Abstract
Background:
Cancer-induced bone pain (CIBP) is a debilitating complication with few effective treatments. Microglial activation contributes to the progression of CIBP. 2-deoxy-D-glucose (2-DG), a glycolytic inhibitor, could inhibit microglial activation. Its effect on CIBP remains unclear. This study aims to investigate the role of 2-DG in CIBP mice and underlying mechanisms.
Methods:
In this research, we established a CIBP mouse model by injecting Lewis lung cancer (LLC) cells into the bone marrow of the femur. Relevant pain behaviors were assessed by measuring the paw withdrawal threshold and spontaneous hind limb lifting. Additionally, the glycolysis inhibitor 2-DG was intrathecally administered to treat CIBP in mice. Western blotting and immunofluorescence techniques were employed to analyze microglial activation and M1/M2 phenotype markers in the spinal cord.
Results:
Our findings demonstrated significant microglial activation and polarization toward the M1 phenotype in the spinal cord of CIBP mice. Intrathecal administration of 2-DG effectively alleviated pain-related behaviors in CIBP mice. Furthermore, this treatment suppressed microglial activation and M1 polarization, while significantly restoring levels of the M2 phenotype. Additionally, 2-DG attenuated the production of pro-inflammatory factors such as tumor necrosis factor-α (TNF-α), interleukin-1β (IL-1β), and interleukin-6 (IL-6), while boosting the secretion of the anti-inflammatory factor (IL-10) in the spinal cord of CIBP mice. Notably, 2-DG effectively suppresses microglia activation and M1 polarization in LPS + IFN-γ-induced BV-2 cells by downregulating CD86, iNOS expression, TNF-α, IL-1β, IL-6 levels while upregulating Arg-1, CD206 expression and IL-10 level.
Conclusion:
These results suggest that 2-DG ameliorates mechanical allodynia, spontaneous pain and neuroinflammation in the spinal cord of CIBP mice by promoting the transition from the M1 phenotype to the M2 phenotype. This study may provide a novel strategy for the treatment of CIBP.
Introduction
Cancer-induced bone pain (CIBP) is a common and debilitating complication in patients with primary or metastatic bone cancer such as breast, lung, prostate, and bladder cancer.1,2 Approximately 40% of early or intermediate-stage cancer patients and 90% of those with advanced cancer experience moderate to severe pain that greatly impacts their quality of life. 3 Unfortunately, up to 70% of patients with CIBP do not obtain adequate pain relief. 3 Clinically available treatment options for CIBP, including nonsteroidal anti-inflammatory drugs, palliative radiation, bisphosphonates, denosumab, and opioids, are limited by their inability to adequately alleviate pain or side effects.4,5 Currently, CIBP treatment remains a major clinical challenge because the pathophysiological mechanisms are poorly understood. 6 Hence, uncovering new mechanisms that underpin CIBP is critical for the development of novel therapeutic strategies.
Microglia, as integral components of the central nervous system, play a pivotal role in neurological disorders. 7 In general, microglia remain in a resting state, and their overall phenotypes or functional states are regulated by the nature of the inflammatory microenvironment. 8 Microglia can polarize into two phenotypes: the classically activated pro-inflammatory M1 phenotype or the alternatively activated anti-inflammatory M2 phenotype. 9 The M1 microglial phenotype is characterized by the secretion of pro-inflammatory cytokines, such as tumor necrosis factor-alpha (TNF-α), interleukin-1 beta (IL-1β), and interleukin-6 (IL-6), along with increased levels of oxidative metabolites like iNOS and the expression of cell surface markers CD86 and CD68. Conversely, the M2 phenotype microglia release anti-inflammatory mediators including IL-10, transforming growth factor (TGF)-β, arginase 1 (Arg1), and induce the expression of the mannose receptor (CD206). 10 To exert their immune functions, microglia respond to various environmental stimuli and rapidly adopt different phenotypes, which requires unique metabolism and bioenergetics for their dynamic functional states. 11 Emerging evidence indicates that microglial polarization toward the M1 phenotype is often accompanied by a metabolic transition from oxidative phosphorylation to aerobic glycolysis. 12 The M1 microglia exhibit increased glycolytic activity, whereas the M2 microglia tend to primarily depend on oxidative phosphorylation.
One such glycolytic inhibitor is 2-deoxy-D-glucose (2-DG), which is a synthetic glucose analog. It has been used as a potential clinical agent in various conditions, including cancer, viral infection, aging-related morbidity, and epilepsy.13,14 Clinical studies demonstrate that 2-DG is safe and relatively non-toxic in animals and humans. 15 Cheng et al. 12 found that 2-DG abolishes LPS-induced microglial activation. Recent studies have suggested that promoting microglial polarization from the M1 toward the M2 phenotype could be a promising therapeutic approach for CIBP and neuropathic pain.16–20 However, the specific molecular mechanisms of 2-DG treatment in CIBP have not been elucidated. In this study, we hypothesize that 2-DG modulates microglial polarization to alleviate CIBP.
Material and methods
Animals
Male C57BL/6 mice aged 6 to 8 weeks (20–25 g) were obtained from Tongji Medical College, Huazhong University of Science and Technology, Wuhan, China. All mice were housed in a temperature- and humidity-controlled environment with a 12 h light/dark cycle and had access to adequate food and clean water ad libitum. All experimental procedures were carried out following the approval of the ethics committee of the Animal Care and Use Committee of Huazhong University of Science and Technology. The animal ethics approval number for this study is TJH-202307027. All experiments complied with the Animal Research Reporting of in Vivo Experiments (ARRIVE) Guidelines.
Experimental design
Experiment 1: Pain behaviors of CIBP models were evaluated at 3, 7, 14, and 21 days post-surgery. Mice were randomized into the sham group (sacrificed 21 days after PBS injection), CIBP 7-day group (sacrificed 7 days post-surgery), CIBP 14-day group (sacrificed 14 days post-surgery), and CIBP 21-day group (sacrificed 21 days post-surgery). Additionally, paw withdrawal thresholds (PWT) and the number of Spontaneous lifting were examined. Subsequently, the lumbar spinal cord enlargements (L4-L5) were collected accordingly for western blotting and immunofluorescence staining (Figure 1(a)).
Experiment 2: To determine whether a single dose of 2-DG could relieve established CIBP, the drug (vehicle, 2-DG 10, 100, 200 μg, i.t.) was given on day 14 after LLC inoculation. The behavioral test was conducted before drug injection and 0.5, 1, 2, 3, 4, 5 and 6 h after injection (Figure 1(b)).
Experiment 3: To assess the efficacy of repetitive treatment with 2-DG in reversing established CIBP, the drug (vehicle, 2-DG 10, 100, 200 μg, i.t.) was administered once daily from day 14 to day 18. The behavioral evaluation was conducted at day 13 and 2 h post-drug injection each day (Figure 1(c)).

Schematic diagram of the experimental design: (a) changes in pain-related behaviors after LLC inoculation in mice. The spinal cord was collected at indicative days, (b) to determine whether a single administration of 2-DG can attenuate established CIBP in mice, 2-DG at doses of 10, 100, and 200 μg was intrathecally injected on day 14 following LLC inoculation, and (c) to clarify if continuous administration of 2-DG, can attenuate established CIBP in mice, 2-DG (10, 100, 200 μg) or vehicle was intrathecally administered once a day from day 14 for 5 days in a row. All behavioral tests were experienced 2 h after 2-DG administration or vehicle. The spinal cord was removed on day 18 for WB and IF after repeated 2-DG administration and behavioral tests.
Cell culture and treatment
Murine Lewis lung cancer (LLC) cells were resuscitated and seeded in a culture flask, then cultured in Dulbecco’s Modified Eagle Medium (DMEM) (Servicebio, Wuhan, China), supplemented with 10% fetal bovine serum, 100 U/mL penicillin, and 100 μg/mL streptomycin (Servicebio, Wuhan, China). Following a 5-min exposure to 0.25% trypsin in a cell incubator at 37°C, the adherent cells were detached from the culture flask. The detached cells were then neutralized with an equal volume of DMEM and centrifuged for 5 min at 1000 rpm. The resulting cell pellet was washed twice using 5 mL of phosphate-buffered saline (PBS) (Servicebio, Wuhan, China) and then resuspended in a minimal volume of PBS. Finally, the LLC cells were diluted to the desired concentration of 1 × 105 cells per 5 μL in PBS, utilizing a cell counting plate. The cell suspension was kept on ice until injection.
BV-2 mouse microglial cells were cultured in Dulbecco’s Modified Eagle’s Medium (Gibco, USA), supplemented with 10% fetal bovine serum and 1% penicillin/streptomycin, and then incubated at 37°C in a 5% CO2. Following this, the BV-2 cells were seeded into 6-well plates and subsequently treated with LPS (100 ng/mL, Solarbio, China) and interferon-gamma (IFN-γ) (20 ng/mL, Sino Biological, China) for 24 h to induce M1 polarization (M1 group).
Establishment of the CIBP model
The surgical procedure was the same as mentioned previously. 21 In brief, C57BL/6 mice were anesthetized with an intraperitoneal injection of sodium pentobarbital. We then carefully shaved the fur on the left leg of each mouse and disinfected the exposed skin with an iodophor solution. A small incision was made over the anteromedial surface of the knee joint to expose the patellar ligament. Using a 25-gauge needle, we drilled into the distal femur to access the medullary cavity. Subsequently, 5 μL of LLC cells were slowly injected into the femoral cavity using the same gauge needle. For sham-operated mice, an equal volume of PBS without LLC cells was injected. To prevent the leakage of cancer cells, the needle was kept in place for 5 min after injection. The injection hole was sealed with bone wax after the needle was removed. Following surgery, mice were placed in a temperature-controlled recovery unit until they fully regained consciousness and then returned to their respective cages.
Bone X-ray and bone histology staining on mouse femurs
Roentgenography of the ipsilateral femur was performed on the 21st day after inoculation of LLC cells. Mice were exposed to an X-ray source. To assess the extent of tumor invasion and bone destruction, the ipsilateral femur was detected by hematoxylin and eosin (HE) staining. Mice were deeply anesthetized on the 21st day following surgery. Subsequently, the mice were perfused intracardially with ice-cold PBS and 4% paraformaldehyde (PFA). The femora were then extracted and post-fixed for 48 h in the same fixative at 4°C. Following demineralization in EDTA (10%) for 10 days, the femur samples underwent dehydration in an ascending ethanol gradient (30%–100%), were embedded in paraffin, and then cut into 8 μm thick sections. All images were captured using a BX53 microscope (Olympus, Tokyo, Japan).
Behavioral tests
All behavioral tests were conducted between 9:00 a.m. and 5:00 p.m. in a temperature- and humidity-controlled separate room. Prior to testing, mice were habituated to the test conditions for 30 min. The behavioral tests were conducted following the blinding principle.
Mechanical allodynia was assessed by measurement of the paw withdrawal threshold (PWT) using von Frey filaments via the up-down method as previously described.18,22,23 Prior to testing, each mouse was acclimated for 30 min in a clear plastic compartment placed on an elevated wire grid. For testing, a series of von Frey filaments (0.04, 0.07, 0.16, 0.4, 0.6, 1.0, 1.4, 2 g) were applied to the central plantar surface of the left hind paw. The initial bending force was 0.4 g. Each filament was applied perpendicularly for approximately 6 s, with a 15-s inter-stimulus interval. A minimum 5-min interval was maintained between different sets of von Frey filament applications. A positive response was defined as brisk lifting, shaking, or flinching of the paw. The final PWT value was recorded as the lowest force that elicited three or more positive responses out of a total of five applications. The behavioral tests were conducted following the blinding principle.
Spontaneous pain in the hind limbs was assessed by counting the number of spontaneous lifting in 10 min, as previously described.21,24 Briefly, mice were placed in chambers on an elevated glass plate. A spontaneous hind limb lift of the left hind paw was defined as the rapid lifting of the entire hind limb, beginning with hip flexion and including dorsiflexion of the toes, and was not associated with walking or grooming. Each instance of this behavior was considered one lifting.
Drug administration
2-Deoxy-D-glucose (2-DG), purchased from MedChemExpress (HY-13966, USA), was dissolved in sterile saline. To assess the direct analgesic effect of 2-DG on the spinal cord of mice, intrathecal (i.t.) injection was employed. For the administration of the drug via intrathecal injection, the procedure began with shaving and disinfecting the skin over the lumbar region (lower back) of the mice. Subsequently, we used a Hamilton micro-syringe to perform a percutaneous injection directly into the lumbar intrathecal space in conscious mice, as described previously. 25 To evaluate the analgesic effects of 2-DG via intrathecal administration in the CIBP model, mice received a single intrathecal injection of varying doses of 2-DG (10, 100, and 200 μg) on day 14 after inoculation with LLC cells. Pain behaviors were assessed at multiple time points: baseline (one day before surgery), and then at 0, 0.5, 1, 2, 3, 4, 5, and 6 h following the 2-DG injection. To evaluate if repeated administration of 2-DG could alleviate established CIBP, the mice in the relevant groups received a daily intrathecal injection of 2-DG at a dose of 200 μg for five consecutive days from day 14 to day 18 after surgery. For each administration, whether of the vehicle or drugs, a volume of 5 μL was carefully and steadily injected using a 10 μL micro-syringe. The dosages of all medications administered were determined based on previous in-house experiments. Furthermore, a previous study 12 reported that 2-DG was dissolved in sterile saline at a concentration of 1000 μM for in vitro experiments, after which 2-DG at the same concentration was incubated with BV-2 microglial cells stimulated by LPS and IFN-γ.
Western blotting
Mice were deeply anesthetized using an overdose of sodium pentobarbital and sacrificed. Subsequently, L4-5 spinal cords were quickly harvested and homogenized in a mixture of radio-immunoprecipitation assay (RIPA) lysis buffer containing a cocktail of protease inhibitors and protein inhibitors. The homogenate was then centrifuged at 4ºC at 13,600 rpm for 10 min to collect the supernatants. BV-2 microglial cells were lysed on ice for 30 min in RIPA lysis buffer. Protein concentrations in the supernatants were quantified using a BCA Protein Assay Kit (Boster, Wuhan, China). Protein samples (40 μg/lane) were separated by 10% or 12.5% SDS-PAGE gels and transferred onto 0.45 μm polyvinylidene fluoride (PVDF) membranes (Millipore, Billerica, MA, USA). After having been blocked with 5% non-fat milk at room temperature (RT) for 2 h, the PVDF membranes were incubated with primary antibodies overnight at 4ºC. The following primary antibodies were used: rabbit anti-Iba1 antibody (1:500; A19776; Abclonal, Wuhan, China), rabbit anti-CD86 antibody (1:1000; A19026; Abclonal, Wuhan, China), rabbit anti-iNOS antibody (1:1000; ER1706-89; Huabio, Hangzhou, China), rabbit anti-CD206 antibody (1:1000; A8301; Abclonal, Wuhan, China), rabbit anti-ARG1 antibody (1:1000; ET1605-8; Huabio, Hangzhou, China), rabbit anti-NF-κB p65 antibody (1:2000; A22331; Abclonal, Wuhan, China), anti-Phospho-NF-κB antibody (1:1000; AP0123; Abcam, Cambridge, UK), rabbit anti-interleukin-1β (1:500; 511369; Zenbio, Chengdu, China), rabbit anti-IL-10 (1:500; A12255; Abclonal, Wuhan, China), rabbit anti-TNF-α (1:500; R1203-1; Huabio, Hangzhou, China), rabbit anti-IL-6 (1:500; R1412-2; Huabio, Hangzhou, China), rabbit anti-beta-actin antibody (β-actin; 1:50000; AC206; Abclonal, Wuhan, China). The membranes were further treated with HRP Goat Anti-Rabbit IgG (H + L) (1:5000; AS014; Abclonal, Wuhan, China) for 1 h at RT. Immunoblots were detected using Super-Lumia ECL reagents (Abbkine, Cat# K22030, Wuhan, China) in the image analysis system (ChemiDoc XRS1, Hercules, CA, USA). The images generated by image lab software (BioRad Laboratories, Hercules, CA, USA) were analyzed by using Image J (National Institutes of Health, MD, USA). All results were normalized to the loading control β-actin and expressed as a fold of the control.
Immunofluorescence staining
Mice were deeply anesthetized via intraperitoneal injection with sodium pentobarbital, and then perfused intracardially with 50 mL cold of PBS, followed by 50 mL 4% ice-cold paraformaldehyde (PFA). Spinal cord specimens were removed and fixed in 4% PFA overnight at 4°C and then dehydrated in 20% and 30% sucrose solution at 4ºC for 24 h, respectively. The spinal cord sections of L4-5 were transversely cut into slices using a cryostat (CM1900, Leica, Wetzlar, Germany). The sections were then infiltrated with 0.3% Triton X-100 for 15 min and blocked with 5% donkey serum for 1.5 h at RT. Subsequently, the sections were incubated overnight at 4ºC with the following primary antibodies: goat anti-Iba1 antibody (ab5076, 1:200; Abcam, Cambridge, UK). For double staining, corresponding primary antibodies of goat anti-Iba1 antibody (ab5076, 1:200; Abcam, Cambridge, UK), rabbit anti-iNOS antibody (1:200, 18985-1-AP, Proteintech, Wuhan, China), rabbit anti-CD206 antibody (ab300621, 1:200; Abcam, Cambridge, UK) respectively, followed by a mixture of corresponding secondary antibodies. After being washed in PBS for approximately 30 min, the sections were incubated with corresponding secondary antibodies for 2 h at RT. The secondary antibodies were as follows: Rhodamine (TRITC)-conjugated Donkey Anti-Goat IgG (H + L) (1:200; SA00007-3; Proteintech, Wuhan, China), FITC Donkey Anti-Rabbit IgG (H + L) (1:300; AS042; Abclonal, Wuhan, China). Finally, the sections were covered with a 50% glycerol solution and then carefully placed under a cover slip, preparing them for observation under a microscope. The images were captured using a fluorescence microscope (BX53, Olympus, Tokyo, Japan), and the immunofluorescence intensity was quantified using Image J version 2.1.0 software (National Institutes of Health, MD, USA).
Statistical analysis
The data was presented as mean ± SEM. GraphPad Prism version 9.0 (Graph Pad Software, San Diego, CA, USA) was used to conduct all statistical analyses. Behavioral results were analyzed with two-way ANOVA, followed by Tukey post-tests. The protein expressions and fluorescence intensity were assessed with one-way ANOVA, followed by Tukey post hoc test. p values less than 0.05 were considered statistical significance.
Results
Intramedullary inoculation of LLC cells induced mechanical allodynia and spontaneous pain in mice
To assess if intramedullary inoculation of LLC cells could induce CIBP, X-rays and HE staining were used to verify the successful implantation of cancer cells. Femur HE staining and X-ray images (Figure 2(a) and (b)) of the femur taken on day 21 post-surgery indicated the presence of bone destruction and infiltration of tumor cells in the CIBP group.
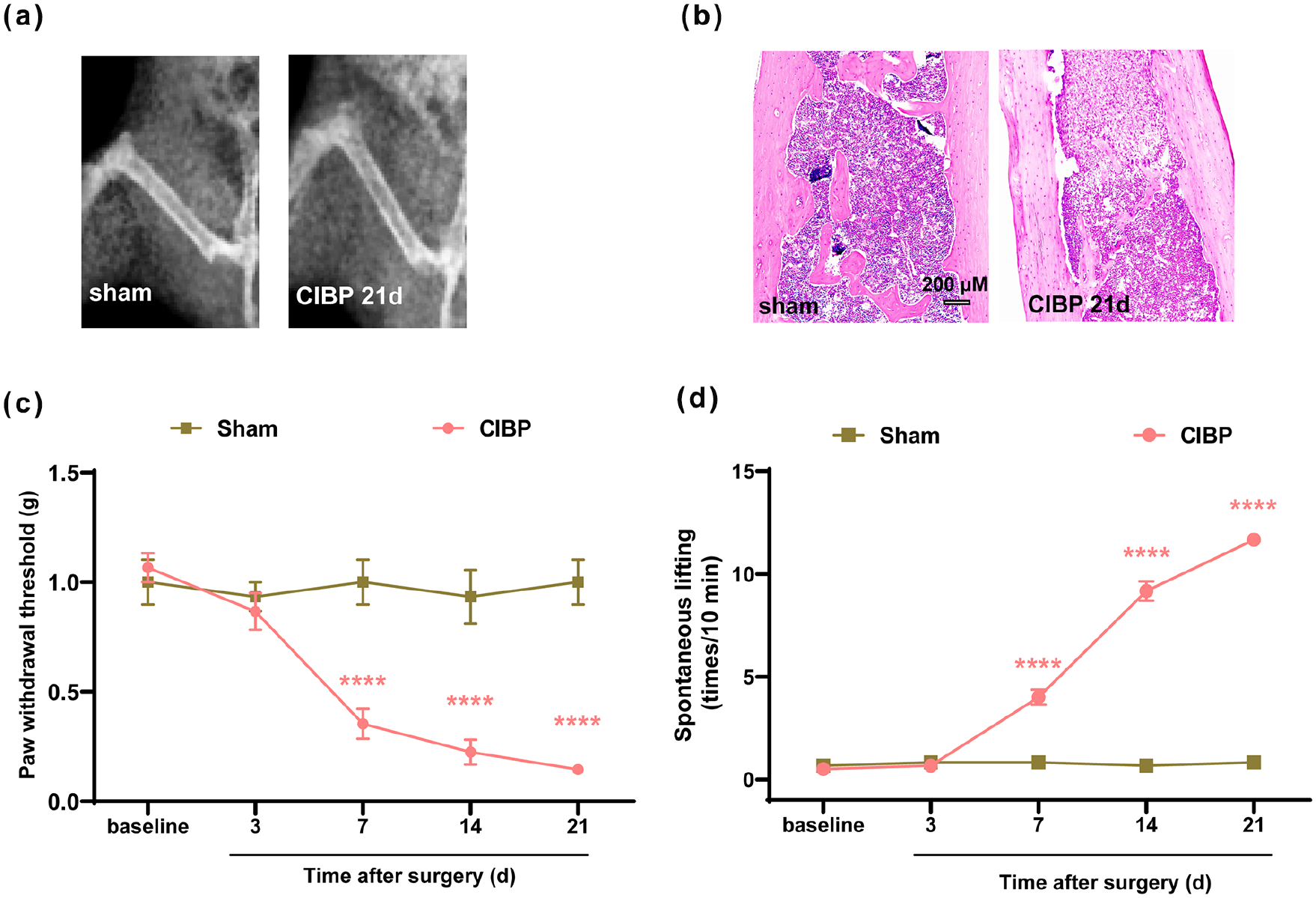
Assessment of pain behaviors over time: (a) representative radiographs of the ipsilateral femur from sham and CIBP mice on day 21 after inoculation of PBS solution or LLC cells, (b) representative images of HE staining of femur sections from sham and CIBP mice, (c) the PWT of the left hind limb in sham mice and CIBP mice in response to von Frey filaments at the indicated time points, and (d) spontaneous lifting of the left hind limb at the indicated time points.
We measured mechanical allodynia through PWT and spontaneous hind limb lifting times to depict pain behaviors over time. PWT and spontaneous hind limb lifting times was evaluated 1 day before LLC cells inoculation (baseline) and on days 3, 7, 14, and 21 after bone cancer induction. Results showed a significant decrease in PWT in the CIBP group from day 7 to day 21 (Figure 2(c): group: F (1, 10) = 25.66, p = 0.0005; time: F (2.671, 26.71) = 20.30, p < 0.0001; interactions: F (4, 40) = 20.53, p < 0.0001) compared to that of the sham group. A statistically significant increase in spontaneous hind limb lifting times was observed from day 7 to day 21 (Figure 2(d): group: F (1, 10) = 645.6, p < 0.0001; time: F (4, 40) = 141.5, p < 0.0001; interactions: F (4, 40) = 140.9, p < 0.0001) after bone cancer induction.
Microglia were markedly activated in the spinal cord of CIBP mice
As shown in Figure 3(a), it is evident that Iba1 protein expression in the spinal cord of CIBP mice exhibited a significant increase from day 7 to day 21 (F (4, 25) = 14.50, p < 0.0001) after bone cancer induction, compared to that of the sham group. Furthermore, the immunofluorescence analysis of the spinal cord indicated a heightened intensity of Iba1 in the ipsilateral spinal dorsal horn (SDH) from day 7 to day 21 (Figure 3(c): F (4, 10) = 60.10, p < 0.0001), as depicted in Figure 3(d). These findings provided evidence that microglia were markedly activated in the spinal cord of CIBP mice.

Microglia is activated in the spinal cord of CIBP mice: (a) representative Western blot results reveal a marked upregulation of spinal Iba1 levels on days 3, 7, 14, and 21 after LLC cells inoculation, (b) and (c) representative immunofluorescence images show activated microglia in the spinal dorsal horn at the indicated time points. The histogram shows the immunofluorescence intensity analysis results.
Microglia polarized toward M1 polarization in the spinal cord of CIBP mice
To further investigate microglial polarization in the spinal cord of CIBP mice, we conducted Western blotting and immunofluorescence assays to assess the levels of M1 and M2 microglial markers. Our findings, as illustrated in Figure 4(a) to (d) (Figure 4(a): F (4, 25) = 9.486, p < 0.0001; Figure 4(b): F (4, 25) = 19.43, p < 0.0001; Figure 4(c): F (4, 25) = 16.25, p < 0.0001; Figure 4(d): F (4, 25) = 24.34, p < 0.0001) demonstrated a significant upregulation of M1 microglial markers (CD86, iNOS) in the spinal cord of CIBP mice from day 7 to day 21, when compared to that of the sham group. Conversely, the expression of M2 microglial markers (CD206, Arg-1) showed a marked decrease in the spinal cord of CIBP mice from day 7 to day 21, in comparison to that of the sham group.

The microglia in the spinal cord of CIBP mice polarized toward the M1 phenotype: (a)–(d) representative Western blotting bands and quantitative analysis show the expression levels of CD86, iNOS, CD206, and Arg1 protein in the Sham group and CIBP group on days 3, 7, 14, and 21 after LLC cells inoculation. β-actin was used as an internal control.
Intrathecal administration of 2-DG attenuated mechanical allodynia and spontaneous pain in CIBP mice
To assess whether a single dose of intrathecally administered 2-DG can attenuate existing mechanical allodynia, mice with CIBP were given 2-DG intrathecally (10, 100, and 200 μg) on day 14. Behavioral tests measuring paw withdrawal threshold (PWT) were performed before 2-DG administration and at 0.5, 1, 2, 3, 4, 5, and 6 h after administration. As shown in Figure 5(a) (group: F (2.135, 10.67) = 110.8, p = 0.0005; time: F (3.406, 17.03) = 59.18, p < 0.0001; interactions: F (4.471, 22.35) = 8.230, p = 0.0002), no significant change in PWT was observed with the 10 μg dose of 2-DG, whereas the 100 and 200 μg doses significantly increased PWT, reaching a peak at 2 h and lasting for approximately 3 h compared with the CIBP + Vehicle group. The numbers of spontaneous lifting in the CIBP + 2-DG (100 and 200 μg) were markedly decreased compared with those of the CIBP + Vehicle group (Figure 5(b): group: F (5, 30) = 662.5, p < 0.0001; time: F (5.600, 168.0) = 239.7, p < 0.0001; interactions: F (40, 240) = 28.32, p < 0.0001). Besides, there were significant differences between the 100 μg and 200 μg 2-DG, suggesting that i.t. 2-DG could reverse mechanical allodynia and spontaneous pain in a dose-dependent manner after CIBP (*p < 0.05, **p < 0.01, and ***p < 0.001 compared with the CIBP + Vehicle group, #p < 0.05, ##p < 0.01, ###p < 0.001 compared with the group treated with 2-DG (100 μg)).

Analgesic effect of intrathecal 2-DG on mechanical allodynia and spontaneous pain in CIBP mice: (a) a single dose of 2-DG (100 and 200 μg, i.t.) markedly increased the PWT in CIBP mice, starting at 1 h and lasting for at least 3 h, (b) a single dose of 2-DG (100 and 200 μg, i.t.) markedly decreased spontaneous lifting of the left hind limb in CIBP mice, starting at 1 h and lasting for at least 3 h, (c) and (d) the PWT and spontaneous lifting of the left hind limb was significantly decreased from day 14 to day 21 in 2-DG-treated CIBP mice compared with vehicle-treated CIBP mice..
To evaluate if repeated injections of 2-DG can alleviate mechanical allodynia and spontaneous pain in CIBP mice, daily treatments were administered for 5 days starting from day 14 to day 18. Behavioral tests assessing PWT and spontaneous lifting times were conducted at 2 h after each 2-DG injection. As shown in Figure 5(c) (group: F (2.249, 11.25) = 54.16, p = 0.0005; time: F (2.507, 12.54) = 36.44, p < 0.0001; interactions: F (3.689, 18.44) = 7.085, p = 0.0015; n = 6/group) and Figure 5(d) (group: F (2.249, 11.25) = 54.16, p = 0.0005; time: F (2.507, 12.54) = 36.44, p < 0.0001; interactions: F (3.689, 18.44) = 7.085, p = 0.0015), PWT and spontaneous hind limb lifting results revealed that spontaneous pain were significantly alleviated from day 14 or day 15 to day 18 after bone cancer induction in CIBP group when compared with that of the CIBP + Vehicle group. Repeated injections of 2-DG (100 and 200 μg, intrathecally) significantly improved pain behaviors in a dose-dependent manner in CIBP mice (**p < 0.01, ***p < 0.001, ***p < 0.001, ****p < 0.0001 versus CIBP + Vehicle group, #p < 0.05, ##p < 0.01, ###p < 0.001 versus the group treated with 2-DG (100 μg). n = 6 per group.
Continuous administration of 2-DG reduced microglial activation in the spinal cord of CIBP mice
At the protein (Figure 6(c): F (3, 20) = 12.36, p < 0.0001) and immunofluorescence intensity levels (Figure 6(b): F (3, 8) = 12.49, p = 0.0022), our results revealed that Iba1 expression levels were significantly upregulated in the CIBP + Vehicle group mice when compared with those of the sham group mice, and it exhibited a marked decrease after 2-DG treatment. These results indicate that 2-DG markedly inhibits CIBP-induced microglial activation.

Continuous administration of 2-DG inhibited microglial activation in the spinal cord of CIBP mice: (a) and (b) representative immunofluorescence images show that intrathecal 2-DG inhibited microglial activation in the spinal dorsal horn of CIBP mice. The histogram shows the immunofluorescence intensity analysis results, and (c) representative Western blotting bands and quantitative analysis show that 2-DG treatment reduced the expression levels of Iba1 in the spinal cord of CIBP mice.
Continuous administration of 2-DG regulated microglia toward M2 polarization in the spinal cord of CIBP mice
To further investigate the underlying mechanisms of the effect of 2-DG on M1/M2 microglial polarization in CIBP mice, we conducted Western blotting experiments and immunofluorescence staining methods to analyze the levels of M1/M2 markers in the spinal cord of each group. We observed an increase in M1 markers (iNOS, CD86) and a decrease in M2 markers (Arg-1, CD206) in the CIBP + Vehicle group compared to those of the sham group (Figure 7(a): F (3, 20) = 11.71, p < 0.0001; Figure 7(b): F (3, 20) = 13.04, p < 0.0001; Figure 7(c): F (3, 20) = 10.92, p = 0.0002; Figure 7(d): F (3, 20) = 7.793, p = 0.0012). Administration of intrathecal 2-DG injections was found to inhibit the M1 phenotype and promote M2 polarization. In response to the 2-DG treatment, the CIBP + 2-DG group exhibited significantly elevated levels of M2 markers Arg-1 and CD206 compared to the CIBP + Vehicle group. Immunofluorescence results (Figure 7(e) and (f)) demonstrated that iba1 co-localized with iNOS and CD206 in the spinal dorsal horn.

Continuous administration of 2-DG reversed the CIBP-induced increase in the M1 phenotype and the CIBP-induced decrease in the M2 phenotype: (a)–(d) representative Western blotting bands and quantitative analysis revealed that spinal M1 microglial markers (CD86, iNOS) were significantly upregulated in CIBP mice. Meanwhile, spinal M2 microglial markers (CD206, Arg-1) displayed an obvious decrease in CIBP mice. While, intrathecal 2-DG could reduce the levels of M1 microglial markers (CD86, iNOS) and increase those of M2 microglial markers (CD206, Arg-1) in the spinal cord in CIBP mice, and (e) representative immunofluorescence images show that Iba1 was colocalized with iNOS and CD206.
Continuous administration of 2-DG attenuated neuroinflammation in the spinal cord of CIBP mice
To evaluate the impact of 2-DG on spinal neuroinflammation in CIBP mice, we measured the levels of inflammation-related factors such as NF-κB, p-NF-κB, pro-inflammatory cytokines (TNF-α, IL-1β, IL-6), and the anti-inflammatory cytokine (IL-10) using Western blotting. NF-κB, as the upstream molecule of robust inflammatory cytokines, plays a key role in microglial activation. Similar to previous reports, 18 cancer cell inoculation did not alter the expression of total NF-κB (Figure 8(a): F (3, 20) = 1.738, p = 0.1914). The expression of the pro-inflammatory gene p-NF-κB p65 was also measured. The increased level of p-NF-κB p65 was reduced by 2-DG treatment compared to the CIBP + Vehicle group, as displayed in Figure 8(b) (F (3, 20) = 9.391, p = 0.0004). The levels of pro-inflammatory cytokines (TNF-α, IL-1β, IL-6) were increased in the CIBP + Vehicle group and reversed by 2-DG treatment. In contrast, the level of the anti-inflammatory cytokine (IL-10) was decreased in the CIBP + Vehicle group and restored by 2-DG treatment (Figure 8(c): F (3, 20) = 9.636, p = 0.0004; Figure 8(d): F (3, 20) = 12.06, p < 0.0001; Figure 8(e): F (3, 20) = 14.09, p < 0.0001; Figure 8(f): F (3, 20) = 7.470, p = 0.0015).
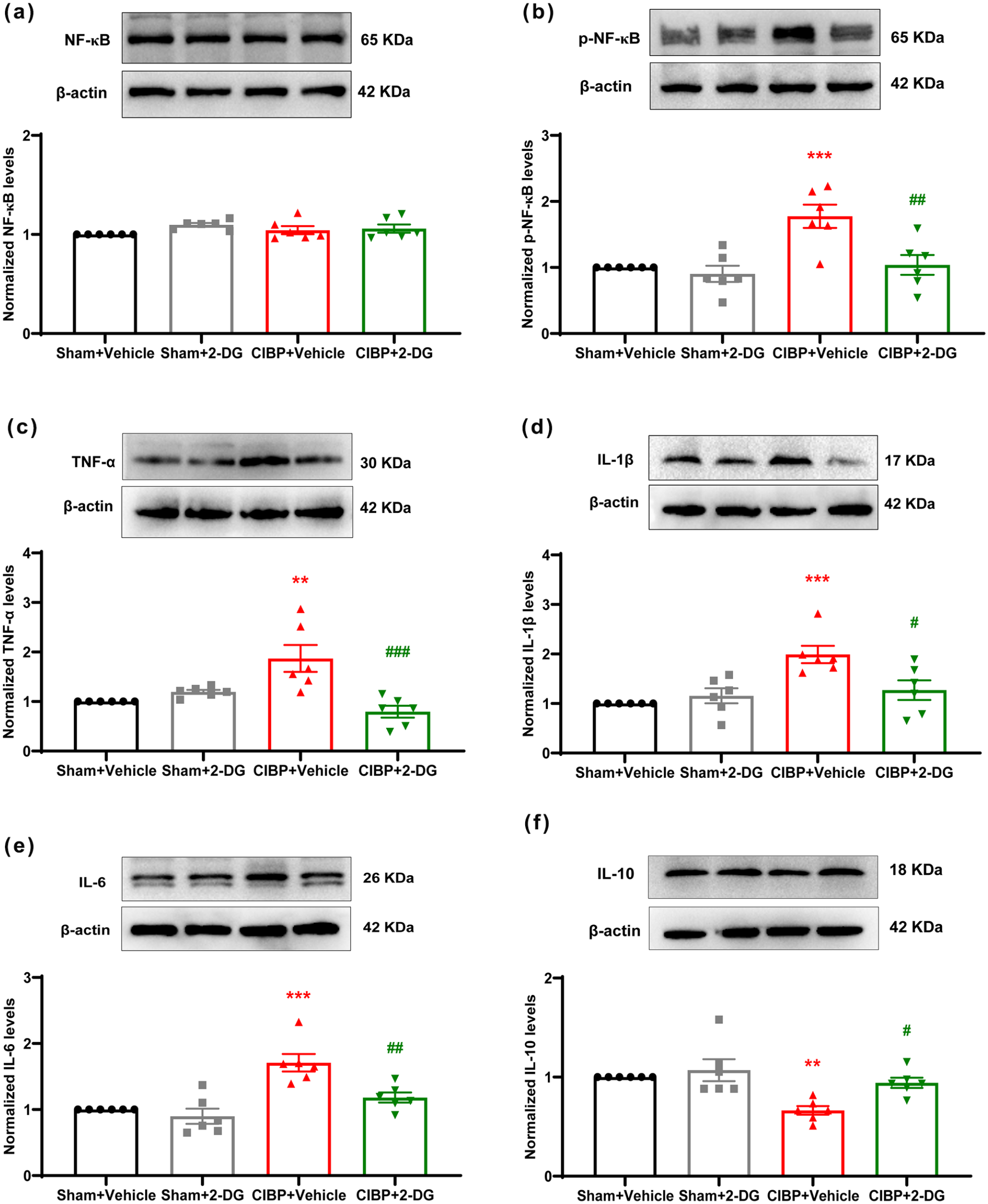
Continuous administration of 2-DG inhibited pro-inflammatory cytokines and improved anti-inflammatory cytokines in CIBP mice: (a)–(f) representative Western blotting bands and quantitative analysis revealed that levels of inflammation-related factors p-NF-κB, pro-inflammatory cytokines (TNF-α, IL-1β, IL-6) were inhibited and anti-inflammatory cytokine (IL-10) was increased by 2-DG in CIBP mice.
In vitro results indicated that 2-DG reduced M1 polarization and restored M2 polarization
In vitro results (Supplemental Figure S1A–C, G–H) demonstrated that 2-DG (1000 μM) downregulated M1 markers, such as CD86 and iNOS, as well as pro-inflammatory cytokines including p-NF-kB, TNF-α, IL-1β, and IL-6 in BV-2 cells treated with LPS and IFN-γ. Furthermore, 2-DG restored the expression of M2 markers, including Arg-1 and CD206, and increased the levels of the anti-inflammatory cytokine IL-10 (Supplemental Figure S1I–K), resulting in a shift in microglial polarization towards the anti-inflammatory M2 phenotype.
Discussion
Based on above results, we demonstrated that (1) microglial activation and polarization was involved in CIBP development; (2) 2-DG significantly alleviated pain behaviors in a mouse model of CIBP; (3) 2-DG administration reduced spinal microglial activation in CIBP mice. (4) 2-DG administration inhibited spinal microglial M1 polarization in CIBP mice. (5) 2-DG attenuated microglial M1 polarization in BV-2 cells. Generally, these data show that 2-DG alleviates pain behaviors in CIBP through inhibiting microglial activation and M1 polarization, as depicted in Figure 9. These findings suggest that 2-DG could be a novel therapeutic agent for treating CIBP.
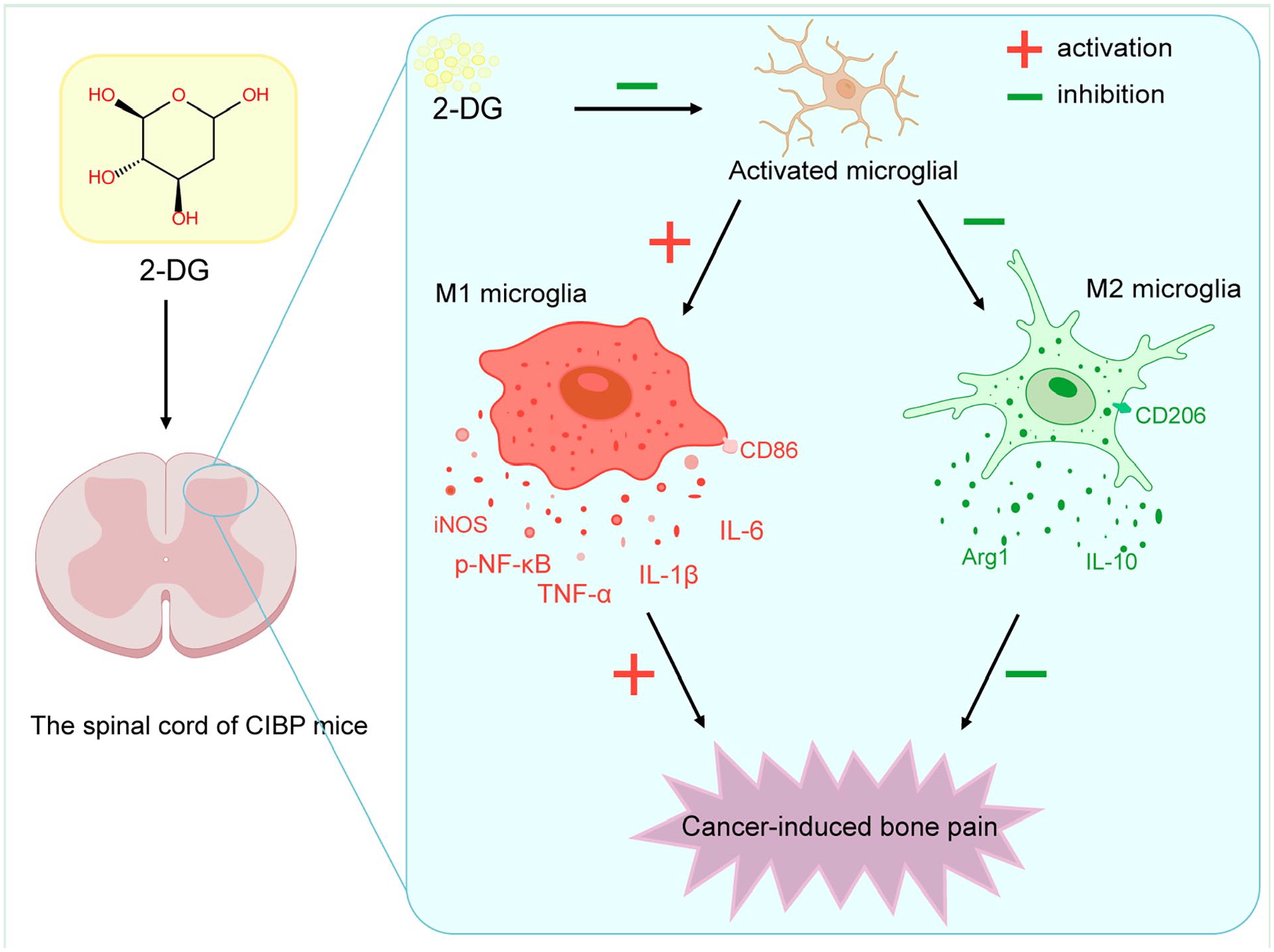
The schematic diagram illustrates that the potential mechanisms by which 2-DG treatment reduces spinal inflammation and alleviates cancer-induced bone pain. Intrathecal administration of 2-DG inhibits microglial polarization towards the M1 phenotype and suppresses neuroinflammation, thereby contributing to the relief of CIBP. −, inhibition; +, activation.
Cancer-induced bone pain is a complex pain condition that combines characteristics of neuropathic and inflammatory pain, resulting in a unique reorganization of neurochemicals in the spinal cord.5,26 Due to its significant impact on the quality of life for cancer patients with bone metastasis, there is a current medical necessity to develop better management strategies and more effective drug treatments for cancer-induced bone pain. Our findings are consistent with many studies, showing that CIBP can activate microglia cells in the spinal cord and induce their polarization to a pro-inflammatory phenotype.
The growing body of evidence indicates that microglial activation plays a crucial role in central sensitization, acting as an essential component of neuroinflammation.27–29 Spinal microglia play a crucial role in the development and progression of chronic pain.29,30 The polarization of microglia is a critical factor in CIBP. Notably, several studies suggest that modulating the polarization of microglia from the pro-inflammatory M1 phenotype to the anti-inflammatory M2 phenotype can be a feasible strategy for CIBP treatment.16–18 Huo et al. 16 found that the imbalanced polarization of microglia toward the M1 phenotype in the spinal cord may contribute to the development of bone cancer pain, while dehydrocorydaline helps alleviate bone cancer pain by shifting microglial polarization toward the M2 phenotype in the spinal cord. Wu et al. 17 reported that inhibition of the P2X7 receptor alleviates CIBP and promotes microglia polarization toward the M2 phenotype while suppressing the M1 phenotype both in vivo and in vitro. Ge et al. 18 demonstrated that shifting microglial polarization from the M1 phenotype to the M2 phenotype reveals a potential option for CIBP treatment. Moreover, our previous study has demonstrated that exogenous intrathecal administration of rDKK3 promotes the transformation of microglia from the M1 phenotype to the M2 phenotype to alleviate neuropathic pain in rats. 19 In vitro, crotalphine downregulated CD86 expression and enhanced CD206 expression in BV-2 cells treated with LPS, leading to a shift in microglial polarization towards the anti-inflammatory M2 phenotype. These results confirm the neuromodulatory role of crotalphine in contributing to its analgesic action. 31 In this study, we found that microglial polarization shift toward the M1 phenotype is thought to cause the development of CIBP, while 2-DG can mitigate the pain of bone cancer through re-shifting microglia polarization towards the M2 phenotype and inhibiting M1 polarization as reflected by augmented expression of M2 microglia marker CD206, Arg-1 and anti-inflammatory cytokine IL-10, in addition to lowered expressions of M1 microglia marker iNOS, CD86 and the proinflammatory cytokines TNF-α, IL-1β and IL-6. Interestingly, promoting microglial polarization to the anti-inflammatory M2 phenotype by 2-DG, suppressed microglia-mediated neuroinflammation and attenuated pain sensation in CIBP mice.
Recent studies have demonstrated the critical regulatory roles of the NF-κB signaling pathway and its downstream inflammatory factors (IL-6, IL-1β, and TNF-α) in the development of cancer pain. During the progression of cancer-induced bone pain (CIBP), we observed an excessive production of TNF-α, IL-1β, and IL-6, alongside a decreased expression of IL-10. To investigate whether NF-κB and its downstream inflammatory factors are involved in the analgesic effects of 2-DG on CIBP, we examined the impact of 2-DG on the expression of NF-κB, IL-1β, TNF-α, and IL-6 in the spinal cord of CIBP mice. Our findings revealed that intrathecal 2-DG treatment led to a reduction in the levels of proinflammatory cytokines (TNF-α, IL-1β, IL-6) while increasing the release of the anti-inflammatory factor IL-10. Thus, targeting neuroinflammation in the spinal cord may present a promising strategy for alleviating pain in CIBP mice.
The use of the glycolytic inhibitor 2-DG as a potential agent to target chemo-resistant hypoxic cancer cells began in the late 1990s. 21 2-DG is structurally similar to glucose but differs in the substitution of a hydrogen atom for a hydroxyl group at the second carbon position. It competes with glucose for uptake into cells. Once inside the cell, it gets phosphorylated to form 2-DG-6-phosphate, which cannot be further metabolized, leading to the inhibition of glycolysis. These effects have been found in studies to ameliorate neuroinflammation. Several studies have indicated that glycolysis inhibition exerts a protective effect on neuroinflammation. 11 2-DG was found to significantly mitigate dopaminergic cell death and microglial activation. 12 Baik et al. 32 showed that metabolic boosting with recombinant interferon-γ treatment reversed the defective glycolytic metabolism and inflammatory functions of microglia. In addition, in cancer cells, 2 – DG has been extensively studied for its potential anti-tumor effects. It can inhibit cancer cell proliferation by interfering with their high-rate glycolysis, which is a characteristic feature of many cancer cells (the Warburg effect).33–36 In immune cells, 2-DG can modulate the function of macrophages, lymphocytes, and dendritic cells.37,38 The current study was strategically designed as a proof-of-concept investigation to establish the fundamental therapeutic potential of 2-DG in CIBP management. Beyond its anti-tumor effects, emerging evidence suggests that 2-DG also modulates neuroinflammation by targeting microglial metabolism. Unlike existing modulators targeting specific receptors (e.g. P2X7), 2-DG uniquely reprograms microglial polarization through metabolic inhibition, offering a dual benefit of suppressing tumor growth and neuroinflammation. Mechanistic studies demonstrated that 2-DG significantly inhibited LPS-induced phosphorylation of mechanistic target of rapamycin (mTOR), an inhibitor of NF-κB kinase subunit beta (IKKβ), and NF-κB inhibitor alpha (IκB-α), degradation of IκBα, nuclear translocation of p65 subunit of NF-κB, and NF-κB transcriptional activity.11,12 In addition, 2-DG significantly inhibited LPS-induced acetylation of p65/RelA on lysine 310, which is mediated by NAD-dependent protein deacetylase sirtuin-1 (SIRT1) and is critical for NF-κB activation. 12 Inhibition of NAD+/SIRT1/p65 acetylation was involved in the anti-neuroinflammatory activity of the glycolytic inhibitor. Therefore, inhibition of glycolysis by 2-DG can ameliorate microglial activation-related neuroinflammatory diseases. 12 In view of its ability to inhibit microglial activation, we investigated its analgesic effects on CIBP mice. 2-DG (200 μg) reduced mechanical allodynia compared to the vehicle group, with similar efficacy in spontaneous pain assays. This provides direct evidence supporting the effects of 2-DG on the spinal cord in CIBP mice.
Our present findings offer substantial evidence demonstrating that 2-DG: (1) effectively promotes microglial phenotype switching from pro-inflammatory M1 (characterized by ↓CD86/iNOS expression and ↓TNF-α/IL-1β/IL-6 secretion) to anti-inflammatory M2 states (evidenced by ↑Arg-1/CD206 expression and ↑IL-10 production); (2) Consistently suppresses neuroinflammatory processes across both in vivo (spinal cord tissue) and in vitro (BV-2 microglial cell line) experimental systems; (3) Produces dose-dependent antinociceptive effects in our CIBP model. However, the precise upstream and downstream molecular mechanisms through which 2-DG modulates microglial polarization remain unclear. We hypothesize these mechanisms may involve glycolysis reprogramming pathways or NF-κB-centered signaling cascades. This represents a limitation of our current study that warrants further investigation.
However, there are some limitations in our study. First, the precise upstream and downstream molecular mechanisms through which 2-DG modulates microglial polarization remain unclear. We hypothesize these mechanisms may involve glycolysis reprogramming pathways or NF-κB-centered signaling cascades. This represents a limitation of our current study that warrants further investigation. Second, it is worth noting that further research is needed to determine if the non-selective effects of 2-DG on various cell types, including neurons and astrocytes. Third, future research is needed to further fractionate and enrich the population analyzed in microglia. Some studies propose a spectrum of activation states or alternative activation phenotypes beyond the M1/M2 paradigm, so a broader range of phenotypes should be included in future research on microglial cells. Fourth, the research only used male mice to establish a CIBP model. Evidence indicates that estrogen could impact the progression of lung cancer in a lung tumor model utilizing LLC cells. 39 Thus, to prevent any potential influence of estrogen fluctuations on tumor growth in our model, only male mice were utilized in this research. Fifth, clinical translation challenges: while intrathecal 2-DG administration demonstrated efficacy in our murine CIBP model, its clinical applicability faces several hurdles. The invasive delivery method necessitates development of alternative strategies (e.g. blood-brain barrier-penetrating formulations) for human use. Our dose-response data, though rigorously established in mice, require validation in higher mammals to determine clinically relevant dosing. Furthermore, the 5-day safety assessment window, while sufficient for proof-of-concept, cannot exclude potential metabolic complications from prolonged glycolytic inhibition that may emerge during chronic treatment regimens.
Conclusion
In summary, our study suggests that 2-DG attenuates CIBP via suppressing M1 polarization of microglia and neuroinflammation in vivo and in vitro. These findings may provide a novel therapeutic option for alleviating CIBP.
Supplemental Material
sj-pdf-1-mpx-10.1177_17448069251348778 – Supplemental material for Administration of 2-deoxy-D-glucose alleviates cancer-induced bone pain by suppressing microglial polarization to the M1 phenotype and neuroinflammation
Supplemental material, sj-pdf-1-mpx-10.1177_17448069251348778 for Administration of 2-deoxy-D-glucose alleviates cancer-induced bone pain by suppressing microglial polarization to the M1 phenotype and neuroinflammation by Lin Liu, Dan-Yang Li, Long-Qing Zhang, Shao-Jie Gao, Fan-He Song, Jia-Yi Wu, Ya-Qun Zhou, Dai-Qiang Liu and Wei Mei in Molecular Pain
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grants from the National Natural Science Foundation of the People’s Republic of China (82101310, 82271291, and 82071556). Wuhan Talents Excellent Youth Program (24-2RSC09033-09) and Key Research Project of Tongji Hospital (2024A07, 2023A20, 2024A19).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
