Abstract
Migraine is a chronic episodic neurological disorder. However, its diagnosis and management remain unclear. The pathogenesis of migraine is intricately linked to the dysfunction of mitochondria and aberrant trigeminal neuronal activity. Here, we established a murine migraine model via intraperitoneal administration of nitroglycerin (NTG) to examine alterations in mitochondria-associated proteins and calcium signaling patterns within trigeminal neurons, while also investigating the underlying mechanisms. NTG-treated mice exhibited marked periorbital allodynia, decreased crossing of the central area, and decreased time spent in the central area in the open field test compared to Veh treated animals. Furthermore, increased calcium signaling in response to adenosine triphosphate (ATP) stimulation was observed in the trigeminal ganglion (TG) of mice with migraine. Meanwhile, mRNA levels of genes including nuclear respiratory factor-1 (Nrf1), nuclear respiratory factor-2 (Nrf2) and peroxisome proliferator-activated receptor gamma coactivator 1-alpha (Pgc-1) were decreased in the TG. Pharmacological regulation of the mitochondrial function affected NTG-induced migraine chronic pain symptoms. TG mitochondria dysfunctions is implicated in the regulation of mechanical hyperalgesia through the modulation of calcium signaling in an NTG-induced migraine animal model.
Introduction
Migraine is a common neurological disorder with varying degrees of headache. In addition to headaches, migraine sufferers also have symptoms such as nausea, vomiting, and sensitivity to light and sound. In general, headache symptoms usually last for hours or days, seriously affecting the patient’s daily life. 1 The etiology of migraine has not been fully elucidated, but alterations in the brainstem and interactions of the trigeminal nerve may play a role in migraine pathophysiology, involving imbalances in brain chemicals such as calcitonin gene-related peptide (CGRP) and serotonin, as well as abnormal firing of trigeminal ganglia (TG) neurons.2,3 Treatment strategies for migraine are hampered by limited therapeutic effectiveness and complex mechanisms. Therefore, discovery of the novel mechanisms and appropriate therapeutic strategies for migraine management is urgent needed.
Mitochondria, as the cellular energy factories, regulates a series of cellular functions, including energy production, calcium homeostasis, and apoptosis.4,5 Previous studies demonstrated the important role of mitochondrial dysfunction in chronic pain development and maintenance of chronic pain.6,7 Disrupted mitochondrial function can interfere with the normal operation of neurons implicated in pain processing, culminating in increased pain sensitivity and the amplification of pain signals. 8 Mitochondrial dysfunction induces reactive oxygen species (ROS) production and oxidative stress. ROS can harm cellular constituents, trigger inflammatory pathways, and contribute to neural sensitization and sustained chronic pain.9,10 Additionally, mitochondria affects pain signaling pathway through regulation of neurotransmitters-release and synaptic transmission. 8 The disruptions of mitochondrial function alter the balance of key neurotransmitters involved in pain modulation, such as glutamate and gamma-aminobutyric acid (GABA), contributing to the initiation and persistence of chronic pain disorders. 11
In addition, Mitochondria also play a key role in maintaining calcium homeostasis and participating in regulation of calcium signaling pathways.12,13 Elevated calcium concentrations in the mitochondria of neurons disrupt the electron transport chain and impair ATP production, leading to insufficient cellular energy and affecting neuronal function, especially in relation to pain perception and chronic pain. 14 Understanding the complex interaction between mitochondria and calcium regulation could help demonstrate the mechanisms of migraine and develop new treatment strategies. In this study, we present evidence demonstrating that dysfunctions in TG mitochondria is implicated in the regulation of mechanical hyperalgesia through the modulation of calcium signaling in a NTG-induced migraine animal model.
Materials and methods
Animals
In this study, Adult (8–12 weeks) male ICR mice, weighing 20–24 g, were selected for the experiments. Animals were kept in a controlled environment with a temperature of 21–22°C and a 12-h light/dark cycle. Five to six mice were kept in a cage, and they were given unlimited access to food and water. All procedures involving animals were reviewed and approved by the Animal Ethical and Welfare Committee of Kangda College of Nanjing Medical University (approval no.: IACUC-22DS001).
Migraine animal model
The migraine mouse model was induced by repeated intraperitoneal (i.p.) injections of nitroglycerin (NTG; Beijing Yimin, China), as described in previous studies.15,16 Briefly, NTG solution was prepared at a concentration of 1 mg/ml in a stock solution. And the stock solution was diluted with 0.9% saline prior to each injection. The veh group was administered 0.9% saline. Mice were injected with either 10 mg/kg NTG or saline (i.p.) on 1st, 3rd, 5th, 7th, and 9th day, over a total duration of 9 days.
TG injection and drugs administration
The mouse was anesthetized with isoflurane and its head was held in one hand. The tip of the needle (0.33 mm × 13 mm) was then inserted through the infraorbital foramen, infraorbital canal, and foramen rotundum, and finally positioned in the TG. Drugs were slowly delivered to the TG through the needle. Various concentrations rotenone (10, 20, and 30 μM) into the TG of mice 1 time/day for 3 days. Different doses (30, 60, and 90 mg/kg) of Schisandra chinensis or mitoquinone (MitoQ) at various concentrations of (2.5, 5, and 10 mg/kg) were intraperitoneally administered to the migraine mice 1 time/day for 3 days.
Nociceptive behavior test
Baseline mechanical sensitivity levels were assessed using von Frey monofilaments on the left side of the face, specifically in the rostral area near the eye, as described in previously. 17 Prior to the administration of NTG or veh over a 9-day period, the mice underwent daily testing to determine their baseline mechanical thresholds. A standard force of 0.16 g periorbital filament curvature was applied to elicit a response behavior. In cases where the mice displayed pain-related behaviors such as facial scratching or rapid withdrawal, a lower force filament was adopted. Conversely, a higher force filament was applied, with a minimum interval of 3 min between stimulations. Six stimulation trials were conducted, and the pain threshold was determined using the up-down method. All behavioral experiments were performed by individuals who were blinded to the treatment.
Open field test
The ANY-Maze software system (Stoelting Co., Illinois) was used to perform the open field behavior experiments. Three days before the test, the mice were habituated to the experimental room for 1 h per day. The ambient noise levels and lighting conditions in the experimental room were carefully controlled to avoid the effect on the mice. Before starting of experiments, the open field (60 cm × 60 cm × 50 cm) was sanitized with 75% alcohol, and the video tracking equipment was calibrated for optimal performance. Each mouse then was placed at the center of the open field and unrestricted movements were recorded for 5 min. The captured data included the total distance covered by the mice, the time spent in the central area, and the frequency of central area crossings. After recording, the open field was cleaned and disinfected with 75% alcohol before the next trial.
Immunofluorescence staining
Immunofluorescence staining protocol was performed according to established procedures. 16 Briefly, mice were deeply anesthetized with isoflurane and perfused through the aorta, firstly with cold saline and then with 4% paraformaldehyde. After paraformaldehyde perfusion, TGs were isolated and fixed in 4% formalin overnight under 4°C. TGs were then dehydrated in 25% and 30% sucrose solution. Subsequently, TG tissues were embedded in optimal cutting temperature (OCT) and sectioned into 20 μm slices. The sections were then incubated in a solution of 2% bovine serum albumin in PBS buffer with 0.01 Triton X for 1 h to block non-specific staining. Following three washes with PBS for 15 min each, the samples were incubated overnight at 4°C with primary antibodies targeting CGRP; (1:500, Santa cruz), and Class III beta-tubulin (TUJ1, 1:1000, Abcam). On the second day, the slices were incubated at room temperature for 10 min. After another 15-min washing with PBS, the sections were incubated with FITC or Cy3-conjugated (1:1000, Boster) secondary antibodies for 1 h at room temperature. The slices were covered with Fluoromount-G and the signals were checked. The immunofluorescence signals were visualized using a Nikon fluorescence microscope and analyzed using ImageJ software. The number of positive cells in TG section slice was counted and values represented the number of cells per 0.2 mm2. Three sections were randomly selected from each mouse for immunofluorescence staining analysis.
Primary culture of TG neurons
The method for the primary culture of TG neurons was according to previously established protocols.16,17 Following isoflurane anesthesia, the mice were perfused with ice-cold artificial cerebrospinal fluid (ACSF) bubbled with 95% O2 + 5% CO2, and the bilateral TG were carefully dissected. Subsequently, the connective tissue surrounding the TGs was meticulously removed in ACSF at 4oC. The TGs were then incubated in ACSF containing collagenase and dispase-II at 37°C for 90 min. The digested cells were pelleted by centrifugation at 100 × g for 10 min and gently triturated using a Pasteur pipette. These cells were plated onto adhesive coverslips and maintained in a 37°C incubator. The TG neurons obtained from this culture were utilized for patch clamp and calcium imaging experiments.
Calcium image
Ca2+ imaging was performed as previously described. 18 The fluorescent Ca2+ indicator Fluo-4 AM was used to quantify the calcium levels in TG neurons. Isolated TG neurons were cultured in 8-well dishes and rinsed with HEPES-buffered Hanks’ balanced salt solution. Following a 40-min incubation with Fluo-4 AM at room temperature, coverslips containing TG cells were transferred to a recording chamber positioned on an inverted fluorescence microscope (Nikon, Japan). ATP stimulation was administered to TG cells from both Veh and chronic migraine mice in the calcium free (EGTA contain) bath solution to exclude possible Ca2+ influx. After finishing recording of intracellular calcium signal, KCl (Ca2+ contain) was applied to check the status of neurons. Only the cell responded to KCl was counted for further analysis. The duration of ATP application was about 30 s. Ca2+ fluctuations of TG neuron were imaged by exciting fluo-4 at 488 nm and detecting emitted fluorescence at 520 nm using a computer-controlled Nikon fluorescence microscope. Corresponding time traces show normalized intensity fluorescence as the ratio of fluorescence to initial fluorescence (F/F0).
RT-qPCR
mRNA samples were prepared and subjected to quantitative real-time polymerase chain reaction (qRT-PCR) following established procedures. 17 Briefly, total RNA from TG tissues was extracted by using TRizol reagent according to the manufacturer’s instruction. cDNA was synthesized using Prime Script TM reagent. The RNA samples were treated with RNase-free DNase I. Quantitative real-time PCR was performed on Step One Plus TM Real-Time PCR System (Applied Biosystems, Carlsbad, CA, USA) using the SYBR Green Master Mix (Vazyme Biotech, China). Relative expression of genes was normalized to the endogenous reference GAPDH by using 2-△△CT method. The sequences of the primers used for qRT-PCR experiments were as follows: NRF1 forward GGCAACAGTAGCCACATTGGCT, NRF1 reverse GTCTGGATGGTCATTTCACCGC; NR-F2 forward CAGCATAGAGCAGGACATGGAG, NRF2 reverse GAACAGCGGTAGTATCAGCCAG; TFAM forward GAGGCAAAGGATGATTCGGCTC, TFAM reverse CGAATCCTATCATCTTTAGCAAGC; PGC-1α forward TATGGAGTGACATAGAGTGTGCT, PGC-1αreverse GTCGCTACACCACTTCAATCC; GAPDH forward CA-TCACTGCCACCCAGAAGACTG, GAPDH, reverse ATGCCAGTGAGCTTCCCGTTCAG.
Determination of reactive oxygen species in TG
Mitochondrial ROS was measured in cultured TG neurons by using MitoSOX Red (GC68230, GLPBIO, China) according to the manufacturer’s instructions. Briefly, primary cultured TG neurons on coverslips were washed with PBS for 3 times and incubated in 10 μM MitoSOX Red at 37℃ in the dark for 15 min. After incubation the neurons were washed 3 times with PBS and then moved to microscope slides. The immunofluorescence signals were visualized using a Nikon fluorescence microscope and analyzed using ImageJ software.
Statistical analysis
All data are presented as the mean ± SEM. Data normality were measured by the Shapiro-Wilk test. If the data fails the normality test, Mann-Whitney nonparametric test was used. The significance of the differences was analyzed using one or two-way analysis of variance (ANOVA) and Student’s t-test. Differences were considered statistically significant at p < 0.05.
Results
NTG injection caused periorbital mechanical hyperalgesia and anxiety-like behaviors
NTG is an easily penetrable nitric oxide donor with lipid solubility that can stimulate vascular smooth muscle, resulting in vasodilation. Previous studies have shown that NTG can induce migraine. 19 Thus, repeated NTG injections were selected to induce migraine-like behavior in mice (Figure 1(a)). Following the establishment of the NTG-induced migraine mouse model, the baseline periorbital mechanical threshold was assessed using a von Frey monofilament. Prior to NTG injection, there were no discernible differences in the mechanical threshold between veh and migraine mice. However, following NTG infusion, the model mice exhibited significant mechanical allodynia from days 1 to 9, indicating the successful establishment of the migraine model (Figure 1(b)). Given the known association between chronic pain and anxiety, we also investigated potential emotional dysfunctions in the model. Utilizing the open-field behavior test, the model mice displayed fewer crossings and spent less time in the center of the open field compared to the veh animals (Figure 1(c)–(e)). These results suggest that NTG injection induced periorbital mechanical hyperalgesia and anxiety-like behaviors in an animal model of NTG-induced migraine.

NTG injection caused periorbital mechanical hyperalgesia and anxiety-like behaviors. (a) Schematic of the timeline for drug treatment and behavioral testing. (b) Periorbital mechanical thresholds of Veh (n = 7) or NTG (n = 7) infusion mice. F(1,12) = 71.41, ***p < 0.001, Two-way ANOVA (repeated measures) with Bonferroni’s post hoc test was used. (c) Number of center entries and (d) the time in central zone decreased after NTG infusion in the open field test. (e) Total movement distance was not affected by NTG infusion. Data are presented as mean ± SEM. Veh, n = 7, NTG n = 7; **p < 0.01, ***p < 0.001 compared with the veh group, student’s t-test.
NTG injection upregulated the expression level of CGRP in TG neurons
Calcitonin gene-related peptide (CGRP) is a neuropeptide with widespread distribution in the trigeminal system that is known to be involved in the occurrence and maintenance of migraine. 20 Thus, we also examined the expression of CGRP in TG neurons of NTG-induced migraine mice using immunofluorescence techniques at day-9 after modeling. Immunofluorescence staining results showed that CGRP was co-expressed with the neuronal marker Tuj1 (Figure 2(a)). Furthermore, a significant increase in the percentage of CGRP-positive neurons was observed in the NTG-treated group compared to the veh group (Figure 2(b)). These results suggest the involvement of CGRP signaling in NTG-induced migraine.

Calcitonin gene-related peptide (CGRP) expression significantly increased in TG neurons from NTG-induced migraine mice. (a) Immunostaining for neuron marker TuJ1 and CGRP in TG. Scale bar: 100 μm. (b) Statistical data show the percentage of CGRP+ neurons in the TG. Data are presented as mean ± SEM. Student’s t-test was performed for two group comparisons. *p < 0.05 compared with the veh group. n = 3 mice per group.
Intracellular Ca2+ signals were enhanced after NTG injection
To investigate the mechanism underlying pain behavior in mice with migraine, we conducted in vitro experiments using cultured TG neurons. Intracellular calcium ions are pivotal in regulating cellular functions under different physiological conditions, and alterations in this signaling pathway may be relevant to migraine pathology. Using calcium imaging techniques, we examined the intracellular calcium signals in these neurons following the induction of migraine conditions. ATP (100 µM) was applied to the cultured TG neurons that were pretreated with Fluo-4 AM, a calcium indicator to induce intracellular calcium response. ATP can initiate IP3 production through a group of G-protein-couple receptors, including purinergic P2Y2 receptor. ATP application induced an increase in calcium transient signals, while the TG neurons from model mice showed a significant increase in intracellular calcium signals compared to the veh group (Figure 3(a) and (b)). These results suggest a potential role for the enhanced intracellular calcium signal pathway in promoting pain initiation and progression in migraine.

Intracellular Ca2+ signals were enhanced after NTG injection (a) Ca2+ response fluorescence intensity representative trace in TG neurons from veh group and NTG treatment group. (b) Statistical summary show Ca2+ response fluorescence intensity in TG neurons was significantly increased after NTG injection. Data are presented as mean ± SEM. Mann-Whitney test was performed for two group comparisons. ***p < 0.001 compared with the veh group. n = 186 cells for veh group, n = 214 cells for NTG group. n = 4 mice per group.
Dysfunction of mitochondrial biogenesis was involved in NTG induced migraine
Mitochondria serve as the primary source of energy in eukaryotic cells, and abnormalities in neuronal mitochondrial function can affect pain processing. Recent studies have highlighted the crucial roles of mitochondrial biogenesis in migraine pathophysiology.17,21,22 In order to explore potential changes in the mitochondrial biogenesis of TG neurons from the NTG-induced migraine model, we examined the expression levels of key regulators associated with mitochondrial biogenesis, including NRF1, NRF2, TFAM, and PGC-1α, in TG neurons. Quantitative PCR analysis showed a progressive decrease in the mRNA levels of NRF1, NRF2 and PGC-1α, following NTG injection compared to those in the veh-injected group (Figure 4(a)–(c)). The mRNA level of TFAM was not significantly changed (p = 0.093; Figure 4(d)). These findings indicate a significant impairment in mitochondrial biogenesis within TG neurons in NTG-induced migraine mice.
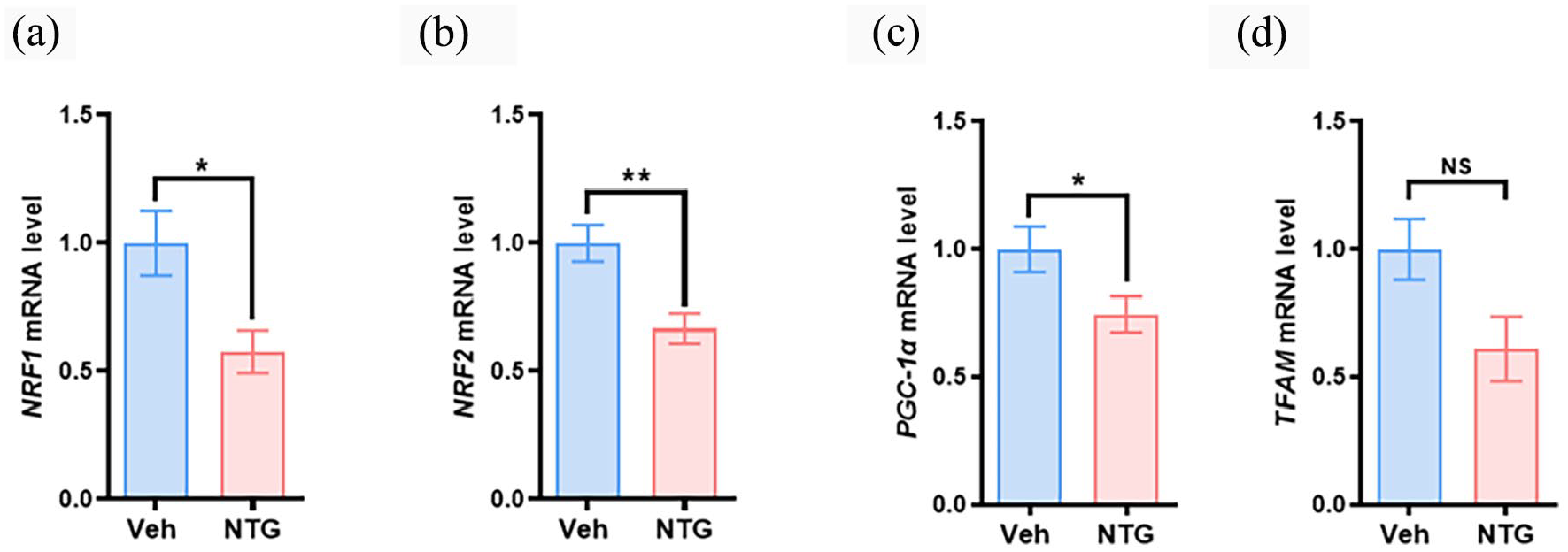
NTG injection caused impairment of mitochondrial biogenesis in TG. Real-time quantitative PCR analysis of (a) NRF1, (b) NRF2, (c) PGC-1, and (d) TFAM mRNA levels in TG from veh and NTG injection mice. Data are presented as mean ± SEM. Unpaired t-test or Mann-Whitney test was performed for two group comparisons. *p < 0.05, **p < 0.01, NSp > 0.05, compared with the veh group, Student’s t-test. n = 6 mice per group.
Regulation of mitochondrial function affects nociceptive behavior in NTG induced chronic migraine
Given the critical role of mitochondria in the pathogenesis of NTG-induced migraine, we attempted to modulate mitochondrial function using a combination of mitochondrial inhibitor and protectant to validate our hypothesis. Rotenone, a potent mitochondrial inhibitor, disrupts mitochondrial electron transport chain and alters mitochondrial function. 23 After injection of various concentrations rotenone (10, 20, and 30 μM) into the TG of mice 1 time/day for 3 days (at d1, d3, d7), the mice showed a dose-dependent induction of periorbital hyperalgesia(Figure 5(a)). Additionally, we tested the anti-nociceptive effects of Schisandra chinensis. Schisandra chinensis is a traditional herbal medicine known for its mitochondrial protective properties. 24 Different doses (30, 60, and 90 mg/kg) of Schisandra chinensis were administered to the migraine mice 1 time/day for 3 days (days d1, d3, d7). Intraperitoneal administration of Schisandra chinensis significantly alleviated migraine-like symptoms induced by NTG injection in mice (Figure 5(b)). To further confirm the role of mitochondrial function on NTG-induced migraine pain behavior, we also applied mitoquinone (MitoQ) at various concentrations of (2.5, 5, and 10 mg/kg) to mice to evaluate its anti-nociceptive effects. MitoQ is a TPP-based mitochondria-targeting antioxidant that blocks H2O2-induced intracellular ROS reaction and prevents oxidative damage. 25 As shown in Figure 5(c), intraperitoneal administration of MitoQ significantly relieved NTG-induced chronic pain in migraine mice. We then also investigated the effect of application of MitoQ on mitochondrial ROS level of TG neuron. Mitochondrial dysfunction induces reactive oxygen species (ROS) production and oxidative stress. The TG neurons from NTG-induced migraine show high level of mitochondrial ROS compared to those from sham mice, however, middle dose of MitoQ treatment reduced the ROS expression level (Figure 5(d)). These results suggest the involvement of mitochondrial dysfunction in the pathogenesis of migraine-like behaviors and highlight the potential analgesic effects of preserving mitochondrial function.

Regulation of mitochondrial function affects nociceptive behavior in NTG-induced chronic migraine. (a) Change in periorbital mechanical allodynia after various doses of rotenone (mitochondrial inhibitor) injection. n = 6 mice/group, F(3,20) = 12.23, p < 0.001, at day-1 veh versus high dose *p < 0.05; at day-5 veh versus high dose ***p < 0.001; at day-9, veh versus middle dose or veh versus high dose ***p < 0.001, Two-way ANOVA (repeated measures) with Bonferroni’s post hoc test was used. (b) Change in periorbital mechanical allodynia of NTG-induced migraine model mice treated with different doses of Schisandra chinensis (mitochondrial protectant). n = 6 mice/group, F(3,20) = 12.02, p < 0.001, at day-1 and day-5, veh versus high dose *p < 0.05; at day-9 veh versus high dose ***p < 0.001, Two-way ANOVA (repeated measures) with Bonferroni’s post hoc test was used. (c) Change in periorbital mechanical allodynia of NTG-induced migraine model mice treated with different doses of mitoquinone (mitochondria-targeting antioxidant). n = 6 mice/group, F(3,20) = 42.02, p < 0.001, at day-1, veh versus high dose ***p < 0.001, Two-way ANOVA (repeated measures) with Bonferroni’s post hoc test was used. Rotenone, Schisandra chinensis and MitoQ was separately administered to the mice 1 time/day for 3 days (at d1, d3, d7). The application of drugs was 4 h before the behavior test. (d) Change in MitoSOX in TG from sham + Veh, model + Veh, and model + MitoQ gourp mice. n = 3, F(2,6) = 154.2, p < 0.001, sham + Veh versus model + Veh ***p < 0.001; model + Veh versus model + MitoQ ***p < 0.001, one-way ANOVA with Bonferroni’s post hoc test was used.
Discussion
In the current study, we found that mitochondrial abnormalities are involved in the regulation of nociceptive behavior through the modulation of intracellular calcium signaling in an NTG-induced migraine mouse model. Our results revealed that repeated administration of NTG lead to pronounced periorbital allodynia and a decline in spatial exploration capacity. Alterations in intracellular calcium signaling were observed in TG neurons after repeated NTG administration, which were potentially associated with abnormal mitochondrial biogenesis. Additionally, the analgesic properties of the Chinese herbal extract Schisandrae (a mitochondrial protector) were evaluated. Schisandrae significantly alleviates NTG-induced migraine-like behavior in mice.
Neuro-inflammation triggered by dysfunction of the trigeminovascular system is a key factor in the pathogenesis of migraine. 26 In the present study, chronic migraine behavior was induced in mice by repeated administration of NTG. NTG, an easily penetrable nitric oxide donor with lipid solubility, can stimulate vascular smooth muscle, resulting in vasodilation. 27 Upon injection, NTG can activate the trigeminal nerve vascular system, leading to the direct activation of the second messenger cyclic guanosine phosphate (cGMP) by the nitric oxide it produces. This activation subsequently induces vasodilation by modulating downstream signaling pathways, stimulating nociceptive neurons in the TG, and triggering the release of the vasoactive peptide CGRP, ultimately culminating in pain sensation. 28 In our NTG-induced mouse model of migraine, we observed a significant increase in the number of CGRP-positive neurons in the TG. Behavioral assessments revealed periorbital mechanical hyperalgesia and reduced exploratory activity in mice following repeated NTG injections. Mechanical allodynia is indicative of trigeminovascular sensitization, which is associated with the underlying pathogenesis of migraines.
Intracellular Ca2+, as a secondary messengers, participates in various pathological and physiological processes. 29 The stabilization of intracellular Ca2+ homeostasis is crucial for cellular function and survival. Ca2+ also play a significant roles in acute or chronic pain modulation. 30 During acute pain, mechanical stimuli causes changes in the cell membrane potential and lead to the opening of voltage-gated calcium ion channels and influx of calcium ions into the cell. In chronic pain, local inflammatory reactions lead to enhanced intracellular calcium signaling pathways. Pain stimuli increase in intracellular calcium ion concentrations, activating a series of molecular effects related to pain perception, ultimately leading to chronic pain. 31 In the NTG-induced migraine model, intracellular Ca2+ signaling was significantly increased, indicating abnormal intracellular calcium signaling pathways in migraine. Additionally, CGRP release is dependent on intracellular calcium ions, because an increase in the intracellular calcium ion concentration promotes CGRP release. 32 In a migraine model, a key mechanism of CGRP action is the sensitization of nociceptive trigeminal neurons, which exhibit exaggerated responses to various stimuli. Meanwhile, increase of Ca2+ can also activate various signaling pathways that modulate ion channel activity. For instance, the Ca2+ influx can activate Ca2+-activated K+ channels, which affects the membrane potential and action potential firing rates of neurons. 33 Changes in intracellular Ca2+ are also involved in the phosphorylation of ion channels, further influencing ion conductance and the neural excitability. 34 This further indicates that pain hypersensitivity induced by NTG is due to abnormal calcium signaling and increased excitability of trigeminal neurons.
Mitochondria are double-membrane organelles of the cell and the power source responsible for energy production. Mitochondria release energy through respiration to meet the needs of cell activity. 9 Mitochondrial biogenesis is the process by which cells increase their individual mitochondrial mass and copy number to meet increased energy requirements. This process, which includes the synthesis of new mitochondrial proteins, lipids and DNA, is essential for maintaining cellular energy homeostasis.35,36 Several key factors were involved in mitochondrial biogenesis include: PGC-1α (Peroxisome proliferator-activated receptor gamma coactivator 1-alpha): A critical regulator that activates genes involved in energy metabolism; NRF1 and NRF2 (Nuclear respiratory factors): Transcription factors that activate the expression of mitochondrial genes; TFAM (Mitochondrial transcription factor A): Necessary for the transcription and replication of mitochondrial DNA (mtDNA). Mitochondrial biogenesis is stimulated by various factors, including oxidative stress, cell renewal and cell division. 36 The impairment of mitochondrial biogenesis likely results in mitochondrial dysfunction and has been associated with a various of diseases including neurodegenerative, 37 metabolic 38 and cardiovascular diseases. 39 In addition, some indirect evidence suggests that mitochondrial biogenesis is impaired in other pain conditions, Sun et al. 40 observed that Sestrin2 overexpression attenuates osteoarthritis-related pain by inducing AMP-activated protein kinase (AMPK) -mediated mitochondrial biogenesis, highlighting the importance of this pathway in modulating pain and inflammation. In the current study, we also found that impairment of mitochondrial biogenesis occurs in the NTG-induced migraine model, which is consistent with a previous study in an inflammation induced migraine model. 8
Disrupted mitochondrial function also significantly impact Ca2+ regulation within cells.7,12 Mitochondria normally take up Ca2+ ions from the cytoplasm to regulate intracellular calcium levels, facilitated by the mitochondrial calcium uniporter protein complex. Calcium stored within the mitochondria can be released back into the cytoplasm when necessary. However, impaired mitochondrial function can disturb calcium balance, leading to altered cellular calcium handling and abnormal intracellular calcium levels. 12 In the NTG-induced migraine model, the dysfunction of TG neurons results in disturbances in intracellular Ca2+ homeostasis. Moreover, high intracellular Ca2+ levels can harm the mitochondria, increasing in cytosolic Ca2+ concentration. 41 Elevated cytosolic calcium levels can activate various calcium-dependent enzymes and signaling pathways, causing cellular dysfunction and potentially contributing to the development of pathological conditions, including chronic pain. 42 The role of mitochondrial dysfunction in pain can be validated using a mitochondrial function inhibitor such as rotenone, to induce migraine-like behaviors in mice. Rotenone induces hypersensitivity through its interaction with mitochondrial function. The involved mechanism may be relative to the inhibition of mitochondrial complex I and increase of reactive oxygen species (ROS), which needs further investigation. Conversely, Schisandra, a mitochondrial protectant, alleviates hypersensitivity in NTG-injected mice. The protective effects of Schisandra on mitochondrial function have been explored in various studies. 43 A previous study 44 indicates that Schisandra has been linked to the enhancement of mitochondrial antioxidant status. This enhancement is believed to provide a generalized protective effect against both internal and external stressors, thereby supporting the overall function of mitochondria, which are critical for energy production in cells. This suggests that Schisandra may play a significant role in maintaining mitochondrial health and function. Furthermore, a randomized clinical trial reported that a mixture of Schisandra chinensis extract significantly improved glycemic control in individuals with hyperglycemia, as evidenced by reductions in fasting plasma glucose and other metabolic markers, 45 which suggests a potential role for Schisandra chinensis in type 2 diabetes. In our study, we also found that Schisandra could relieve NTG-induced migraine pain sensitivity, indicating its potential therapeutic effect on chronic migraines.
Conclusion
Our results suggest that intracellular calcium signaling imbalance and mitochondrial dysfunction in TG neurons are among the causes of NTG-induced migraine-like behaviors, which may be related to mitochondrial damage. Repetitive NTG exposure alters the balance of mitochondrial function and impairs mitochondrial biogenesis in TG neurons. Manipulation of mitochondrial dynamics and biogenesis signaling may indicate a novel mitochondria-targeted strategy to alleviate metabolic defects in migraine. Further research is needed to uncover the mechanisms underlying mitochondrial involvement in pain perception and develop effective interventions for migraine.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by National Natural Science Foundation of China (82001177), the Qing Lan Project of Jiangsu Province, Science and Technology Plan (Social Development) Project of Lianyungang City (KJSKJJZC24013).
