to patients with SLE for bone loss include low levels of C4, absence of anti-Smith autoantibodies, presence of anti-Ro/SSA, presence of lupus anticoagulant, use of anticoagulants, use of anti-epileptic medications, FOK-I vitamin D receptor FF or Ff genotype and low serum levels of dehydroepiandrosterone. As in RA, the likelihood of low BMD increases with decreasing body weight, increased disease activity, and use of GCs, particularly in doses >7.5 mg daily or for prolonged periods. Lupus nephritis and subsequent aberrations in parathyroid hormone, calcium, and vitamin D are further risk factors for low BMD in patients with SLE. 92
Spondyloarthropathies
The spondyloarthropathies including psoriatic arthritis and ankylosing spondylitis are characterized by increased bony proliferation, rather than loss of BMD as is seen in RA. However, low BMD is found in patients early in the disease process. Reduction in physical activity, spinal mobility, ankylosis and decreased functional capacity place patients with axial spondylarthritis at substantial risk for vertebral fractures after very minimal trauma. In fact, the risk of vertebral fractures in this patient group is higher than that found in patients with RA (odds ratio of 7.1 vs 2.7). 93 Fractures occur more frequently in the cervical spine than in patients with primary osteoporosis, leading to a higher risk of neurologic compromise. Thankfully, the risk of fractures elsewhere such as the wrist or the hip is not raised to the same degree as that of vertebral fractures. 94
Fusion of the cervical spine and decreased range of motion at the hips and lumbar spine increase the risk for falls in this population (Figure 4). These factors also make accurate assessment of fracture risk difficult by dual-energy X-ray absorptiometry or bone densitometry and trabecular bone score, and therefore, the use of fracture risk evaluations such as FRAX® (Fracture Risk Assessment Tool) scoring may help more accurately identify patients at risk for fragility fractures. High disease activity, longer disease duration, low weight, low body mass index, and male gender were positively associated with OSP. 95 As with other chronic inflammatory diseases, reduction in disease activity with biologic agents reduces the likelihood of loss of BMD in this patient population. 93

Plain radiograph of the cervical spine in a patient with ankylosing spondylitis (AS). The image shows significant anterior and posterior osseous fusion (ankylosis) of the C3-5 vertebrae. Despite the proliferative bone changes, patients with spondyloarthropathies, including AS, are more prone to cervical spine fractures than patients with primary osteoporosis. The resulting decreased range of motion of the spine also increases the risk for falls and thus fractures in this population.
Vasculitis
Even prior to treatment with GCs, there is an increased prevalence of osteopenia in patients with anti-neutrophil cytoplasmic antibody or ANCA-associated vasculitis, suggesting a mechanism driven by a chronic inflammatory state. 96 However, due to the organ and life-threatening nature of the diseases, vasculitis patients tend to stay on GC therapy at some level for long periods with standard of care being at least 52 weeks, even when other treatment modalities are used concurrently. 97 Furthermore, relapses are common in vasculitides, particularly large vessel vasculitis, for which GCs are largely used. 98 In patients with Giant Cell Arteritis and PMR, the frequency of OSP has been found to be up to 85% and the incidence of fractures in this group was found to be 11%–14%. 99
Inflammatory myopathy
BMD is decreased in patients with inflammatory myopathies through mechanisms similar to what is described above in relation to chronic inflammation. Demineralization due to immobility contributes to poor bone health as well. Like the vasculitides, GC therapy is a cornerstone of treatment for these conditions and is another driver of poor skeletal health in this patient group.
Arthroplasties in the aging female population
It is impossible to talk about bone health in aging female rheumatic disease populations without delving into the prevalence and impact of joint arthroplasties.
Arthritis often leads to disability and poor quality of life. In the United States, it is the most common cause of disability, affecting approximately 54.4 million (22.7%) adults. It is projected that this number will rise to 78.4 million by the year 2040. 100 As arthritis progresses, it may lead to joint destruction causing end-stage arthritis, severe pain, and functional disability needing surgical joint replacement. Arthritis and arthralgia are major manifestations of rheumatic musculoskeletal diseases, whether or not autoimmune or inflammatory in nature. The use of joint arthroplasties may mitigate bone loss in these populations, as they improve physical functionality including the ability to ambulate in these patients. The most common indication for joint replacement surgery is osteoarthritis (OA). However, patients with ARDs are disproportionately at risk for requiring joint replacements than the general population. This is likely due to the underlying inflammatory pathology that brings about more rapid destruction of the joint compared to OA. Those with psoriatic arthritis and RA are two and four times, respectively, more likely to undergo joint replacement surgery compared to non-RA patients.101,102 Similarly, patients with SLE are at higher risk and tend to be younger and female compared to patients with OA needing arthroplasty perhaps due to the increased risk for osteonecrosis in this population. 103
The overall prevalence of total hip arthroplasties (THA) and total knee arthroplasties (TKA) in the United States is 0.83% and 1.52%, respectively. 104 Projections on the overall use of these surgeries are predicted to increase by 284% and 401% by year 2040. 105 The increase in the aging population and the obesity epidemic has been linked to an increase in OA cases, thus the rise in joint replacements. 106 Of note, the prevalence of arthritis is higher in females than males (26% vs 19.1%), if adjusted for age. 107 Consequently, the rate of joint replacements is higher in older women. 104 Furthermore, female gender was found to be an independent risk factor for readmission, reoperation, and wound infection after total hip replacement, while male gender was an independent risk for the same outcomes but for total knee replacement surgery. In either case, females tend to require more hospital length of stay than males. 108 Frail patients who receive joint replacements tend to be female and sicker which leads to higher mortality and perioperative complications following primary and revision THA.109,110
Overall, the outcomes of joint replacement surgery are good and are associated with significant improvement in quality of life. 111 This is also observed even in those with rheumatic musculoskeletal diseases. Interestingly, patients with RA are not at higher risk of acute surgical site infection after TKA compared to those with OA. 112 Similarly, SLE was not found to be an independent risk factor for poor short-term pain or function after either hip or knee arthroplasty. 113 Over the past 20 years with the increasing use of DMARDs especially the biologic agents, the proportion of patients with inflammatory arthritis who needed to undergo joint replacement surgeries has decreased. 114 This contrasts with an increase in joint replacements in patients with OA over time. 115
Conclusion
The risks of CVD and osteoporosis and resultant fractures in aging female rheumatic disease populations, especially those with ARD, are increased. Changes in the immune system in aging populations need to be considered especially among patients with ARD. Immunosenescence is closely aligned to reduced adaptive immunity and increased non-specific innate immunity leading to chronic inflammation of inflammaging. The effective use of DMARDs to control ARD, may also mitigate factors leading to CVD and osteoporosis. Rheumatic diseases which largely manifest with arthritis predispose patients to premature joint degeneration and poor bone health and therefore have a higher risk of developing end-stage arthritis and the need for joint arthroplasties sooner or more often than other patients without rheumatic disease.
