Abstract
Immunomodulatory molecules play a crucial role in the establishment and maintenance of chronic pain. Among these, anti-inflammatory interleukin-10 (IL-10) has emerged as one of therapeutic options to ameliorate pain state. Some components reduced pain through stimulating endogenous IL-10. However, IL-10 has a short half-life, which limits its long-term treatment of chronic pain. Gene therapy targeting
Introduction
Chronic pain is a significant clinical challenge affecting millions of individuals worldwide. 1 Currently, the effective approaches for chronic pain treatment remains limited. Therefore, new therapeutic strategies are required. Neuroinflammation, characterized by the activation of neural immune cells and their release of pro-inflammatory factors, is increasingly recognized as an important molecular mechanism in chronic pain pathogenesis. A growing body of evidence shows that the anti-inflammatory cytokine interleukin 10 (IL-10) can inhibit proinflammatory cytokines and reduces pain.2–4 Therefore, balancing inflammatory cytokines using IL-10 has recently emerged as promising therapeutic approaches for chronic pain.4–6
Gene therapy implies for many facets of animal and human diseases. 7 In gene therapy, gene expression is targeted to peripheral or central nervous system (CNS) regions to provide sustained long-term efficacy.7–10 There are two approaches to achieving successful transfection of target genes into cells – viral and non-viral strategies. Although non-viral systems have continued to be utilized as vehicles for gene delivery,11,12 viral vectors-mediated gene transfer remains generally more effective in delivering nucleic acids to a variety of target cell populations. 13 Viral vector-mediated strategies for pain have been developed for years.14–17 Recombinant herpes simplex virus (HSV), adeno-associated virus (AAV), adenovirus (AV), and lentivirus (LV)-based vectors are main examples of commonly used viral vectors in gene therapy for chronic pain.7,10 Watkins and Milligan groups have investigated and well reviewed the non-viral mediated IL-10 delivery for preclinical pain treatment, especially focusing on plasmid DNA encoding interleukin-10.12,18,19 Therefore, this review emphasizes the application of IL-10 mediated by viral vector for chronic pain.
Chronic pain and IL-10
Neuroinflammation of chronic pain
Nerve injury and tissue damage including inflammation induce release of proinflammatory molecules from activated nociceptors of sensory neurons (e.g. peripheral terminals of C fibers and A-delta fibers) or non-neural cells (such as macrophages, T cells, and neutrophils) that reside within or infiltrate into the injured area.20–23 These factors include neurotransmitters, neuropeptides (substance P, calcitonin gene-related peptide, bradykinin, etc.), proinflammatory cytokines (e.g. interleukin-1β (IL-1β) and IL-6, and tumor necrosis factor alpha (TNFα)), chemokines, and related lipids (such as prostaglandins), neurotrophins, reactive oxygen and nitrogen species, as well as extracellular proteases and protons, etc., referred to as the “inflammatory soup.”21,23,24 Many factors in the “inflammatory soup” bind on the receptors of the peripheral nociceptor terminals, such as G protein-coupled receptors, by activating transient receptor potential channels, acid-sensitive ion channels, two-pore potassium channels, and receptor tyrosine kinases.20,23,25 This process leads to enhanced excitability and sensitization to previously subthreshold stimulation in the first-order sensory neurons, termed peripheral sensitization.12,20,26 Neuroinflammation in the spinal dorsal horn is a key contributor to the onset and persistence of chronic pain, driven by the activation of glial cells including microglia and astrocytes in response to tissue damage, nerve injury, viral infection, and chemotherapy, triggering the release of pro-inflammatory mediators such as TNFα, IL-1β, etc.27–30 In the spinal dorsal horn, the second order neurons express receptors for many of these factors, for example, TNF receptors.12,25,31 It is reported that TNFα through neuronal TNF receptor I, induced mechanical allodynia (pain in response to a stimulus that does not normally provoke pain, a form of nociceptive hypersensitivity) in neuropathic pain, 32 regulating spinal cord synaptic plasticity and central sensitization (a state of enhanced transmission in nociceptive projection neurons that is characterized by a decreased activation threshold and an increased responsiveness to nociceptive stimuli. 26 ). TNF receptors play a distinct role in regulating different phases of inflammatory pain. 33 TNFα and IL-1β enhanced the frequency of spontaneous excitatory postsynaptic current in regulating synaptic and neuronal activity in the superficial spinal cord. 34 A great body of evidence reveals chemokines as important players in pain control.35–38 There are bidirectional activities in chemokine-mediated neuron-glia interactions. Neurons can produce chemokines, and glial cells express the respective receptors of these chemokines. Glial cells also express chemokines, and neurons express their respective receptors.23,37 Additionally, glia secrete brain-derived neurotrophic factor (BDNF), increasing neuronal excitability by altering chloride ion gradients. 39 Neurons themselves contribute to the circuits by releasing neurotransmitters and neuropeptides that activate glial cells, creating a self-sustaining inflammatory loop.40,41 Through ascending pain pathways, the second-order neurons in the spinal cord dorsal horn receive signals from the first-order neurons and then project to the brain. 20
IL-10 and its mechanisms of action on pain
Human IL-10, a 35-kD homodimer, has two non-covalently bonded monomers. 42 Early evidence shows that IL-10 reduces the release of proinflammatory cytokines such as TNFα and IL-1β from human polymorphonuclear leukocytes. 43 IL-10 exerts its anti-inflammatory effects primarily through activation of IL-10 receptor complex. IL-10 receptor is tetrameric, consisting of IL-10 receptor α (IL-10Rα) and IL-10 receptor β (IL-10Rβ).44,45 IL-10 receptor induces the activity of transcription factor STAT3, 46 leading to the inhibition of proinflammatory signals that activate the innate immune response. 47 IL-10 induces the expression of suppressor of cytokine signaling-3 (SOCS3), which negatively regulates JAK/STAT activation, further dampening inflammatory responses. 42 IL-10 signaling induces SOCS3 suppressing proinflammatory cytokines by targeting the p65 NF-κB subunit for degradation. 42 IL-10 also has potent anti-inflammatory activity, through the suppression of gene activation by TLR signaling pathways. 48 Meanwhile, IL-10 induces expression of MAPK phosphatases, inhibiting MAPK signaling and hence transcription of proinflammatory cytokines (see Reviews42,49). Evidence showed that IL-10 inhibits the LPS- and/or IFN-γ-induced secretion of TNF-α, IL-1β, IL-6, IL-8, G-CSF, and GM-CSF in immune cells. 49 Niiro et al. 50 reported that IL-10 inhibited LPS-induced cyclooxygenase (COX) activity in neutrophils. Recent report showed that genetic depletion of IL-10 elevated M1 like phenotype (CD86+ CD206-) and increased release of proinflammatory cytokines and chemokines under proinflammatory conditions, suggesting that it promotes the polarization of microglia into M1-prone phenotype under proinflammatory conditions (see Figure 1). 51 Gonzalez et al. 52 reported that IL-10 and IL-10RI expressions in injured areas were mainly found in microglia/macrophages and in reactive astrocytes after an excitotoxic injury to the postnatal rat brain, suggesting that this expression may have anti-inflammatory effects in distinct anatomic sites. The detailed actions and pathophysiology of IL-10 and IL-10 receptors have been extensively reviewed elsewhere.53–57
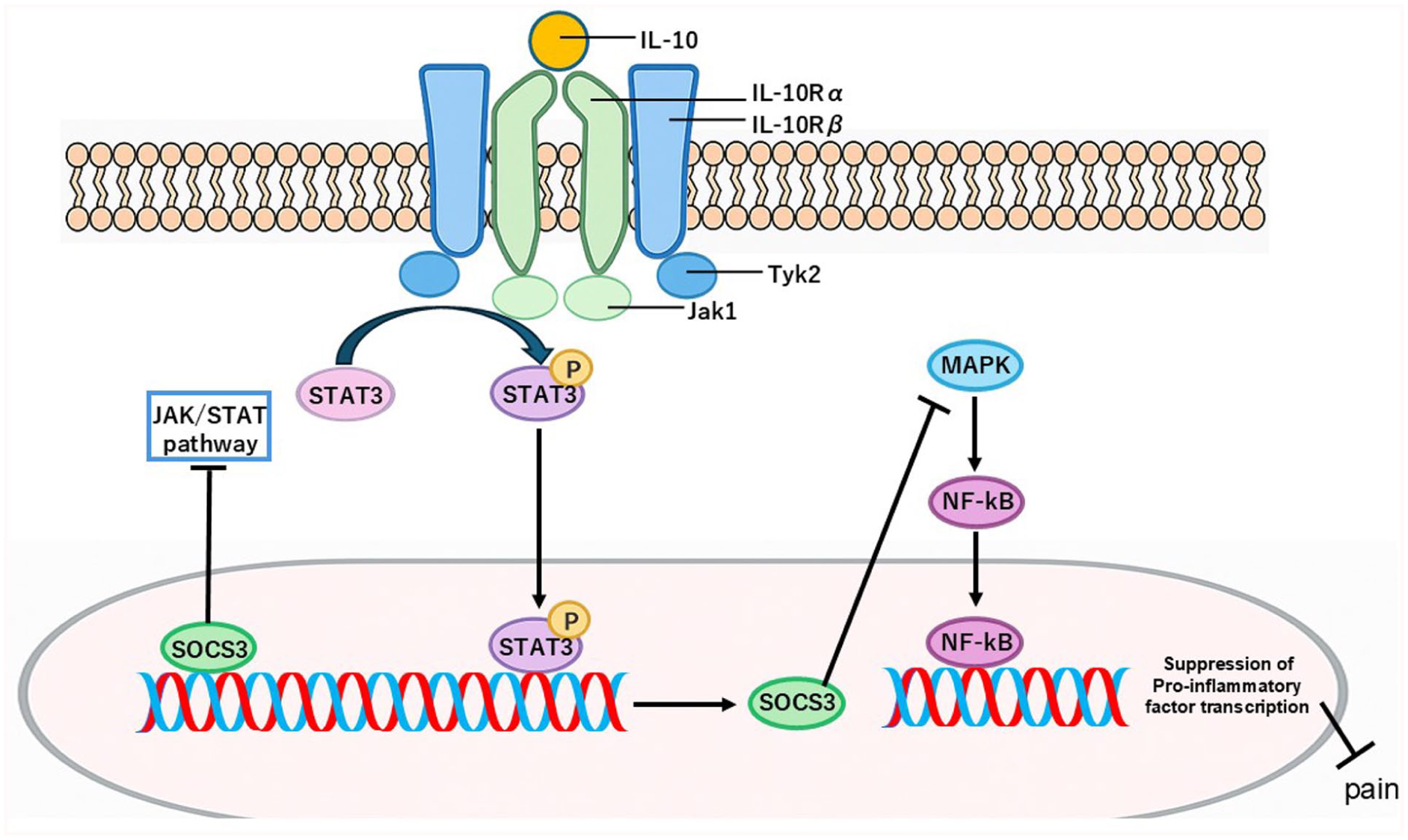
The IL-10 possible signaling pathway in pain. IL-10 binding to its IL-10 receptor leads to the phosphorylation of STAT3. Phospho-STAT3 binds to STAT-binding elements of STAT3-responsive genes in the nucleus and induces the transcription of suppressor of cytokine signaling 3 (SOCS3). SOCS3 may inhibit the MAPK – NF-kB signal, leading to downregulation of proinflammatory molecular expression, finally reducing pain.
Pain therapeutic potentials of IL-10
IL-10 has been extensively investigated in preclinical models of chronic pain. Due to a short half-life of IL-10 protein, intrathecal IL-10 protein can transiently reverse neuropathic pain. 58 Based on its potent anti-inflammatory and neuroprotective properties, IL-10 has been regarded as a promising target for pain management. 42 Despite its therapeutic promise, challenges remain in optimizing IL-10 delivery methods, ensuring long-term efficacy, and minimizing potential off-target effects.42,59
Endogenous IL-10 is also involved in the mechanisms of chemicals in pain treatment. Thalidomide, for example, primarily inhibits TNF synthesis. In a nerve-injury induced pain model, thalidomide reduced pain, reversed the increase in endoneurial TNFα, and also increases endogenous interleukin-10. 60 Anandamide, a lipid mediator that acts as an endogenous ligand of CB1 receptors enhances LPS/IFN gamma-induced IL-10 production in microglia. 61 It is reported that BIRT-377, a small molecule non-competitive lymphocyte function-associated antigen-1 antagonist, relieved allodynia in neuropathic rats through decreasing IL-1β and concurrent increasing IL-10 immunoreactivity. 62 Similarly, BIRT377 reversed pain, reduced IL-1β and TNF, and increased IL-10 in neuropathic pain mice. 63 IL-10 transgene plasmid DNA (XT-150) injected intrathecally is currently in phase I clinical trial for preliminary evaluation of safety, tolerability, and efficacy for the treatment of painful lumbar disk disease (ClinicalTrials.gov NCT04466410).
Gene therapy for pain management
Gene therapy strategies attempt to adjust molecular changes through directly delivering nucleic acids such as DNA, siRNA, shRNA, miRNA, and antisense oligonucleotides into cells to modulate gene expression.10,64 The most important factor in gene therapy is effectively delivering nucleic acids into the target cells, this is exceptionally challenging. 65 There are mainly two approaches to achieving successful transfection to cells, viral and non-viral methods. Non-viral gene delivery can be broadly separated into physical and chemical methods.64,65 Physical non-viral methods include microinjection, ballistic gene delivery, electroporation, sonoporation, laser irradiation, magnetofection, and electric field-induced molecular vibration 64 ; chemical approaches use cationic lipids, cationic polymers, and cell-penetrating peptides that can be engineered to deliver target nucleic acids into cells locally or systemically.64,65 Generally, main disadvantage of non-viral methods is lower delivery efficacies than viral gene delivery methods. Although novel non-viral systems have continued to apply as useful vehicles for gene delivery,11,12 viral vectors-mediated gene transfer remain generally highly effective methods in delivering nucleic acids to a variety of target cell populations.13,66 Gene therapy offers a unique approach to modulating chronic pain by delivering therapeutic genes directly to the affected tissues.7,8,10,67 Because IL-10 has a short half-life and it is rapidly cleared and attains low target concentrations, 12 viral-vector-mediated gene therapy is a more desirable route of administration to require local and long-term IL-10 expression for chronic pain management. Watkins and Milligan groups have well investigated and reviewed the non-viral mediated IL-10 delivery for preclinical pain treatment, especially focusing on plasmid DNA encoding interleukin-10.12,18,19 Here, in this review we focused on the utility of viral vector-mediated IL-10 expressions in preclinical pain models. Current main viral vector systems including herpes simplex virus (HSV), adeno-associated viruses (AAV), adenoviruses (AV), and lentiviruses, have been employed to achieve efficient and sustained expression of target genes.65,68–73
Literature for gene therapy approach by viral vector-mediated IL-10 for chronic pain in animal models
A comprehensive literature search was conducted in January 2025 across PubMed, bioRxiv, and the Cochrane Database of Systematic Reviews (CDSR). The PubMed search covering studies from 1990 to 2025 initially yielded 41,421 records for the term “

PRISMA flow diagram. Database searching yielded 43,262 records: PubMed (
Summary of gene therapy approach by HSV-mediated IL-10 pain in animal models.
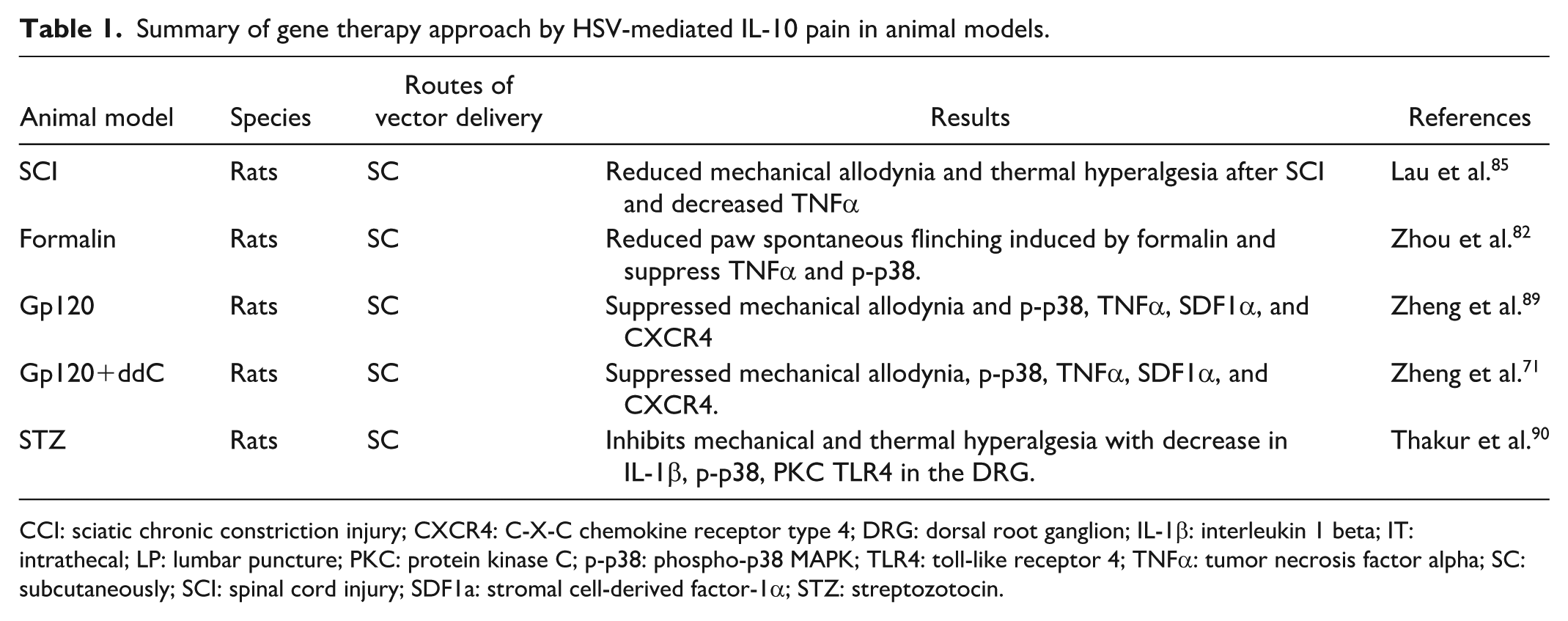
CCI: sciatic chronic constriction injury; CXCR4: C-X-C chemokine receptor type 4; DRG: dorsal root ganglion; IL-1β: interleukin 1 beta; IT: intrathecal; LP: lumbar puncture; PKC: protein kinase C; p-p38: phospho-p38 MAPK; TLR4: toll-like receptor 4; TNFα: tumor necrosis factor alpha; SC: subcutaneously; SCI: spinal cord injury; SDF1a: stromal cell-derived factor-1α; STZ: streptozotocin.
Summary of gene therapy approach by AAV-mediated IL-10 pain in animal models.

AAV: adeno associated viruses; CCI: sciatic chronic constriction injury; IT: intrathecal; LP: lumbar puncture; SCI: spinal cord injury; SIN: Sciatic inflammatory neuropathy; SNI: spared nerve injury; SNL: spinal nerve ligation; TF: transforaminal epidural Injection.
Summary of gene therapy approach by AV and LV vector-mediated IL-10 pain in animal models.

AV: adenoviruses; CCI: sciatic chronic constriction injury; IL-1β: interleukin 1 beta; IL-6: interleukin 6; IT: intrathecal; Local use at SCI lesion: local delivery at spinal cord injury lesion area using poly(lactide-co-glycolide) scaffold; LV: lentivirus vector; SCI: spinal cord injury; SIN: Sciatic inflammatory neuropathy; SNI: spared nerve injury; SNL: spinal nerve ligation; TNFα: tumor necrosis factor-alpha.
HSV vector
HSV is a neurotropic virus with double-stranded linear 152-kb DNA with a 40-kb genome capacity. 8 Recombinant HSV vectors are incapable of viral replication in vivo but these vectors, like the wild-type parental virus, can establish a latent state in neurons in the skin or mucous membranes, from which the transgene products are expressed in vivo. 74 After non-replicating HSV vectors are injected into the skin, they are taken up by the peripheral terminals of dorsal root ganglion (DRG) neurons and transported retrograde along the axon to the cell bodies of DRG neurons. 75 Thus, HSV can be effectively utilized to express target genes following subcutaneous administration to modify the nervous system. 76
HSV vectors can be used effectively to transfer target genes into DRG neurons from subcutaneous inoculation to modulate pain-related behaviors through anti-inflammatory and/or inhibitory neurotransmitters.77–80 Rats receive a subcutaneous injection of formalin into the plantar aspect of the hind paw induces a standard model of tonic pain, the so-called formalin test. 81 Zhou et al. reported that HSV-mediated IL-10 reduced the number of paw flinches in phase 1 (1–10 min) and phase 2 (10–60 min) in formalin test; the antinociceptive effect of IL-10 correlated with decrease in phosphorylation of p38 and TNFα in spinal microglia, 82 indicating that gene therapy targeting the neuroimmune component of chronic pain, further supporting the therapeutic versatility of gene-based cytokine modulation. 83
It is known that neuropathic pain involves chronic neuroinflammatory response driven by activated glia releasing pro-inflammatory cytokines.41,84 Lau et al. 85 reported that HSV-mediated IL-10 expression significantly reduced neuropathic pain following spinal cord injury (SCI), in which right hemilateral spinal cord damage was created in rats by blunt impact through a T12 laminectomy. Subcutaneous inoculation of IL10 vector 1 week after SCI reduced mechanical and thermal hyperalgesia, and mechanical allodynia by 2 weeks after injury, and the anti-nociceptive effect lasted throughout the 4-week period beginning 1 week after injection. 85 Evidence showed that IL-10 reduces the neurotoxic effects of glutamate on spinal cord neurons in vitro and enhances expression of Bcl-2 and Bcl-x(L), suggesting that IL-10 has direct neuronal effects with important implications for neuroprotection. 86 HSV-based vector to express IL-10 in spinal cord after lateral hemisection injury increased neuronal survival of the spinal cord and improved motor function up to 6 weeks after injury, that correlated with increased expression of Bcl-2 and Bcl-x(L) in neurons. 87 IL-10 mediated by HSV blocks the activation of TNFα and p-p38 MAPK induced by LPS. 82 Persistent inflammatory pain induced by formalin increased TNFα in the spinal cord. 82 Transgene-mediated expression of IL-10 by HSV attenuated TNFα expression and p-p38 MAPK in dorsal horn of spinal cord. 82 Human immunodeficiency virus (HIV)-related chronic pain is primarily induced by the HIV-related distal sensory polyneuropathy and antiretroviral toxic neuropathies (see Review 88 ). HIV peripheral neuropathy induces glia activity and releases proinflammatory factors. 88 Zheng et al. 89 provided evidence demonstrating that IL-10 mediated by HSV vectors suppressed neuropathic pain induced by HIV gp120 application into the sciatic nerve in rats, adding valuable insight into its therapeutic role in neuropathic conditions. The anti-allodynic effect was apparent on day 3 after vector inoculation and lasted more than 28 days. 89 IL-10 expressed by HSV reduced p-p38, TNFα, stromal cell-derived factor-1α (SDF-1α), and C-X-C chemokine receptor type 4 (CXCR4) in the DRG and/or the spinal dorsal horn. Zheng et al. 71 also demonstrated that IL-10 mediated by HSV vector significantly reduced neuropathic pain induced by HIV gp120 application to the sciatic nerve combined with intraperitoneal ddC (a drug to treat HIV) injection in rats, further validating the potential of IL-10 in treating complex neuropathic conditions. IL-10 expression mediated by the HSV vectors increased mechanical threshold, and the anti-allodynic effect of IL-10 lasted more than 3 weeks. 71 And IL-10 reduced expression of p-p38, TNFα, SDF1α, and CXCR4 in the spinal cord or DRG. 71 Diabetic neuropathy is a painful complication. Evidence implicates the role of proinflammatory mediators in the pathogenesis of neuropathic pain. Thakur et al. 90 showed that delivery of IL10 in the nerve fibers achieved by HSV vector mediated transduction of dorsal root ganglion (DRG) in animals with Type 1 diabetes induced by intraperitoneal injection of streptozotocin (STZ; 50 mg/kg). IL10 mediated by HSV improved hind-paw thermal and mechanical pain thresholds measured 6 weeks after the onset of diabetes and 4 weeks after vector inoculation. 90 They found that IL-10 blocked the nociceptive responses in the DRG neurons, and reduced IL1β expression along with inhibition of p-p38 MAPK, protein kinase C (PKC), TLR4 expression in the DRG, highlighting its application in diabetes-induced neuropathic pain. 90
Adeno-associated viral vectors
AAV is a single-strand DNA virus with a 4.7-kb genome; it has low immunogenicity, important for the application in human gene therapy.68,91 AAV integrates in the host genome and is transmitted to the progeny of the cells. 7 AAV vectors have the tissue tropism of different serotypes. 67 Subtypes of AAV 1, 2, 4, 5, 7, 8, and 9 are naturally-occurring serotypes and have the tropism to CNS (see more information, at Addgene.org and vectorbuilder.com).
Recent advances in gene therapy have improved the feasibility of IL-10-based treatments for chronic pain.
30
Milligan et al.
58
demonstrated the potential of AAV2 to drive IL-10 production for controlling neuropathic and pathological pain. Peri-sciatic injection of an immune activator, such as zymosan, around sciatic nerve induces sciatic inflammatory neuropathy (SIN) and chronic pain,
92
which creates robust mechanical allodynia. Intrathecal AAV-IL10 blocked development of SIN-induced mechanical allodynia for 10 days.
58
Sciatic chronic constriction injury (CCI) is a classic model of neuropathic pain induced by placing loosely constrictive ligatures around the common sciatic nerve.
93
Intrathecal AAV-IL10 reversed established CCI-induced thermal hyperalgesia and attenuated established CCI-induced mechanical allodynia around 2 weeks.
58
In addition to these behavioral outcomes, AAV-mediated IL-10 delivery has also been associated with suppression of inflammatory mediators. Milligan et al.
58
discussed that intrathecal AAV-IL-10 reversed neuropathic pain in parallel with reduced spinal expression of pro-inflammatory cytokines such as TNFα and IL-1β. Storek et al.
94
introduced a self-complementary recombinant AAV8 vector for lumbar puncture delivery, targeting sensory neurons to manage chronic pain. The antiallodynic effect of sc-rAAV8 expressing nonopioid analgesic gene IL-10, alone or in combination with beta-endorphin was equally effective for about 3 months.
94
The spared nerve injury (SNI) is a model neuropathic pain model involving a lesion of two of the three branches of the sciatic nerve (namely, the tibial and common peroneal nerves) leaving the remaining sural nerve intact.95,96 Kim et al.
97
reported that transforaminal epidural injection of AAV vector expressing IL-10 reduced mechanical allodynia at 4 weeks after AAV, and that a combination of three AAVs (AAV5-
Unger et al.98,99 demonstrated the intrathecal delivery of human IL-10 using self-complementary AAV8 vectors in swine, and also evaluated that safety studies of IL-10 or related therapeutics may require syngeneic large animal models, suggesting that swine are not idiosyncratically sensitive to intrathecal IL-10 because, recapitulating previous reports in dogs, they suffered no clinical neurotoxicity from the human ortholog.
Adenovirus vectors
Adenoviruses discovered in 1950s are DNA viruses carrying linear double-stranded DNA of approximately 36-kb. 100 Early gene 1 (EG1) in the adenovirus genome is essential for replication. Deletion of the region coding for EG1 can make the virus replication-defective, which reduces the risk of genotoxicity, and provides sufficient space for target gene insertions.101,102 AV does not integrate into the chromosome of host cells, therefore, it could decrease the likelihood of insertion mutation.67,101
A replication-defective adenoviral vector containing the cDNA encoding for human IL10 (AD-h-IL10) was used in neuropathic pain models including CCI, SIN, and HIV gp120-induced neuropathic pain. 72 Intrathecal AD-h-IL10 elevated lumbosacral cerebrospinal fluid (CSF) levels of human IL-10, reversing thermal hyperalgesia and mechanical allodynia for more than 1 week in CCI-induced pain state. 72 Intrathecal AD-h-IL10 was given 5 days before acute sciatic inflammatory neuropathy, preventing SIN-induced mechanical allodynia. 72 Adenoviral IL-10 attenuates chronic SIN-induced territorial, extra-territorial and mirror-image mechanical allodynia for more than 1 week. 72 Pretreatment with intrathecal AD-h-IL10 for 5 days prevented acute gp120-induced mechanical allodynia for more than 100 min. 72 AD-h-IL10 lowered CSF levels of IL-1 protein. 72 Johnston et al. 6 reported that pretreatment with intrathecal adenovirus expressing interleukin-10 (ADIL-10) for 5 days potentiated acute morphine analgesia on day 1 and attenuated the development of analgesic tolerance apparent on day 5, and reduced chronic morphine-induced thermal hyperalgesia and tactile allodynia for more than 5 days.
Lentivirus vector
Lentivirus vector (LV) is derived from HIV. It contains a single-stranded RNA genome with ~ 8-kB. 7 Upon transduction, it is reversely transcribed into DNA in the host cell’s cytoplasm. 103 It integrates into the chromosome of host cells, 7 therefore, long-term expression of the transgene can be achieved. 104 LV vectors are capable of efficiently transducing a wide variety of both dividing and non-dividing cells. 103 Park et al. 73 found that local transgene expression of IL-10 by delivery of lentiviral vectors in spinal cord tissues lasted for a month, decreased mechanical and thermal hypersensitivities for more than 12 weeks and downregulated the levels of Sensory Perception of Pain-associated genes such as TNFα, transient receptor potential ankyrin 1 (Trpa1) and vanilloid 1 after spinal cord injury. He et al. 105 evaluated the effect of a lentivirus delivering human IL-10 gene administered intrathecally on allodynia and hyperalgesia for more than 4 weeks in CCI rat model of neuropathic pain, indicating that intrathecal hIL-10 reversed enhanced pain states, and inhibited activation of TNFα, IL-1β, IL-6, and HMGB1-RAGE pathway in the CCI rat model.
While viral vector gene therapy represents a very promising strategy for the treatment of chronic pain, current major concerns in the clinic are related to the safety of viral vectors including toxicity, immunogenicity, and oncogenesis from insertional mutagenesis. 64 Although gene therapy offers many advantages for treatment of chronic pain in preclinical studies, it may not be appropriate for all patients. We have to consider the balance between homeostasis versus immunocompromise. For example, IL-10 over-expression can inhibit a sufficient pro-inflammatory response to pathogens, likely effectively enabling the pathogens to escape immune control. 12
Although a growing body of preclinical evidence suggests that viral vectors are promising therapeutic tools for managing chronic pain, the number of viral vectors that have entered clinical trials remains small. A nonreplicating HSV-based vector expressing enkephalin in patients with cancer pain for phase 1 and 2 trial was carried out for safety evaluation (NCT00804076). 70 AAV encoding an IL-1 receptor antagonist has been used for Phase 1 trial in patients with osteoarthritis pain (ClinicalTrials.gov NCT02790723 NCT05835895).106–108 In other phase 1/2 trials (NCT05454566 and NCT04875754) for alleviating osteoarthritis pain, a recombinant AAV engineered to express a truncated form of the human transcription factor NKX3.2 was used; patients with osteoarthritis pain received a single intra-articular injection of the vectors, indicating that six of eight treated subjects had reduced knee pain. 109 Adenovirus vectors expressing IL-1Rα by intra-articular injection has been initiating the knee osteoarthritis pain in phase I clinical trial (NCT04119687, NCT06884865).109,110 In a cohort study of phase I trial (NCT03282656), a single infusion of autologous bone marrow derived CD34+ HSC cells transduced with the lentiviral vector containing a short-hairpin RNA targeting BCL11A was carried out for patients with severe painful sickle cell disease. 111 Although these early-phase clinical trials initial clinical translation of viral vectors-based therapies for pain, further research is required to refine safety and efficacy.
Conclusion
In the past three decades, the development of viral vectors for the treatment of nervous system disorders has followed a complex track.7,14,112 The success of gene therapy is dependent on effective gene delivery, in which many factors such as, level and duration of transgene expression, cellular specificity, and safety affect target gene expression. In the current review, we searched for original research articles that utilized viral vectors expressing IL-10, including HSV, AV, AAV, and LV for the treatment of chronic pain, indicating that viral vector mediated gene therapy for chronic pain will be a promising approach.7,70 However, challenges such as immune responses to viral vectors and variability in gene expression levels persist. Overall, viral vector gene therapy targeting IL-10 represents a very promising strategy for the treatment of chronic pain. By addressing the underlying neuroinflammatory mechanisms, this approach has the potential to provide long-lasting relief and improve patient outcomes.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: We thank the support from the Department of Anesthesiology, University of Miami, FL. This work was partially supported by grants from the NIH R01NS066792 (SH), R01DA34749 (SH), R01DA047089 (SH), R01DA047157 (SH), 5I01BX005114 (SH), and by JSPS KAKENHI Grant JP23K08397, JP22K09016, and JP21K08940.
