Abstract
Patients with cancer perineural invasion (PNI) report greater spontaneous pain and mechanical allodynia. Here, we examine the impact of the disease on the peripheral sensory system, the excitability changes induced by PNI at the dorsal root ganglia, and the potential protective role of the absence of Tumor Necrosis Factor-α Receptor 1 (TNFR1). To study these effects, we use a murine model generated by injecting mouse oral cancer squamous cell carcinoma (MOC2) into the sciatic nerve (MOC2-PNI) in both male and female mice. We found that MOC2-PNI induces a profound change in the somatosensory landscape by deactivating/blocking the peripheral inputs while modulating the afferent’s sensibility (tactile desensitization with concurrent nociceptive sensitization) and demyelination without inducing spontaneous activity. All these changes caused by MOC2-PNI are unmitigated by the absence of TNFR1. We conclude that MOC2-PNI induces an aberrant neuronal excitability state and triggers extreme gender-specific neuronal plasticity. These data allow us to speculate on the role of such plasticity as a powerful defense mechanism to prevent terminal sensory dysfunction, the rise of chronic pain, and extend animals’ survivability.
Introduction
Cancer invading in, around, or along the nerve, a process termed PNI correlates with worse pain and poor prognosis.1,2 This is particularly important for a subset of patients exhibiting symptoms of peripheral neuropathy, such as intractable pain, function impairment, numbness, and formication. 3 In a recent multi-center study, we found that PNI is an independent predictor for function-evoked but not spontaneous pain in patients with oral cancer that significantly impairs quality of life. 4
However, studying the pathophysiological mechanism of pain under these conditions is often extremely complicated and challenging due to two main factors: a) Although similar to human symptoms, animals’ response to the injury seems to be of lesser intensity, 5 and b) it involves several cellular subtypes (both neuronal and non-neuronal). Tumors and their surroundings (sensory neurons, fibroblasts, Schwann, muscle, immune, and endothelial cells) create a highly interactive microenvironment. This interaction leads to the release of many pro-inflammatory and pro-nociceptive mediators thought to be critical for cancer pain and tumor progression. 6
Together, these factors reduce our ability to appropriately evaluate the effects of pharmacological or molecular manipulations (e.g. the use of transgenic animals). In this context, it has been shown that some cancer cell lines may invade the nerves and cause demyelination, decreasing the nerve conduction velocity and inducing sensory neuron hyperexcitability. 7 Furthermore, axonal damage deeply modulates the peripheral signal, reducing tactile mechanical inputs while reducing nociceptive thresholds and increasing pain perception.2,8
In this context, it has been proposed that TNF-α is a key mediator of this process. 9 Released by the Schwan, tumoral, and immune cells (monocytes and macrophages), 10 TNF-α seems to function as a “master regulator” cytokine, critically triggering a cytokine-mediated inflammatory cascade. 11 TNF-α activation of its receptor TNFR1 has been shown to mediate mechanical pain and promote tumor progression, in addition to their pro-inflammatory functions (for review, see Ref. 42).
The primary goal of the current study is to expand previous observations on the effects of cancer PNI on primary afferent plasticity and the putative role of TNFRs in preventing these effects and tumor growth. Based on our earlier studies on PNI and nerve injury models,2,5,8 we hypothesize that a more aggressive cell line (mouse oral squamous cell carcinoma, MOC2) 12 will induce a more profound disruption of the primary sensory neuron activity (at L4), in a syngeneic, immune-competent mouse model of PNI. We also expected that the absence of one of these receptors (TNFR1) will positively impact the overall electrical status of these afferents by reducing this disruption. Due to a potential gender bias of oral cancer pain, 13 we also expect that females will be more affected than males, returning peripheral activation patterns similar to males but with a higher magnitude. We aim to correlate the tumor burden with the behavioral (reflexive and non-reflexive) and peripheral sensibility of the affected afferents to establish the putative protective role of the TNFR1 deficiency, preventing functional and electrical peripheral damage.
Methods
Thirty-two mice, 4–6 weeks of age, of two breeds (C57BL/6J termed wild type (WT) and Tnfrsf1atm1Mak (TNFR1 -/- termed KO)) (Jackson Laboratory, Bar Harbord, ME. USA) with four experimental groups (Sham, MOC2-PNI, males, and females, 8 animals per group) were used to study the putative protective role of the absence of TNFR1 on the development of the MOC2-PNI-induced neuropathy. Cell culture procedures and MOC2 cell implantation on the animals’ sciatic nerves were performed at New York University (NYU) facilities. They were then transported to Wake Forest University (WFU) for behavioral, electrophysiological (sensorial and biophysical), and molecular evaluation following a linear consecutive experimental design (Figure 1(a)). Due to its size and detail our study will be presented in two parts: 1) Animals behavior (reflexive and non-reflexive) and peripheral sensory electrophysiology (distribution, sensibility, and afferents conduction velocity (CV)) (Part 1.) and 2) peripheral sensory neurons somatic biophysics (active and passive) and gene expression (Part. 2) (Figure 1(a) gray). In both facilities, the animals were housed in pairs under a 12-h light/dark cycle in a climate-controlled room. The use and handling of animals were under guidelines provided by the National Institutes of Health and the International Association for the Study of Pain, and the Institutional Animal Care and Use Committees of New York University and Wake Forest University Health Sciences approved the procedures and experiments.

(a) Schematic of the experimental design performed by New York University (NYU) and Wake Forest University (WFU), (b) Diagram of the electrophysiological (ePhys) intracellular recordings performed in the lumbar 4 (L4) ganglia, the area of the L4 dermatome (purple) searched for cellular receptor fields and the approximate location of the MOC2-PNI (green) along the sciatic nerve (SN). The gray box indicates the pos hoc analysis to be presented in Part 2 of the study, included here for informative purposes only. (C: contralateral, I: ipsilateral).
Cell culture
MOC2 cell line (Sigma Chemicals) were cultured in IMDM/F12 (2:1 mixture) with 5% FBS (Fisher Scientific), 1% penicillin/streptomycin, 1% amphotericin, 5 ng/mL EGF (Millipore), 400 ng/mL hydrocortisone and 5 mg/mL insulin.
MOC2 implantation and sham surgery
The animals (WT, KO, male, and female) were anesthetized using isoflurane, and their right sciatic nerve was exposed. 3000 MOC2 mouse oral carcinoma cells in 3 μl of serum-free media or vehicle (serum-free growth medium, for the sham group) were injected into the sciatic nerve, distal to the bifurcation of the tibial and common peroneal nerves. The formation of a bulb in the injection area indicates a good injection. After gently removing the needle, the nerves were then covered with the underlying muscles, and the skin was closed with skin closure clips (Reflex 7). Mice were observed until they fully recovered.
Behavioral testing
Reflexive behavioral testing: Paw withdrawal threshold (PWT)
Three weeks after the implantation of MOC2 cells in the sciatic nerve (or sham surgery), mice were placed on a mesh surface in a plastic cage and were acclimated for 20 min before testing. The paw withdrawal threshold (PWT) was assessed by application through the mesh flooring of calibrated von Frey filaments to the footpad until the filaments bent. This was done by a person blinded to the surgical treatment. The von Frey filaments used were 3.84, 4.08, 4.31, 4.56, 4.74, 4.93, 5.18, 5.46, and 5.88, corresponding to 0.5, 0.9, 1.7, 3.7, 5.5, 8.0, 12.4, 21.5, and 53.0 g, respectively. A filament was applied three times, separated by 5 s, with a response considered positive if it resulted in a brisk withdrawal of the paw on any of the three applications. The force resulting in a 50% probability of withdrawal was determined using the up-down method as previously described. 14
Non-reflexive behavioral testing: gait analysis
After PWT testing, the animals were moved to the CatWalk Automated Gait Analysis System (Noldus Information Technology, Wageningen, Gelderland, The Netherlands) and software version XT 10.6) for conditioning and non-reflexive behavioral analysis. Each animal completed a minimum of three compliant runs. 15 Compliant runs for each animal were set for a minimum and maximum run duration of 0.5 and 5 s, respectively, necessary to complete a defined path length (20 × 10 cm). CatWalk system was used to measure MOC2-induced peripheral nerve injury/protection by measuring various parameters (stand, print area, stride length, swing speed) within different categories, including pain-related and coordination-related categories. 16
Electrophysiology (ePhys)
After the Gait analysis was completed, the animals were moved to the ePhys facility for in vivo intracellular recordings (Figure 1(b)). In this facility, the animals were anesthetized with isoflurane 3%. The trachea was intubated, and the lungs were ventilated using pressure-controlled ventilation (Inspira PCV, Harvard Apparatus, Holliston, MA, USA) with isoflurane in humidified oxygen. Heart rate and noninvasive blood pressure were monitored throughout as a guide to the depth of anesthesia. Anesthetized animals were immobilized with pancuronium bromide (2 mg/kg), and end-tidal isoflurane concentration was maintained at 2% throughout the study. A dorsal incision was made in the lumbar midline, and the L4 dorsal root ganglion (DRG) and adjacent spinal cord were exposed by laminectomy, as previously described. 17 The exposed nervous tissue was continuously superfused with oxygenated artificial cerebrospinal fluid [aCSF (in mM): 127.0 NaCl, 1.9 KCl, 1.2 KH2PO4, 1.3 MgSO4, 2.4 CaCl2, 26.0 NaHCO3, and 10.0 D-glucose]. The spinal column was secured using custom clamps, and the anesthetized animal was transferred to a preheated (32–34°C) recording chamber where the superfusate was slowly raised to 37°C (MPRE8, Cell MicroControls, Norfolk, VA USA), and its flow rate kept at 1 ml per minute (bath exchange time of ~5 s). A thermocouple (IT-23, Physitemp, Clifton, NJ, USA) monitored the pool temperature adjacent to the DRG.
The electrophysiological recording session from each animal was limited to a maximum duration of 60 min to diminish the likelihood that surgery and experimental manipulation would result in primary sensory afferent sensitization. A total of 12 cells per animal per hour were targeted (1 every 5 min) to provide as much information as possible on 1) the cell’s type distribution (see cellular classification protocol), 2) peripheral mechanical sensibility, and 3) afferents conduction velocity. DRG neuronal somata were impaled with borosilicate micropipettes (80–250 MΩ) containing 1 M potassium acetate. DC output from an Axoclamp 2B amplifier (Axon Instruments/Molecular Devices, Sunnyvale, CA, USA) was digitized and analyzed offline using Spike2 (CED, Cambridge, UK). The sampling rate for intracellular recordings was 21 kHz throughout (MicroPower, CED).
Cellular classification protocol
The general procedure for classifying primary sensory afferents was applied elsewhere for DRG neurons. 18 The receptive field (RF) was searched, starting on the animal’s paw in a longitudinal pattern, covering the digit, progressing proximally to the hairy skin, and ending 5 mm distal to the surgical incision. Neurons with receptive field (RF) located near the skin incision were excluded because of the potential sensitization of those afferents by surgery. RFs were located with the aid of a stereomicroscope using increasing mechanical stimulation; the latter progressed from light touch with a fine sable hair paintbrush to searching with a blunt probe (back of the paintbrush) and ultimately gentle to intense pinching with fine-tipped forceps. Based on their mechanical RF (or absence thereof) and their capability to generate a somatic action potential (AP), these cells were classified as follows:
a) Cells with RF: Based on the combination of their mechanical threshold, conduction velocity (CV), and dynamic response (phasic; on-off; tonic), neurons were classified into three groups: LTMRs (low-threshold mechanoreceptors, rapidly adapting (RA) and slow adapting (SA) tactile afferents), muscular spindles (MS), A-HT (A fiber high-threshold mechanoreceptors), and C-HT (C fiber high-threshold mechanoreceptors) innervating skin.
b) Cells without RF: Electrically excitable Cells but without mechanical RF were separated into two different populations based on the shape of the action potential (AP)29–31 as neurons with inflection in the repolarizing phase (slow or S-type neurons) and neurons without this inflection (fast, or F-type neurons). To more clearly determine the presence of this inflection, the first derivative records of the AP were used (presence or absence of a second additional negative component in the time course of the AP first derivative). Since RF properties, especially response characteristics, were used to define differences in the fast-conducting afferents (those without inflected APs), the ability to define and categorize these three populations further was not possible.
All included cells satisfied the following requirements: Resting membrane potential (Em) more negative than −40 mV, evoked AP amplitude ≥30 mV, and the presence of afterhyperpolarization (AHP). Passive membrane properties indicative of poor impalement (extremely low input resistance (Ri), extremely short time constant (tau, τ)), and unstable impalement were also reasons for exclusion. CV was measured at the end of the recording.
Mechanical sensibility and conduction velocity
Sensibility
After identifying the cellular RF area responsive to search stimuli, the area was marked using a red fine-point marker. This initial procedure was performed gently to avoid damaging the skin (as assessed visually by lack of development of erythema, edema, glossiness, etc.). The presence of spontaneous activity was assessed during this procedure. Resting mechanical threshold (MT) was determined in each afferent with calibrated von Frey filaments (Stoelting, Wood Dale, IL) as the minimum force to generate at least one AP, testing the most sensitive area of the cellular RF. The presence or absence of post-discharge hyperpolarization (PDH) 19 was determined during this procedure.
Conduction Velocity (CV)
Spike latency was obtained by stimulating the RF at the skin surface using a bipolar electrode (0.5 Hz, current range: 0.1–1.2 mA) and a stimulus isolator (A360LA, WPI, Sarasota, FL, USA). This was performed following mechanical stimulation to prevent potential alterations in RF properties by electrical stimulation. All measurements were obtained using the absolute minimum intensity required to excite neurons consistently without jitter. This variability (jitter) in the AP generation latency, particularly at significantly shorter latencies seen at two- to three-fold threshold intensity, has been presumed to reflect spread to more proximal sites along axons. Stimuli ranged in duration from 50 to 100 µs; utilization time was not considered. The distance was measured for each afferent on the termination of the experiment by inserting a pin through the RF (marked with ink at the time of recording) and carefully measuring the distance to the L4 DRG along the closest nerve (Figure 1(b)).
Statistical analysis
Before analysis, parametric assumptions were evaluated for all variables using histograms, identifying outliers with boxplots, descriptive statistics, and the Shapiro–Wilk test for normality. Data are reported as median (range or quartiles) if not normally distributed or mean (standard error) if normally distributed. Student’s t-test and repeated measures analysis of variance (ANOVA) were used for normally distributed data, and the Friedman test and Mann Whitney U-test were used for not normally distributed data. Changes in Em in A-HT over time were analyzed using repeated-measures ANOVA with Greenhouse & Geisser sphericity correction as distributions at each time point proved parametric, and there were no significant outliers. Friedman tests were run on the number of APs per stimuli and duration data as the distributions were non-parametric at one or more time points in each dependent variable. For all analyses, p was set at 0.05 for statistical significance. All post hoc analyses were Bonferroni adjusted. Analyses were conducted using SPSS Statistics for Windows, version 22 (IBM Corp, Armonk, NY), and OriginPro 9.5 (Northampton, MA).
Results
Effects of MOC2-PNI on the animal’s behavior
Reflexive. Paw withdrawal threshold (PWT)
Thirty-two mice (male 16/32, female 16/32) from both breeds (WT 8/16 and KO 8/16) were used 3 weeks after MOC2 implantation or Sham surgery. As presented in Figure 2(a), it has grouped both genders and shown that PNI induces a significant (p < 0.001) reduction in the PWT in both strains. However, the WT Sham group also showed a significant (p < 0.001) effect of the surgical procedure (contralateral (C) vs ipsilateral (I)). This effect was not observed in the KO Sham group.
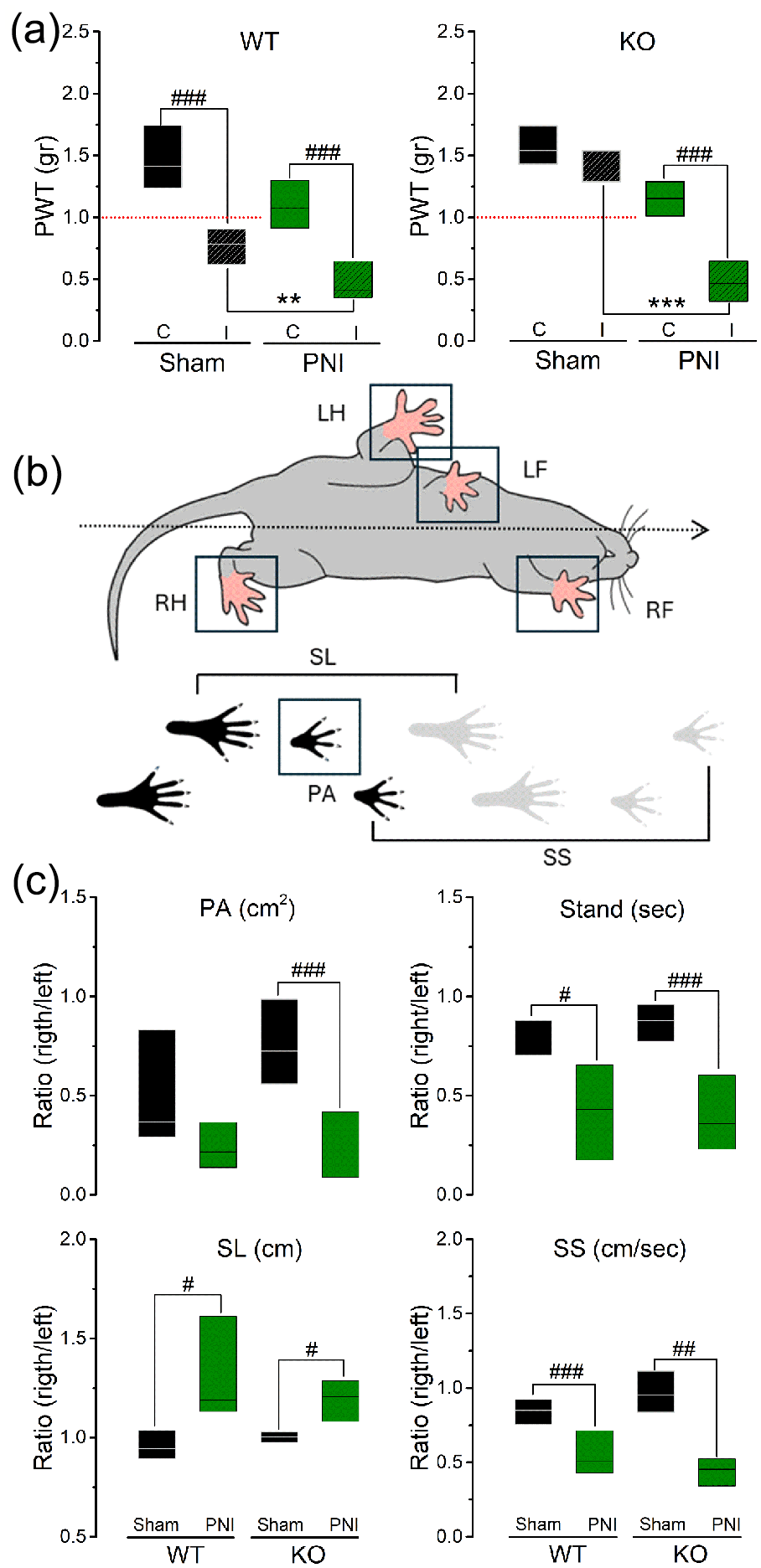
MOC2-PNI affects both reflexive and non-reflexive behavior of WT and KO animals. (a) Effect of MOC2-PNI in the paw withdrawal threshold of WT and KO animals. Males and females were included in the experimental groups (Sham and PNI), and both limbs were studied (C: contralateral; I; ipsilateral). (b) Schematic of the Gait parameters (LH: left hind paw, RH: right hind paw, LF: left front paw, RF: right front paw) included in this study (SL: stride length, SS: swing speed, PA: paw area). (c
Non-reflexive (gait)
As shown in Figure 2(b), several components of the mice’s gait and locomotor behavior were analyzed. These parameters included print area (PA, cm2), stance time (Stand, seconds), stride length (SL, cm), and swing speed (SS, cm/s). All these parameters were significantly (from p < 0.05 out to p < 0.001) affected in both strains, with one notable exception for the PA on WT (Figure 2(c)).
Effect of MOC2-PNI on sensory neurons innervating the L4 dermatome
Following the behavior studies (reflexive and non-reflexive), the animals were processed for electrophysiological analysis. As indicated in the Methods section, this analysis focused on several aspects of the afferents innervating the L4 dermatome in both genders (males and females) and strains (WT and KO). The results of this analysis have been grouped to provide a comprehensive description of the effects of MOC2-PNI on A) the sensory peripheral terminals innervating the dermatome (distribution per modality (%), mechanical sensibility (mN), conduction velocity (m/s)) and B) Its effect on the somatic electrical signature of recorded afferents (somatic membrane-active and passive properties). A total of 365 afferents were recorded from the L4 ganglia of the above-mentioned 32 animals (males 183/365 and females 182/365).
Effect of MOC2-PNI on the afferents peripheral terminals
Except for MS afferents, no spontaneous activity was observed. Afferents responded to mechanical stimulation only, and no detectable PDH was observed in the collected nociceptive afferents regardless of CV (A- or C-).
Distribution per modality
As presented in Figure 3(a) and (b), the percentage of available mechanosensitive afferents (LT, A-HT, C-HT, and MS) was higher in Sham animals than those with PNI (Males WT Sham: 70%, Males KO Sham: 85%, Females WT Sham: 76%, Females KO Sham: 66%). The presence of MOC2-PNI severely reduced the mechanosensitive afferents in both genders, particularly affecting the KO animals (Males WT PNI: 54%, Males KO PNI: 25%, Females WT PNI: 34%, Females KO PNI: 25%). This reduction in the available mechanosensitive afferents occurs in a non-modality-specific manner, concurrently increasing the number of mechano-insensitive afferents (F-type and S-type). Notably, muscle spindles (MS) seem unaffected in males, while their presence in the afferents distribution is reduced in females.

MOC2-PNI disrupts the peripheral mechanosensory system by changing the balance between tactile (LT) and nociceptive (HT) afferents availability and sensibility (distribution switch). Effect of MOC2-PNI on (A and B). Distribution (%) of L4 recorded afferents per gender and strain. (C) and (D) Mechanical threshold (MT) of subtypes of afferents innervating the L4 dermatome per gender and strain. (E and F) Conduction velocity (CV) of the recorded mechanosensitive afferents per gender and strain. Data are medians (vertical bars) with boxes representing the 25 and 75 percentiles. Dotted lines represent the maximal tactile and minimal nociceptive observed sensibility. Circles represent maximal and minimal values. #: compare among groups, * compare between groups. #/*: p < 0.05, ##/**: p < 0.01, ###/***: p < 0.001.
Mechanical sensibility
In both genders and strains, sham animals’ nociceptive afferents were found to have mechanical thresholds (MT) above 13.7 mN, while tactile afferents were below 0.7 mN without overlap between modalities regardless of the afferents conduction velocity (Figure 2(c) and (d)). MOC2-PNI induces a significant (p<0.05) increase in the overall nociceptive sensibility while desensitizing tactile afferents. This tactile desensitization effect is particularly important in males KO (p < 0.001) and females (WT and KO, p < 0.01), inducing a complete sensibility overlap or even crossover between modalities in these animals.
Conduction velocity (CV)
As presented in Figure 3(e) and (f), the afferents (tactile and nociceptive) collected from Sham animals (both genders and strains) show CV raking Aβ (above 11.5 m/s) and Aδ (above 1.2 m/s). MOC2-PNI induces a significant (p < 0.05 to p < 0.001) reduction on the CV of the recorded A-afferents (tactile and nociceptive), eliminating the Aβ signaling (cells above 11.5 m/s). This reduction affects both tactile and A-HT afferents in an equally significant (p < 0.01) magnitude but without fully invading the C-afferents conduction range (below 1.2 m/s).
Discussion
This study has established that cancer PNI induces profound changes in the somatosensory system’s activity. These changes include (but are not restricted to) modulating the afferents’ availability, sensibility, and electrical activity in a modality-specific manner.2,8,17 Arguably, these changes parallel the human patient’s symptomatology but often fail to match its magnitude. The principal observations of our studies are that 1) the allograft model of MOC2-PNI successfully recreates several aspects of the sensorial neuropathy and symptomatology as observed in human patients, 8 2) that the absence of TNFR1 does not alleviate this MOC2-PNI-induced sensorial neuropathy and 3) that gender is a fundamental factor driving both the magnitude and prevalence of this neuropathy. 2 We have also observed that MOC2-PNI induces peripheral deactivation (silencing) that is not caused by a degenerative process (cell conduction block or apoptosis) (see Part 2), as observed in several animal models of peripheral injury. 17
Together, our observations addressed several fundamental questions related to the pivotal role of the peripheral somatosensory system plasticity in the genesis and maintenance of neuropathic pain states. Our results also suggest that, unlike humans, mice can control peripherally driven hypersensitivity by reducing the effective number of hyperexcitable nociceptive afferents. These results allow us to speculate on the role of the observed modulation of different modalities (tactile and nociceptive) and their impact on the spinal circuitry integration of somatosensory information.
Technical considerations
Although the number of cells recorded per experiment is appropriate, the behavioral data must be interpreted cautiously (due to the limited number of animals used as a consequence of the technical difficulty of the intraneural injections). This study focused on the cellular consequences of MOC2-PNI, and the experimental design of our behavior study was not powered to detect meaningful behavioral differences between genders. Furthermore, and to be comparable to similar studies, only the mechanical sensibility of the afferents was studied. While helpful, these data only partially describe the likely greater peripheral effects of MOC2-PNI at advanced stages of the disease. MOC2 is an aggressive cell line; only one time point was used for data collection. Previously, in an immunodeficient model of sciatic nerve PNI using a slowly growing human cell line HSC-3, 2 sham surgery did not induce long-lasting nociceptive behavioral alterations, suggesting that the pain induced by sham in this study could be due to an immune response or accidental nerve damage induced by sham operations that likely obscured the behavioral outcome. However, this caveat still supports our interpretation of the generalizability of sex differences in electrophysiological responses to nerve injury, regardless of the nature of the injury.
Effects of MOC2-PNI on the somatosensory system and pain perception
Does the PNI injury model induce spontaneous pain behaviors?
Curiously, data on the relation between specific pain behavior and pain intensity in human patients is often rare. 20 However, the importance of guarding is associated with self-reported pain intensity 21 and may even be a contributing factor to persistent pain in humans. 22 In animals, spontaneous pain is particularly difficult to measure and largely unchanged in different models of nerve injury and tissue damage. 23
Far less explored is the common observation that mild injury triggered instinctive responses (in humans and animals alike), such as wound-licking and rubbing, and how these behaviors are related to rapid control of pain perception.24,25 This area of research is in dire need of further exploration, as the literature on the matter is rather scattered, and the general interpretation has very little relation to the peripheral system activation or the time frame of these behaviors (usually triggered rapidly after injury). Available information relates these instincts to the healing properties of the saliva 26 or a distraction (mediated by central processing) 27 as the substrate for its analgesic properties, despite its consistency and rapid onset across the mammalian evolutive line.
From a peripheral activation standpoint, both behaviors (wound-licking and rubbing) are expected to correlate with the magnitude of the injury. Indeed, in our previous study on PNI and using a less aggressive cancer cell line (HSC-3), we observed both. 2 However, in the current study, only guarding was present (although not quantified). Our reflexive studies did not capture this divergence between cell-line models. The PWT was similar regardless of the cell line model (HSC-3 vs MOC2) used and tumor size (MDB personal observation). On the other hand, the non-reflexive data suggested the presence of mechanical allodynia, which corroborates the presence of the injury generated by PNI (both cell lines). However, in both cases, we also noticed very few changes in the animal’s normal feeding patterns or general mobility, which raises the question of whether our models capture the real magnitude of the pain as perceived in humans with similar injuries (central vs peripheral compartments). 28 This observation significantly impacts our understanding of human pain perception because it jeopardizes pre-clinical studies’ translatability.
Does the animal behavior correlate with the peripheral somatosensory disruptions?
This is an important question for two reasons: a) it has been suggested that while many peripherally-induced neuropathic conditions are initiated by damage to the peripheral nervous system (peripheral compartment), their persistence is suggested to be induced by relying on maladaptive processes within the central nervous system (central compartment) 28 and b) despite the above-mentioned limitations of behavioral studies in dealing with this dichotomy, they are often used as the sole mean to evaluate pain pathologies in animal models. 28
The cornerstone of studies aiming at the peripheral component of pain perception has remained unchanged for decades. As such, it is widely believed that peripheral terminals of pain processing are unmyelinated C-fibers and thinly myelinated Aδ that spur pain after damage (tissular or metabolic), leading the cells to hyperexcitability and altering channel/receptors expression and composition,30–34 resulting in ectopic firing and faulty signal transmission. 35
Based on this overall description and the degenerative, traumatic nature of the PNI models, in our initial attempt to study this model, 2 we expected to find plenty of slow-conducting nociceptive afferents (C- and Aδ) with spontaneous activity (SA). However, no SA was detected in our recordings. Among many other possibilities, these somehow surprising results are attributed to the type of cell line used (HSC-3) and not the model. However, in this study, we used a similar (but more aggressive) cell line (MOC2), and we corroborated those results. Again, no SA was found, and likewise, the density of effective mechano-nociceptive afferents (C- and A) deteriorated (%) to the point where only a few recorded afferents were still mechanosensitive. This effect, particularly important in female WT and KO animals, occurred in a manner analogous to our initial study 2 and the phenomenological effects of postherpetic neuralgia, 31 still-active nociceptive afferents were deeply sensitized (to mechanical stimulation) but not spontaneously active.
In addition, we were also able to corroborate the effects of the pathology on the non-nociceptive afferents (tactile). As observed using the HSC-3 cell line, 2 but in a greater magnitude, MOC2-PNI also induced a reduction in the availability (%) of low-threshold mechanosensitive afferents (Female WT and KO animals) concurrent with a notable increase in their activation thresholds (desensitization).
Together, the ePhys observations highly correlate with the behavioral data. As such, both parts of the study indicate that sham WT animals were affected by the surgery differently from the KOs. These effects were greater in female animals (ePhys data only), and the absence of TNFR1 receptors (KO animals) does not offer any detectable advantage to the animals, except for more resiliency to the mild effect of the surgical procedure itself.
Conclusions
Overall, this study shows no benefit in the absence of TNFR1 in protecting the peripheral system against the effect of advanced MOC2-PNI. However, it does indicate that this receptor may be important in the initial development of the disease or after mild injury.
The current study also corroborates the presence of extreme peripheral plasticity (tactile desensitization with concurrent nociceptive sensitization) in our MOC2-PNI model. This peripherally mediated modulatory process will likely condition the spinal integration, the underlined spinal circuitry, and central nervous system response to injury. Due to its preservation across several injury models, corroborating its existence in human patients is paramount.
Footnotes
Acknowledgements
In the loving memory of Rafaela Endara-Maldonado and Juan Bernardo Boada-Bustos.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Supported by grants R01 DE029493 to YY and 1P01NS119159-01A1 to MDB from the National Institutes of Health, Bethesda, MD, USA.
