Abstract
Purpose:
To investigate the analgesic effects and mechanisms of BMSCs-IL-10+ administered via intrathecal injection in CCD rats.
Patients and methods:
After CCD surgery, rats were administered intrathecal injections of PBS, BMSCs, BMSCs-IL-10+, BMSCs-IL-10++Anti-IL-10, LV-IRF-8, and LV-IRF-8+IL-10. Pain was assessed by measuring the mechanical withdrawal threshold (MWT) and thermal withdrawal latency (TWL). Glial cell activation and M2 microglial polarization were evaluated by immunofluorescence staining of Iba-1, GFAP, and Arg-1, and by WB for Iba-1 and Arg-1. Spinal cord inflammation was assessed by PCR analysis of TGF-β, TNF-α, and IL-1β expression.
Results:
Compared with the CCD+PBS group, intrathecal injection of both BMSCs and BMSCs-IL-10+ significantly alleviated CCD-induced mechanical and thermal pain. However, the analgesic effect of the BMSCs group markedly decreased after 4 days, while the BMSCs-IL-10+ group lasted at least 14 days. The BMSCs-IL-10+ group significantly upregulated the expression of TGF-β while downregulating TNF-α and IL-1β, inhibiting glial cell activation and promoting M2 microglia polarization. These effects were superior to the BMSCs group and could be abolished by anti-IL-10 antibody. IRF-8 overexpression exacerbated pain and inflammation in CCD rats, but the combined application of IL-10 protein reversed this impact.
Conclusion:
IL-10 is a key cytokine mediating the analgesic effects of BMSCs. Transplantation of BMSCs-IL-10+ cells reduces glial activation, alleviates neuroinflammation, and relieves neuropathic pain by enhancing IL-10 expression and suppressing IRF-8.
Keywords
Introduction
Neuropathic pain (NP) is a chronic pain syndrome caused by nerve damage or disease, significantly impairing patients’ quality of life.1,2 Its pathogenesis is complex, and conventional analgesics demonstrate limited efficacy with considerable side effects in treating NP. 3 Bone marrow mesenchymal stem cells (BMSCs) have received considerable attention in NP therapy due to their immunomodulatory properties and proliferative potential.4,5 Intrathecal injection of BMSCs alleviates NP by suppressing glial cell activation, inhibiting monocyte infiltration, and secreting paracrine factors.4,6 However, the short survival time of transplanted cells and the gradual decline of paracrine effects often limit their long-term therapeutic efficacy. 7
Interleukin-10 (IL-10) plays a crucial role in NP regulation as a key anti-inflammatory cytokine,8,9 and intrathecal injection of IL-10 antibodies can reverse the alleviating effect of BMSCs on NP. 10 Based on this, combining both approaches by genetically modifying BMSCs to overexpress IL-10 holds promise for prolonging cell survival, enhancing paracrine effects, and further suppressing spinal glial cell activation. This strategy may achieve more sustained and effective analgesia in NP treatment.
Interferon Regulatory Factor 8 (IRF-8), a transcription factor, 11 plays a crucial role in NP following peripheral nerve injury (PNI).12,13 In the spinal cord, IRF-8 expression is normally low but is robustly induced predominantly in microglia.14,15 Accumulating evidence indicates that IRF8 is both a key regulator of microglia and a modulator of a reactive microglia phenotype associated with neuropathic pain.16,17 Studies have indicated that IL-10 can suppress the expression of IRF-8, but whether this regulatory effect exists in the spinal cord remains unclear.18–20
This study systematically investigates for the first time the analgesic and anti-inflammatory effects of BMSCs and IL-10-overexpressing BMSCs (BMSCs-IL-10+) in a rat model of chronic compression of the dorsal root ganglion (CCD), focusing on the potential regulatory mechanism of IRF-8. The aim is to provide novel cell therapy strategies and molecular targets for NP treatment. We hypothesize that BMSCs-IL-10+ cells reduce NP by enhancing IL-10 expression and suppressing IRF-8, thereby decreasing glial cell activation and alleviating neuroinflammation.
Material and methods
Experimental animals
Fifty-six adult male SPF Wistar rats (8 weeks) were kept in a specific environment. 21 Experiments were conducted after a 5-day acclimatization period. This study was approved by the Ethics Committee of the Laboratory Animal Center, Qilu Hospital of Shandong University, Approval No.: DWLL-2023-099.
Isolation and culture of BMSCs
BMSCs were isolated from the femurs and tibias of healthy adult Wistar rats. Briefly, bilateral femurs and tibias were aseptically harvested, and bone marrow was flushed with sterile α-minimum essential medium (α-MEM). The cell suspension was filtered through a 70-μm strainer, collected by centrifugation, resuspended in complete α-MEM supplemented with fetal bovine serum and antibiotics, and plated in culture flasks. Non-adherent cells were removed after 24 h, and adherent cells were expanded with medium changes every 2–3 days. BMSCs from the 3rd to 5th passages were selected for intervention and rat transplantation. 10
Viral transfection
Cells were seeded in a 12-well plate. After 24 h, transfection with the LV-IL-10 was performed. 22 After 72 h of transfection, cells were selected using 4 μg/ml puromycin. 23 After continuous selection for 7–14 days, fluorescent expression was observed under a fluorescence microscope, and IL-10 expression was further confirmed by qPCR and Western blotting. 24
Establishment of the CCD rat model
Rats were anesthetized by inhalation of isoflurane in oxygen. The right L4 and L5 intervertebral foramina were exposed, and two L-shaped rods (diameter: 0.6 mm) were inserted at a 30° angle along the foramen toward the spine, thereby compressing the dorsal root ganglion (DRG).25,26 The other end of the rod remained outside the intervertebral foramina. The position and depth of rod insertion were kept consistent across animals to ensure reproducible and sustained compression of the DRG without penetrating the spinal canal. All surgical procedures were performed under aseptic conditions by the same experimenter.
Grouping and intrathecal injection intervention
To verify the role of IL-10 following BMSCs transplantation, 32 rats were randomly divided into 4 groups (Figure 1): CCD+PBS, CCD+BMSCs, CCD+BMSCs-IL-10+, and CCD+BMSCs-IL-10++Anti-IL-10 (n = 8 per group). Three days post-CCD, the CCD+PBS group received 20 μl PBS, the CCD+BMSCs group received 20 μl BMSCs suspension (1 × 106 cells), 27 the CCD+BMSCs-IL-10+ group received 20 μl LV-IL-10-transfected BMSCs (1 × 106 cells), and the CCD+BMSCs-IL-10++Anti-IL-10 group received 20 μl LV-IL-10-transfected BMSCs (1 × 106 cells) in combination with 20 μl IL-10 antibody (R&D Systems, AF519, USA). 10

Experimental schematic and timeline.
To assess whether IL-10 exerts analgesic effects by downregulating IRF-8, 24 rats were randomly divided into 3 groups (Figure 1): CCD+PBS, CCD+IRF-8, and CCD+IRF-8+IL-10 (n = 8 per group). Rats received daily intrathecal injections for 3 days post-surgery. The CCD+PBS group received 20 μl PBS, the CCD+IRF-8 group received 10 μl LV-IRF-8 plus 10 μl PBS, and the CCD+IRF-8+IL-10 group received 10 μl LV-IRF-8 plus 10 μl IL-10 protein (sino biological, 80082-RNAE, China).
Intrathecal injection method
Rats were fixed on a small animal anesthesia apparatus and anesthetized via inhalation of a mixture of isoflurane and oxygen. After disinfecting the dorsal skin, the L4/L5 intervertebral space was localized at the intersection of the bilateral iliac crest line and the spinal midline, and the needle was advanced downward into the L5/L6 intervertebral space. 28 Slowly inject at a rate of 1 μl/s. 29
Behavioral assessment
Mechanical withdrawal threshold (MWT) 30 : After 20 min of adaptation, MWT was assessed using an electronic von Frey apparatus (XR-XZD, Shanghai XinXin Information Technology, China). The von frey was applied vertically to the area between the third and fourth toes on the rat’s footpad. Paw withdrawal or licking was considered a positive reaction, and the force (g) was noted.
Thermal withdrawal latency (TWL) 31 : Adjust the temperature of the thermal plate pain tester (Model XR1700, Shanghai Xinxin Information Technology, China) to 45–65°C. The reaction time was recorded as the latency from the moment the rat was set on the hot plate to its first paw-licking response.
All behavioral assessments were performed by an experimenter blinded to the experimental group assignments.
Tissue collection and processing
Fourteen days 32 after CCD modeling, rats were euthanized in a CO₂ chamber. After shaving, the right atrial appendage was opened for perfusion. Five rats per group were perfused with 200 ml cool saline, and the lumbar spinal cord segment on the right side was isolated for protein and RNA extraction. Another three rats per group were perfused with saline and 4% paraformaldehyde, post-fixed for 48 h, dehydrated in graded sucrose (15%, 22%, 30%), and embedded in OCT for histological analysis.
Western blot
Tissue samples (~30 mg) were lysed in protein buffer and homogenized ultrasonically. And then transferred to membranes, blocked, and incubated with primary antibodies against β-actin (1:3000, Affinity, AF7018, England), Arg-1 (1:10000, Proteintech, 66129-1-Ig, USA), Iba-1 (1:5000, HUABIO, ET1705-78, China), IRF-8 (1:500, Affinity, DF13627, England), and IL-10 (1:3000, R&D Systems, AF519, USA). After 1 h with HRP-conjugated secondary antibodies, bands were visualized by ECL and quantified with ImageJ.
Quantitative real-time PCR (qPCR)
RNA was isolated from spinal cord samples using Trizol, and RNA concentration and purity were determined with a micro-spectrophotometer (A260/A280). Using 1 μg of total RNA as a template, cDNA synthesis was performed in a 20 μl reaction with reverse primers, cDNA template, and DEPC water (Table 1).
Primer sequences.
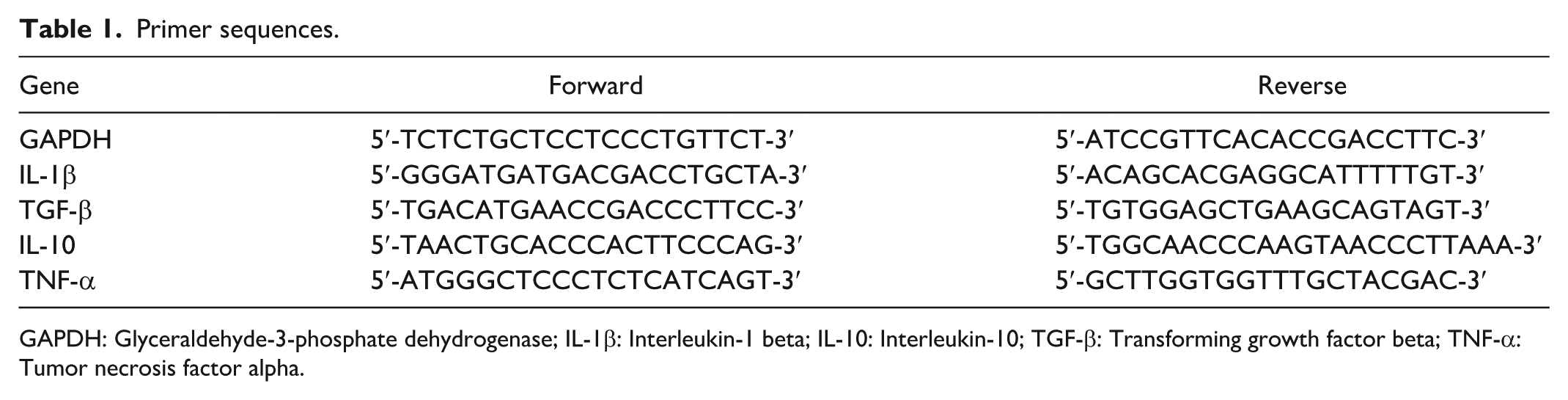
GAPDH: Glyceraldehyde-3-phosphate dehydrogenase; IL-1β: Interleukin-1 beta; IL-10: Interleukin-10; TGF-β: Transforming growth factor beta; TNF-α: Tumor necrosis factor alpha.
Immunofluorescence
Frozen spinal cord tissues were sectioned at 10 μm using a cryostat. Sections were permeabilized and blocked. The sections were then incubated with primary antibodies against Iba-1 (1:100, HUABIO, ET1705-78, China), GFAP (1:300, HUABIO, EM140707, China), and Arg-1 (1:300, Proteintech, 66129-1-Ig, USA) and secondary antibodies.
Statistical analysis
GraphPad Prism 9.0 was used to analyze the experimental data. Immunofluorescence images were quantified for mean fluorescence intensity and positive area using ImageJ. Western blot, PCR, and immunofluorescence data were compared between groups using one-way ANOVA. Behavioral data were analyzed using repeated measures ANOVA. All results are presented as mean ± SEM, and p < 0.05 was considered statistically significant.
Results
Stable expression of IL-10 in BMSCs post-transfection and increased IL-10 expression at the lumbar enlargement of the injured spinal cord following intrathecal injection of BMSCs-IL-10+ in CCD rats
Significant GFP expression was observed in most BMSCs post-transfection (Figure 2(a)), with markedly elevated IL-10 mRNA and protein expression levels in BMSCs (Figure 2(b) and (c)). Compared with the CCD+PBS group, IL-10 protein expression in the lumbar enlargement of the spinal cord was significantly increased in both the BMSCs and the BMSCs-IL-10+ groups, with the increase in the BMSCs-IL-10+ group being markedly higher than in the BMSCs group alone. Co-administration of IL-10 antibody with BMSCs-IL-10+ resulted in a reduction of IL-10 protein expression (Figure 2(d) and (e)).
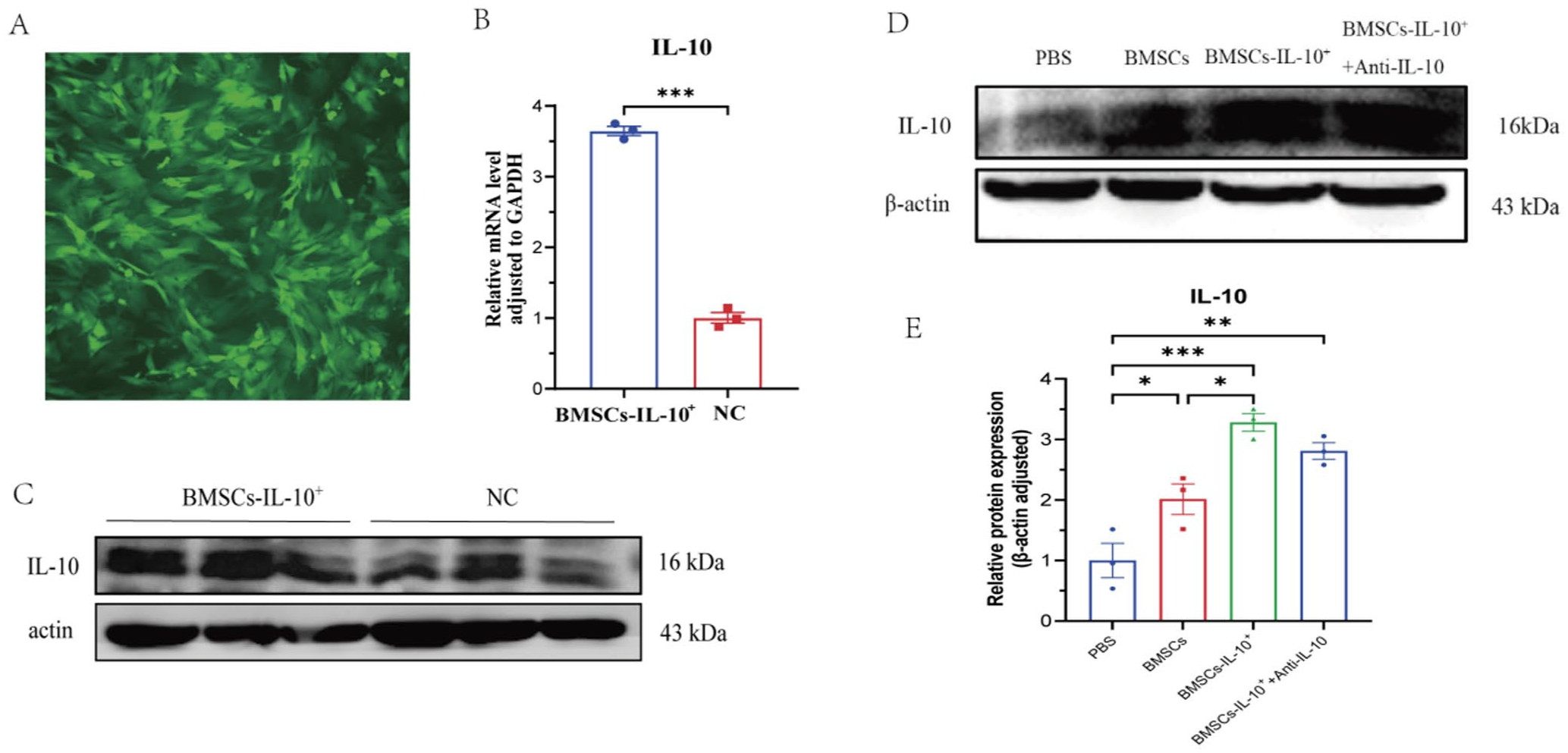
Stable IL-10 expression in BMSCs and upregulation in the injured spinal cord after BMSCs-IL-10+ administration in CCD rats. (a) Significant GFP expression in most BMSCs after transfection with IL-10 lentivirus. (b) IL-10 mRNA levels in BMSCs after LV-IL-10 transfection (n = 3). ***p < 0.001 compared to the non-transfected group. (c) Increased IL-10 protein expression in BMSCs after LV-IL-10 transfection. (d–e) Effects of different interventions on IL-10 protein expression in the lumbar enlargement of the injured side in CCD rats (n = 3). ***p < 0.001, **p < 0.01, *p < 0.05; comparisons are between connected groups.
Intrathecal injection of BMSCs transduced with LV-IL-10 alleviates mechanical and thermal hyperalgesia in CCD rats
Compared with the CCD+PBS group, both the BMSCs group and the BMSCs-IL-10+ group significantly suppressed CCD-induced mechanical and thermal hyperalgesia. The analgesic effect of BMSCs group began to decline markedly at 7 days post-surgery, whereas the BMSCs-IL-10+ group maintained suppression of both mechanical and thermal pain for at least 14 days. Furthermore, co-administration of IL-10 antibody to specifically reduce IL-10 expression significantly attenuated the analgesic effect of BMSCs-IL-10+ (Figure 3(a) and (b)), indicating that IL-10 plays a critical role in alleviating pain in the CCD model.

Intrathecal injection of IL-10–overexpressing BMSCs reduces pain and neuroinflammation in CCD rats more effectively than BMSCs alone. (a–b) Changes in MWT and TWL in CCD rats at 0, 1, 3, 5, 7, and 14 days after different interventions (n = 5). ***p < 0.001, **p < 0.01, *p < 0.05 compared to the CCD+PBS group. (c–e) Spinal cord levels of inflammation-related factors TGF-β, TNF-α, and IL-1β after interventions (n = 5). **p < 0.01, *p < 0.05; comparisons are between connected groups.
Intrathecal injection of BMSCs-IL-10+ alleviates neuroinflammation
Compared with the CCD+PBS group, the BMSCs-IL-10+ group exhibited significantly increased expression of the anti-inflammatory factor TGF-β and markedly reduced levels of pro-inflammatory factors TNF-α and IL-1β in the spinal cord. This anti-inflammatory effect was significantly diminished when IL-10 expression was suppressed using anti-IL-10 antibodies (Figure 3(c)–(e)). However, the anti-inflammatory effect was not significant in the BMSCs group. This indicates that IL-10 can alleviate neuroinflammation in the spinal cord of CCD rats.
Intrathecal injection of BMSCs transduced with LV-IL-10 reduces glial reactivity and promotes M2 microglial polarization
Following intrathecal injection of BMSCs and BMSCs-IL-10+, CCD rats exhibited significantly reduced expression levels of Iba-1 (Figures 4(a) and 5(a)) and GFAP (Figure 5(b)) in the injured spinal cord, along with significantly increased expression of Arg-1 (Figures 4(b) and 5(c)). The BMSCs-IL-10+ group demonstrated more pronounced regulatory effects compared to the BMSCs group. This suppression of glial overactivation and promotion of M2 microglial polarization (Figures 4(a)–(b) and 5(a)–(c)) was notably attenuated when IL-10 antibody was co-administered, indicating that IL-10 plays a critical role in this regulation.
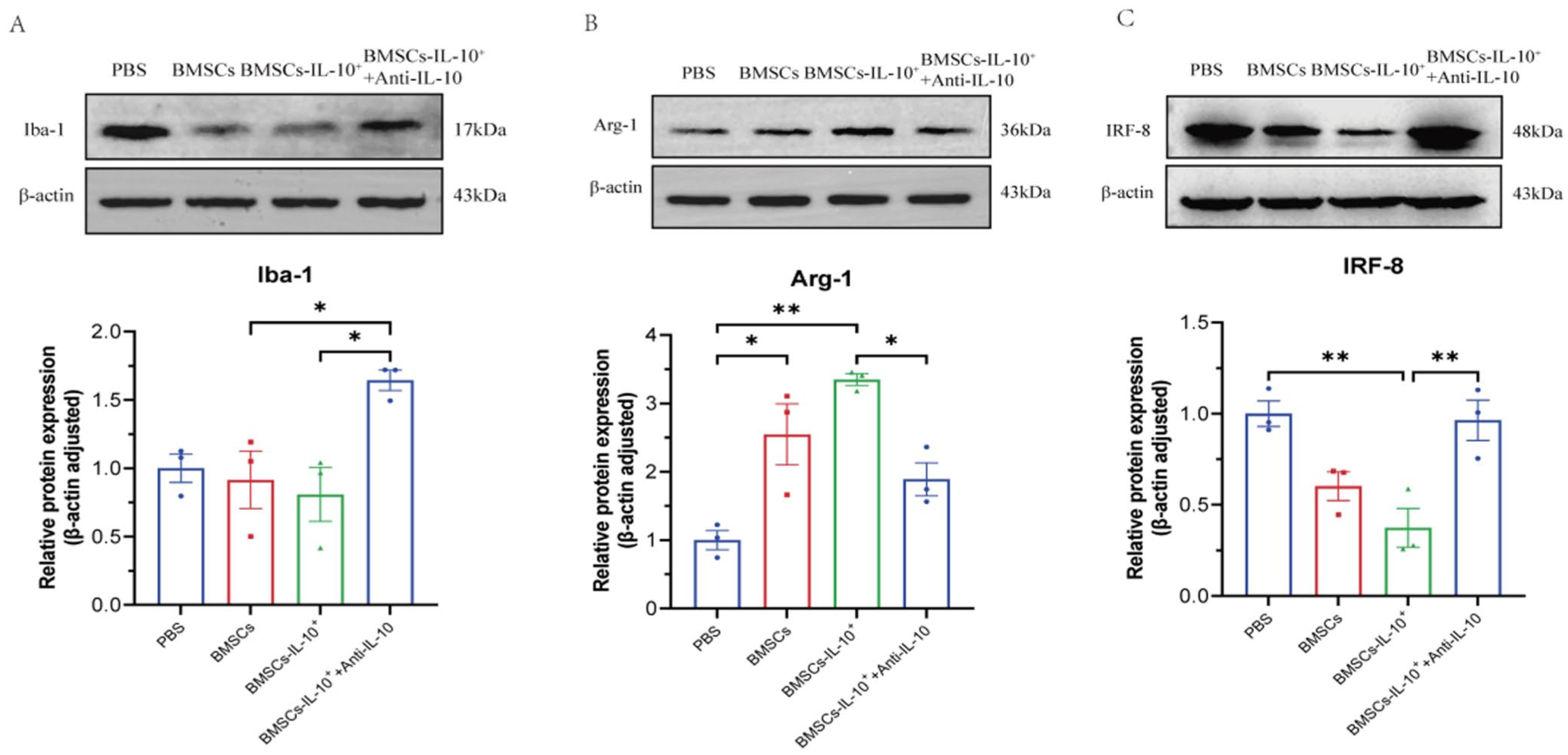
Effects of different interventions on spinal Iba-1, Arg-1, and IRF-8 protein expression in CCD rats. (a) Protein expression levels of Iba-1 in the spinal cord. (b) Protein expression levels of the M2 microglial marker Arg-1 in the spinal cord. (c) Protein expression levels of IRF-8 in the spinal cord (n = 3). ***p < 0.001, **p < 0.01, *p < 0.05; comparisons are between connected groups.

Effects of different interventions on spinal Iba-1, GFAP, and Arg-1 expression in CCD rats. (a) Expression levels of Iba-1 in the spinal cord. (b) Expression levels of GFAP in the spinal cord. (c) Expression levels of the M2 microglial marker Arg-1 in the spinal cord (n = 4). ***p < 0.001, **p < 0.01, *p < 0.05; comparisons are between connected groups. Scale bar: 50 μm.
Intrathecal LV-IRF-8 injection increases IRF-8 expression in the lumbar enlargement of CCD rats, whereas IL-10 suppresses IRF-8 expression
IL-10 levels influenced IRF-8 protein expression (Figure 4c). To further clarify the relationship between IL-10 and IRF-8, we administered LV-IRF-8 intrathecally to CCD rats 1–3 days post-surgery and examined changes in IRF-8 protein expression in the ipsilateral lumbar enlargement region 14 days post-surgery (Figure 6(a) and (b)). Results showed that IRF-8 expression in the CCD+IRF-8 group was significantly higher than in the CCD+PBS group, confirming that intrathecal injection of LV-IRF-8 effectively upregulates IRF-8 expression in the spinal cord. When IL-10 protein was co-administered with LV-IRF-8, IRF-8 expression was significantly reduced compared with the LV-IRF-8 group alone (*p < 0.05), indicating that IL-10 can effectively suppress IRF-8 expression in the spinal cord.

Intrathecal injection of IRF-8 lentivirus increases pain and spinal inflammation in CCD rats, which can be reversed by IL-10. (a–b) Spinal IRF-8 protein levels in CCD rats after IRF-8 lentivirus and IL-10 protein interventions (n = 3). ***p < 0.001, **p < 0.01, *p < 0.05; comparisons are between connected groups. (c–d) MWT and TWL in CCD rats at 0, 1, 3, 5, 7, and 14 days post-interventions (n = 5). ***p < 0.001, **p < 0.01, *p < 0.05 compared to the CCD+PBS group. (e–g) Spinal levels of inflammatory factors TGF-β, TNF-α, and IL-1β after interventions (n = 5). **p < 0.01, *p < 0.05; comparisons are between connected groups.
Intrathecal injection of IRF-8 lentivirus exacerbates mechanical and thermal pain in CCD rats, while IL-10 reverses the effects of IRF-8
Intrathecal administration of LV-IRF-8 significantly aggravated mechanical and thermal hyperalgesia in CCD rats compared with the CCD+PBS group. However, when combined with IL-10 protein intervention, this IRF-8-mediated hyperalgesic effect was significantly suppressed, suggesting that IL-10 can alleviate neuropathic pain by inhibiting IRF-8 expression (Figure 6(c) and (d)).
Overexpression of IRF-8 exacerbates neuroinflammation, while IL-10 suppresses its effects
Compared to the CCD+PBS group, the IRF-8 overexpression group exhibited significantly reduced expression of the anti-inflammatory factor TGF-β in spinal cord tissue, while pro-inflammatory factors TNF-α and IL-1β showed markedly elevated expression. In the IRF-8+IL-10 co-treated group, TGF-β expression was significantly upregulated compared to the IRF-8 group, while TNF-α and IL-1β expression levels showed a marked downward trend (Figure 6(e)–(g)). This indicates that IL-10 can alleviate neuroinflammation in the spinal cord of CCD rats by regulating IRF-8 expression.
IRF-8 overexpression increases glial reactivity and suppresses M2 microglial polarization, which is reversed by IL-10
Following intrathecal injection of IRF-8, CCD rats exhibited significantly increased expression of Iba-1 (Figure 7(a)) and GFAP (Figure 7(b)) in the injured spinal cord, alongside decreased expression of Arg-1 (Figure 7(c)). The regulation of glial cells by IRF-8 was reversible following IL-10 protein administration (Figure 7(a)–(c)), indicating that IL-10 suppresses activation of microglia and astrocytes by modulating IRF-8 expression, meanwhile promoting the increase of M2 microglia.

Effects of different interventions on spinal cord Iba-1, GFAP, and Arg-1 expression in CCD rats at 14 days post-surgery. (a) Expression levels of Iba-1 in the spinal cord. (b) Expression levels of the GFAP in the spinal cord. (c) Expression levels of the M2 microglial marker Arg-1 in the spinal cord (n = 4). ***p < 0.001, **p < 0.01, *p < 0.05; comparisons are between connected groups. Scale bar: 50 μm.
Discussion
In this study, we demonstrated that intrathecal injection of BMSCs-IL-10+ significantly reduced mechanical and thermal hyperalgesia induced by CCD, with analgesic effects lasting over 14 days – longer than those of BMSCs alone. This analgesia mechanism involves inhibiting IRF-8-mediated glial cell activation and neuroinflammation. This is the first study to increase BMSCs’ analgesic impact through IL-10 gene modification and investigate its underlying mechanism, resulting in a novel cellular therapy strategy for neuropathic pain (NP).
Our study found that BMSCs-IL-10+ significantly prolonged the duration of improved mechanical and thermal pain sensitivity, whereas the analgesic effect in the BMSCs group began to decline markedly on postoperative day 7. This suggests that IL-10 gene modification extends the duration of BMSCs-mediated analgesia, likely through sustained IL-10 release that maintains an anti-inflammatory environment in the spinal cord. Consistently, studies in the spinal nerve ligation (SNL) model have shown that intrathecal IL-10 injection suppresses thermal hyperalgesia and mechanical allodynia in rats, and IL-10 antibody pretreatment completely abolishes the inhibitory effects of IL-10 on pain hypersensitivity, indicating that IL-10 is a key mediator of analgesia. 15
Compared with the CCD+PBS group, the anti-inflammatory effect of unmodified BMSCs was not significant, possibly because BMSCs lose viability within 14 days after intrathecal injection, leading to insufficient secretion of paracrine factors, including IL-10, and failing to sustain inflammation suppression. Previous studies have indicated that in CCI mice receiving intrathecal injections of fluorescently labeled BMSCs, the number of BMSCs peaked on day 3 post-injection and then gradually declined. 33 Our study found that intrathecal injection of BMSCs-IL-10+ significantly increased the anti-inflammatory cytokine TGF-β and decreased pro-inflammatory cytokines TNF-α and IL-1β in CCD rats. This anti-inflammatory effect was markedly reduced when IL-10 expression was neutralized with an antibody, confirming the critical role of IL-10 in modulating spinal neuroinflammation in CCD rats. These results are consistent with earlier research showing that intrathecal IL-10 gene therapy alleviates paclitaxel-induced NP by reducing pro-inflammatory cytokine levels. 34 Following BMSCs-IL-10+ injection, spinal dorsal horn glial cell activation was reduced and the number of M2 anti-inflammatory microglia increased, further supporting the regulatory effect of IL-10 on glial cell activation.35,36
Interferon Regulatory Factor 8 (IRF-8), a key transcription factor, is significantly upregulated following PNI. Previous studies have unequivocally demonstrated that IRF-8 is expressed specifically in spinal dorsal horn microglia and plays a central role in regulating microglial activation and gene expression remodeling of dorsal horn microglia in the spinal cord after PNI, driving microglia toward a reactive phenotype. 15 Building upon this foundation, the present study investigaties how modulation of IRF-8 by IL-10 influences glial activation states and associated pain behaviors. We report for the first time that intrathecal administration of IL-10 significantly downregulates IRF-8 expression in the spinal cord, effectively alleviating mechanical and thermal pain in CCD rats, reducing neuroinflammation, and suppressing microglial activation. Notably, although our analyses were performed at the whole-spinal-cord tissue level, LV-IRF-8 was associated with decreased expression of Iba-1 and GFAP, alongside increased expression of the anti-inflammatory marker Arg-1, suggesting altered activation states in both microglia and astrocytes. Future studies employing cell-type-specific approaches will be essential to definitively delineate the cellular origin of IRF-8 and its precise functional contribution to neuropathic pain pathogenesis. These findings establish the IL-10-IRF-8 axis as a key regulatory pathway in neuropathic pain and identify a novel therapeutic target.
While IL-10-mediated immunomodulation plays a central role in the observed analgesic effects, sustained overexpression of IL-10 may also carry potential risks. Chronic suppression of spinal immune activity could alter physiological neuroimmune homeostasis or increase susceptibility to local immune dysregulation. Although no apparent adverse effects were detected within the experimental timeframe of this study, longer-term and repeated administration paradigms were not examined. Further studies incorporating long-term behavioral, immunological, and histological assessments will be necessary to better characterize the safety profile of sustained IL-10 expression in the spinal cord.
This study has certain limitations. Due to the lack of specific labeling of BMSCs and IL-10, we were unable to trace their distribution and survival time after intrathecal injection. Future studies will employ fluorescent labeling to systematically investigate their distribution and dynamic changes in the spinal microenvironment, further elucidating the underlying mechanisms. Additionally, although this study observed an IL-10-mediated suppression of IRF-8-related glial cell activation and neuroinflammation, it did not directly investigate the molecular mechanism linking IL-10 and IRF-8. It is also important to consider that the present conclusions are derived from male animals. Accumulating evidence indicates that neuropathic pain processing exhibits marked sex-specific differences, with microglia-dependent mechanisms predominating in males, while alternative immune pathways may contribute to pain hypersensitivity in females. 37 These sex-dependent differences may influence the generalizability of the present findings, and future studies incorporating female animals will be necessary to fully elucidate sex-specific mechanisms.
Conclusion
This study demonstrates that BMSCs-IL-10+ markedly alleviate neuropathic pain in CCD rats, with a more sustained analgesic effect compared to conventional BMSCs. The underlying mechanism may involve upregulation of the anti-inflammatory cytokine IL-10, suppression of IRF-8 expression, and reduction of glial cell activation and neuroinflammation. For the first time, this study highlights the critical regulatory role of the “IL-10-IRF-8” axis in neuropathic pain, providing a theoretical basis for stem cell therapy and combined IRF-8-targeted interventions in the treatment of neuropathic pain.
Footnotes
Acknowledgements
We thank Research Center for Basic Medical Science of Qilu hospital affilitated to Shandong University for consultation and instrument availability that supported this work.
Author contributions
Yican Yang was responsible for experimental operation, data organization, and manuscript drafting. Mutong Wang assisted in conducting experiments and participated in data analysis. Yan Feng contributed to manuscript drafting. Yang Zhang participated in manuscript revision and review. Shouwei Yue oversaw experimental guidance, and participated in manuscript revision and finalization. All authors approved the final edited version.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work is supported by the Natural Science Foundation of Shandong Province of China (No. ZR2021MH023), the Key Research and Development Program of Shandong Province (No. 2022ZLGX03) and the Scientific Research Innovation Team Project of the Affiliated Hospital of Shandong University of Traditional Chinese Medicine (No. 2024-72).
Ethical considerations
This study was approved by the Ethics Committee of the Laboratory Animal Center, Qilu Hospital of Shandong University, Approval No.: DWLL-2023-099.
