Abstract
Preclinical studies on pathological pain rely on the von Frey test to examine changes in mechanical thresholds and the acetone spray test to determine alterations in cold sensitivity in rodents. These tests are typically conducted on rodent hindpaws, where animals with pathological pain show reliable nocifensive responses to von Frey filaments and acetone drops applied to the hindpaws. Pathological pain in orofacial regions is also an important clinical problem and has been investigated with rodents. However, performing the von Frey and acetone spray tests in the orofacial region has been challenging, largely due to the high mobility of the head of testing animals. To solve this problem, we implemented a sheltering tube method to assess orofacial nociception in mice. In experiments, mice were sheltered in elevated tubes, where they were well accommodated because the tubes provided safe shelters for mice. Examiners could reliably apply mechanical stimuli with von Frey filament, cold stimuli with acetone spray, and light stimuli with a laser beam to the orofacial regions. We validated this method in Nav1.8-ChR2 mice treated with oxaliplatin that induced peripheral neuropathy. Using the von Frey test, orofacial response frequencies and nociceptive response scores were significantly increased in Nav1.8-ChR2 mice treated with oxaliplatin. In the acetone spray test, the duration of orofacial responses was significantly prolonged in oxaliplatin-treated mice. The response frequencies to laser light stimulation were significantly increased in Nav1.8-ChR2 mice treated with oxaliplatin. Our sheltering tube method allows us to reliably perform the von Frey, acetone spray, and optogenetic tests in orofacial regions to investigate orofacial pain.
Keywords
Introduction
Pain in the orofacial regions, including the face, jaw, and mouth, is a significant clinical problem as it impacts speech, swallowing, eating, and facial expression. 1 Orofacial pain can be caused by various disorders, such as trigeminal neuralgia, temporal mandibular joints (TMJ) disorders, and chemotherapy-induced peripheral neuropathy (CIPN).1–3 Collectively, these disorders affect around 20% of the population, and pain severely reduces the quality of life in these patients.4,5 There is a knowledge gap in understanding the mechanisms of orofacial pain, which limits the development of effective treatments. Our understanding of the mechanisms of pain in the body heavily relies on rodent pain models and pain assessment methods. Classically, rodent pain assessments were primarily performed in the hindpaws of rats and mice, and reliable behavioral outcomes can be obtained to help understand pain mechanisms.6–8 In contrast to the pain assessment in hindpaws, the assessment of orofacial pain in rodents remains a challenge, and current methods have limitations, highlighting the need for better approaches for assessing orofacial pain in rodents.
Several rodent models of orofacial pain have been created, including the infraorbital nerve chronic constrictive nerve ligation (ION-CCI), CFA-induced inflammation in TMJ, and orofacial pain induced by chemotherapy drugs such as oxaliplatin.9–12 In these orofacial pain models, animals display mechanical and thermal hypersensitivity in their orofacial regions, resembling clinical orofacial pain in patients.7,13–15 Adopted from pain assessment in the hindpaw of rodents, previous studies have used the von Frey test to assess mechanical sensitivity and the acetone spray test to assess cold sensitivity in orofacial regions in rodents.7,8,16–19 For example, in a study performed in rats whose infraorbital nerve was partially transected, the acetone spray test revealed an increased duration of wiping behavior, suggesting a heightened cold sensitivity. 17 Also, heightened mechanical sensitivity was observed, as determined by the von Frey test. 17 We have previously shown in rats that treating animals with the chemotherapy drug oxaliplatin resulted in mechanical and cold allodinia and hyperalgesia.19,20 However, a challenge in performing sensory tests such as the von Frey and acetone spray tests on orofacial regions is the high mobility of the head of testing animals, making it extremely difficult to apply sensory stimuli to orofacial regions reliably. In attempting to address this issue, in a previous study, mice were held and habituated within the experimenter’s hand, and sensory tests were performed on orofacial regions after an extended acclimation period to reduce the mobility of animals’ heads. 21 However, holding testing animals in the experimenter’s hand restricts head movement and stresses animals, which impacts animals’ behavioral responses to orofacial sensory stimuli. In other studies, mice were placed in small wire-meshed cages or a cage with a gap at the center, and von Frey filaments were applied to orofacial regions for mechanical sensitivity.17,22,23 However, the wire-meshed cages make it difficult to access orofacial regions using von Frey filaments or the acetone spray. Additionally, the head mobility of the mice remains an issue in this experiment setting. In a different study, rats were positioned on a raised platform, and mechanical sensitivity was assessed by the von Frey test. 24 However, standing on an elevated platform is a stressful experience for an animal, which may impact orofacial behavioral responses. Additionally, head mobility remains an issue for an animal standing on an elevated platform, opening in every direction. Previous studies in our lab have used operant tests to measure changes in mechanical and cold sensitivity in the orofacial region of rats. 20 While reliable, the orofacial operant tests require rats to undergo a long training period, and mice seem more difficult to train for the operant test. Therefore, thus far, no experimental approach has been available to allow performing sensory tests on orofacial regions in rodents, particularly in mice, in a manner less stressful for animals, reliable for sensory stimuli, and convenient for experimenters.
In the present study, to overcome the aforementioned technical limitations, we implemented a new method, the sheltering tube method, to study behavioral responses to mechanical stimulation with von Frey filaments and cold stimulation with acetone spray in the orofacial regions of mice. We used the NaV1.8-ChR2 mouse line, 25 which allowed us to perform an optogenetic test to stimulate NaV1.8-expressing trigeminal nerves using blue laser light. Furthermore, using the sheltering tube method, we investigated changes in mechanical, cold, and light sensitivity in the orofacial regions of the NaV1.8-ChR2 mice treated with the chemotherapy drug oxaliplatin.
Materials and methods
Animals
Heterozygous Nav1.8-ChR2 mice were generated by crossing Scn10aCre and Ai32(RCL-ChR2(H134R)/EYFP) transgenic mice. Scn10aCre mice were gifts from Dr John Wood at University College London and transferred to us from Dr Stephen Waxman’s lab at Yale University. Ai32 mice were purchased from Jackson Labs. The mice used in the present study were aged between 13 and 21 weeks, and only male mice were included. All animal care and experimental procedures were conducted following the guidelines established by the National Institutes of Health (NIH) for the care and use of experimental animals. Approval for the experimental protocols employed in this study was granted by the Institutional Animal Care and Use Committee (IACUC) at the University of Alabama at Birmingham.
Animals were divided into oxaliplatin-treated and control groups. For the oxaliplatin-treated group, each mouse was injected with oxaliplatin intraperitoneally at a dose of 2 mg/kg for five consecutive days (an accumulative dose of 10 mg/kg), a dosing regimen adopted from a previous study. 20 The mice in the control group were injected with saline. One day before orofacial behavioral tests, the hair in mice’s orofacial regions and whiskers was trimmed off with a trimmer and shaved with a razor. The reason for trimming off the whiskers was to avoid the interference of whiskers when stimuli were applied to orofacial regions.
The setup of sheltering tubes for orofacial behavioral assessment
The sheltering tubes each consist of a one-end closed cylinder plastic tube with an inner diameter of 3.8 cm and a depth of 11 cm. The actual depth of the sheltering tube was adjusted to 5.6 to 3.5 cm by adding the lids of Falcon tubes so that the sheltering tubes could accommodate mice of different body sizes for orofacial behavioral assessment. Each sheltering tube was mounted on a stand placed on a lab bench, with the sheltering tube slightly tiled 15° upward. Mice underwent two sessions, each 45 min, habituation in two consecutive days. In the first habituation session, the sheltering tube depth was set at 5.6 cm to fit animals of different sizes. In the second session of the habituation and the orofacial behavioral tests in the following days, the depth of the sheltering tubes was adjusted to 3.5 to 4.2 cm so that the orofacial regions of the mice were accessible by stimuli such as von Frey filaments. One day before the second session of the habituation or orofacial behavioral tests, the hair in the orofacial regions of mice was trimmed off with a trimmer (Dapsang Eyebrow) and shaved with a razor (BIC Sensitive Single Blade). In the second session of the habituation, mice were also subjected to 4 to 5 times of trial tests with Von Frey filaments. On the orofacial behavioral assessment day, mice were brought to the behavioral test room and acclimated for 30 min in the home cages. Mice were placed in the sheltering tubes and acclimated for 10 min before applying stimuli for orofacial behavioral tests. After completing the aforementioned habituation or the orofacial behavioral tests described below, the sheltering tubes were thoroughly cleaned with soap and water for the next uses.
The von Frey test in orofacial regions for mice in sheltering tubes
To examine the mechanical sensitivity in the orofacial regions of testing animals, von Frey filaments were applied on the right side of the face, in the cheek areas, and behind the whisker pad. The von Frey filaments were applied to the orofacial regions when the head of the mouse stayed still in the sheltering tube. Three von Frey filaments (North Coast Medical, NC12775-99), with bending forces of 0.02 g, 0.04 g, and 0.07 g, were sequentially applied to the orofacial region to determine the response frequencies. For each of the von Frey filaments, it was applied by perpendicularly poking the skin of orofacial regions until the von Frey filament was bent, and this stimulation was repeated 10 times at the interval of 1-2 min. Positive responses were considered when animals rapidly moved their faces (heads) away from the von Frey filaments, pushed the filaments with their forepaws, guarded their orofacial regions with their forepaws, or bit the filaments. No responses were considered when animals did not show the aforementioned behaviors. In addition to measuring the response frequency, the response to mechanical stimulation was further assessed using the 0.07 g von Frey filaments to score the nociceptive responses. In this set of experiments, the von Frey filament (0.07 g) was applied 5 times with an interval of 1 minute between the mechanical stimulation. Nociceptive responses were scored as follows: 0, no response; 1, moves away the face from the filament or pushes away the filament one time with forepaws; 2, pushes away the filament with forepaws multiple times; 3, pushes away the filament with forepaws multiple times followed by guarding the orofacial areas with the forepaws; and 4, directly attack/bite the filament.
Acetone spray tests on orofacial regions of mice in sheltering tubes
Cold sensitivity was accessed using the same sheltering tube method described above for the von Frey test. In Brief, acetone (90%) was prepared, and 40 µl of the acetone solution was gently spray-applied to the orofacial regions using a 100-µl micropipette (ErgoOne® Single Channel). As a control for acetone, distilled water (room temperature) was applied in the same manner to the orofacial region. Mice responded to the acetone spray by rubbing their orofacial region against the floor of the sheltering tube and repeatedly wiping their cheek with their forepaws. To quantitatively describe the orofacial responses in this set of experiments, we measured the duration of animals’ rubbing and wiping behaviors following acetone spray. The orofacial behavioral responses to acetone spray were recorded by a video camera and analyzed offline.
Light stimulation to orofacial regions of mice in sheltering tubes
To investigate the response to light stimulation, the Nav1.8-ChR2 mice were placed in the sheltering tube. A blue laser beam was applied to the orofacial region with an optical fiber (diameter: 0.2 mm: Laserglow technologies). The light intensities were calibrated with an optical power and energy meter (PM100D, Thorlab). For our experiments, laser light stimulation was applied to the orofacial regions at the intensities of 15, 25, and 40/mm2, and the duration of each stimulus was 50 ms. Light stimulation at each intensity was applied 5 times at the time interval of 1 minute. Positive responses were the jerking movement of the heads of mice immediately following the light stimulation. The fraction of positive responses in the 10 times of the stimulation at a given laser intensity, i.e., the response frequency, was determined.
Open field test
The Open Field Test was conducted to assess exploratory activity in both saline-injected mice (control group) and mice injected with oxaliplatin. In each experiment, a testing mouse was placed in a squared open chamber at the dimensions of 40 (l) x 40 (w) x 30 (h) cm (MazeEngineer). The activity of each animal within the periphery and center regions of the chamber was recorded for 10 min using a video camera. The video was then analyzed offline using the DeepLabCut and Behavior Depot26,27 to determine the exploratory distance and time spent in the periphery and center regions of the open chamber.
Data analysis
Experiments and data analyses were performed in a blinded manner in which the control and oxaliplatin-injected groups were unknown by the person who performed orofacial behavioral assessment and data analyses. Data were analyzed using Graphpad Prism 10 software, represented as the Mean ± S.E.M. Statistical analyses were performed using one-way ANOVA, two-way ANOVA with Šídák’s multiple comparisons test, or Student's t-test, ns, no significant difference, *p < .05, **p < .01, and ***p < .001.
Results
To achieve a reliable experimental condition for applying mechanical, cold, and light stimulation to orofacial regions of Nav1.8-ChR2 mice, we made sheltering tubes for animals to reside in during behavioral tests (Figure 1(a)–(d)). Residing in a hole is an innate behavior for a mouse and other rodents, and our sheltering tubes created such a hole-like environment for the testing mice. In our study, each animal first underwent habituation in a sheltering tube (45 min daily for 2 days, Figure 1(a)). When a mouse was placed in a sheltering tube, it could freely turn its body with its head either inward or outward in the tube. All mice turned their body with their head outward after a few times of body position adjustment during habituation. Animals usually keep their heads outward throughout the experiments (Figure 1(b)–(d)). Animals adapted to the sheltering tube rapidly after two times of acclamation training, and they did not show any observable stressful behaviors while residing in the sheltering tubes. This created a desirable experimental condition for investigating behavioral responses to stimulations applied to the orofacial regions of the mice (Figure 1(b)–(d)). Assessment of orofacial behavioral responses of Nav1.8-ChR2 mice using the sheltering tube method (a) Brief description of the habituation schedule for each animal before behavioral tests. The days for habituation are not accounted for as experimental days. The first experiment day could be any day after the habitation, as the dashed line indicates. (b)–(d) Images show the sheltering tube method for testing the mechanical sensitivity using the von Frey test (b), the cold sensitivity using the acetone test (c), and the light sensitivity using the blue laser light (d) in the orofacial regions. Arrows in (b)–(d) indicate von Frey filament for mechanical stimulation (b), pipette tip for spraying acetone (90%) (c), and laser probe for shining blue laser light (d). Mice were placed in the sheltering tubes and accommodated for 10 min before the tests. (e) Response frequency to mechanical stimulation with 0.02-g, 0.04-g, and 0.07-g von Frey filaments applied to the orofacial regions. The von Frey tests were performed in the 1st, 2nd, 3rd, and 4th week (n = 8). (f) Response duration to the spray of acetone (90%, 40 µl) on the orofacial region in the 1st, 2nd, 3rd, and 4th week (n = 8). Control is the spray of distilled water (40 µl) at room temperature. (g) Frequency of avoidance response evoked by blue laser light stimulation applied to orofacial regions at the intensity of 15, 25, 40, and 60 mW/mm2 (n = 8). The light stimulation at each intensity was tested 5 times for measuring response frequency. Data represent mean ± SEM.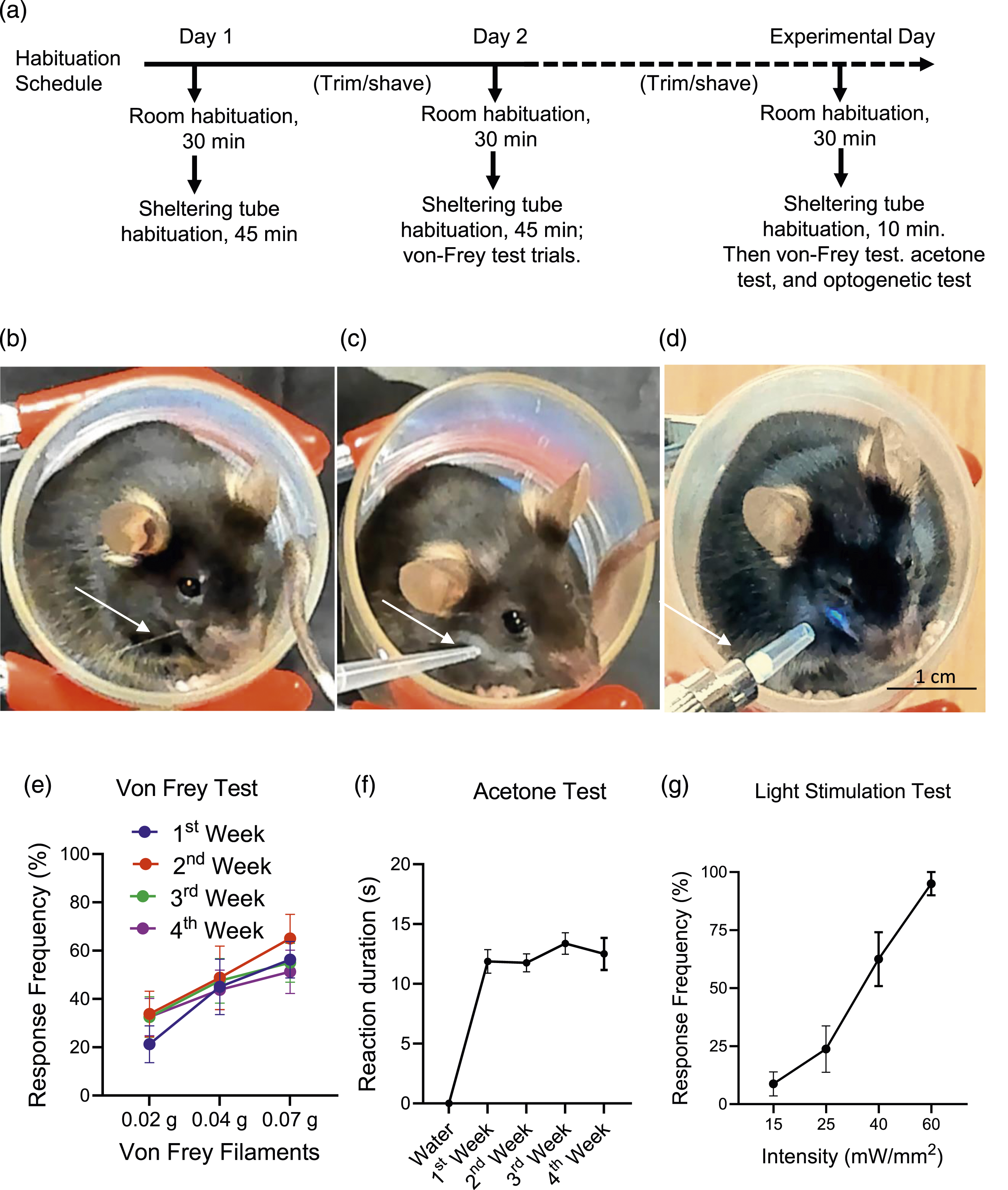
In the present study, we used Nav1.8-ChR2 mice because we wanted to not only investigate the orofacial behavioral responses to mechanical stimulation with the von Frey test (Figure 1(b)) and cold stimulation with the acetone test (Figure 1(c)) but also test light sensitivity using the optogenetic approach with Nav1.8-ChR2 mice (Figure 1(d)).
We conducted the von Frey test to determine the mechanical sensitivity in the orofacial region of naïve mice. In these experiments, mechanical stimulations with von Frey filaments of 0.02 g, 0.04 g, and 0.07 g were applied to the orofacial regions of the mice resided in the sheltering tube. Each von Frey filament was applied 10 times to determine the response frequency. As shown in Figure 1(e), the response frequency was increased with the enhanced forces of von Frey filaments from 0.02 to 0.07 g. The von Frey tests were performed in the 1st (n = 8), 2nd (n = 8), 3rd (n = 8), and 4th (n = 8) week, and the response frequency to a given filament force tested at different times was not significantly different. This result suggests a high reproducibility over time using this sheltering tube method for the von Frey test (Figure 1(e)).
We assessed the cold sensitivity on the orofacial region of mice that resided in the sheltering tube using the acetone spray method. In this set of experiments, a drop of acetone (40 µl) in a pipette was gently sprayed onto the orofacial region of the testing animals. Animals responded to the acetone spray by rubbing their faces against the walls of the sheltering tubes and wiping their cheek with their forepaws. We used the duration an animal rubbed and wiped its face to indicate the response to cold stimulation. In the consecutive 3 weeks, the response duration was consistent and not significantly different at different times with acetone spray (n = 8). We also sprayed 40 µl distilled water (room temperature) onto the orofacial regions, and no animal exhibited the rubbering and wiping responses (n = 7). These results suggest that acetone spray produced reliable behavioral responses in mice residing in the sheltering tubes, and no significant differences in cold sensitivity were observed over the 4 weeks tested in the present study (Figure 1(f)). On the other hand, water at room temperature was not a cold stimulation to the orofacial regions of mice.
We examined behavioral responses to blue laser light stimulation applied to the orofacial region because Nav1.8-ChR2 is expressed in many nociceptors in the mice used in the present study. In these experiments, we applied a blue laser beam to the orofacial region with a light intensity ranging from 15 to 40 mW/mm2. Light stimulation could elicit rapid avoidance reactions or nocifensive responses (Figure 1(g)). When response frequency was measured with 5 times stimuli for a given light intensity, the response frequencies were low at the light intensity of 15 mW/mm2, and the response frequency increased in a light intensity-dependent manner (n = 8, Figure 1(g)). With the light intensity of 40 mW/mm2, the response frequency reached nearly 100% (n = 8, Figure 1(g)). These results suggest that the sheltering tube method allows for a quantitative study of nociceptor activation by light stimulation in the Nav1.8-ChR2 mice (Figure 1(g)).
With the establishment of the sheltering tube method for investigating mechanical, cold, and light sensitivity, we used this method to investigate orofacial pain following the treatment of mice with the chemotherapy drug oxaliplatin. Figure 2(a) illustrates the schedule of behavioral tests before and after the intraperitoneal injections of oxaliplatin at the daily dose of 2 mg/kg for five consecutive days. We monitored the mice’s body weights throughout the experimental timeline, from baseline through week 3, for the saline-injection group (control, n = 10) and oxaliplatin-injection group (n = 10). We observe a tendency of body weight reduction in the first week (p = .058) in the oxaliplatin-injected group compared to the saline-injected group (Figure 2(b)). Evaluation of the effects of oxaliplatin on body weight and the open-field test (a) The experiment schedule. Following baseline behavioral best, oxaliplatin was i.p. administrated at 2 mg/kg daily for five consecutive days. Behavioral tests were then performed on the days indicated. (b) Body weights measured before, in the 1st, 2nd, and 3rd week after the administration of saline (control, n = 10) or oxaliplatin (n = 10). (c) The open field test shows the time spent in the center (blue symbols) and the periphery (red symbols) in animals before (baseline) and after administering saline (circles, n = 6) or oxaliplatin (squares, n = 7). (d) Similar to (c), except for the distances covered in the center and the periphery for the saline group (circles, n = 6) and the oxaliplatin group (squares, n = 7). Data represent mean ± SEM, *p < .05, ns, not significantly different, two-way ANOVA with the Bonferroni post hoc test.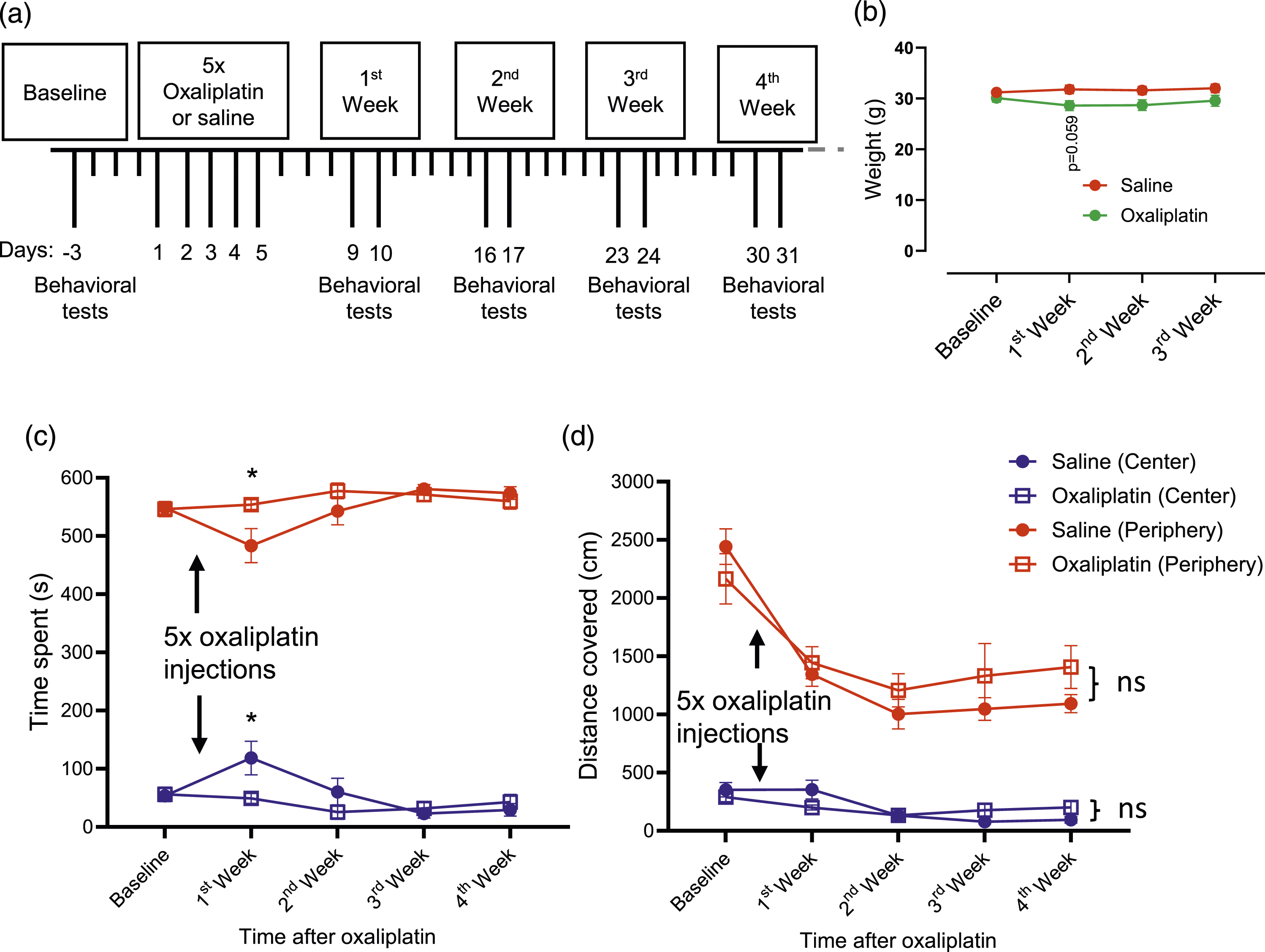
We performed the open field test to examine whether oxaliplatin injection affects general locomotor activity, anxiety, and willingness to explore the environment in the mice. In the present study, each animal was placed in an open field box to allow it to move and explore the environment freely, and its movement in the open field was video-recorded for 10 min. The video data were then analyzed using the DeepLabCut and Behavior Depot. The accumulative time an animal spent (Figure 2(c)) at the center and the periphery in the open field box was determined by tracking the central position on the dorsal side of the body. Except for the first week after the oxaliplatin, there was no significant difference in the time spent at the center or the periphery between the saline-injected (n = 7) and oxaliplatin-injected (n = 7) groups in the 4-weeks experiments (Figure 2(c)). For the first week, the saline-injected group spent less time at the peripheral and more time at the center than the oxaliplatin-injected group. However, this difference seems to be because the data in 1st week deviated from the baseline and other time points in the saline group. For the distance covered at the center and the periphery in the open field box, no significant difference was observed in the distance covered at the center or the periphery between the saline-injected (n = 7) and oxaliplatin-injected (n = 7) groups throughout 4 weeks of the experiments (Figure 2(d)). These results suggested that animals’ locomotor activity and mental status were similar in the two groups of animals. Therefore, locomotor activity and mental status were unlikely confounding factors in the orofacial nociceptive behavioral tests in oxaliplatin-injected animals described below.
We assessed mechanical sensitivity in the orofacial region of mice before and after oxaliplatin injections using the von Frey test. In these experiments, mice resided in the sheltering tube, and mechanical stimulation was applied with 0.02-g, 0.04-g, and 0.07-g von Frey filaments 10 times each, and the response frequency was then determined. At the baseline before oxaliplatin injections, response frequencies were 30.00 ± 20.82%, 47.14 ± 17.99%, and 62.86 ± 23.60% with 0.02-g, 0.04-g, and 0.07 g von Frey filaments, respectively (Figure 3(a), n = 7). The response frequencies were significantly increased in the 1st to 4th week after the injections of oxaliplatin (Figure 3(a)). For example. In the third week following the injections of oxaliplatin, the response frequencies were 82.86 ± 11.13% (n = 7), 91.43 ± 9.0% (n = 7), and 98.57 ± 3.78% (n = 7) with 0.02-g, 0.04-g, and 0.07 g von Frey filaments, respectively (Figure 3(a)). The response frequencies to the mechanical stimulation returned to a level not significantly different from that of the baseline (Figure 3(a), (n = 7) in the 5th to 6th week. Assessment of mechanical response frequency in the orofacial region following oxaliplatin treatment (a) Mechanical response frequency before (baseline, control) and in the 1st to the 6th week following the administration of oxaliplatin. The mechanical response frequency was measured with the von Frey filaments of 0.02 g (blue, n = 7), 0.04 g (red, n = 7), and 0.07 g (green, n = 7), each applied 10 times to the orofacial region. The response frequency after oxaliplatin was compared with the baseline; one-way ANOVA with the Sídak’s multiple comparisons test for 0.02 g (F (6, 42) = 8.398, p < .0001), 0.04 g (F (6, 42) = 9.361, p < .0001), and 0.07 g (F (6, 42) = 8.883, p < .0001). (b)–(d) Mechanical response frequency in the saline-injected group (control, blue, n = 10) and the oxaliplatin-injected group (red, n = 10) in the 1st week (F (1,18) = 47.11, p < .001, (b), 2nd week (F (1,18) = 77.25, p < .001, (c), and 3rd week (F (1,17) = 55.83, p < .001, (d). The mechanical response frequency was measured with the von Frey filaments of 0.02 g (n = 7), 0.04 g (n = 7), and 0.07 g (n = 7), each applied 10 times to the orofacial region. Data represent individual observations and mean ± SEM, *p < .05, **p < .01, ***p < .001, two-way ANOVA with Sidak’s post hoc test (b)–(d).
In different experiments, we compared mechanical sensitivity in the orofacial regions between saline-injected and oxaliplatin-injected groups. Mechanical sensitivity was tested by stimulating the orofacial regions of mice with 0.02-g, 0.04-g, and 0.07-g von Frey filaments 10 times each, and response frequency was determined. In the 1st week after the injection of saline, the response frequencies were 27.00 ± 13.37% (n = 10), 48.00 ± 19.32% (n = 10), and 59.00 ± 16.63% (n = 10) with 0.02-g, 0.04-g, and 0.07 g von Frey filaments, respectively (Figure 3(b)). In contrast, in the 1st week after the injection of oxaliplatin, the response frequencies were 73.00 ± 24.97% (n = 10), 86.00 ± 10.75% (n = 10), and 93.00 ± 8.23% (n = 10) with 0.02-g, 0.04-g, and 0.07 g von Frey filaments, respectively (Figure 3(b)), and were significantly higher than the saline-injected group (Figure 3(b)). Similarly, the mechanical sensitivity in the orofacial regions, as measured by the response frequency in the von Frey test, was also significantly increased in the oxaliplatin-injected mice in the 2nd (Figure 3(c)) and 3rd (Figure 3(d)) week after the injections of oxaliplatin.
We measured mechanical sensitivity using the avoidance response frequency in the above experiments. However, in response to mechanical stimulation, animals exhibited several behaviors, including moving the face away from the von Frey filament, pushing the filament away with their forepaws, guarding the orofacial areas with the forepaws, and directly attacking and biting the filament. We scored the above nocifensive responses evoked by the 0.07-g von Frey filament to assess mechanical hyperalgesia in animals after oxaliplatin injections. As shown in Figure 4(a), at the baseline before oxaliplatin injections, the response score was 1.11 ± 0.8 (n = 7), mainly representing the reflexively moving away the face from the von Frey filament. However, in the 1st, 2nd, and 3rd weeks after the injections of oxaliplatin, the scores were 2.66 ± 0.81 (n = 7), 3.11 ± 0.44 (n = 7), and 2.83 ± 0.24 (n = 7), respectively, which were significantly higher than that before the injection of oxaliplatin. The response scores in the 4th, 5th, and 6th weeks after oxaliplatin injection returned to levels not significantly different from the baseline (Figure 4(a)) Assessment of mechanical response score in the orofacial region following oxaliplatin treatment (a) Mechanical response scores were determined by applying mechanical stimulation to the orofacial region using a 0.07 g-von Frey filament. The test was performed before (control) and in the 1st to 6th week after oxaliplatin injection (F(6, 42) = 14.30, p < .0001, n = 7). The mechanical response scores following oxaliplatin were compared with the baseline scores. (b)–(d) Mechanical response scores were determined in animals injected with saline (control, n = 10) and oxaliplatin (n = 10). The tests were performed, and the mechanical response was scored at the 1st week (t = 3.906, p = .001, (b), 2nd week (t = 7.485, p < .0001, (c), and 3rd-week (t = 7.337, p < .0001, (d) post-injection of saline or oxaliplatin. Data represent mean ± SEM, **p < .01, ***p < .001, one-way ANOVA with Sidak’s post hoc test (a) or Student’s t-test (b)–(d).
We compared the mechanical response scores in the orofacial regions between saline-injected and oxaliplatin-injected groups. In the 1st week after the oxaliplatin injection, the response scores were 2.80 ± 0.67 (n = 10), significantly higher than those in the saline group (1.54 ± 0.77, n = 10, Figure 4(b)). Similarly, the response scores were also significantly higher in the oxaliplatin-injected mice (n = 10) than in saline-injected mice (n = 10) in the 2nd (Figure 4(c)) and 3rd (Figure 4(d)) week after the injections of oxaliplatin.
We assessed cold sensitivity in the orofacial region in mice before and after oxaliplatin injections by using the acetone spray test. In these experiments, mice resided in the sheltering tubes, and a drop of acetone (40 µl, 90%) was sprayed onto the orofacial region of mice. Mice responded to the acetone spray by rubbing their orofacial region against the floor of the sheltering tube and repeatedly wiping their cheek with their forepaws in the orofacial region. The duration of the aforementioned responses was determined. As shown in Figure 5(a), at the baseline before oxaliplatin injections, the response duration was 12.14 ± 2.55 (n = 7). However, in the 1st, 2nd, and 3rd weeks after the injections of oxaliplatin, the durations were 17.29 ± 1.60 (n = 7), 17.29 ± 3.45 (n = 7), and 16.29 ± 3.35 (n = 7), respectively, which were significantly higher than that before the injection of oxaliplatin (Figure 5(a)). The reaction durations in the 4th, 5th, and 6th weeks after oxaliplatin injection returned to levels not significantly different from the baseline (Figure 5(a)). Assessment of cold sensitivity in the orofacial region following oxaliplatin treatment (a) Cold sensitivity determined by the acetone test with acetone (40 µl) sprayed to the orofacial region. The cold response was quantified by the duration of the facial rubbing reaction following the acetone spray. The tests were performed before (baseline) and post-oxaliplatin injection in the 1st to 6th week (F(6, 42) = 4.513, p = .0013, n = 7). The cold responses after oxaliplatin were compared with the baseline. (b)–(d) The acetone test for cold sensitivity was determined in animals injected with saline (control, n = 10) and oxaliplatin (n = 10). The acetone tests were performed at 1st (t = 4.523, p = .0003, (b), 2nd (t = 3.750, p = .0015, (c), and 3rd (t = 4.416, p = .0004, (d) week post-injection of saline (blue) or oxaliplatin (red). Data represent mean ± SEM, *p < .05, **p < .01, ***p < .001, one-way ANOVA with Sidak’s post hoc test (a) or Student’s t-test (b)–(d).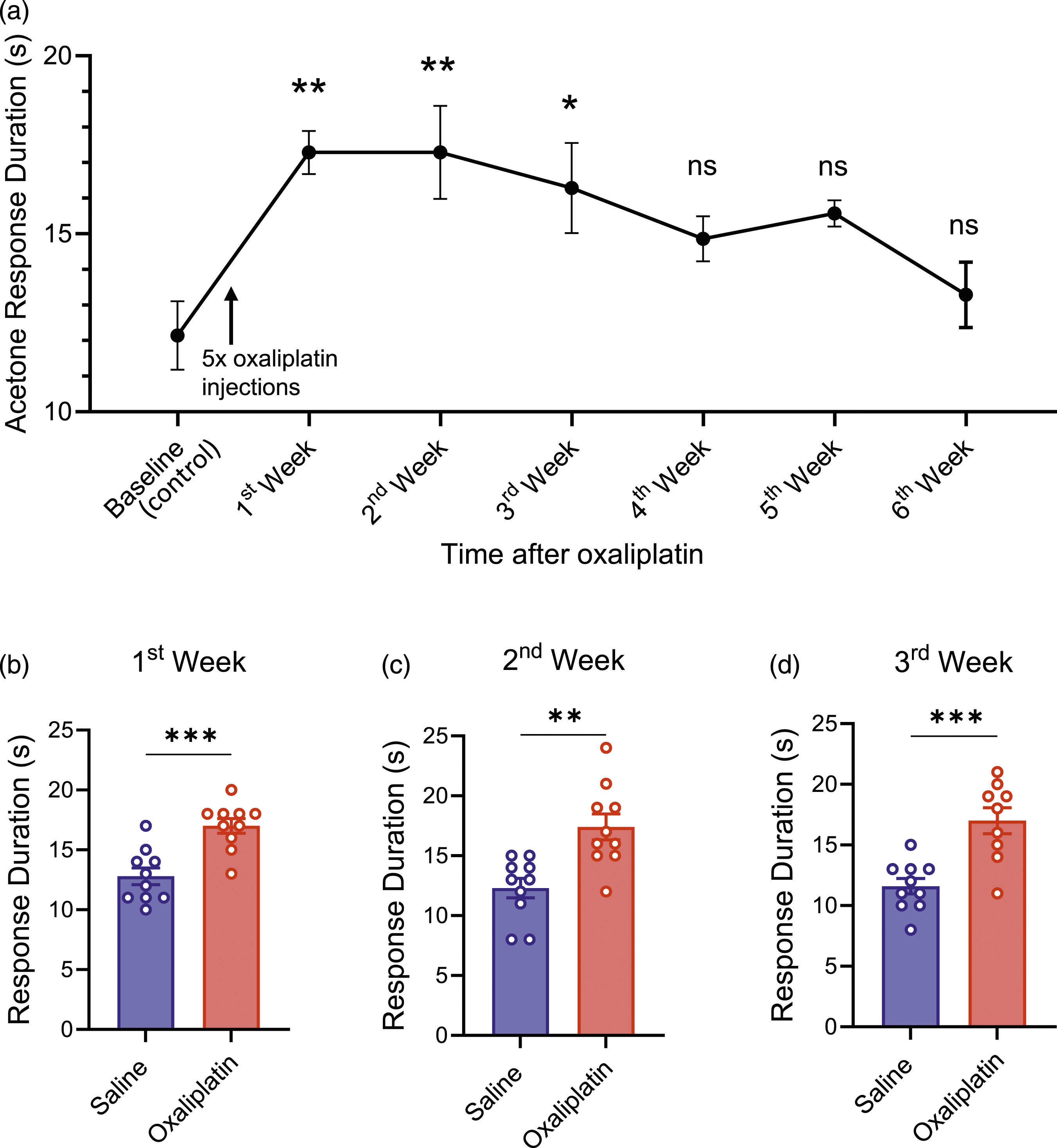
We compared the cold sensitivity in the orofacial regions between saline-injected and oxaliplatin-injected groups. In the 1st week after the oxaliplatin injection, the response durations were 17.00 ± 1.94 (n = 10), significantly higher than those in the saline group (12.80 ± 2.20, n = 10, Figure 5(b)). Similarly, the response duration was also significantly higher in the oxaliplatin-injected mice (n = 10) than in saline-injected mice (n = 10) in the 2nd (Figure 5(c)) and 3rd (Figure 5(d)) week after the injections of oxaliplatin.
In a different set of experiments, we applied a blue laser beam to the orofacial region of the Nav1.8-ChR2 mice (Figure 6(a) and (b)), and positive responses were considered when the mice exhibited avoidance or jerking response to the blue laser beam (Figure 6(b)). We compared the behavior responses to blue light stimulation in the orofacial regions between saline-injected and oxaliplatin-injected groups. Light sensitivity was tested by stimulating the orofacial regions of mice with light intensity of 15 mW/mm2, 25 mW/mm2, and 40 mW/mm2 each 5 times, and response frequency was determined. In the 2nd week after the injection of saline, the response frequencies were 26.67 ± 24.22% (n = 6), 46.67 ± 20.66% (n = 6), and 83.33 ± 8.17% (n = 6) with light intensity of 15 mW/mm2, 25 mW/mm2, and 40 mW/mm2, respectively (Figure 6(c)). In contrast, in the 2nd week after oxaliplatin injection, the response frequencies were 53.33 ± 24.22% (n = 6), 83.33 ± 8.17% (n = 6), and 100.00 ± 0.00% (n = 10) with 15 mW/mm2, 25 mW/mm2, and 40 mW/mm2, respectively (Figure 6(c)), and were significantly higher than the saline-injected group with the light intensity of 15 mW/mm2 and 25 mW/mm2, but not significantly different with the light intensity of 40 mW/mm2 (Figure 6(c)). Assessment of light sensitivity in the orofacial region following oxaliplatin treatment. (a) & (b) Images illustrate the head positions before (a) and immediately after the shining of blue laser light on the orofacial region (b). The avoidance reaction in response to the light stimulation was evidenced in (b) (c) Response frequency to the light stimulation in animals after 9 days (2nd week) following saline (control, n = 6, blue) and oxaliplatin (n = 6, red) injections; the light stimulation was applied to the orofacial regions at 15, 25, and 40 mW/mm2; F(1, 30) = 22.15, p < .0001. Data represent individual observations and mean ± SEM, *p < .05, **p < .01, ns, not significantly different, two-way ANOVA with Sidak’s post hoc test.
Discussion
In the present study, we have implemented a sheltering tube method for performing sensory tests, including the von Frey test, the acetone spray test, and the optogenetic test in mice’s orofacial regions. This new method provides testing animals with a less stressful environment, allows easy and precise application of sensory stimuli to orofacial regions, and generates highly reproducible behavioral outcomes. By using the sheltering tube method with the von Frey test, the acetone spray test, and the optogenetic test in Nav1.8-ChR2 mice, we have demonstrated orofacial hypersensitivity to mechanical, cold, and light stimulation in animals following the treatment with the chemotherapy drug oxaliplatin. The new approach offers a new and convenient tool for pain research to study pathological pain in the orofacial regions of mice.
In the present study, we placed testing mice in sheltering tubes for sensory tests in the orofacial regions because these mice could rapidly accommodate the environment in the sheltering tubes. Previously, sensory tests for mechanical and cold sensitivity in the orofacial regions were performed by holding a testing animal in the experimenter’s hand, 21 putting a testing animal in a small mesh-wired cage,17,23 or placing a testing animal on an elevated platform. 24 However, under these previous conditions, mice were stressed and poorly accommodated in the experimental environment. The von Frey and acetone spray tests were difficult and unreliable under these conditions. To overcome the difficulties of the orofacial sensory tests, we implemented the sheltering tube method because sheltering tubes created a hole-like environment where mice innately like to reside and feel secure. The sheltering tube may still be considered as a form of restraint. However, in our sheltering tubes, animals have enough space to turn around their bodies. To further reduce any potential stress in the sheltering tube, we let each animal undergo habituation in the sheltering tubes. Therefore, this new approach allows animals to accommodate the experimental environment with less stress and allows experimenters to apply sensory stimuli to the orofacial area of mice easily and reliably.
Our mechanical sensitivity tests on naïve Nav1.8-ChR2 mice showed increased response frequency after applying von Frey filaments at increased forces onto the orofacial regions. The graded response is generally consistent with the tests on mice’s hindpaws.7,8,25 However, we observed a response frequency of ∼30% with 0.02-g, ∼45% with 0.04-g, and ∼60% with 0.07-g von Frey filaments, and the response thresholds appear much lower than the previous von Frey tests performed on the glabrous skin of the mice’s hindpaws. 28 For example, previous studies have shown that the response frequency was 15 to 20%, about a three-fold difference from our values with 0.07-g von Frey filament. This discrepancy could be due to several reasons. (1) the hindpaws bear body weights while orofacial regions do not, making behavioral responses to tactile stimuli different in these two regions; (2) the structure of the skin in orofacial regions (hairy skin) is different from that in the glabrous skin of the hindpaws, resulting in differences in skin mechanics and force transduction, and (3) the sensitivity and density of the mechanoreceptors may be different between the orofacial skin and the glabrous skin. A previous study performed the von Frey test in orofacial regions while animals were held in experimenters’ hands 21 ; the mechanical response thresholds were significantly higher in that study than in our present study. The discrepancy may be due to the differences in the stress level between the two methods.
For the acetone spray test, spraying a drop of room-temperature water did not evoke any avoidance responses. However, spraying a drop of acetone evoked an avoidance response, then rubbering and wiping the facial areas. We interpreted these behavioral responses as animals’s reactions to cooling stimulation caused by acetone evaporation. 7 We repeated the von Frey and acetone spray tests for 4 weeks and demonstrated stable responses in the naïve mice. This result suggests good consistency of behavioral responses with the sheltering tube method. For the optogenetic tests with laser light stimulation to orofacial regions in our NaV1.8-ChR2 mice, the stable head position in the sheltering tube is important because laser light may shine into animals’ eyes if their heads are unstable. Our optogenetic tests showed an increased frequency of avoidance response with increased laser light intensity from 15 to 40 mW/mm2. This is consistent with another study on the hindpaw, where response frequency increased with higher light stimulation intensity. However, in that study, the response frequency reached 100% at 10 mW/mm2 in the hindpaws. 25 In contrast, it reached 100% at 40 mW/mm2 in the orofacial region in the present study, suggesting that orofacial regions are less sensitive in the optogenetic test. It is known that ChR2 is selectively expressed in NaV1.8-positive afferent neurons in NaV1.8-ChR2 mice, 29 and NaV1.8-positive afferent neurons are primarily nociceptors. 25 Therefore, the avoidance responses evoked by the blue laser lights were nocifensive responses due to the direct excitation of nociceptors by the laser lights.
We have validated the usefulness of the sheltering tube method in studying orofacial neuropathic pain by performing the sensory tests in NaV1.8-ChR2 mice following the treatment of these animals with the chemotherapy drug oxaliplatin. Clinically, oxaliplatin and other chemotherapy drugs induce peripheral neuropathy in cancer patients,14,20 and orofacial pain is a common and debilitating symptom in cancer patients undergoing chemotherapy treatment.30,31 Previous studies have reported that oxaliplatin-induced neuropathic pain in orofacial regions could be observed in up to 70% of patients.32,33 Since pain in orofacial regions can affect important daily tasks such as speaking, eating, and drinking, studying chemotherapy-induced orofacial pain in rodent models is clinically relevant. Pain in CIPN is characterized by mechanical and cold allodynia/hyperalgesia in the body’s peripheral regions, including orofacial regions.11,12,34,35 The chemotherapy-induced neuropathic pain could be recaptured in animal models in rats and mice by treating animals with chemotherapy drugs. Sensory tests, including the von Frey test and acetone spray test in the animal models of chemotherapy-induced neuropathic pain, were commonly performed in the glabrous skin of the hindwaws of rats and mice to demonstrate the presence of mechanical and cold allodynia/hyperalgesia in the hindpaws.7,8,25,36 Previous studies also demonstrated mechanical and cold allodynia/hyperalgesia in orofacial regions using the orofacial operant test.19,20 In the present study, we treated NaV1.8-ChR2 mice with oxaliplatin and investigated mechanical, cold, and light sensitivity in these animals’ orofacial regions. We show that oxaliplatin treatment increases the response frequency to von Frey filament stimulation, suggesting the presence of mechanical allodynia/hyperalgesia in the orofacial regions of oxaliplatin-treatment mice. These animals also showed increased response duration in the acetone spray tests, suggesting the presence of cold allodynia in the orofacial in these animals. The mechanical and cold hypersensitivity observed in the present study is consistent with our previous studies using the orofacial operant test.19,20 In the optogenetic test on the oxaliplatin-treated animals, we showed that the response frequency was significantly increased with laser light stimulation intensity at 15 and 25 mW/mm2 compared to the saline-injected animals. However, at the high intensity of 40 mW/mm2, we did not see significant differences in the increases of the response frequency between oxaliplatin-treated and saline-injected groups, most likely because animals’ response has been near the maximal responses with the high-intensity light stimulation. The optogenetic test in the present study may suggest that NaV1.8 nociceptors display hyperexcitability following oxaliplatin treatment. This result may suggest that the enhanced excitability at the terminals of nociceptors may be an underlying mechanism for the mechanical and cold allodynia/hyperalgesia.
In summary, the sheltering tube method presented here provides a reliable and convenient approach to studying orofacial pain and revealing mechanical and cold allodynia and hyperalgesia in CIPN animals and possibly other pathological pain models17,21–24 used for orofacial pain research.
Footnotes
Acknowledgements
We thank Dr R. Vaden for his technical support.
Author contributions
J.G.G. conceived the research project and designed experiments. S.G. performed experiments. S.G. and J.L. created and maintained the transgenic mice. S.G. and J.G.G. analyzed data and participated in data interpretation. J.G.G. and SG wrote the paper.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by National Institute of Dental and Craniofacial Research (DE018661 and DE023090) and NIH grants NS109059, DE018661 and DE023090 to J.G.G.
