Abstract

Intervention versus Invention
As I was preparing to write this Perspectives piece towards a more international audience, I was curious to see what would turn up in my search results with the key terms ‘United Kingdom Nursing Invention’. I was amused to see that the search engine asked me, ‘Did you mean: United Kingdom Nursing Intervention? (Figure 1)’ The irony didn’t escape me that Florence Nightingale, founder of modern nursing practice, hailed from England and invented the pie chart. It made me contemplate the differences and similarities between nursing interventions and inventions.
Search engine results.
Throughout our history, nurses have adapted care to meet the needs of patients. Many nurses are known for inventing products to improve the care and experience of both patients and clinicians, such as ostomy appliances, crash carts, and neonatal phototherapy (Cosier, 2017). The ostomy pouch was invented in 1954 by a Danish nurse, Elise Sørensen, who was inspired to make the device for her sister who had a colostomy and refused to leave her home for fear that her stoma might leak. Sørensen found a solution by working with a plastic manufacturer and eventually invented the ostomy pouch (Danmark, n.d.). The crash cart, initially called the ‘crisis’ cart, was invented by Emergency Room nurse Anita Dorr in 1967, and was later patented by a physician (Nuvara, 2021). Neonatal phototherapy was inadvertently discovered by a nurse observing that infants that were exposed to outside and sunshine were more inclined to recover from jaundice faster. These are only a few examples of nurse invention interventions that have made significant enhancements in the healthcare field. Today, nurses are involved in a myriad of ways in product development, including inventing, research and development, and entrepreneurialism.
A concept not addressed clearly in the nursing literature is how one might progress from the conception of a novel idea to the creation of a product, usable to those it was intended to help. The following narrative describes a process that was made possible by a team of nurses and an occupational therapist in a large Midwest academic medical center and their journey in creating an INVENTION for patient care.
The problem
As part of a workgroup tasked with decreasing hospital-acquired Catheter-Associated Urinary Tract Infections, a team of nurses identified that there were no standard products or practices to ensure optimal urinary drainage (foley) bag placement when patients are ambulating. Improper placement of the foley bag above the bladder can enable urinary output to backwash into the bladder, increasing the patient’s risk for infection (Stratton and Zieve, 2021). Despite the use of modern urinary drainage bags for over 60 years, some healthcare providers (HCPs) find workarounds and creative solutions to secure urinary drainage bags when ambulating patients with catheters. They may use items, like disposable gloves or tourniquets, to tie around an IV pole to create a makeshift hanger for the urinary drainage bag (Figure 2). Other situations may include the HCP hooking the drainage bag into their trouser pocket while assisting the patient in ambulation (Figure 3). It has been observed that patients also hang their urinary drainage bag from their shirt pockets (Figure 4). Improper management of the drainage device may also lead to inadvertent removal of the catheter, which may cause trauma to patients’ urethra. Frankly, and remarkably, there is no known device on the market to alleviate this problem.
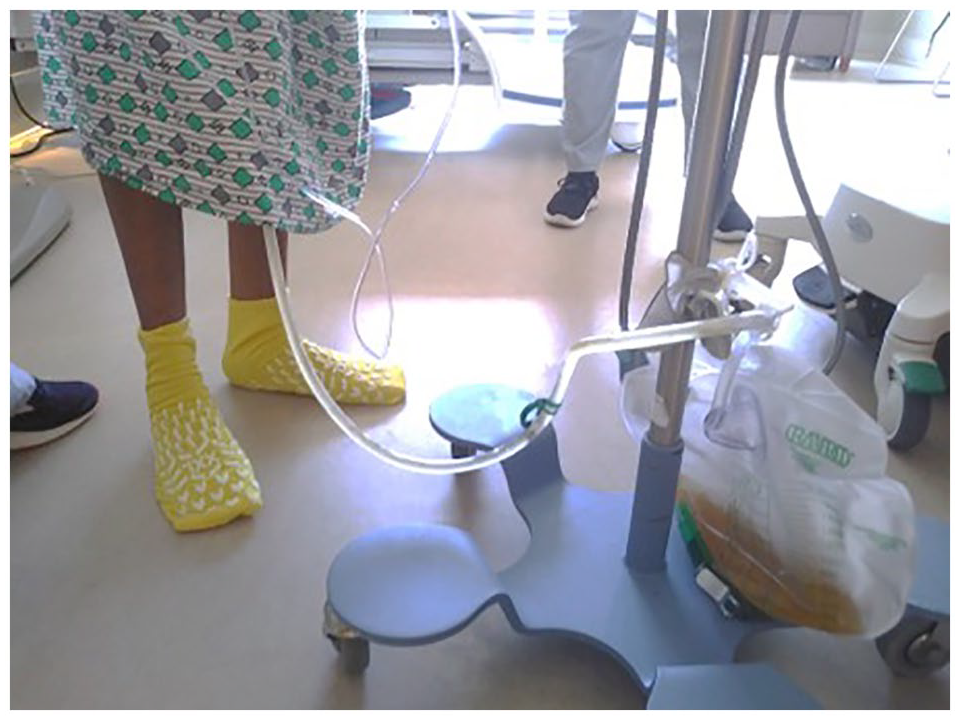
A urinary drainage bag hooked onto a glove tied around an IV pole.
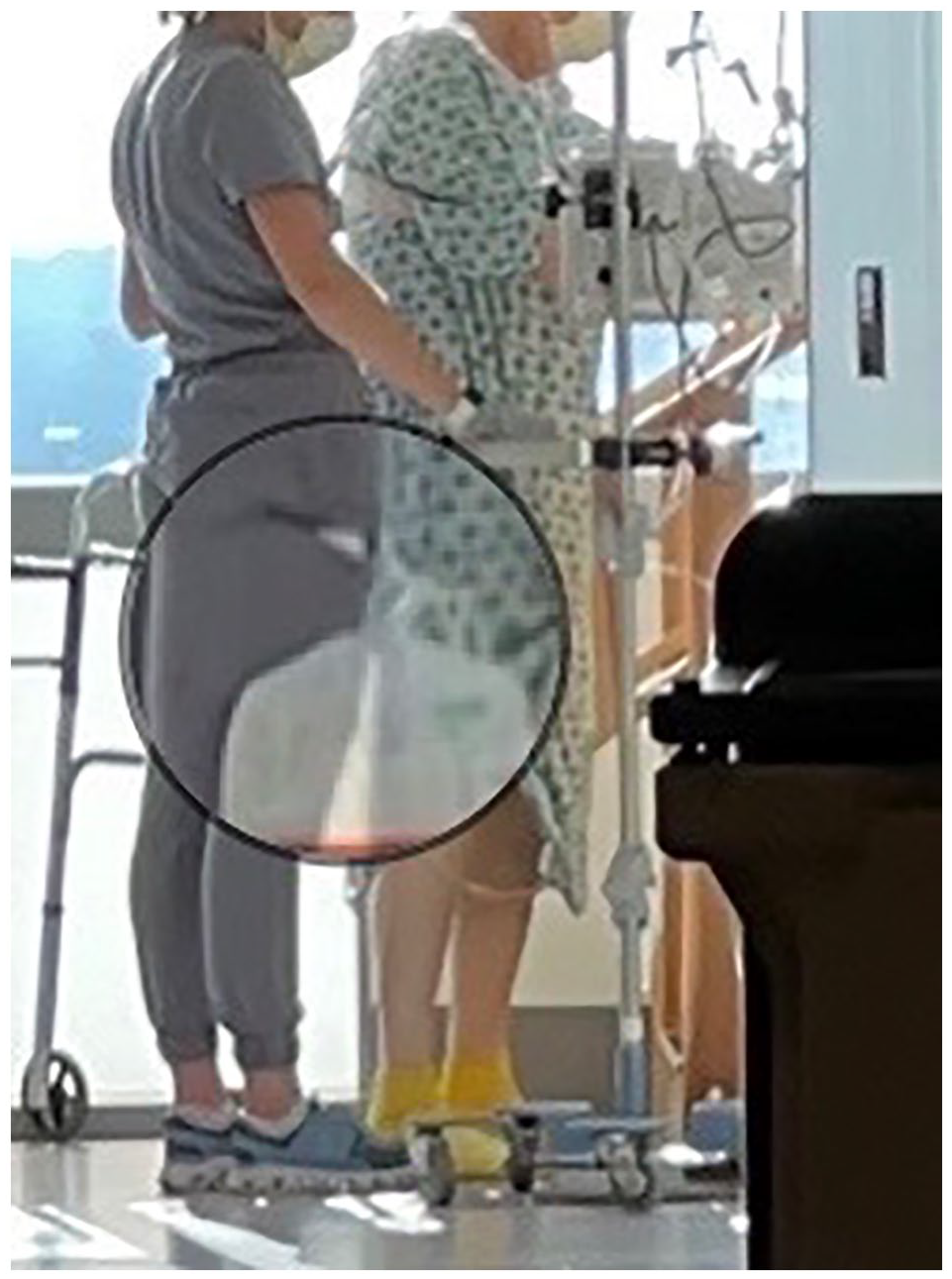
A urinary drainage bag hooked into the pant pocket of a therapist.
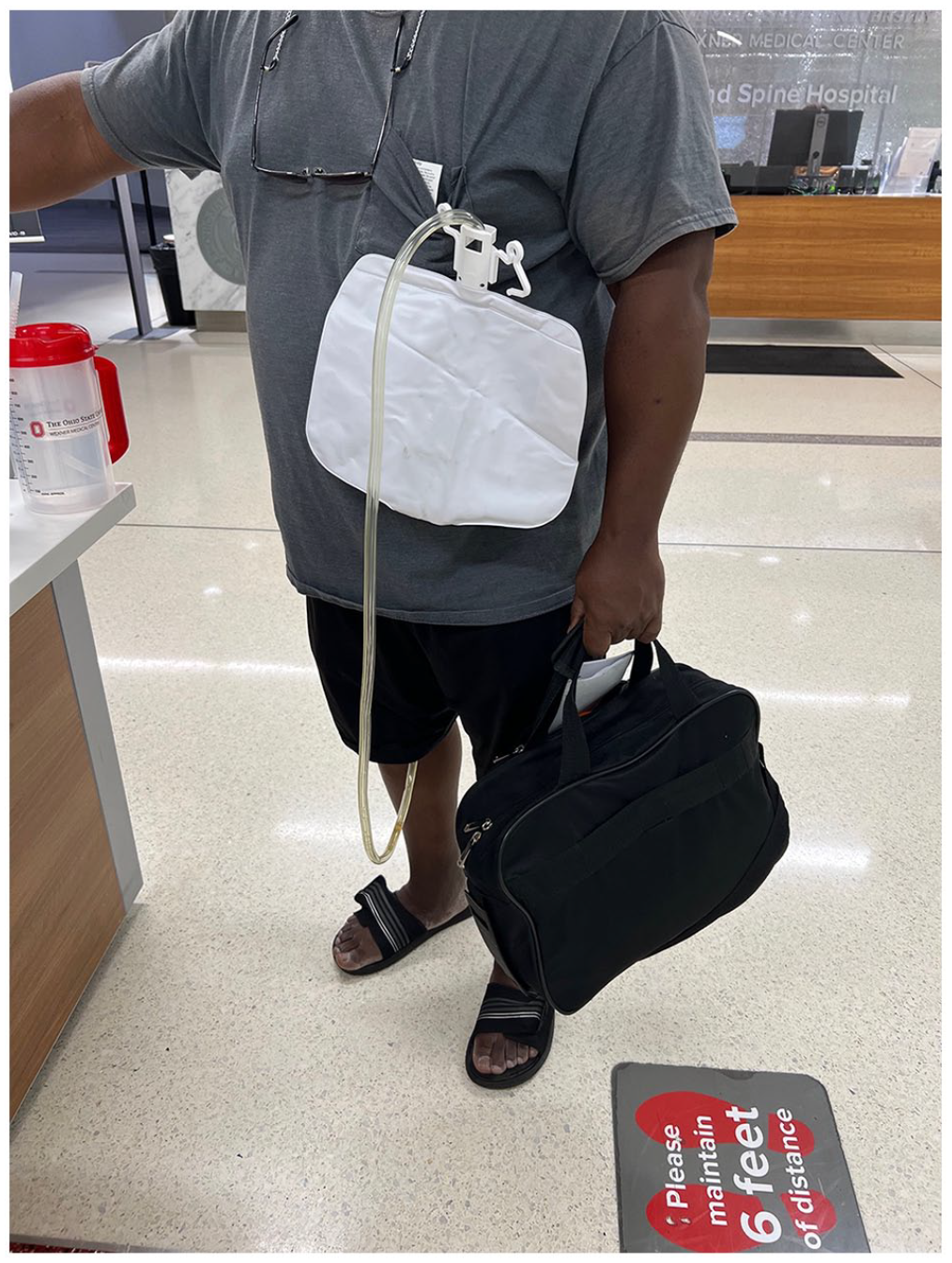
A patient hanging their urinary drainage bag from shirt pocket.
The solution
To address this patient problem, a team of nurses and an occupational therapist worked together to conceptualise a device to hold urinary drainage bags in an optimal position while patients are ambulating, formally called the Bag Below Bladder (B3) Buddy. Many organisations are starting to support creative spaces, or ‘maker spaces’ to allow for HCPs to develop grass roots innovation like the B3 Buddy. At The Ohio State University, the Innovation Studio was created to foster interdisciplinary collaboration in creating healthcare solutions. This studio is open to all students, faculty, and staff to utilise tools such as 3D printers, laser cutters, and other resources to prototype products. The studio also provides opportunity to pitch an idea to procure funding and mentorship. Our team worked with an engineering student to develop a prototype and were able to produce Computer-Aided Design (CAD) drawings (Figure 5) and 3D printings of the B3 Buddy. This process took about 2 years to complete.
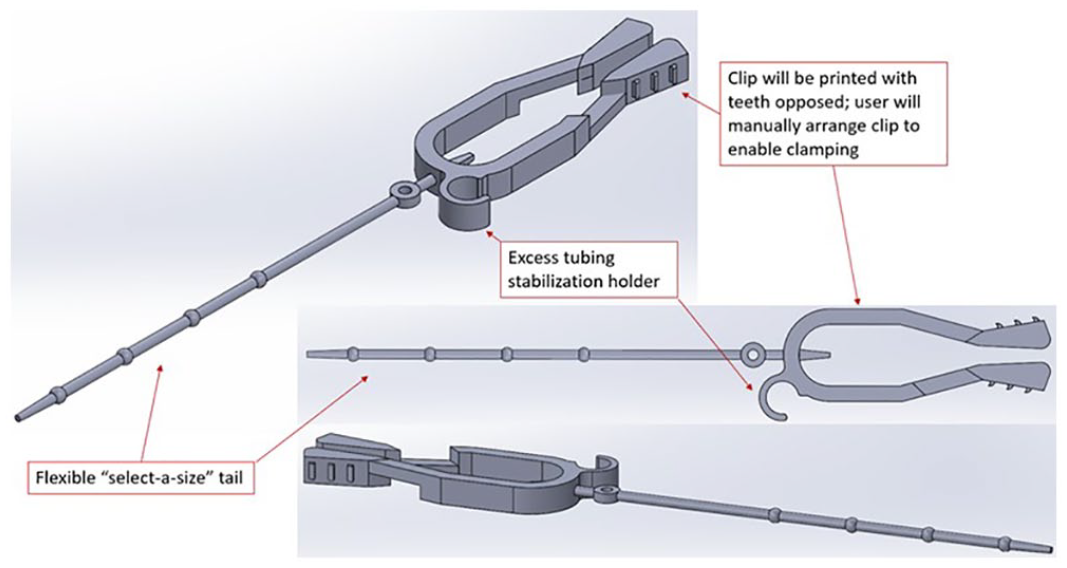
Computer-aided design drawing of the B3 Buddy.
The B3 Buddy is designed to be a flexible, easy to use, and safe device to help patients and frontline clinicians keep catheter drainage bags at an appropriate level. This device can adapt to a variety of patient situations (Figures 6 and 7). Since there are many different urinary drainage bags out on the market, the B3 Buddy demonstrates its versatility using a dual-function ‘select-a-size’ tailed hook on one end and a strong clamp on the other. It is designed to hang off the patient’s gown, clothing, IV pole or ambulatory assistive device while securing the catheter bag on the other end.

B3 Buddy clipped to patient with urinary drainage bag hooked to securement area.

B3 Buddy looped around IV pole and securing urinary drainage bags two different ways.
Building a team
With most of our team’s expertise being in health care, it was necessary to seek collaboration with experts in areas that are not typical in the everyday work of a nurse. For example, after conceptualising an idea and simple prototype, we required more advanced printing so that we could test the product on patients. Our invention team was able to connect with engineers from The Ohio State University’s College of Engineering, specifically the Center for Design and Manufacturing Excellence (CDME). With the CDME’s expertise in product design and development, we were able to create a final prototype that could be printed and tested on patients (with approval from the organisation’s Institutional Review Board).
Throughout the invention process, we also consulted with experts in the fields of product development, FDA compliance, research, and commercialisation. All these consultations were important factors to help our team move in a process that is undefined and unknown to a team of clinicians. In the end, our patent application included four nurses, an occupational therapist, a software engineering student, and a lead engineer. It is pivotal to ensure an invention team has the right players involved to achieve the end goal of successful product development.
Funding for research and development
Developing an invention isn’t without cost. There are ample opportunities within an academic medical centre to procure funding for the innovation of a product and the research to support it. At The Ohio State University, the Innovation Studio provides seed money to eligible teams that ‘pitch’ their project. This process is available to students, faculty, and staff on a quarterly basis. The Innovation Studio provided $1000 of seed money to continue product development with the CDME. Internally, the invention and research team applied for an Innovation and Research Grant available to nurses in the hospital organisation and was awarded $2000 for the final design and production of the B3 Buddy device.
It is also pertinent to share that the American Nurses Association has launched campaigns such as ‘The Gutsy Innovator’ and the ‘ANA Innovation Awards’ to spearhead innovative ideas and products developed by nurses. The National Science Foundation also has a campaign called ‘America’s Seed Fund’ to jump start projects that will enhance the lives of others. These are just a few examples of ways to procure funding for projects.
Device testing and research
It’s important to note that research in the usability of nursing inventions is a novel concept and not well defined by scientific literature. Usability can be defined as a measurement of a product to achieve its intended purpose with effectiveness, efficiency, and satisfaction in a specified context (Bitkina et al., 2020; Brooke, 1995; Schmettow et al., 2017). As the invention team drew closer to a usable product, there came a challenge on how to appropriately test the effectiveness with patients. We did perform formative testing in a lab environment and found the results reassuring and performed as expected. After consultation with the above-mentioned experts, it became clear that a research study with IRB approval was necessary to test the B3 Buddy on patients. At this juncture, we were advised that the B3 Buddy was considered a medical device. To test the B3 Buddy on patients, we had to develop a research protocol to determine the usability and acceptability of this invention.
The definition of a medical device is surprisingly complex and varies dependent on the regulatory body that defines it, such as the United States Food and Drug Administration (FDA) and the European Medicines Agency. Aronson et al., (2020) proposed a simple, yet comprehensive definition to describe medical devices: ‘a contrivance designed and manufactured for use in healthcare, and not solely medicinal or nutritional’ (p.85). Furthermore, the FDA considers a product to be a medical device if it meets the following definition,
Per Section 201(h)(1) of the Food, Drug, and Cosmetic Act, a device is: An instrument, apparatus, implement, machine, contrivance, implant, in vitro reagent, or other similar or related article, including a component part, or accessory which is: (A) recognized in the official National Formulary, or the United States Pharmacopoeia, or any supplement to them, (B) intended for use in the diagnosis of disease or other conditions, or in the cure, mitigation, treatment, or prevention of disease, in man or other animals, or (C) intended to affect the structure or any function of the body of man or other animals, and which does not achieve its primary intended purposes through chemical action within or on the body of man or other animals and which does not achieve its primary intended purposes through chemical action within or on the body of man or other animals and which is not dependent upon being metabolized for the achievement of its primary intended purposes. The term ‘device’ does not include software functions excluded pursuant to section 520(o) (United States Food and Drug Administration (USFDA), 2022).
The FDA also stipulates that testing a medical device requires at least 15 test participants for adequate human factors validation testing (United States Food and Drug Administration, 2016). This allows for assessment of whether the device is effective for the intended users. The FDA regulates the introduction of medical devices for commercial use (Wehde, 2020). Typically, device manufacturers conduct clinical trials to obtain data for marketing clearance from the FDA. If the device is considered a high-risk device, the FDA may need to provide approval prior to IRB protocol submission. For those devices with low risk, such as Class I devices like tongue depressors and gloves, no FDA approval is required. After testing is complete, the FDA may require either a 510(k) premarket notification to prove that it is equivalent to other similar devices in the market or a Premarket Approval, to show that the device is safe and effective (Wehde, 2020). After appropriate clinical testing and approval from the FDA, the medical device is then ready to be marketed to the healthcare industry.
Our team received IRB approval for the research project called ‘Feasibility Testing of a Urinary Drainage Bag Securement Device to Improve Patient and Clinician Ambulation Experiences’. During the IRB application and approval phases, it was determined that the B3 Buddy is considered a device study and did not meet the FDA definition of a medical device. We are moving forward with this feasibility study to determine whether the invention is both usable and acceptable to the end users. Bitkina et al. (2020) state that current literature suggests that research related to usability or user experience in medical-device design and healthcare technology is inadequate. Our team hopes that by testing the B3 Buddy on patients, we can ensure a design that works well for the end user.
Obtaining a patent and commercialisation
Most academic centers have a technology commercialisation department since intellectual property is an inherent outcome of inventions and copyright materials. At The Ohio State University, for example, we have a Technology Commercialization Office. In the Fall of 2021, we disclosed the B3 Buddy invention through the Technology Commercialization Office, who have since facilitated the submission of a provisional patent to protect the intellectual property of this device on behalf of the university.
Although it may seem like a negative experience with a university ‘owning’ your intellectual property, it in fact has many benefits. For example, the university can use its resources to help develop and move your intellectual property into fruition such as finding corporate partners that may want to fund or grow your project. And on the other end, if there were legal consequences to the product, the larger entity would take responsibility of the repercussions since the intellectual property is in the university’s name (as opposed to a single person). The university benefits in this relationship by being named the organisational originator of the product and appreciating a percentage of royalties.
Conclusion
The way of an inventor is a convoluted and meandering path, much like a labyrinth. Those that have the gumption to set upon this path need to embody the traits of curiosity, determination and adaptability to be successful. An inventor needs to build a strong team and be prepared to explore outside their comfort zone. Nursing research is prime for more studies in device usability, especially in areas where nursing practice and patient care is affected. The B3 Buddy Team is currently recruiting patients for a feasibility study of the invention. Future publications will be disseminated on usability and acceptability of B3 Buddy.
Footnotes
Acknowledgements
I would like to acknowledge my B3 Buddy team members, Bridgette Kreuz, Michelle Kuhn, Barbara Potts, Lindsay Ring, Garrett Carder, and Ryan Brune for making this product possible. A special thank you goes to my research mentor, Carl Brown, who has guided me through the development of the above-mentioned research protocol. I am also grateful for the opportunities that The James Cancer Hospital and The Ohio State University provide to support projects such as the B3 Buddy.
