Abstract
Heart failure (HF) is a prevalent clinical syndrome that causes significant physical limitations. Osteoporosis is also an important cause of loss of functionality, and it mainly affects women. There are several reports linking HF and osteoporosis, and both share risk factors. Most of the data available so far point to bone fragility as a consequence of HF, and several mechanisms have been identified to explain this relationship. Among the proposed pathophysiological mechanisms are the hyperactivation of the renin–angiotensin–aldosterone system and the increase in parathyroid hormone, functional limitation, production of inflammatory mediators and the use of drugs for HF. The role of osteoprotegerin has gained attention owing to its cardiovascular and skeletal effects, its observed deficiency during the postmenopausal period along with its compensatory increases in HF and severe osteoporosis. The objective of this review was to perform a literature search for the main evidence on skeletal impairment in HF, with emphasis on women. As for epidemiological studies, we selected data from 3 meta-analyses and 20 individual observational studies, which together showed the interrelationship between the two clinical conditions in terms of both decreased bone density and increased fracture risk. In conclusion, HF and osteoporosis are interrelated conditions mediated by complex pathophysiological mechanisms which may be more relevant for postmenopausal women, considered to be a vulnerable population for both cardiovascular diseases and bone fragility.
Introduction
Heart failure (HF) is a complex clinical syndrome arising from structural or functional heart abnormalities that result in high filling pressures or inadequate cardiac output.1,2 These changes cause cardinal clinical manifestations, such as dyspnoea, peripheral oedema, pulmonary congestion and fatigue. 1 As the syndrome is underdiagnosed, it is estimated that HF affects 64.3 million people worldwide, with approximately 50% of the cases occurring in women, in whom there is a greater tendency to diastolic dysfunction and better prognosis, despite the fact that they receive less treatment than men.1–5
Osteoporosis is the most common bone metabolic disorder and is characterized by the deterioration of the skeletal microarchitecture, which compromises bone strength thus increasing the risk of fractures.6–8 Osteoporosis is more common in women than in men, especially in the postmenopausal period, and causes one in two women over 50 years of age to have a fracture in their lifetime. This finding demonstrates its high impact on health costs, quality of life and morbidity and mortality.8–10
Despite the scarcity of studies that evaluate them together, some data point to an important interrelationship between these two clinical entities, with shared risk factors and important pathophysiological mechanisms.11,12 In this review, we aimed to compile the main scientific evidence on skeletal impairment in HF, with emphasis on women.
Literature search strategy
PubMed and Cochrane Library databases were accessed to select the articles used in this review. The following descriptors were used, all contained in the Medical Subject Headings (MeSH): ‘heart failure’ AND osteoporosis OR osteopenia OR ‘bone density’ OR ‘bone diseases, metabolic’ OR ‘fractures, bone’ OR ‘bone remodeling’ when advanced searches were performed. As a search refinement criterion, articles published in the last 20 years were selected.
Subsequently, a qualitative analysis of the articles was performed, selecting those with full allusion to the proposed theme, which had well-defined inclusion and exclusion criteria, clarity in methodology and data treatment, and reproducibility. For the epidemiological studies, the articles that mandatorily included women patients were selected. This procedure was initially performed by two different authors in a double-blind system, and in case of disagreement, there was a third author analysis. Articles that aimed to elucidate the functioning of the pathophysiological mechanisms were included in the study in order to assist in their detailing.
Common risk factors for osteoporosis and heart failure
There are some coincident risk factors that may help to explain a probable interrelationship between HF and osteoporosis, such as ageing, diabetes mellitus, hypertension, sedentary lifestyle, sex hormone deficiency and changes in inflammatory mediators.8,13,14 A detailed assessment of the role of some of these risk factors, performed in the next section, may help in understanding the pathophysiology of osteoporosis in patients with HF.
Bone abnormalities in heart failure
The pathophysiological mechanisms that interrelate HF and osteoporosis are not completely understood; however, there are data that point to the roles of some changes seen in HF that may contribute to bone loss. 14
HF is one of the several cardiovascular disorders that can cause a certain degree of tissue hypoxia.14,15 Hypoxia causes a progressive increase in osteoclastogenesis and resorption in the bone tissue, which is one of the possible mechanisms that contribute to the development of osteoporosis in individuals with HF. 16
Hyperactivation of the renin–angiotensin–aldosterone system (RAAS) is the central feature in the pathophysiology of HF. RAAS acts as a compensatory mechanism in response to reduced cardiac function, which in the long run activates signalling pathways associated with myocyte hypertrophy and apoptosis, thus feeding a vicious cycle that culminates in myocardial function deterioration.17,18 This mechanism causes a large part of the therapeutic arsenal against HF to act by blocking some points in the RAAS.1,2,17,18 As regards to bone metabolism, RAAS hyperactivation increases urinary and faecal excretion of calcium and magnesium, which contributes to the elevation of parathyroid hormone (PTH) levels, an essential peptide for calcium homeostasis, which promotes an increase in bone catabolism when chronically elevated. PTH levels also seem to be increased by the direct action of aldosterone on mineralocorticoid receptors (MR) present in the parathyroid glands. Furthermore, the increase in oxidative stress associated with hyperaldosteronism leads to secondary elevations in PTH, which is one of the ways by which HF causes damage to bone health. 19 The identification of MR in bone cells also indicates the direct action of aldosterone in this tissue.19,20 Other causes of secondary hyperparathyroidism commonly seen in patients with HF, such as chronic kidney disease and vitamin D deficiency, may also contribute to the onset of osteoporosis.21,22 HF is a relevant cause of functional limitation.11,23 Immobility, which may result from this loss of functionality, is capable of leading to significant loss of bone and muscle mass.24–26
In women, the decrease in osteoprotegerin (OPG) is related to the development of postmenopausal osteoporosis because physiologically oestrogen deficiency decreases the expression of this protein, which competes with the receptor activator of nuclear factor κB ligand (RANKL) and decreases the osteoclastic activity.27,28 In addition to the transcriptional action of oestrogen on OPG, there are data that attest to its action at the post-transcriptional level. 28 Similar to what occurs in the bone tissue, the protective action of OPG was observed at physiological levels in cardiovascular health. The action of OPG in blood vessels is mainly due to its connection to the tumour necrosis factor–related apoptosis-inducing ligand. OPG acts on the endothelium, thus restricting the calcification of the arterial wall. 29 OPG-deficient mice present early-onset osteoporosis and arterial calcification. 30 However, the complexity of the role of OPG was obvious when observing that high concentrations of this protein are directly related to the severity of bone loss.31–34 High concentrations of OPG were also associated with greater severity of coronary artery disease and cardiovascular mortality.29,35,36,37 In patients with HF, higher levels of OPG were linked to greater symptoms and lower bone mineral density (BMD) 34 as well as an increase in all-cause mortality (in individuals hospitalized for HF). 37 Therefore, OPG appears to have a beneficial effect on bone and cardiovascular health when present at homeostatic levels. This observation is consistent with the increase in cardiovascular risk and bone mass loss observed in the postmenopausal period, when there is a drop in oestrogen and consequently OPG levels. This finding contrasts the clear harmful effect of persistently high concentrations of this biomarker observed in cardiovascular diseases and severe osteoporosis, possibly due to compensatory mechanisms.
Pro-inflammatory cytokines, such as tumour necrosis factor alpha (TNF-α) and interleukin-6 (IL-6), are elevated in patients with HF, contributing to macrophage activation, microvascular dysfunction, contractile changes and fibrogenesis. 38 In the bone tissue, these inflammatory cytokines are shown to potentiate bone resorption, thereby increasing the risk of fractures.39,40 TNF-α is shown to be a bone catabolic agent, by stimulating osteoclastogenesis (especially through RANKL activation), suppressing the recruitment of osteoblast precursors and the expression of bone matrix protein genes, and apparently providing some degree of resistance to calcitriol (the biologically active form of vitamin D).39,41,42 Serum levels of soluble TNF-α receptors 1 and 2, which are shown to be the most reliable biomarkers for long-term exposure to TNF-α, have already been shown to be associated with a higher risk of hip fracture independently of other risk factors. 39 IL-6 is a pleiotropic cytokine with an important role in autoimmunity, acute inflammation and chronic inflammatory states. 43 IL-6 plays a relevant role in bone resorption, by stimulating the expression of RANKL, especially in pathological conditions that directly affect the bone, such as multiple myeloma, Paget’s disease and osteoporosis itself. Factors such as PTH, lipopolysaccharides, calcitriol and TNF-α stimulate greater transcription of IL-6 in bone tissue cells, leading to a predominance of osteoclastic activity.43,44 IL-6 exerts its effects through direct binding to a transmembrane receptor (cis pathway), or through binding to a soluble receptor (trans pathway). The latter seems to be the most important for the osteoclastogenic response induced by chronic inflammation of extraosseous origin, as healthy individuals apparently do not have sufficient levels of soluble IL-6 receptors to amplify bone resorption mediated by this cytokine, unlike individuals with rheumatoid arthritis, colitis and oestrogen deficiency.43–45 As IL-6 is one of the main inflammatory mediators involved in the pathophysiology of HF, 38 this is another possible mechanism linked to bone involvement in individuals with HF.
Finally, the use of drugs in the treatment of HF can have deleterious effects on bone health. Loop diuretics are prominent as they promote greater renal calcium excretion.11,14,46,47 Their prolonged use has already been associated with a higher risk of fractures in postmenopausal women 46 as well as an increase in bone turnover and loss of BMD. 47 The hypotensive effect of other classes of drugs used in HF therapy can also exacerbate the risk of falls and, consequently, fractures, especially in elderly patients. 11 Figure 1. summarizes the potential pathophysiological mechanisms presented here.

Potential pathophysiological mechanisms for bone involvement in HF.
Heart failure, bone mineral density and fractures in women – meta-analyses of observational studies
When searching for clinical trials, randomized clinical trials, systematic reviews and meta-analyses, six articles related to the topic were identified, of which three were excluded because they included only men. Thus, three studies were selected,14,48,49 all of which were meta-analysis (Table 1).
Meta-analyses that addressed heart failure and bone health in women.
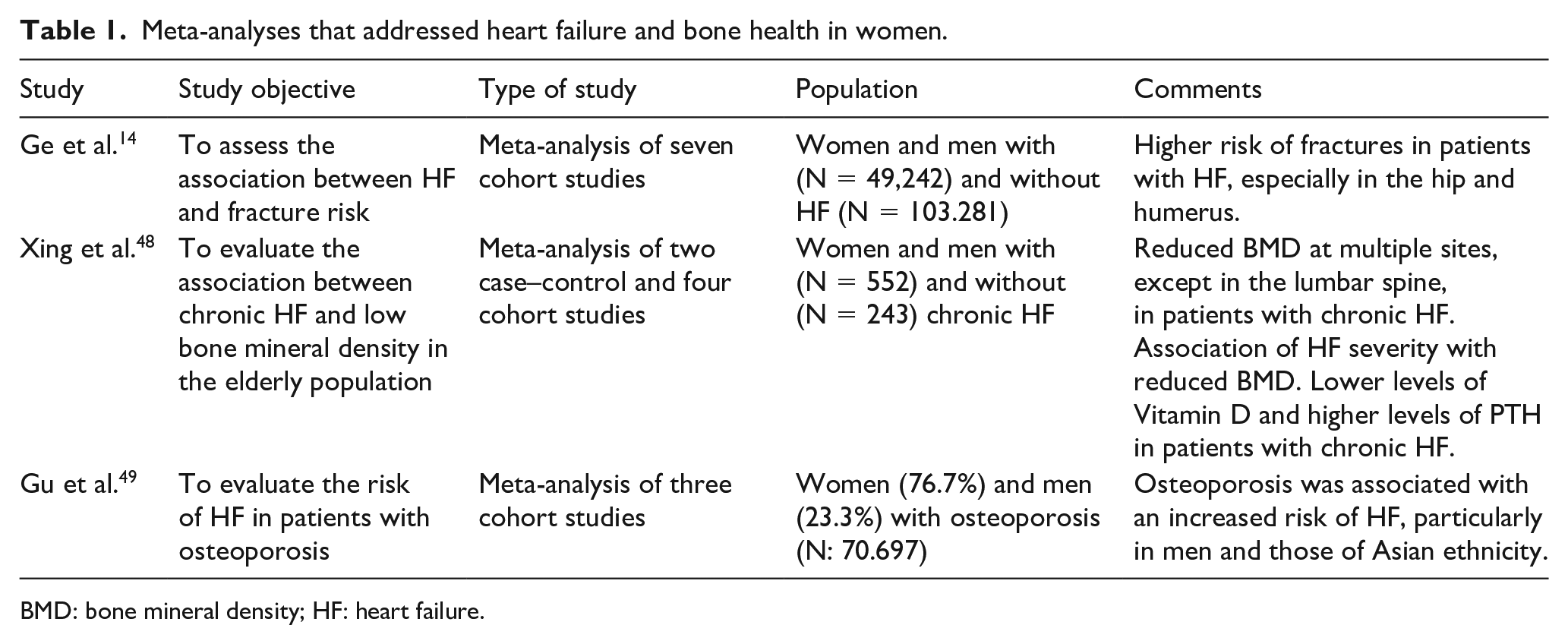
BMD: bone mineral density; HF: heart failure.
A meta-analysis of seven cohort studies found that patients with HF were at increased risk for any fracture (relative risk (RR): 1.66, confidence interval (CI) 95%: 1.14–2.43, P = 0.008); hip fracture (RR: 3.45, 95% CI: 1.86–6.4, P < 0.0001) and humerus fracture (RR: 1.91, 95% CI: 1.07–3.4, P = 0, 03), in addition to a trend towards an increased risk of vertebrae fractures (RR: 1.78, 95% CI: 0.88–3.58, P = 0.11) and forearm fracture (RR: 1.06, 95% CI: 0.80–1.41, P = 0.69). 14
Two case-control studies and four cohort studies were included in the meta-analysis by Xing et al., 48 which aimed to evaluate the association between low BMD and chronic HF. Decreased BMD was observed in the whole body, hips, trunk, arms and legs, but not in the lumbar spine, in patients with chronic HF. An association between the severity of HF, evaluated using the New York Heart Association (NYHA-FC) functional classification, and low bone mass was also observed in addition to higher levels of PTH and lower levels of vitamin D, which could be related to impairment of physical capacity and low sun exposure.
On the contrary, the meta-analysis by Gu et al. 49 evaluated the risk of HF in patients with osteoporosis. There was a mean increase of 17% in the overall risk of HF in individuals with osteoporosis; however, in women, this risk was only slightly increased, without statistical significance (hazard ratio: 1.14, 95% CI 0.94–1.37, P = 0.19). Such data, along with the numerous risk factors in common that interrelate osteoporosis and HF, show the need for further studies to establish possible causal relationships.
Heart failure, bone mineral density and fractures in women – observational studies
Twenty-five observational studies addressing bone health and HF were identified, of which five were excluded because they evaluated only men, thus 20 articles were selected. Low BMD was observed in patients with HF when compared with the control groups.50–54 HF was associated with a higher prevalence of osteoporosis and worsened the age-related loss of BMD. 50 Lower BMD was linked to lower levels of vitamin D and higher rates of frailty, with a clear acceleration in the rate of bone loss in those with polymorphism of the vitamin D receptor gene.51–53
There are not enough data to allow a detailed stratification of bone consequences according to the different HF phenotypes; however, there are indications for greater bone loss in those with lower left ventricular ejection fraction and with a higher degree of clinical decompensation. Left ventricular ejection fraction was independently associated with lower BMD in elderly women, with values ⩽ 49% being the best cut-off point to predict osteoporosis. 54 Similarly, low BMD has been shown to be related to left ventricular diastolic dysfunction. 55 The impact of HF severity on bone mass was evaluated using the blood levels of the N-terminal fragment of the B-type natriuretic peptide (NT-proBNP) and NYHA-FC.56,57 A relationship was observed between low BMD and high levels of NT-proBNP 56 as well as high levels of NYHA-FC. 57
HF was shown to be directly associated with a higher risk of osteoporotic fractures, especially of the hip.58–60 A 12% prevalence of vertebral fractures identified on radiographs was found in a cross-sectional study conducted in a HF clinic, with multiple fractures in more than half of the affected portion. 61 The presence of secondary hyperparathyroidism was associated with the occurrence of vertebral fractures in middle-aged patients with HF. 62
When evaluating the serum OPG levels of individuals with HF, an inverse association with trochanteric BMD was observed, as previously discussed. 34 However, when RANKL levels were assessed in the bone marrow microenvironment of individuals with HF, high levels were observed to the point of raising the RANKL/OPG ratio, which promoted the osteoclastic activity and increased the risk of osteoporotic fractures. 63
When examining the role of body mass index (BMI) and body composition and their relationship to BMD in patients with HF, it was found that higher BMI and mineral-free lean mass were associated with lower odds of osteoporosis and osteopenia, with fat mass not being associated with BMD. This finding shows the important role of muscle mass in maintaining the bone mass in individuals with HF. 64
In agreement with the pathophysiological showed above, the use of loop diuretics was also associated with osteoporosis in different studies.50,65 As for bone turnover markers, it was observed that the implantation of a ventricular assist device in patients with advanced HF resulted in a decrease in bone resorption markers such as type I collagen C-telopeptide and type I collagen N-telopeptide (CTX and NTX) and the formation marker, procollagen type 1 amino-terminal propeptide (P1NP), thus indicating that the hemodynamic improvement led to the improvement in bone remodelling. 66
The evaluation of a possible reverse causality, where osteoporosis predisposes an individual to HF, was also evaluated in two different studies, one retrospective 67 and one prospective, 68 both observing a higher incidence of HF in individuals with osteoporosis.
Thus, the scarcity and heterogeneity of the literature on this topic still does not allow the establishment of an irrefutable causal relationship, but the pathophysiological mechanisms give plausibility to the hypothesis that HF can cause significant bone involvement, as well as data from available epidemiological studies. Large prospective studies with rigorous methodologies are needed to bridge this gap.
Conclusion
Heart Failure and osteoporosis are interrelated conditions mediated by complex pathophysiological mechanisms which may be more relevant for postmenopausal women, considered to be a vulnerable population for both cardiovascular diseases and bone fragility.
