Abstract
This study investigated the role of brain-derived neurotrophic factor (BDNF) in patients with degenerative lumbar stenosis, focusing on its expression and correlation with pain intensity. The study examined 96 patients with lumbar stenosis and 85 control participants. BDNF levels in the yellow ligamentum flavum were measured using reverse transcription quantitative polymerase chain reaction (RT-qPCR), enzyme-linked immunosorbent assay (ELISA), and western blot analysis. The results showed significantly higher BDNF expression at both messenger ribonucleic acid (mRNA; fold change = +1.35 ± 0.23; p < 0.05) and protein levels in patients (28.98 ± 6.40 pg/mg) compared to controls (4.56 ± 1.98 pg/mg; p < 0.05). Furthermore, BDNF levels correlated positively with pain intensity reported by patients, with higher expression observed in those experiencing more severe pain. The study also explored the influence of lifestyle factors, such as smoking and alcohol consumption, and related diseases, such as diabetes, on BDNF expression. Smoking, alcohol use, and diabetes were associated with significantly elevated BDNF levels (p < 0.05). These findings suggest that BDNF could serve as a biomarker for pain severity in degenerative lumbar stenosis at the protein level, although this was not consistently observed at the mRNA level; this highlights the potential for BDNF-targeted therapies in managing pain. Future research should involve larger longitudinal studies to validate these findings and explore therapeutic interventions. This study underscores the importance of considering molecular and lifestyle factors in the treatment of degenerative lumbar stenosis, aiming to improve patient outcomes through comprehensive, targeted approaches.
Keywords
Introduction
Lumbosacral (L/S) spine stenosis is the narrowing of the spinal canal, resulting in compression of the neural and vascular elements within the lumbar spine.1,2 Clinical symptoms are characterized by pain in the buttocks and lower limbs; in some cases, lower back pain may also occur.3,4 Neurogenic claudication, which is characteristic of lumbar stenosis, occurs during standing and walking, and subsides in sitting, lying, and forward-bending positions.4–6
Lumbar stenosis most commonly affects the L4/L5 level, followed by L3/L4 and L2/L3, while the L5/S1 and L1/L2 spaces are less frequently involved. 7
Patients with both central stenosis and lateral recess stenosis complain of resting and nighttime pain, as well as pain during sneezing. Many patients who have not developed significant neurological symptoms can be treated conservatively with rehabilitation and analgesics. Surgical treatment is reserved for patients who are resistant to pain management and have increasing disability and neurological symptoms.8,9
Surgical treatment of lumbar stenosis involves decompressing neural elements. For optimal decompression, a decompressive laminectomy is commonly used, which involves removal of the vertebral arch and the yellow ligament, trimming of the joint, and widening of the nerve root canal.8,9 The clinical manifestation of degenerative stenosis of the L/S spine also has a molecular basis. 10 It is well documented that various bioactive compounds, including brain-derived neurotrophic factor (BDNF), glial cell line–derived neurotrophic factor (GDNF), substance P, cyclooxygenase 2 (COX2), prostaglandin E2 (PGE2), and Immediate Early Gene 6 (IE-6), are involved in neuropathic pain.11,12 In addition, neurotrophic factors have been investigated as neurotransmitters implicated in the processes of pain generation, modulation, and nerve injury repair. 13
Nevertheless, to the best of the authors’ knowledge, changes in the BDNF expression pattern in the yellow ligament of humans have not been previously evaluated. Only Li et al. 14 assessed BDNF expression in the dorsal root ganglia of a lumbar spinal stenosis model in rats. Most studies on BDNF have focused on degenerative changes within the knee joints.15–17
BDNF (also known as ANON2 or BULN2), along with ex vivo inflammatory and nociceptive factors that induce neuronal plasticity and can actively diffuse, leading to neonephrosis and pain in vivo.18–21 BDNF is a neurotrophin synthesized in central and peripheral nervous system cells. It is crucial for neuron development, growth, memory processes, apoptosis, neurogenesis, and neuroregeneration. 22 It belongs to the neurotrophin family, which includes NT-3, NT-4/5, NT-7, and glial cell–derived neurotrophic factor, all produced in the brain and other tissues. 23
Notably, BDNF activity significantly depends on other neurotrophic factors, such as NT-3 and NT-4/5, which also exhibit neuroregenerative properties.18,24 The synergistic interaction between NT-4/5 and BDNF in dopaminergic neuron cultures has been shown to enhance neuron survival more than BDNF alone.13,25 These growth and neurotrophic factors are crucial in modeling and producing pain, including discogenic pain.13,25 For instance, Maynard et al. 25 demonstrated the significant role of neurotrophic factors in discogenic pain induction and intensification in advanced intervertebral disc (IVD) degeneration stages.
BDNF mRNA expression in the peripheral nervous system increases during damage, both in motor neurons and Schwann cells in the distal part of the severed nerve stump, persisting only for the initial days after the damage ceases. 26 BDNF also promotes increased macrophage and mast cell migration into inflamed IVD, aiding in the repair processes of the damaged annulus fibrosus. 27 Experimental studies have shown that peripheral nerve damage induces BDNF expression at the damage site, promoting neuron regeneration. BDNF is initially produced in an inactive form, pro-BDNF, which is enzymatically converted by plasmin and metalloproteinases MMP-2 and MMP-9 into the mature BDNF (mBDNF) protein. 28
Therefore, this study aimed to evaluate the variations in BDNF concentrations in the yellow ligamentum flavum of the L/S section based on the severity of degenerative changes, pain intensity, habits, lifestyle factors, and comorbidities.
Materials and methods
This study was built on the work carried out in our previous papers.29–31
Ethical considerations
Approval for this study was granted by the local Bioethics Committee at the District Medical Chamber in Kraków (224/KBL/OIL/2022). All procedures adhered to the guidelines outlined in the 2013 Declaration of Helsinki, and patient data were pseudonymized. Written, voluntary consent was obtained from each participant before their inclusion in the study group. For the control group, clinical material was obtained postmortem in accordance with the Act of July 1, 2005, on the Collection, Storage, and Transplantation of Cells, Tissues, and Organs (Journal of Laws of 2020, item 2134). Article 5 of this Act operates on an opt-out basis for organ donation. 32
Characteristics of the study group participants
The study group comprised 96 patients (46 women, or 48%; 50 men, or 52%) with an average age of 68.3 ± 2.4 years who were scheduled for extended fenestration and foraminotomy neurosurgical procedures. The inclusion and exclusion criteria are detailed in Table 1. Participants in the experimental group self-reported their smoking and alcohol consumption status, indicating whether they currently smoked or consumed alcohol. However, no additional information was gathered on the duration or amount of consumption. Spinal stenosis of the L/S section was diagnosed based on clinical interviews, physical examinations, and MRI results in various sequences and planes, utilizing 3 mm and 4 mm slices.
Inclusion and exclusion criteria for the study group.

Pain assessment in the study group
Pain intensity in the study group was evaluated using a 10-point visual analog scale (VAS), with 0 indicating no pain and 10 indicating severe pain. None of the patients reported experiencing pain levels between 0 and 3 on the VAS scale. A total of 19 patients experienced level 4 pain, 22 patients experienced level 5 pain, 23 patients experienced level 6 pain, 9 patients experienced level 7 pain, 8 patients experienced both levels 8 and 9 pain, and 7 patients experienced level 10 pain.
Description of neurosurgical procedure
Extended fenestration and foraminotomy were performed under general endotracheal anesthesia. A skin incision was made over the affected area, followed by dissection of the paraspinal muscles and removal of the hypertrophied ligamentum flavum using Kerrison bone biters. Foraminotomy and decompression of the dural sac and nerve roots were conducted, followed by saline irrigation and skin suturing. The procedures were assisted by an operating microscope. Patients without early postoperative complications were discharged on the third day postsurgery, with a follow-up scheduled at the Neurosurgical Outpatient Clinic 4 weeks later.
Characteristics of the control group participants
The control group included 85 participants (39 women, or 46%; 46 men, or 54%), with an average age of 49.17 ± 2.65 years. Control samples were obtained during forensic autopsy or organ donation, adhering to the criteria outlined in Table 2. For the control postmortem samples, we know which individuals smoked, consumed alcohol, or had diabetes; however, we did not have detailed information on the duration or quantity, such as how long they smoked, the frequency or amount of alcohol consumed, or specifics about their diabetes management. Hematoxylin & eosin (H&E) staining was used to confirm the absence of degenerative changes, with two neurosurgery specialists (D.S. and R.S.) independently qualifying ligamentum flavum samples for the control group.
Inclusion and exclusion criteria for the control group.

Securing of collected material for molecular testing
Ligamentum flavum samples from both groups were thoroughly washed and placed in sterile Eppendorf tubes with RNAlater reagent (Invitrogen Life Technologies, Carlsbad, CA, USA), and then stored at −80°C until molecular analysis commenced.
RNA extraction and assessment
Total RNA extraction was performed using a modified Chomczyński-Sacchi method with TRIzol reagent (Invitrogen Life Technologies, Carlsbad, CA, USA). Samples were homogenized (T18 Digital Ultra-Turrax, IKA Polska Sp. z o.o., Warsaw, Poland), incubated with TRIzol (Invitrogen Life Technologies, Carlsbad, CA, USA) and chloroform (POL-AURA, Dywity, Poland), and centrifuged to separate the RNA. Isopropyl alcohol (POL-AURA, Dywity, Poland) was added for RNA precipitation, followed by washing with 70% ethanol. RNA isolates were purified using DNAse I and the RNeasy Mini Kit (Qiagen, Valencia, CA, USA), dried, and stored at −80°C.
Qualitative and quantitative assessments of RNA were conducted using agarose gel electrophoresis stained with ethidium bromide at a concentration of 0.5 mg/mL (Sigma-Aldrich, St. Louis, MO, USA) and spectrophotometry (Nanodrop, Thermo Fisher Scientific, Waltham, MA, USA), respectively. The correct results showed visible bands corresponding to 28SrRNA and 18SrRNA, with RNA purity ratios between 1.80 and 2.00.
Reverse transcription quantitative polymerase chain reaction (RT-qPCR)
RT-qPCR was performed in a 50 µL reaction mixture with specific thermal profiles: reverse transcription (45°C, 10 min); polymerase activation (95°C, 2 min); and 40 three-step cycles, comprising denaturation (95°C, 5 s), hybridization (60°C, 10 s), and annealing (72°C, 5 s). Primers for BDNF were as follows: Forward: 5′-AATGGGTTTAAGGTAGGTTTAAGAG-3′; Reverse: 5′-TTTTATATTCCTCCAACAAAAAAAA-3′. Beta-actin (ACTB) (Forward: 5′- TCACCCACACTGTGCCCATCTACGA-3′; Reverse 5′-CAGCGGAACCGCTCATTGCCAATGG-3′), and 18S rRNA (forward: 5′ CGGACAGGATTGACAGATTGA 3′, Reverse: 5′ GCCAGAGTCTCGTTCGTTAT 3′), served as an endogenous control. The RT-qPCR reaction was conducted using Sensi-Fast One-Step Probe Assay reagents (Bioline, London, UK). We used a one-step RT-qPCR protocol for cDNA analysis with 50 ng/µL RNA input per reaction to ensure consistency. Relative mRNA expression was calculated using the 2−∆∆Ct method. A value greater than 1 indicates upregulation of the target gene, and a value less than 1 indicates downregulation in the experimental sample compared to the control.
Test enzyme-linked immunosorbent assay (ELISA)
Samples of yellow ligamentum flavum were collected from both the study group patients and the control group participants. These samples were finely minced with a scalpel, placed in new Eppendorf tubes, and weighed. These prepared samples were then incubated with a solution containing 4M guanidine hydrochloride (Sigma-Aldrich, St. Louis, MO, USA), 1M sodium acetate (Sigma-Aldrich, St. Louis, MO, USA), 2% Triton (Sigma-Aldrich, St. Louis, MO, USA), and a protease inhibitor cocktail (Sigma-Aldrich, St. Louis, MO, USA) for 12 h at 4°C on a laboratory rocker. After incubation, the samples were centrifuged, and the supernatant was stored at −20°C for subsequent analysis. For the ELISA, a polyclonal anti-BDNF antibody (bs-4989R, STI, Poznan, Poland; 1:500 dilution) was used according to the manufacturer’s instructions. A sample without the primary anti-BDNF antibody served as a negative control to rule out nonspecific or autofluorescence signals, while a human HeLa cervical carcinoma cell line was used as a positive control. Each sample was analyzed in triplicate, and the mean values were used for analysis. Comprehensive protocols for the ELISA and subsequent western blot procedures were previously published.33,34
Western blot analysis
Initially, yellow ligamentum flavum samples were collected from both study group patients and control group participants. These samples were rinsed with Phosphate-Buffered Saline (PBS) solution and placed into new Eppendorf tubes. Each tube received 0.50 mL of radioimmunoprecipitation assay (RIPA) buffer supplemented with a cocktail of protease and phosphatase inhibitors (all from Sigma-Aldrich, St. Louis, MO, USA). The samples were homogenized using a hand-held homogenizer (T18 Digital Ultra-Turrax, IKA Polska Sp. z o. o., Warsaw, Poland) until no solid fragments remained. Following homogenization, the tubes were placed on ice and gently rocked for 60 min. The samples were then centrifuged, and the supernatant was collected and stored at −80°C for further analysis.
Upon thawing, the total protein concentration in the samples was determined using a bicinchoninic acid (BCA) assay kit (Thermo Fisher, Waltham, MA, USA) according to the manufacturer’s instructions. Protein concentrations varied between 20 and 100 μg of total protein. Measurements were calculated using a standard curve based on bovine serum albumin (BSA) standard solutions, comprising six standard points (0, 250, 500, 1000, 1500, and 2000 µg/mL; Sigma-Aldrich, St. Louis, MO, USA).
A polyclonal anti-BDNF antibody (bs-4989R, STI, Poznan, Poland; molecular weight 14 kDa; 1:300 dilution) was employed as per the manufacturer’s protocol. B-actin (ACTB; Santa Cruz Biotech, Dallas, TX, USA, molecular weight 37 kDa; catalog number sc-25778; 1:500 dilution) was used as the endogenous control protein. The secondary antibody used was HRP-conjugated goat antirabbit IgG (BioRad, Milan, Italy; catalog number 1706515; 1:3000 dilution).
Equal amounts of protein (20 µg) were separated by sodium dodecyl sulfate polyacrylamide gel electrophoresis (POL-AURA, Dywity, Poland). The proteins were then transferred onto a polyvinylidene difluoride (PVDF) membrane (0.45 µm pore size, Thermo Fisher, Waltham, MA, USA). The optical density of each blot lane was measured using Kodak MI 4.5SE software (Kodak, Rochester, NY, USA).
Samples of yellow ligamentum flavum that did not include the primary mouse monoclonal IgG1 κ pro BDNF antibody served as a negative control, while a human HeLa cervical carcinoma cell line was used as a positive control.
Immunohistochemical (IHC) analysis
Tissue specimens were sliced at a thickness of 8.0 µm using a microtome (Leica Microsystems, Wetzlar, Germany). Subsequent processing steps, including dehydration, antigen retrieval, antibody incubations, and staining, were carried out according to the manufacturer’s guidelines provided in the instruction manuals for the DAB Substrate Kit (Peroxidase, HRP; Vector Laboratories, Newark, CA, USA) and the IHC-Paraffin Protocol (IHC-P; Abcam plc, Cambridge, UK).
The resulting immunohistochemical reactions were observed and captured using a Nikon Coolpix fluorescent optical system. The cellular localization and quantity of the selected proteins were analyzed through computer image analysis utilizing the ImageJ software. A total of 15 photographs were taken from 3 slides per patient under 200× magnification. Using the IHC-Profiler plug-in in ImageJ, the optical density of the DAB reaction products was assessed in areas where the immunohistochemical reaction occurred in response to the presence of the selected proteins. Additionally, the average percentage of the DAB-stained area was calculated relative to the background values in each field.
Statistical analysis
All statistical analyses were performed using the Statplus software (AnalystSoft Inc., Brandon, FL, USA), with significance set at p < 0.05. Data distribution normality was assessed with the Shapiro–Wilk test, and parametric tests followed accordingly. For group comparisons, one-way analysis of variance (ANOVA) and Scheffe’s post-hoc test were employed to identify differences in BDNF levels across VAS pain scores and Body Mass Index (BMI) categories, while a Student’s t-test was used for pairwise comparisons between binary groups (e.g. smokers vs nonsmokers). To explore the relationships between individual factors (VAS score, BMI, smoking, alcohol use, and diabetes) and BDNF levels, univariate linear regression was first applied for simple associations, followed by multivariate regression to assess the combined influence of these variables on BDNF expression. Interaction terms were included in the multivariate model to evaluate the potential moderating effects of lifestyle factors on the VAS–BDNF relationship. Additionally, multicollinearity was assessed to confirm the reliability of each predictor, and separate models were considered for the study and control groups to explore any differential impact of lifestyle factors on BDNF expression in patients with stenosis compared to controls.
Results
Expression changes in the mRNA BDNF in the control and test samples
At the mRNA level, we demonstrated BDNF overexpression in the examined samples compared to the control group (FC = +1.35 ± 0.23; p < 0.05). Subsequently, we assessed whether the transcriptional activity of BDNF mRNA was dependent on the severity of pain symptoms. In yellow ligament samples obtained from patients reporting pain levels of 4 and 5, BDNF expression was close to the control levels. This expression increased to FC = 1.57 ± 0.12 when patients reported a pain severity of 10 on the VAS scale. However, the ANOVA revealed that BDNF transcriptional activity did not vary significantly by pain severity (F(6,18) = 2.13, p > 0.05; Figure 1; p > 0.05).

Transcriptional activity of BDNF mRNA in the yellow ligamentum flavum obtained from the test group, including pain severity (RT-qPCR).
Expression changes in the protein levels of BDNF in the samples obtained by ELISA
At the protein level, we also observed significantly higher BDNF concentrations in ligaments obtained from the study group compared to the control group (F(6,95) = 83.20, p < 0.0001). Specifically, ELISA measurements revealed that the BDNF concentration in the yellow ligaments from the study group was 28.98 pg/mg ± 6.40 pg/mg, while in the control group, it was 4.56 pg/mg ± 1.98 pg/mg (p < 0.05).
Figure 2 illustrates the changes in BDNF concentrations in samples collected from the study group according to the severity of pain symptoms. We noted that BDNF concentration increased with the intensity of pain reported by patients (one-way ANOVA F(6,95) = 83.20, p < 0.05). A subsequent post-hoc test indicated statistically significant differences in BDNF concentrations between patients reporting pain intensity at level 4 compared to those reporting pain at level 6 and above (Figure 2; p < 0.05), as well as between patients with pain at level 5 compared to those at level 8 and higher (Figure 2; p < 0.05).

The concentration of BDNF in the yellow ligamentum flavum from the control and study groups, including the pain severity (ELISA results).
In addition, Table 3 summarizes the ELISA results, including the outcomes of the statistical analyses.
Concentration of BDNF in the study and Control groups, with differences based on VAS pain severity levels (ELISA results).

Data are presented as mean ± standard deviation.
Statistically significance differences between BDNF expression in the study and control groups (Student’s t-test).
Statistically significant differences between BDNF expression in VAS 4 and VAS 6 (Scheffe’s post-hoc).
Statistically significant differences between BDNF expression in VAS 4 and VAS 7 (Scheffe’s post-hoc).
Statistically significant differences between BDNF expression in VAS 4 and VAS 8 (Scheffe’s post-hoc).
Statistically significant differences between BDNF expression in VAS 4 and VAS 9 (Scheffe’s post-hoc).
Statistically significant differences between BDNF expression in VAS 4 and VAS 10 (Scheffe’s post-hoc).
Statistically significant differences between BDNF expression in VAS 5 and VAS 8 (Scheffe’s post-hoc).
Statistically significant differences between BDNF expression in VAS 5 and VAS 9 (Scheffe’s post-hoc).
Statistically significant differences between BDNF expression in VAS 5 and VAS 10 (Scheffe’s post-hoc).
statistically significant differences between BDNF expression in VAS 6 and VAS 9 (Scheffe’s post-hoc).
Expression changes in the protein levels of BDNF in the samples obtained by Western blot analysis
The expression profile of BDNF in degenerated and control yellow ligamentous flavum, determined using the western blot technique, was the same as that noted in the ELISA procedure. Figure 3 presents an example electropherogram that confirms the specificity of the reaction, as well as the nativity of the samples (based on the ACTB result; molecular mass of 42 kDa). The normalized optical density of BDNF (molecular weight 28 kDa) relative to ACTB in the study samples was 1.10 ± 0.67 compared to 0.39 ± 0.24 in the control samples, which was statistically significant (t(94) = 28.43, p < 0.05).
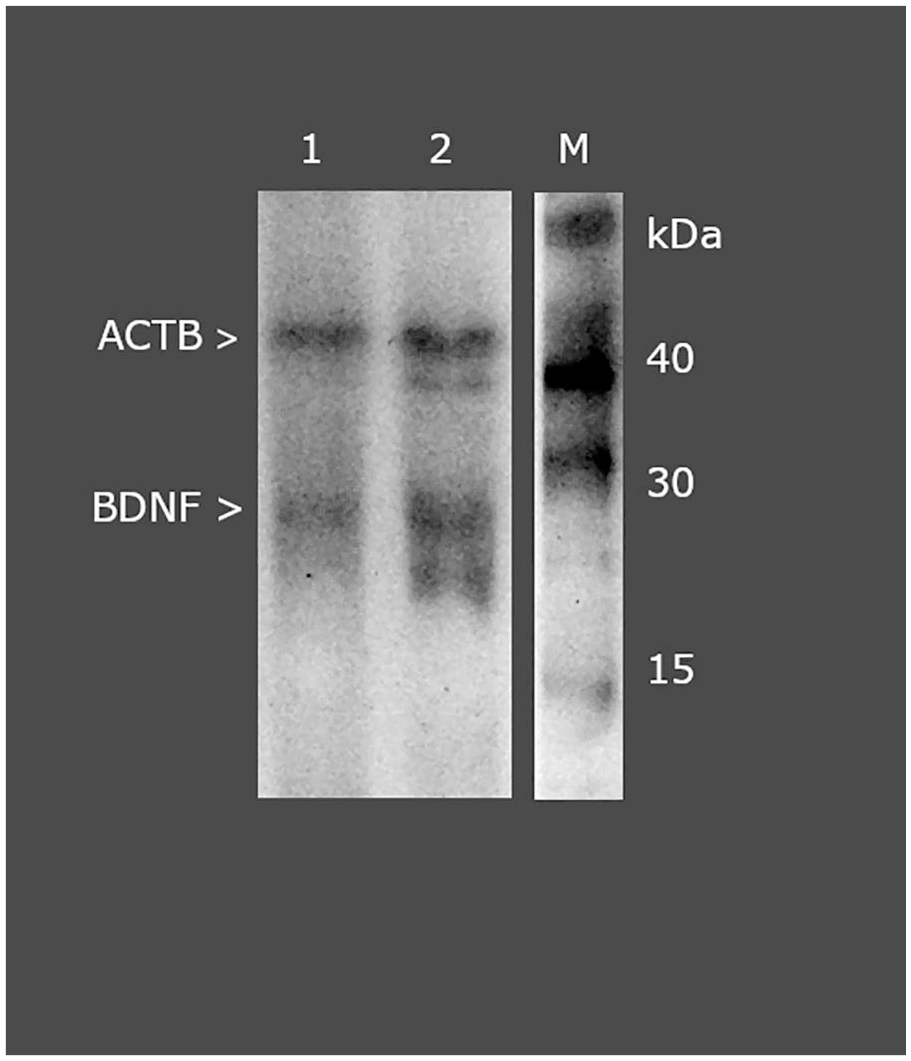
Normalized expression of BDNF in ligamentum flavum normalized against ACTB expression (western blot).
The normalized band optical density of BDNF in degenerated and control yellow ligamentum flavum is presented in Figure 4.
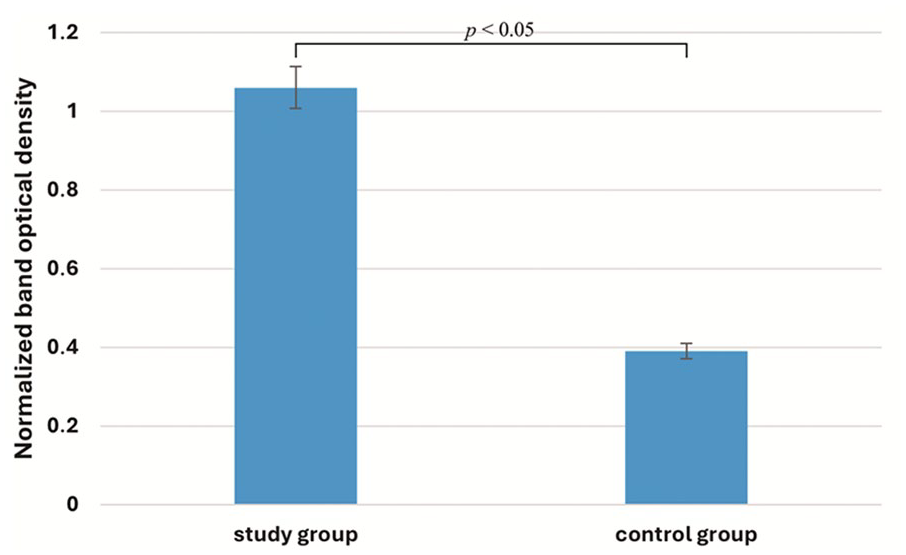
Band optical density of BDNF in L/S spine yellow ligamentous flavum collected from the study and control groups, determined using western blotting.
Differences according to lifestyle in the expression profiles of BDNF at the mRNA and protein levels in degenerated yellow ligamentous flavum samples
Table 4 presents BDNF expression profiles at the mRNA and protein levels in yellow ligamentum flavum samples from control and study groups, stratified by gender, BMI, diabetes, smoking, and alcohol consumption. In general, BDNF protein expression was markedly higher in the study group across all categories, indicating a substantial increase in the degenerated samples. For instance, females in the study group exhibited a protein level of 31.16 ± 3.45 pg/mg compared to 4.72 ± 1.85 pg/mg in controls. Obese individuals also showed notably elevated BDNF levels, particularly at the protein level, with the study group reaching 35.06 ± 6.22 pg/mg. Diabetes, smoking, and alcohol use were consistently associated with increased BDNF expression, with smokers in the study group demonstrating the highest protein levels at 42.17 ± 4.44 pg/mg. These findings suggest that lifestyle factors like obesity, diabetes, smoking, and alcohol consumption may amplify BDNF upregulation in degenerative conditions, with the study group displaying consistently higher expression profiles compared to controls.
Expression of BDNF at the mRNA and protein levels in yellow ligamentous flavum samples obtained from the study and control groups.

BDNF: brain-derived neurotrophic factor; BMI: body mass index.
, p-Value of student’s two-tailed t-Test.
Regression analysis of variables potentially associated with BDNF levels in yellow ligamentum flavum from the study groups
Table 5 summarizes the associations between various characteristics and BDNF expression levels at mRNA and protein levels in control and study groups using univariate and multivariate regression analyses. For gender, neither mRNA nor protein expression showed significant associations in either group, with low r-values and p-values above 0.3. In contrast, BMI demonstrated moderate to strong correlations, particularly in protein expression, with the study group showing a higher correlation (r = 0.76, R² = 0.58, p < 0.0001) than the control (r = 0.63, R² = 0.40, p = 0.0042). Diabetes was also significantly correlated with BDNF, showing a stronger relationship in the study group for both mRNA (r = 0.74, R² = 0.55, p = 0.021) and protein (r = 0.69, R² = 0.48, p = 0.028) compared to controls. Smoking and alcohol consumption were both strongly associated with increased BDNF expression in the study group, particularly at the protein level (smoking: r = 0.83, R² = 0.69, p = 0.022; alcohol: r = 0.92, R² = 0.85, p < 0.0001). Overall, lifestyle factors such as BMI, diabetes, smoking, and alcohol consumption were consistently more strongly correlated with BDNF expression in the study group, especially at the protein level, suggesting a compounding effect of these factors in degenerative conditions. Variables found to be insignificant using linear regression were not included in the multiple regression model.
Univariate and multivariate regression analyses of variables associated with BDNF levels in control and study group yellow ligamentum flavum.

BDNF: brain-derived neurotrophic factor; BMI: body mass index; r: correlation coefficient.
The multivariate regression analysis indicated a significant association between VAS pain scores and elevated BDNF protein levels (coefficient = 2.86, p < 0.001), highlighting pain severity as a primary factor influencing BDNF expression. Univariate analyses also showed positive associations between lifestyle factors – such as BMI, smoking, alcohol consumption, and diabetes – and BDNF protein levels, with each factor linked to elevated protein levels (p < 0.05 for each factor in Table 5). However, in the multivariate model, these lifestyle factors did not display statistically significant independent effects on BDNF protein levels when controlling for VAS pain scores (e.g. smoking: coefficient = 0.24, p = 0.807; alcohol: coefficient = 0.35, p = 0.721). This finding suggests that pain intensity primarily mediates the observed relationship between lifestyle factors and BDNF protein levels. Additionally, BDNF mRNA levels remained stable across lifestyle factors, with no significant changes associated with BMI, smoking, alcohol, or diabetes (p > 0.05 in Table 4). Therefore, while lifestyle factors correlate with BDNF protein expression, pain intensity emerges as the predominant factor driving BDNF protein level changes in this dataset (Table 6).
Summary of multivariate regression analysis on BDNF protein levels in relation to pain and lifestyle factors.

BDNF expression profile determined by IHC
The IHC analysis demonstrated visible BDNF expression in both the control and degenerated yellow ligamentum flavum samples, with distinct staining observed in the degenerated sections. Quantification of BDNF expression, measured by optical density, indicated a significant increase in the degenerated samples compared to the controls. The baseline optical density for BDNF in the control group was set at 100%, while the degenerated group showed an optical density reaching 135.72% of the control group, indicating a 35.72% increase in BDNF expression in the degenerated ligamentum flavum. Statistical analysis was performed using a Student’s t-test, confirming the significance of this difference with a p-value of <0.05. Figure 5 provides representative images of the IHC staining, illustrating this elevated expression in degenerated samples (Figure 5(b)) compared to controls (Figure 5(a)).

Immunochemical expression of BDNF in the control (a) and study (b) samples.
Discussion
Significant advancements in molecular biology have reshaped our approach to understanding and managing complex conditions, such as spinal degenerative stenosis.35,36 Our study assessed BDNF expression in the ligamentum flavum of patients with L/S degenerative stenosis, examining its association with pain severity. Our findings demonstrated significantly elevated BDNF levels in patients with higher pain scores (p < 0.05), positioning BDNF as both a marker and a potential mediator of pain in degenerative spinal conditions. 37 The elevated BDNF expression we observed highlights the role of BDNF in modulating neural plasticity, which is crucial in the context of pain. Cortical processing of pain involves substantial synaptic changes, with BDNF playing a pivotal role in enhancing neural connectivity in response to painful stimuli. Increased BDNF levels in the spinal dorsal horn and brainstem have been shown to amplify pain signaling, leading to heightened pain sensitivity, manifesting as hyperalgesia or allodynia.38,39 This aligns with Baumbauer et al., 40 who identified the BDNF SNP rs6265 (Val66Met) as linked to reduced synaptic BDNF release. Carriers of the minor allele (A) exhibit greater sensitivity to pain and require higher doses of analgesics. 40 This underscores the complex interplay between genetic factors and pain processing, which BDNF may drive at central levels. The potential of BDNF as a biomarker for pain is substantial, particularly given the limitations of subjective pain assessments, which vary with individual pain sensitivity and can be influenced by psychological and social factors. 41 Our findings suggest that BDNF could provide an objective measure of pain, supplementing patient-reported scores with biologically grounded insights. This could pave the way for a more standardized assessment of pain severity in degenerative spinal stenosis and may support the development of mechanism-based treatments, reducing dependence on opioids and enhancing patients’ quality of life.42–44
BDNF also plays a critical role in peripheral nerve regeneration. It was demonstrated that administering exogenous BDNF to injury sites in mice improved axon regeneration, while blocking BDNF in injured tissues limited connective tissue proliferation without impacting axon growth.45,46 These findings are significant, as they highlight BDNF’s role not only in supporting nerve repair, but also in maladaptive processes, such as pain sensitization in degenerative conditions. 47
Moreover, Orita et al. 48 found that injecting BDNF-neutralizing antibodies into damaged IVDs in mice reduced the number of Calcitonin Gene-Related Peptide (CGRP)-positive neurons in the dorsal ganglia, indicating a possible link between increased local BDNF and discogenic pain. 48 Our results align with those of Henry et al., 49 who found that elevated BDNF levels in degenerating disks correlate with higher concentrations of proinflammatory cytokines, such as IL-1β and Vascular Endothelial Growth Factor (VEGF).
These findings suggest that BDNF’s role in degenerative spinal pain may be partly mediated by inflammatory pathways, which are also known to exacerbate pain. The potential of BDNF as a biochemical marker for lumbar spine pain severity has been emphasized by Khan et al. 50 and Kartha et al., 51 who observed that BDNF could support tailored treatment approaches by helping gauge pain severity. Lee et al. 52 corroborated these findings by detecting BDNF expression in 24 out of 25 cases of degenerative lumbosacral disks; this suggests a consistent role for BDNF across degenerative tissues.
BDNF’s role across diverse tissue types highlights its involvement in various forms of pain, such as musculoskeletal and visceral pain. Fay et al. 53 suggested that BDNF elevation in degenerative tissues may reflect its release from other cells, such as thrombocytes, rather than an ongoing regenerative process. This reinforces the importance of examining BDNF as a broad marker of pain in degenerative conditions, which has been well documented in osteoarthritis studies.23,54,55 In osteoarthritis patients, higher BDNF levels have been correlated with pain severity, suggesting that BDNF may influence pain through inflammatory processes in the musculoskeletal system. 56 The presence of BDNF in bone fracture healing further underscores its relevance across multiple pain types. 57
Our findings indicate that lifestyle factors, including smoking, alcohol consumption, and diabetes, significantly influence BDNF expression. Patients with these lifestyle factors exhibited elevated BDNF levels compared to those without, suggesting that these behaviors may aggravate pain through BDNF-related mechanisms. Smoking, for example, is a well-documented risk factor for spinal degeneration and has been shown to exacerbate degenerative changes through oxidative stress and inflammation, which likely stimulate BDNF upregulation as a maladaptive response.58,59 Smoking impairs recovery from injuries, increases bone fragility, and is associated with both intervertebral disc degeneration and osteoporosis, compounding pain and spinal degeneration symptoms.50,60–66
Similarly, alcohol-induced dehydration can contribute to disk degeneration by reducing hydration in intervertebral discs, impairing their structural integrity, 67 and compounding the degenerative effects of poor dietary and sleep habits.68–70
Our findings that diabetic patients have higher BDNF levels also align with research linking diabetes to spinal degeneration. Mahmoud et al. 71 and Russo et al. 72 found that hyperglycemia in diabetic patients increases apoptosis and degradation in disk cells, accelerating intervertebral disc degeneration. In addition, studies by Park et al. 73 and Ruiz-Fernández et al. 74 revealed that metabolic and inflammatory dysregulation linked to obesity and diabetes can lead to an increased risk of spinal degeneration mediated by factors such as BDNF. The influence of body weight on BDNF levels, as noted by Seifert et al., 75 suggests that metabolic health and lifestyle interventions could be crucial in managing BDNF expression and associated pain.76–80
The clinical implications of BDNF as a therapeutic target are promising, especially in the context of reducing opioid dependency. Targeted interventions to modulate BDNF levels could provide a nonaddictive alternative for pain management. This could include pharmacological approaches that use BDNF-neutralizing agents to reduce its pronociceptive effects or lifestyle-based interventions to naturally decrease BDNF levels. 47 For example, studies have shown that interventions such as exercise or dietary adjustments may lower BDNF levels in chronic pain conditions, suggesting a multimodal approach to managing pain.15,75 As noted in Simão et al., 15 BDNF levels correlated with pain during functional activities, suggesting that BDNF may also be linked to movement-related pain exacerbations; this could inform activity-based therapy approaches for managing spinal pain. 15
Future research should investigate the longitudinal effects of BDNF modulation in patients with degenerative spine conditions, particularly to determine whether altering BDNF expression could slow degeneration progression. In addition, investigating the synergistic effects of BDNF with other neurotrophic factors, such as Nerve Growth Factor (NGF), and cytokines, such as IL-1β, may offer new therapeutic strategies that target multiple pathways in pain generation and inflammation. 57 Recent studies suggest that BDNF plays a role in metabolic regulation, influencing glucose and lipid metabolism, which could inform a more integrated approach to managing spinal degeneration in diabetic or obese patients.81,82 For instance, Duan et al. 83 observed that dietary restrictions increase BDNF secretion, suggesting that metabolic interventions could potentially regulate BDNF expression and, consequently, pain perception.
This study has several limitations that should be acknowledged. First, the sample size, while adequate for the initial findings, may limit the generalizability of the results. A larger cohort would provide more robust data and allow for more definitive conclusions. Second, the study is cross-sectional in nature, capturing data at a single point in time, which prevents the assessment of causality or changes in BDNF expression over time. Longitudinal studies are needed to understand the dynamics of BDNF levels and their relationship with pain progression and treatment outcomes. Third, the study relies on self-reported pain measurements, which can be subjective and influenced by individual pain tolerance and psychological factors. Incorporating objective pain assessment tools and biomarkers would strengthen the validity of the findings. Finally, while the study investigated the influence of various lifestyle factors on BDNF expression, it did not account for other potential confounding variables, such as genetic predispositions, medication use, or the presence of other comorbid conditions. Future research should aim to address these limitations to provide a more comprehensive understanding of the role of BDNF in degenerative lumbar stenosis and its potential as a therapeutic target.
Conclusion
This study provides significant insights into the role of BDNF in degenerative lumbar stenosis, particularly in relation to pain severity. Our findings demonstrate that BDNF expression is markedly elevated in the yellow ligamentum flavum of patients with lumbar stenosis compared to control subjects, and that this increase is positively correlated with the intensity of pain reported by patients. Additionally, lifestyle factors such as smoking, alcohol consumption, and the presence of diabetes significantly influence BDNF levels, highlighting the multifaceted nature of pain modulation in this condition. These results suggest that BDNF could serve as a valuable biomarker for assessing pain severity and potentially guiding treatment strategies. Moreover, targeting BDNF signaling pathways may offer new therapeutic avenues for managing pain in patients with degenerative spinal conditions. However, further research is needed to confirm these findings in larger longitudinal studies and to explore the therapeutic potential of modulating BDNF expression. Overall, this study underscores the importance of considering both molecular and lifestyle factors in the management of degenerative lumbar stenosis and opens new directions for research and treatment aimed at improving patient outcomes through targeted interventions.
Footnotes
Acknowledgements
We would like to thank Nikola Zmarzły for their assistance with the preparation of the figures.
Author contributions
Conceptualization, Dawid Sobański and B.O.G.; methodology, Dawid Sobański; formal analysis, R.S. and Dawid Sobański; resources, Damian Strojny and M.S.; data curation, Dawid Sobański and B.O.G.; writing – original draft preparation, Dawid Sobański, Damian Strojny, and B.O.G.; writing – review and editing Dawid Sobański, R.S., and B.O.G.; supervision, Dawid Sobański; B.O.G.; visualization, M.S. project administration, Dawid Sobański; B.O.G. All authors have read and agreed to the published version of the manuscript.
Consent to participate
Written, voluntary consent was obtained from each participant before their inclusion in the study group. This ensured that participants were fully informed about the nature, purpose, and procedures of the study, as well as their rights to withdraw at any time without consequence.
Consent for publication
Written, voluntary consent was obtained from each participant for the use of anonymized data collected during the study in academic publications. Participants were assured that no personal or identifying information would be disclosed.
Data availability statement
The data used to support the findings of this study are included in the article. The data cannot be shared due to third-party rights and commercial confidentiality.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical considerations
Approval for this study was granted by the local Bioethics Committee at the District Medical Chamber in Kraków (224/KBL/OIL/2022). All procedures adhered to the guidelines outlined in the 2013 Declaration of Helsinki, and patient data were pseudonymized. For the control group, clinical material was obtained postmortem in accordance with the Act of July 1, 2005, on the Collection, Storage, and Transplantation of Cells, Tissues, and Organs (Journal of Laws of 2020, item 2134). Article 5 of this Act operates on an opt-out basis for organ donation.
