Abstract
The cellular and molecular mechanisms of acupuncture have been investigated across various tissues in multiple animal models. However, the dynamic cellular and molecular changes at human acupuncture points remain unexplored. The primary challenge preventing such a study is the practical difficulty of obtaining sufficient cells from acupoints. To address this, we developed a new needle manipulation technique that enables the collection of sufficient cell number from the acupuncture needle during the treatment. Using this approach and single-cell technology, we identified eight cell types at the acupoint BL23: inflammatory fibroblast, myofibroblast, skeletal muscle cell, endothelial cell, smooth muscle cell, adipocyte, macrophage, and a novel cell type characterized by marker genes CNTNAP2 and CSMD1. Remarkably, this novel cell population was significantly enriched during the pain relief phase compared to the pain state, while the other seven cell types were significantly reduced following acupuncture analgesia. Transcriptomic analysis suggested that these novel cells are involved in synapse assembly and synaptic plasticity. This study presents the first characterization of cellular and transcriptional dynamics at the acupoint BL23, offering new insights into the mechanism underlying acupuncture-induced pain relief.
Introduction
Acupuncture, originated in ancient China, has gained widespread adoption in the Western world over recent decades due to its demonstrated therapeutic benefits. In many countries, it is now recognized by healthcare systems and covered by insurance. One of its most common clinical applications is for pain relief. 1 According to traditional Chinese medicine, low back pain (LBP), a common symptom, 2 is believed to result from internal disharmony, which causes a blockage of the body’s vital energy, known as Qi, along certain meridians. Acupuncture analgesia is thought to restore the flow of Qi by stimulating specific acupoints and facilitating neuronal signal transmission.3–5 Among these meridians, acupoints along the bladder meridian are frequently used in clinical practice to manage musculoskeletal pain and dysfunction. 6
Although acupoints are thought to contain multiple cell types that work synergistically, the specific cell types and their roles remain unclear. Moreover, despite advances in modern anatomical studies, the physical presence of acupuncture points has yet to be confirmed. 7 Thus, understanding the specific cell types at the acupoints is crucial to explaining the cellular or molecular events that lead to acupuncture’s therapeutic effects.
To understand the cell composition at acupoints, ongoing efforts are focused on developing methodologies to analyze cells near or directly from acupoints. Efforts to date include transcriptomic profiling of skin cells near acupoints following stimulation,8,9 investigations into the role of mast cells in acupuncture-induced analgesia,10–12 and the isolation of primo vascular system cells from acupoints.13,14 Recently, Liu et al. 15 revealed a neuroanatomical basis for acupoints in driving specific autonomic pathways. However, these studies have been conducted in animal models. In humans, most research at acupoints has focused on histological or anatomical features, such as nerve distribution, connective tissue density, blood vessels, and fascial structures. 16 True cellular-level analysis (like single-cell RNA seq, spatial transcriptomics, or even flow cytometry) of human acupoints has never been done.
In this study, we developed a needle biopsy technique that enables the retrieval of cells directly from the tip of acupuncture needles and applied single-cell transcriptomic analysis to examine the cellular heterogeneity at the acupoint BL23 (Shenshu), both before and after LBP relief. We identified eight cell types, including a novel cell population enriched in the post-treatment state. Additionally, we characterized the roles of fibroblasts, endothelial cells, and macrophages in acupuncture at BL23. This work represents the first comprehensive cellular and molecular characterization of an acupoint and offers new insight into the cellular landscape at the acupoint BL23 in relation to the therapeutic effects of acupuncture.
Materials and methods
Patient and sample collection
This study included two patients with chronic LBP and two patients with acute LBP (lumbar sprain). The acupuncture therapy was administered twice weekly for 2–4 weeks. Cells adhered to acupuncture needles were collected at the first time intervention and again 2 weeks later from the BL23 (or UB23, Shenshu) acupoint as paired samples. To collect cells, following needle insertion, rotational stimulation was applied to allow fascial fibers to wrap around the needle. Rapid needle withdrawal to extract the tissue biopsy from the acupoint. Using the needle biopsy technique, viable cells were consistently obtained unless the pain had resolved; no cells are recovered from healthy acupoints. Demographic characteristics of the patients are provided in Table 1.
Demographic characteristics of individuals included in this study.
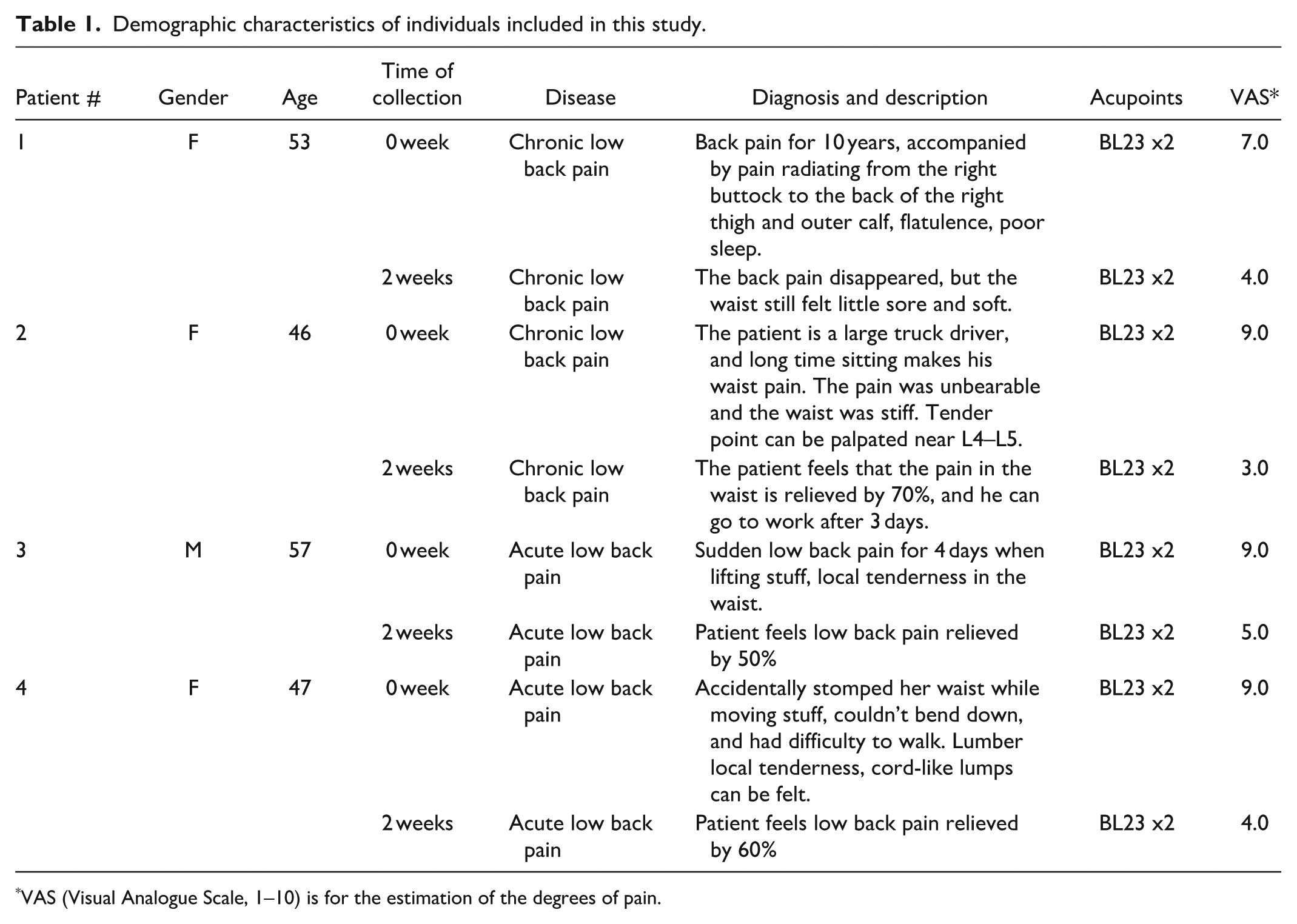
VAS (Visual Analogue Scale, 1–10) is for the estimation of the degrees of pain.
Needles and reagents
HuanQiu® Copper Handle Sterile Acupuncture Needles (30 gauge, 40 mm length) are from SAMA Co. Ltd., China. Tissue collection buffer (1X PBS, pH7.4). Homogenization buffer (Tris-Cl 10 mM, NaCl 10 mM, MgCl2 3 mM, NP40 0.1%, Colleganase-7 0.01 mg/mL). Wash buffer (1X PBS, 1% BSA, RNase Inhibitor 0.2 U/μL).
Nuclei isolation
Tissues were placed in tissue collection buffer immediately after collection and snap frozen in dry ice for later nuclei isolation. For processing, tissues were thawed in 500 μL homogenization buffer in a Kimble® 2-mL Glass Dounce Tissue Grinder (885300-0002) and incubate at room temperature with gentle rotation for 5 min. Samples were then transferred to ice to cool rapidly, then homogenize and centrifuge at 500 × g for 5 min at 4℃. Remove the supernatant and resuspend the pellet with 500 μL wash buffer, followed by centrifugation at 500 × g for 5 min at 4℃. After discarding the supernatant, the pellet was resuspended in 40 µL of wash buffer. Nuclei concentration was determined using the Countess II Automated Cell Counter (Thermo Fisher Scientific) with 0.4% Trypan Blue staining.
Library prep and sequencing
The single nuclei were partitioned into nanoliter-scale Gel Bead-In-EMulsions (GEMs) using Chromium™ Single Cell 3′ GEM Kit v3.1 & Gel Bead Kit v3.1 (10X Genomics) and Chromium™ Controller (10X Genomics). Barcoded cDNAs are generated through Reverse transcription reaction within GEMs. Sequencing ready libraries are generated from barcoded cDNA by using Chromium™ Library Construction Kit (10X Genomics). 28 + 90 bp pair-end reads were sequenced on Illumina NovaSeq 6000 sequencer (Illumina, Inc), and data was de-multiplexed using CellRanger mkfastq v7.2.0 (10X Genomics).
Data analysis
The acupuncture snRNA-seq data were analyzed using Python package Scanpy version.1.10.2. First, the cells with low numbers of features (<100 genes/cell) or a high number of mitochondrial genes (>2%) were filtered out. The four pairs of snRNA-seq datasets were integrated using the Scanorama algorithm, which incorporates batch correction. 17 A low-dimensional embedding of each cell was computed using Scanorama, after which cell clustering was performed using a resolution of 0.75. Cell identities were subsequently assigned based on the expression of known marker genes obtained from referenced expression data or commonly used markers in literature. Differentially expressed genes (DEGs) were computed using wilcoxon method (P-adj < 0.05). PAGA connectivity between cell types was performed to show the lineage relations. Shiny GO 0.81 18 and Enrichr 19 are used for the Gene Ontology (GO) Biological Process analysis.
Results
Acupuncture needle biopsy and tissue sampling
To investigate the cellular changes associated with acupuncture-induced pain relief in LBP patients (Figure 1(a)), our acupuncturist uses a novel needle manipulation technique, which enables the collection of minute tissue samples directly from the needle tip during acupuncture. The method reveals a fascinating link between pain and tissue availability. As pain subsides, the size of the collected tissue progressively decreases, ultimately becoming impossible to collect once patients have experienced full recovery. This unique feature suggests a dynamic interplay between the therapeutic effects and the changes in cellular characteristics at the acupoint BL23 (Figure 1(b)).

Characters of acupuncture samples from BL23. (a) Location of acupoint of BL23. (b) Pathological acupoint specimen of BL23 from deep interstitial connective tissue of back pain patients. (c) Formalin-fixed paraffin embedded acupuncture sample is histologically stained by HE and Sirius-H (400X).
To further characterize the tissue obtained through the needle biopsy at BL23, H&E, and Sirius-red&H staining were performed and revealed numerous fusiform nuclei, indicative of elongated fibroblast cells, while Sirius-red staining highlighted abundant collagen fibers (Figure 1(c)). These observations suggest that BL23 is composed of dense connective tissue, playing a structural role in acupuncture-mediated therapy. 20
Single-cell RNA-seq unveils cellular diversity at the acupoint BL23
To explore the cellular composition and their functions at the acupoint BL23 in acupuncture-induced pain relief, single-cell RNA-seq was performed on the collected cells from the BL23 acupoint. Consistent with the decrease in tissue collection after pain relief, cell yield was significantly lower in the Post state (after pain relief) compared to the Pre state (before pain relief; Table 2). This suggests that cells at BL23 exhibit increased adhesion during the inflammatory state associated with LBP, potentially mediated by local changes in the extracellular matrix (ECM) environment.
The numbers of isolated nuclei from the acupuncture sample of BL23.

Unbiased clustering of single-cell data revealed eight distinct cell populations at BL23 (Figure 2(a)). A drop-plot of normalized top variable genes from these populations highlighted their distinct functional profiles and marker genes (Figure 2(b)). Accordingly, these cell populations were annotated as inflammatory fibroblast, myofibroblast, endothelial cells, vascular smooth muscle cells, skeletal muscle cells, adipocytes, macrophages, and an unidentified cell type. The most intriguing finding of this study is the identification of a previously unknown cell type. To characterize it, we followed three recognized standards: (1) Unique gene expression profile: The genes from this cell population are mainly related to synaptic assembly (CNTNAP2, CSMD1), regulation of synapse organization (GRID2, NRG1), and pre/post-synaptic cell adhesion (TENM2, CTNNA2; Figure 2(c)). This unique gene combination has only been characterized in neuron, astrocyte, microglia, and oligodendrocyte that play a role of synaptic assembly. (2) Lack of traditional markers: The absence of typical markers for the established cell types, combined with the presence of genes associated with synaptic functions, supports the notion of a distinct cell type. (3) Reproducibility: The gene expression profile is observed in all samples from LBP patients. Therefore, we name this new cell population as
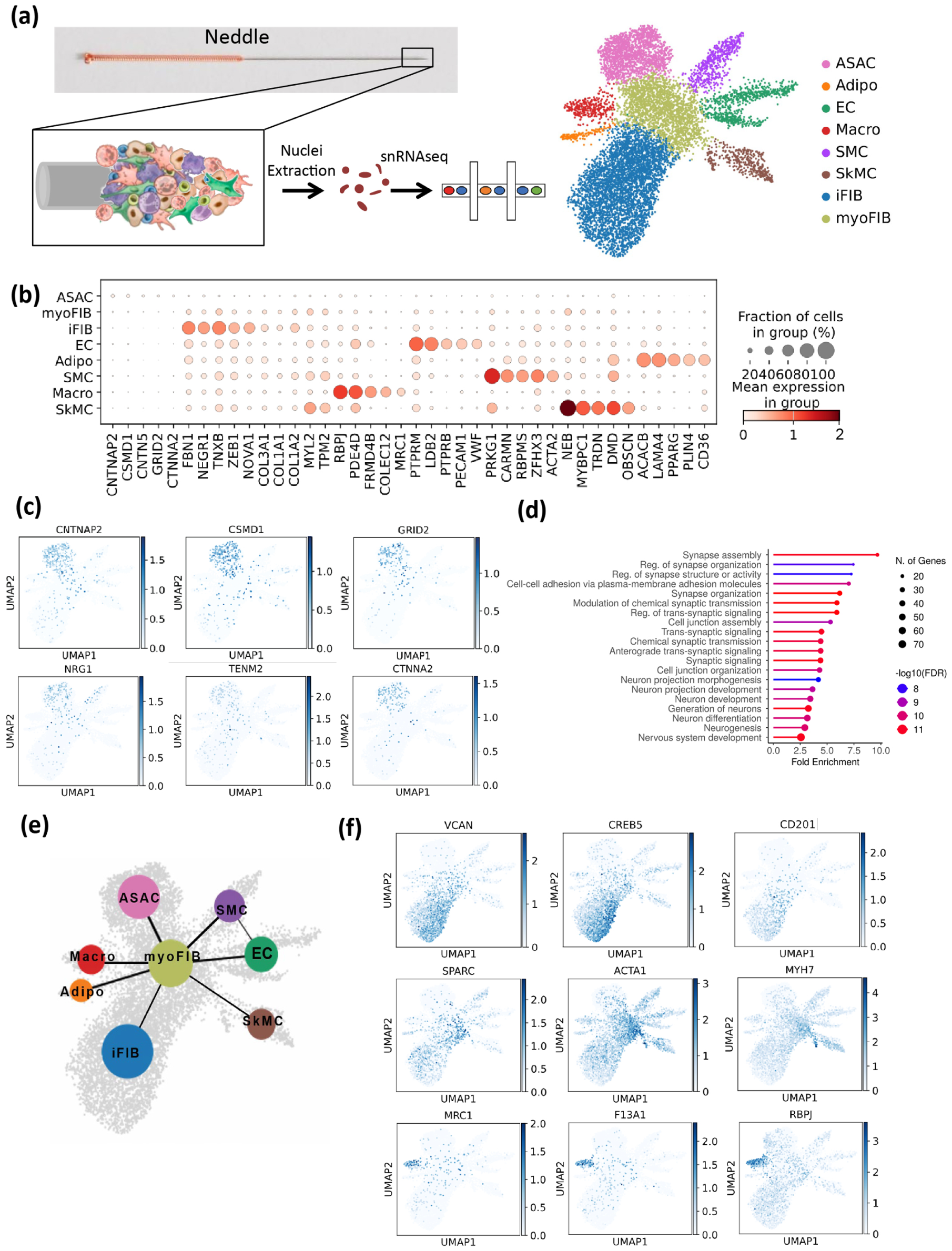
Identification of cell populations at acupoint BL23 of patients undergoing low back pain. (a) A flowchart depicting the snRNAseq and UMAP delineate eight major cell clusters from acupoint BL23. (b) Dropplot of normalized expression of the top variable genes in acupoint cell types. (c) Synapse assambly/plasticity genes (CNTNAP2, CSMD1, GRID2, NRG1), gene coding presynaptic cell adhesion protein (TENM2) and postsynaptic cell adhesion protein (CTNNA2) in ASAC. (d) Gene Ontology (GO) analysis of Biological Processes for ASACs. FDR < 0.05, P-adj < 0.05. (e) PAGA connectivity of the cell types. (f) Inflammatory genes (VCAN, CREB5, CD201) in inflammatory fibroblast (iFIB); Myocytic genes (SPARC, ACTA1, MYH7) in myofibroblast (myoFIB); Tissue remodeling genes (MRC1, F13A1, RBPJ) in marcrophage (Macro).
Moreover, Gene Ontology (GO) Biological Process analysis revealed enrichment for Synaptic Assembly (GO:0007416), Reg. of Synapse Organization (GO:0050807), and Cell-Cell Adhesion via Plasma-Membrane Adhesion Molecules (GO:0098742), advocating for a neuron repair function (Figure 2(d)). PAGA connectivity analysis showed no significant lineage relationships between ASACs and other cell types except with myofibroblast, further supporting the notion that ASACs represent a novel cell type (Figure 2(e)).
Regarding other cell types, inflammatory fibroblasts were enriched with pro-inflammatory genes, including VCAN and CREB5, while myofibroblasts expressed markers such as SPARC and ACTA1 (α-skeletal actin; Figure 2(f)). Notably, myofibroblasts exhibited dual expression of ACTA1 and ACTA2 (α-smooth muscle actin). Since ACTA1 is typically expressed in striated muscle cells, where it contributes to cellular movement and morphological changes, 21 while ACTA2 is involved in the formation of stress fibers, 22 this co-expression suggests a heightened contractile capacity for wound closure, potentially relevant to the pathological state of LB. 23 Additionally, CD201+ fibroblast progenitors, previously reported to respond to pro-inflammatory fibroblast/myofibroblast differentiation, 24 were primarily located in the inflammatory fibroblast cluster (Figure 2(f)), hinting at their involvement in spatiotemporal regulation of fibroblast differentiation at the acupoint. Acupoint macrophages, characterized by MRC1, F13A1, and RBPJ (Figure 2(f)), likely contribute to tissue remodeling and wound healing, given their association with extracellular matrix formation and repair processes. 25
Cellular changes before and after pain relief
Cell-type-specific analysis revealed significant alterations in cell abundance between the Pre and Post states (Figure 3(a) and (b)). Needle captured fibroblasts (inflammatory fibroblast and myofibroblast), vascular cells (endothelial cell and smooth muscle cell), and skeletal muscle cells decreased dramatically in the Post state, indicating a reduced inflammation, ECM remodeling and needle-adhesion characteristics in the Post state.26,27 However, ASACs were significantly enriched in the Post state, suggesting enhanced neural connectivity and synaptic plasticity in response to acupuncture. Therefore, we postulate the inflammatory environment associated with LBP inhibits the local ASACs activity and impairs their roles in neuron synapse assembly and plasticity functions that may be linked to the flow of Qi through meridians and are restored by acupuncture stimulation.

Distinct patterns of cell populations in different states at acupoint BL23. (a) Cell clusters and distribution of the cell numbers in Pre and Post. (b) Total cell numbers in Pre and Post states.
Gene expression changes in response to acupuncture at BL23
Next, we sought to understand the effect of acupuncture on the gene expression across distinct cell types. A heatmap of gene expression highlighted dynamic changes in synaptic plasticity, angiogenesis, tissue repair, inflammatory, and ECM genes profile among various cell populations in the Pre and Post states (Figure 4(a)). For ASACs, the volcano-plot of differentially expressed genes (DEGs) shows upregulated expression of synaptic assembly genes CNTNAP2/CSMD1 in the Post ASACs suggested a potential role as establishing and refining synapses, while the upregulated expression of morphological remodeling genes MYH7/TNNT1 in the Pre ASACs may provide mechanical and structural support for the synaptic environment by maintaining cytoskeletal organization (Figure 4(b)). This is also confirmed by GO analysis of the DEGs (Figure 4(c)).

Functional heterogeneity in fibroblats, macrophages, endothelial cells, and ASACs at acupoint BL23. (a) Heat map of feature genes in fibroblasts, macrophages, endothelial cells, and ASACs. (b) Volcano plot shows DEGs of ASACs in Pre versus Post. (c) GO analysis of Biological Processes for the upregulated DEGs in Post ASACs and Pre ASACs. DEGs with Padj < 0.05. (d) Relative abundance of expression of inflammatory genes (CREB5, VCAN) in iFIB, and myocytic genes (SPACR, MYH7) in myoFIB, Pre (blue) versus Post (red). (e) Relative abundance of expression of angiogenesis genes (EPAS1, ELT1) in EC, Pre versus Post. (f) Relative abundance of expression of M1-classical activation genes (TLR4, CD86) and M2-tissue repair genes (CSF1R, CD163L1) in Macro, Pre versus Post.
Notably, fibroblasts exhibited significant functional gene expression changes following acupuncture. Inflammatory fibroblasts showed a reduced abundance of expressing pro-inflammatory markers CREB5 and VCAN, whereas myofibroblasts displayed an increased abundance of expressing myocytic genes, like SPARC and MYH7 in Post state (Figure 4(d)). Beyond fibroblasts, endothelial cells also demonstrated an increased abundance of expressing angiogenesis markers EPAS1 (HIF2α) and FLT1 (VEGFR1) in the Pre state (Figure 4(e)), consistent with an early response to tissue injury and suggestive of a role in tissue repair.28–30 These findings suggest that acupuncture modulates inflammatory responses and facilitates tissue recovery, key mechanisms underlying pain relief of LBP.
Acupuncture also modulated macrophage function. Given the well-known plasticity of tissue-resident macrophages in response to environmental cues,31,32 we observed a shift in macrophage activation states following acupuncture treatment. In the Post state, macrophages exhibited a reduced abundance of expressing classical activation markers TLR4 and CD86, and increased tissue repair markers CSF1R and CD163L1 (Figure 4(f)). This transition suggests an anti-inflammatory and tissue remodeling effect, reinforcing roles of macrophages in acupuncture’s therapeutic effect. 25 Notably, the total number of tissue-resident macrophages remained unchanged between the Pre and Post states.
Additional evidence for the anti-inflammatory role of acupuncture includes the upregulation of the lncRNA IL12A-AS1 in Post state of ASACs, which is known to inhibit IL12 secretion and thereby suppress inflammatory macrophage activation 33 (Figure 4(a)). The elevated IL10RA expression in Pre state macrophages may reflect a compensatory anti-inflammatory response through increasing their sensitivity to IL-10 to limit excessive inflammation and prevent tissue damage 34 ; this expression diminishes upon pain relief in the Post state (Figure 4(a)). Beyond their immune-regulatory functions, Post state ASACs also expressed genes associated with synaptic plasticity (CNTNAP2, RBFOX1) and pain relief (GRM5, KCNJ6), highlighting their potential contribution to neuronal circuit restoration and analgesia (Figure 4(a)). Recent studies have reported that teneurin (TENM) signaling stabilizes axon branches during synaptic partner matching. 35 Consistently, our data showed increased TENM2 expression in Post state ASACs (Figure 4(a)), suggesting that ASACs may facilitate synapse formation within specific subcellular compartments through TENM signaling.
Comparison of acute and chronic LBP in response to acupuncture at BL23
Lastly, given that acupuncture tends to be more clinically effective in acute LBP than in chronic LBP, especially in the early stages of treatment,36,37 we investigated whether this difference correlates with the increase of ASACs. Our analysis revealed a greater increase of ASACs in patients with acute LBP compared to those with chronic LBP (Figure 5(a) and (c)). Furthermore, in comparison of Pre versus Post states, acute LBP patients exhibited a substantially increase in ASACs in the Post state than did chronic LBP patients (Figure 5(b) and (d)). While the overall cell attachment to the acupuncture needle likely depends on the level of inflammatory adhesion proteins, the behavior of ASACs may be governed by their specialized role in synaptic adhesion. Together, these findings suggest that the distinct cellular compositions between acute and chronic LBP may underlie their differential therapeutic responses to acupuncture, with ASACs potentially serving as key mediators of effective intervention.

Cell populations in different diagnosis at acupoint BL23. (a) Cell clusters in acute LBP patients. (b) Cell clusters of acute LBP in different stages, Pre versus Post. (c) Cell clusters in chronic LBP patients. (d) Cell clusters of chronic LBP in different stages, Pre versus Post.
Discussion
To elucidate the cellular nature of acupoint, this study utilized single-cell RNA sequencing to construct a comprehensive cellular atlas of acupoint BL23 in patients with LBP. The findings highlight how acupuncture reshapes the local cellular landscape to mediate pain relief. A key technique we developed in this study is successful collection of sufficient cells directly from the tip of the acupuncture needle at the human acupoint BL23, making single-cell analysis technically possible at acupoints.
Through our single-cell analysis, we identified eight cell types at the acupoint BL23. Seven of these are well known components of fascia or deep interstitial connective tissue—structures recognized as key anatomical substrates for acupoints.38–40 While previous animal studies have reported mast cells within the hypodermis at acupoint sites,10–12 we did not detect mast cells in our human BL23 samples. In contrast, we discovered a novel cell type with unique gene expression profile, named Acupoint Synapse Assembly Cells (ASACs). ASACs comprised 21% of all captured cells and 59.2% of cells in the Post group in BL23, and appear to play key roles in: (1) forming, strengthening, or remodeling synaptic connections in response to the needle stimulation, contributing to synaptic plasticity and subsequent pain relief, and (2) expressing cell adhesion molecules (e.g. cadherins, protocadherins, neurexins, and neuroligins) that are critical for the initial formation and stabilization of synaptic contacts. 41 Together with the GO Biological Process analysis and PAGA connectivity analysis, these findings suggest that although acupuncture clinically manifests as the restoration of both “Qi flow” and tenderness upon palpation through the conversion of mechanical stimuli into neural and tissue-level responses, the enrichment of ASACs in the post-treatment state observed in this study provides strong evidence that synapse assembly may play a critical role in mediating analgesia. Regarding the origin of ASACs, needle-induced mechanical stimulation may trigger their amplification, differentiation from mesenchymal cells, or recruitment from peripheral region guided by neurons. However, the precise mechanisms underlying the origin of ASACs require further investigation.
To investigate the molecular changes of acupuncture treatment at the acupoint BL23, the functional gene expression profiles at both Pre and Post states were analyzed. Our data revealed a significant upregulation of synapse assembly and organization genes in ASACs following treatment. Additionally, other cell types also exhibited distinct wound healing response-related genes across states. For example, an increased number of fibroblasts expressing inflammatory genes and extracellular matrix remodeling genes were detected in Pre state; more endothelial cells displayed higher angiogenesis genes also presented in Pre state, suggesting early tissue repair activity. Moreover, tissue-resident macrophages demonstrated a phenotypic shift from pro-inflammatory M1 to tissue repair M2 states in the Post state. Together, these diverse cell populations coordinate to mediate neural plasticity, analgesia, and wound healing, ultimately contribute to the therapeutic effects of acupuncture.
This pilot study involved a limited number of patients due to the challenges of sample acquisition. Nevertheless, the reproducible cellular features and differential expression patterns observed across subjects suggest that the findings are biologically meaningful and merit further investigation in larger cohorts. Furthermore, future work employing spatial transcriptomics, multiplex immunostaining, or organoid-based co-culture systems will be warranted to validate ASAC identity and function. Additionally, exploring the interactions between stromal cells and neurons at acupoint may provide deeper insights into the broader physiological mechanisms underlying acupuncture therapy.
In conclusion, this study identifies ASACs as a novel cell type, likely critical to acupuncture-induced pain relief. By comparing the Pre and Post states in LBP patients, we propose that the Pre state represents an inhibitory phase characterized by elevated inflammation and disrupted meridian energy flow. In contrast, the Post state reflects a recovery phase, mediated in part by raised ASACs, enabling local meridian energy flow. We hypothesize that ASACs are activated by acupuncture stimulation and participate in the formation of new synaptic connections with local neurons that were impaired during the pain state. ASACs at the acupoint BL23 may serve as specialized organizers of synapse formation for analgesia. This study advances our understanding of cellular dynamics at human acupoint and offers critical insights into the cellular and molecular mechanisms of acupuncture, guiding future strategies for enhancing clinical efficacy and therapeutic outcomes.
Footnotes
Author contributions
Conceptualization: G.Z. and X.L. Methodology: G.Z., Shunfa.H., and X.W. Investigation: G.Z. and X.L. Resources: Shunfa.H., Shihui.H., G.Z., S.C. and X.Z. Formal analysis: M.D. and X.W. Data curation: M.D. and X.W. Validation: H.L., R.L. and K.B. Writing:original draft: G.Z. and X.W. Writing:review and editing: X.L. Supervision: X.L. Project administration: G.Z. and X. L.
Data availability statement
All data relevant to this study are included in the main text. Raw sequencing datasets produced for this study have been archived in the GEO database and are accessible under accession number GSE312510.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by NIH grans R01DE29173, P30CA016042, and U54AG089335.
Ethical approval
This study has been approved by UCLA Research Administration Department webIRB (OHRPP). The IRB number is 23-001773.
Consent to participate
All patients provided informed consent to participate and tissue collection via acupuncture needle.
