Abstract
Preemptive analgesia is used for postoperative pain management, providing pain relief with few adverse effects. In this study, the effect of a preemptive regime on rat behavior and c-fos expression in the spinal cord of the uterine surgical pain model was evaluated. It was a lab-based experimental study in which 60 female Sprague-Dawley rats; eight to 10 weeks old, weighing 150–300 gm were used. The rats were divided into two main groups: (i) superficial pain group (SG) (with skin incision only), (ii) deep pain group (with skin and uterine incisions). Each group was further divided into three subgroups based on the type of preemptive analgesia administered i.e., “tramadol, buprenorphine, and saline subgroups.” Pain behavior was evaluated using the “Rat Grimace Scale” (RGS) at 2, 4, 6, 9 and 24 h post-surgery. Additionally, c-fos immunohistochemistry was performed on sections from spinal dorsal horn (T12-L2), and its expression was evaluated using optical density and mean cell count 2 hours postoperatively. Significant reduction in the RGS was noted in both the superficial and deep pain groups within the tramadol and buprenorphine subgroups when compared to the saline subgroup (p ≤ .05). There was a significant decrease in c-fos expression both in terms of number of c-fos positive cells and the optical density across the superficial laminae and lamina X of the spinal dorsal horn in both SD and DG (p ≤ .05). In contrast, the saline group exhibited c-fos expression primarily in laminae I-II and III-IV for both superficial and deep pain groups and lamina X in the deep pain group only (p ≤ .05). Hence, a preemptive regimen results in significant suppression of both superficial and deep components of pain transmission. These findings provide compelling evidence of the analgesic efficacy of preemptive treatment in alleviating pain response associated with uterine surgery.
Introduction
Postoperative pain is still a challenging issue, regarded as one of the principle concerns of both surgeons and anesthetist.1,2 Patients feel severe pain post-operatively due to the tissue damage caused by the surgery.3,4 A new concept of preemptive analgesia emerged in pursuit of post-operative pain relief, suggests that the preemptive regimen is based on the notion that blockage of pain transmission before the application of painful stimulus reduces postoperative pain. 4 The preemptive regime is a combination of different pharmacological agents that are administered before the onset of a pain stimulus (surgery/incision), acting at different sites within the central and peripheral nervous systems. This regime, when administered before surgical intervention, may help in reducing the general anesthesia (GA) dose-related side effects, which are critical in elderly, hypertensive patients who are unfit for prolonged induction of GA.5,6 Moreover, several side effects and addiction issues, related to the use of opioid analgesia postoperatively, can be managed by use of preemptive regimes and ensuring pain–free surgery. 7 GA alone is insufficient in relieving pain. Using GA in combination with local anesthesia (LA) and analgesia may attenuate pain transmission significantly. 8
Postoperative pain leads to the activation of pain receptors, distributed throughout the body’s surface (superficial or somatic pain receptors) and organs (deep or visceral pain receptors). These activated receptors send pain signals to the spinal cord and subsequently to higher brain centers, activating pain markers such as c-fos, jun, and pERK in the nociceptive neurons across the nervous system. 9 Among these pain-makers, c-fos is an immediate response gene. It is a proto-oncogene, and its cellular variant, v-fos, has been identified in the murine Osteogenic Sarcoma virus. 10 These genes are rapidly activated by growth factors and neurotransmitters like substance P, glutamate opening of voltage-gated calcium channels, typically within a few minutes and leads to activation of noxious stimulation.11,12 Consequently, the sensation of pain triggers the expression of c-fos in neurons, within a timeframe of 30 to 60 min. 13
The topographical distribution and detection of c-fos expression in both the brain and spinal cord have proven to be a dependable approach for evaluating the effectiveness of pain-inducing and preventive interventions, such as preemptive analgesia. 14 Additionally, the combination of c-fos expression analysis and the assessment of behavioral response may further enhance the evaluation of postoperative pain and the efficacy of different preemptive regimens. Usually, Rat Grimace Scale (RGS), which quantifies the nociceptive pain in the non-verbal human population is utilized to assess the behavioral response to pain. 14 The RGS involves the scale which computes the frequency of four facial “action units” (orbital constriction, nose or cheeks bulging, whisker alterations, and ear position) with lack of need for subject preparation and specialized equipment. This integrated approach using both c-fos and RGS may provide a more comprehensive understanding of pain modulation, facilitating an accurate assessment of the impact of interventions in pain management. 15
Pain preventive measures suppress c-fos expression in the CNS and lower RGS rating, but neither analgesia nor anesthesia alone cause marked suppression of c-fos expression or complete alleviation of pain. Several studies have supported the efficacy of preemptive analgesia (multimodal regimen) for intra & postoperative pain. 7 However, the efficacy of preemptive analgesia remains questionable due to several factors, such as inconsistency in prescription of preemptive analgesia (type, duration, route, and timing), difference in the type of surgical stress and the lack of an objective measurement of pain. 16 This incongruence may also result from incomplete afferent blockade using LA or GA. Therefore, further research is required to explore these aspects in detail. 5
In this study, different types of analgesia (tramadol and buprenorphine) will be combined with LA and GA in a rat’s uterine surgical model, compassing both somatic and visceral pain transmission mechanisms, to examine the morphological, molecular, and behavioral changes occurring in the spinal cord of the rats.
Methods
The study protocol was approved by Advanced Study and Research Board (ASRB) KMU Peshawar, dated 14/10/21 notification no DIR/KMU-EB/MB/000755. All experiments were conducted according to the guidelines for animal care provided by the National Institutes of Health and the International Association for the Study of Pain. 17
Animals
Sixty Sprague-Dawley female rats, weighing 150-250g, were included in the study that was calculated from resource equation for multiple observation applied for behavioral study (RGS) and single observation for molecular study (c-fos expression). 18 Animals were housed in groups of three per cage and kept on a 12-h light-dark cycle. The room temperature was maintained at 22 °C with free access to food and water. Rats were accustomed to the housing amenities for 1 week before the start of the study.
Surgery
The animals were anesthetized using 5 %isoflurane for induction, followed by maintenance with 2% isoflurane throughout the surgical procedure. 19 Preemptive injections, (tramadol + lidocaine or buprenorphine + lidocaine) and saline injections were administered preoperatively. A lower abdominal transverse incision was performed to expose the uterus. A one-cm transverse incision was made after retracting the bladder from the uterus, and the uterus was opened. All incisions were closed in reverse order with Vicryl 2/0. For the superficial pain group, only skin incisions were given and closed with Vicryl 2/0. The animals were allowed to recover completely and were transferred to the behavioral room for monitoring.19,20
Experimental groups
Sixty Sprague-Dawley rats were divided into two main groups: superficial pain group (SG) (n = 30) and deep pain group (DG) (n = 30). Each group was further subdivided into three subgroups of 10 rats each, based on the preemptive regime used. The deep pain group was further divided into (a) Tramadol and Lidocaine subgroup (TD) (n = 10). (b) Buprenorphine and Lidocaine subgroup (BD) (n = 10) and (c) Saline and Lidocaine subgroup (SD) (n = 10). The superficial pain group was further divided into (a) the Tramadol and Lidocaine subgroup (TS) (n = 10), (b) the Buprenorphine and Lidocaine subgroup (BS) (n = 10), and (c) the saline and Lidocaine subgroup (SS) (n = 10). Each subgroup consisted of 10 rats in total with seven rats for behavioral studies and three rats for immunohistochemistry. The sample size was calculated using the resource equation. 18
Rate Grimace scale
Each rat was placed in a plastic chamber with two transparent and two opaque walls. Nociceptive responses were recorded for 30 minutes at an interval of 2, 4, 6, 9, and 24 h after the surgery, through two cameras [Hikvision model DS-2CE16HOT-IT3F 3.6 mm, 5MP/0.01 Lux @ F1.2,0 lux with IR DNR/D-WDR//IP67/OSD menu, IR distance 49m, 12V, 4.3 W MAX, material code 300509598 China] simultaneously, placed orthogonally. Up to 10 still images were captured for each animal in each thirty-minute video recording by using GOM player 2003 software (https://www.gomcorp.com). Images were not selected if an animal was found sleeping or grooming. At times, images were captured manually, if the rat appeared to have occupied an unusual position. Each image was scored for four facial features: orbital tightening, nose/cheek flattening, ear changes, and whisker change, scored over a scale as “absent” (score = 0), “moderate” (score = 1), or “severe” (score = 2), with an average score (range 0–2). 21
Images were randomized and scored independently by a single observer at two different time points after image selection with proper coding for binding purpose. The generated scores were used to assess intra-rater reliability via Cronbach’s alpha, with scores of 0.7 or above being considered reliable. 22
Immunohistochemistry
For immunohistochemical analysis, three rats from each subgroup were euthanized by cardiac puncture and decapitation 2 hours post-surgical procedure while the remaining animals were euthanized at 4, 6, 9 and 24 h post surgery following behavioral assessment. The thoracolumbar spinal cord was removed and processed for immunohistochemistry analysis. Paraffin blocks were made following standard protocols, 23 and 5 µm thin sections were obtained using a microtome. Three to four sections from the thoracic and lumber spinal cords were randomly selected for analysis. The sections were sequentially dewaxed in a hot air oven for 15 minutes, followed by the clearing of xylene in decreasing concentrations of alcohol, and washed with Phosphate-Buffered-Saline (PBS) thrice. A mouse monoclonal c-fos antibody (ab 208942) was diluted to a concentration of 1:100 in 0.01 M PBS containing 10% normal goat serum and 0.3% Triton X-100 after optimization.
A 2% retrieval (Unmasking) solution with a pH of 6.5 was used. The retrieval solution, along with the slide, was heated for 40 minutes in a microwave (heating to mask the antibody site). The sections were then washed with distilled water thrice. Permeabilization was done with 1% PBS +0.4% Triton X for 15 minutes at room temperature, followed by two washes with distilled water. Blocking was performed with 5% peroxidase blocking (DM 821, Dako) for 10 minutes at room temperature, followed by a wash with distilled water.
The primary antibody, c-fos, diluted to a concentration of 1:100, was applied to the sections and left for 1 hour. After two washes with distilled water, the secondary antibody horse reddish peroxidase (Deko HRP) was applied for 45 minutes. The sections were washed twice, followed by treatment with diaminobenzidine (DAB)- Chromogen (Deko) for 15 minutes and hematoxylin for 30 seconds. Finally, all sections were rinsed, mounted on glass slides, and cover-slipped with Prolong mounting medium. Slides were examined for c-fos expression under a compound microscope (Nikon ECLIPSE 80/Japan 221765) with digital apparatus (Canadian ICES-003) to capture the images at magnifications of 10X, 20X, and 40X.
Image J analysis
The image J (Fiji) programme was used to analyse the images captured by the compound microscope. The optical density (OD) was used to calculate the mean and standard deviation (SD) of colour intensity of c-fos expression in each group, while c-fos cell count measured the mean and SD of total cell count in each study group. The mean cell count was recorded by image J software (selecting image > type 8-bit > Image > adjust > threshold (85 minimum to 255 Maximum) > processes > make binary > convert to mask > select area I-II, III-IV, lamina X > Analysis particles). (https://youtu.be/1PQprFZ2Byg, https://www.unige.ch/medecine/bioimaging/files/3714/1208/5964/CellCounting.pdf). 24
The results were saved in Excel and SSPS software for further analysis. The optical density was calculated using Image J software using validated protocol: (record a macro, select image > plugin> macro > Record > select > Rectangular shape > to draw on the image, and then click ‘create’ and “save” as macro item”, followed by selecting the images one by one from the file as follows: open file > image -colour - Deconvolution- H-DAB > selected colour 2 for quantification of intensity > plugin-Run-macro. ijm > and then > Analyse – measure). A results window that had been saved in an Excel sheet appeared.
(https://www.researchgate.net/post/Does-anyone-have-a-protocol-for-quantifying-IHC-images-in-ImageJ).
The optical density was calculated by using the following formula:
OD= log of (maximum intensity/mean intensity)
Max intensity = 255 for every 8-bit picture. 25
Statistical analysis
The statistical package for Social Science (SPSS) version 25 and Microsoft Excel 18 were used to conduct the statistical analysis. Pain behavior (RGS) was assessed and compared among the three subgroups at 2, 4, 6, 9 and 24 h using one-way analysis of variance (ANOVA) separately at each interval. Further, repeated measure ANOVA was used to determine the behavioral changes among each subgroup over all time periods. The total number of positive c-fos neurons in lamina I-II, III-IV, and X neurons were counted using the Fiji Image J software and the difference between the groups was analyzed using one-way ANOVA. A post hoc least significant difference (LSD) test was used to compare the significant values across treatment groups, along with the student t test. 26 The significance level was set at p ≤ .05, and all data are shown as the mean ± SD.
Results
In this study, pain responses were assessed in 60 rats, considering both behavioral and histomorphological aspects. Forty-two rats were included in the behavioral study (n = 7 in each group) and 18 were assessed histomorphologically (n = 3 in each group).
Rat Grimace scale
Following surgery, observations were made for 30 minutes, at 2, 4, 6, 9, and 24-h. Intra-reliability was assessed with Cronbach’s alpha, which was found between 0.89 and 0.98. In the deep pain group, the pain behavior response, i.e., the mean Rat Grimace Scale, was statistically significantly different when the saline subgroup was compared to the preemptive subgroups at all the aforementioned post-surgical periods except at 24 h (p ≤ .05 on one way ANOVA, Figure 1). However, the two preemptive regime groups did not differ significantly at all-time points. (p > .05; Figure 1). Furthermore, post hoc analysis followed by Bonferroni correction was performed, according to which at 2 h, a notable statistical reduction in RGS was evident in both TD and BD groups (p < .01; Figure 1), with the BD group displaying the lowest RGS among all (p < .0001; Figure 1), indicating effective pain alleviation. Subsequently, at 4 h, RGS in the BD and TD groups were comparable and statistically significantly lower than the SD group (p < .001; Figure 1). However at, the 6-h time point, the TD group exhibited the lowest RGS score, surpassing both BD and SD (p < .001). Finally, at 9 h, RGS in the BD and TD groups remained similar but markedly lower than the SD group, showing statistically significant result (p ≤ .05). Comparison of RGS among deep pain subgroups. Comparison of RGS in Study groups RGS in the deep pain group (a–e) was assessed at five time points (2, 4, 6, 9 and 24 h) for 30 min. The saline subgroup (SD) was taken as internal control group. The preemptive groups were Tramadol +lidocaine (TD) and Buprenorphine +Lidocaine (BD). Values represent the means ± S.E.M, with n = 7 for all subgroups. The RGS at 2, 4, 6, 9, and 24 h were significant (p < .001) and * indicate statistical significance. The error bar represents the standard error of the mean (SEM). p ≤ .05=*, p ≤ .001=**, p ≤ .0001=***, p ≥ .05=NS.
The difference in mean RGS pain score in rats across all time periods was statistically insignificant in tramadol, buprenorphine and saline subgroup (p > .05: Figure 2) by one way repeated measure ANOVA. However, this change in difference of mean RGS pain score in tramadol was obvious at 6 h postoperatively but it was statistically insignificant when compared with scores of other time points (p > .05: Figure 2). Comparison of drugs across time in deep pain group. The difference in mean RGS sores in the deep pain subgroups was assessed at five-time points (2, 4, 6, 9, and 24 h) for 30 min in tramadol (TD), buprenorphine, and saline subgroups. One-way repeated measure ANOVA shows statistically insignificant result (p > .05=NS). The saline (SD) was used as the internal control subgroup. The preemptive groups were Tramadol +lidocaine (TD) and Buprenorphine +Lidocaine (BD). For each subgroup, the values indicate the means ± S.E.M., with n = 7. The standard error of the mean (SEM) is shown by the error bar.
A comprehensive view of Rat Grimace Scale (RGS) in the superficial pain group (B) at various time points (2, 4, 6, 9, and 24 h) after surgery was also seen. Comparing the preemptive analgesia groups to the control group, a consistent lower RGS score were observed, with buprenorphine + lidocaine showing the most significant pain relief, especially at the 4 and 6-h mark (p ≤ 0.05; Figures 3 and 4). The RGS in the saline subgroup was statistically significantly higher (p ≤ .05; Figures 3 and 4) than the two preemptive regime subgroups, at all the post-surgical time points, as both buprenorphine and tramadol appeared to decrease the RGS statistically significantly as compared to saline-treated rats at 2, 4, 6, 9, and 24 h (p ≤ .05). Comparison of RGS among superficial pain subgroups. The RGS in the superficial pain group (b) at 2, 4, 6, 9, and 24 hours were assessed. The internal control group used was the saline subgroup (SS). The preemptive groups were Tramadol + Lidocaine (TS) and Buprenorphine + Lidocaine (BS). Values represent means ±S.E.M, for all subgroups with n = 7. Significant RGS during 2, 4, 6, 9 and 24 h is p < .05 and indicated by *. The error bar displayed SEM. Preemptive subgroups showed successive reductions in RGS compared to the saline subgroup at all-time points (oneway ANOVA followed by post hoc analysis). The dark line indicates ANOVA and the light line indicates post hoc analysis p ≤ .05=*, p ≤ .001=**, p ≤ .0001=***, p ≥ .05=NS. Effect of the individual drug across all time periods in the superficial pain group Effect of tramadol (TS), buprenorphine (BS) and saline (SS) on the difference of mean RGS score in superficial pain subgroups after 2 h, 4 h, 6 h, 9 h & 24 h period post-surgery. The Saline (SS) subgroup was an internal control subgroup while TS & BS were preemptive groups. Figure displays statistically insignificant results when the mean RGS is analyzed after giving TS, BS & SS subgroups in rats across the all-time periods by one-way repeated measure ANOVA, p > .05=NS.
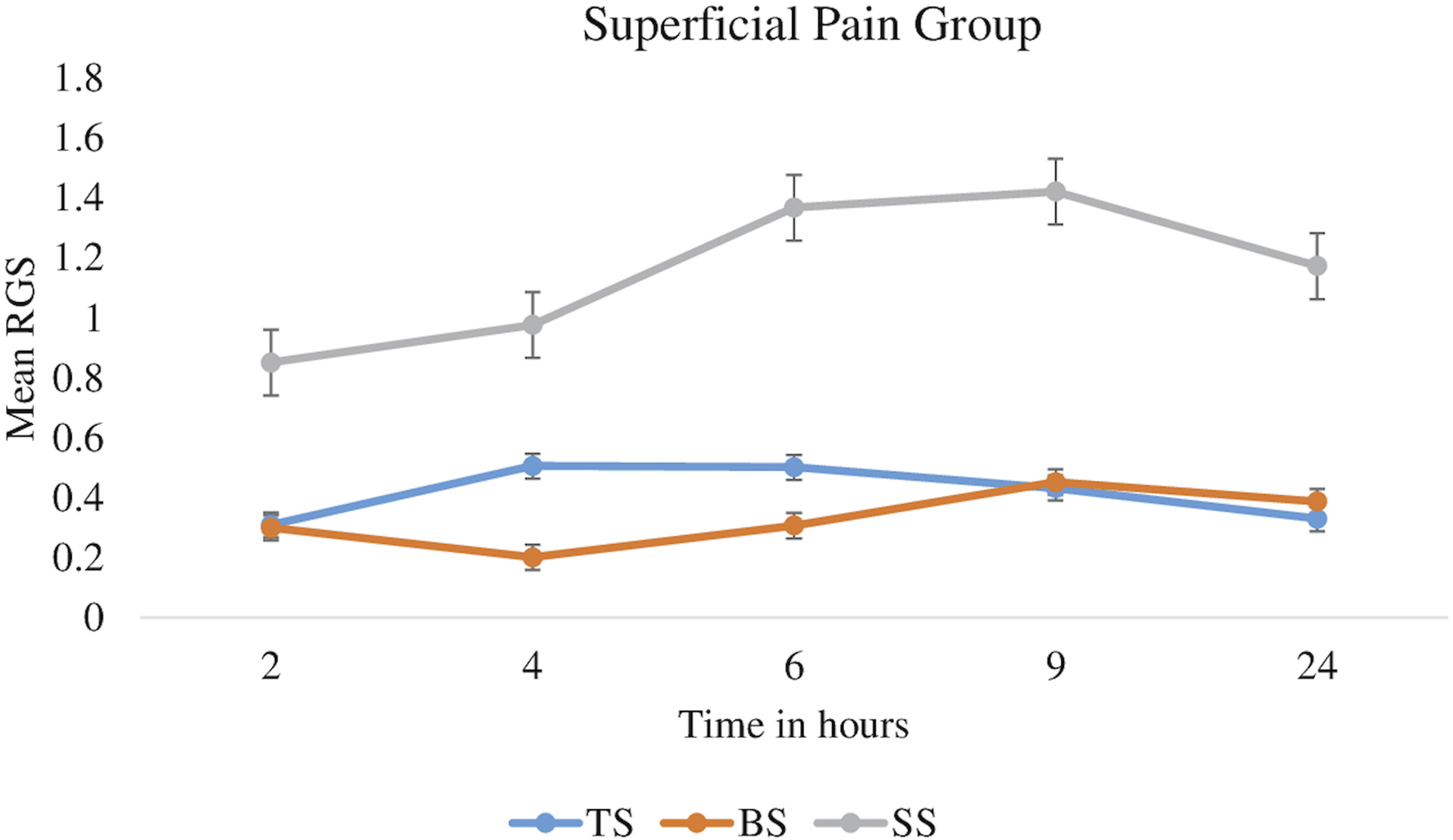
One way repeated measure ANOVA shows statistically insignificant result, when difference in mean RGS pain score of saline, buprenorphine and tramadol was trace across all the time point post-surgery. Although this variation in difference of RGS was more noticeable at 4 and 6 h in preemptive group but result was statistically insignificant (p > .05: Figure 4).
c-fos expression
The quantification of c-fos expression involved tallying the number of c-fos positive cells in lamina I-II, III-IV, and X, along with measuring the optical density of DAB-stained c-fos positive regions. Both analyses assessing the intensity of c-fos positive neurons were performed using Image J software (ANOVA). The photomicrographs (Figures 5 and 6) demonstrate increased c-fos protein expression in specific spinal cord laminae (I-II, III-IV, and X) following the uterine surgery in the deep pain group, displaying heightened neural activity. (Figures 5 and 6) Photomicrographs showing the c-fos expression in the spinal at 4x. It shows the expression of cfos protein in the spinal cord after the surgery. (a) Drawing of a section of the spinal cord adapted from Paxinos and Franklin Altas
27
The dotted area delimitates the lamina I-II, III-IV of the posterior horn and lamina X around the center canal showing the region for counting c-fos positive cells. (b) Presented the areas for measuring the optical density of c-fos expression in lamina I-II, III-IV, and X of the spinal cord. Scale bar is 100µm × 40X. Figure c and d shows the lamina I-II, I-IV and lamina X at 100µm × 100X. Arrows displays c-fos positive cells. Photomicrographs of c-fos expression in the spinal cord among deep pain subgroups. Pictures taken at magnifications of 100 (a–f), 200 (a-f), and 400 (aa-ff), demonstrating the expression of cfos in the lamina I-II, III-IV, and X of thoracolumbar spinal cord sections from the deep pain group of saline subgroup (SD), the tramadol subgroup (TD), and the buprenorphine subgroup (BD) following uterine surgery, figures a to f. A c-fos positive neuron with a dark stain was shown by the arrow. With n = 3 for each group in the profound pain groups. Scale bars for figures (a–f) are 100 µm for 100X, while those for figures (a-f) are 100 µm for 200X and figures (aa-ff) showed a scale bar of 10 µm with magnification of 400X.

In deep pain groups, the saline subgroups exhibited a statistically significant increase in the number of c-fos positive neurons in laminae I-II, III-IV, and X when compared to the tramadol and buprenorphine subgroups (p ≤ .0; Figure 7; Table 1). Although the total number of c-fos positive neurons appeared to be reduced in the tramadol subgroup compared to the buprenorphine subgroup, but this difference was statistically insignificant (p > .05). The preemptive groups showed statistically significantly decreased expression of c-fos in lamina X (p ≤ .05) in the deep pain group but not in the superficial pain group as compared to the saline subgroup (p > .05; Figure 7; Table 1). The c-fos expression measured through the optical density also appeared to be statistically reduced in preemptive subgroups as compared to control group in uterine pain model. (p ≤ .05; Figure 7). Mean cell count and optical density of c-fos immunopositive cells in the spinal cord between deep pain subgroup: (a, c and e) mean cell count / mm2 of c-fos immunoreactive cells in lamina I-II, III-IV, and lamina X of spinal cord in uterine pain model (n = 3). The value reflects the means ± SEM. While graph (b, d and f) displays a semi-quantitative analysis of the optical density of c-fos expression / mm2 in I-II, III-IV and X respectively. At two hours following surgery, the comparison of the SD (saline deep pain group), TD (Tramadol deep pain subgroups), and BD (Buprenorphine deep pain subgroup) for c-fos expression in the uterine model is statistically significant (p ≤ .05). p ≤ .05=*, p ≤ .001=**, p ≤ .0001=***, p ≥ .05=NS. Comparing c-fos expression between the study groups interms of cell count and optical density. Within the group comparison of c-fos cell count (CC) and optical density (OD) by post hoc analysis with Bonferroni correction (p ≤ .02). TD: Tramadol deep Pain subgroup, BD: Buprenorphine deep pain subgroup, SD: Saline deep pain subgroup. While TS: Tramadol superficial pain subgroup, BS: Buprenorphine superficial subgroup, SS: Saline superficial pain subgroup. Lamina I-II and III-IV were the regions of the Posterior horn of the spinal cord, Lamina X is around the central canal of the spinal cord. (n = 3). The bold values are the p values which are computed after paired group mean comparison using Independent T test.
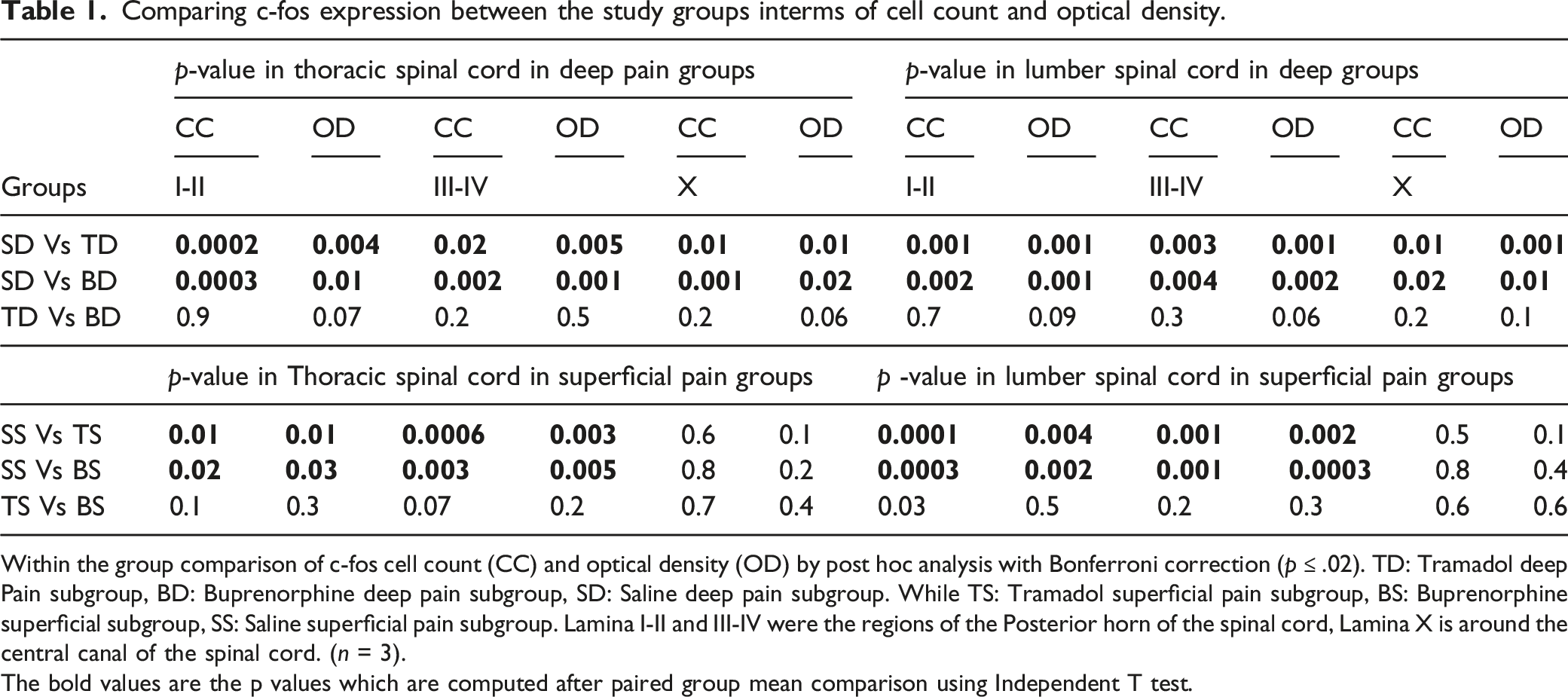
Post hoc analysis with Bonferroni correction was used to analyze the results within the groups. After 2 hours post-surgery, the deep pain subgroups treated with Buprenorphine (BD) and Tramadol (TD) displayed reduced c-fos immunoreactive cell counts and lower optical density (OD) when compared to the saline deep pain group (SD) in laminae I-II, III-IV and X, which were statistically significant (p ≤ .05; Table 1). When buprenorphine was compared with tramadol, it was found statistically insignificant (p > .05; Table 1).
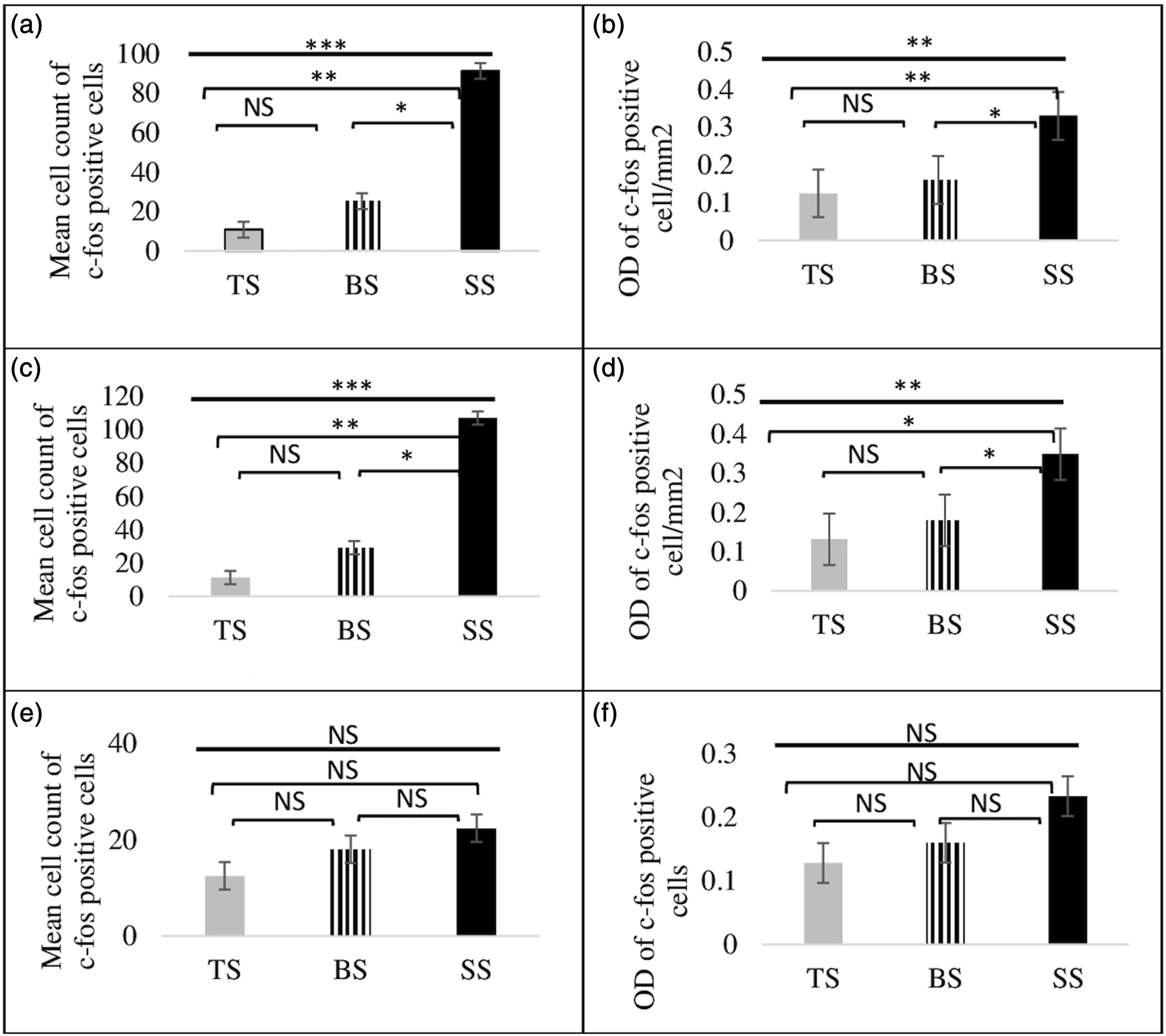
In the superficial pain group, a pronounced statistically significant reduction was observed in the number of c-fos positive neurons within spinal cord laminae I-II and III-IV, 2 hours postoperative when treated with Buprenorphine (BS) and Tramadol (TS) as compared to the saline subgroup (SS) (p < .05; Figures 8 and 9: Table 1).
27
Photomicrographs of the spinal cord showing expression of c-fos positive cells in superficial pain subgroups. Figure showing the expression of c-fos positive neurons in lamina I-II and III-IV in the dorsal horn of rat’s spinal cord following postoperative surgery in the superficial pain subgroup, 100X (figures A-E), 200X (a-f) with scale bar of 100µm, and 400x (aa-ff) with a scale bar of 10µm, demonstrating the expression of c-fos in the lamina I-II, III-IV, and X of thoracolumbar spinal cord sections from the superficial pain group of saline subgroup (SS), tramadol subgroup (TS), and buprenorphine subgroup (BS) following uterine surgery (figures a to f). A c-fos positive neuron with a dark stain was shown by the arrow. With n =3 for each group. Mean cell count and optical density of c-fos immunopositive in the spinal cord between superficial pain subgroup. Figure (a, c, and e) shows average number of c-fos immunoreactive cells mm2 in spinal cord lamina I-II, III-IV, and X (n = 3) in the superficial pain group. Value was SEM of means. Semi-quantitative examination of optical density of c-fos expression/ mm2 shown in graphs (b, d, and f in lamina I-II, III-IV & X respectively). Comparison of c-fos expression in the superficial pain group, between the SS (saline superficial pain subgroup), TS (tramadol d pain subgroups), and BS (buprenorphine superficial pain subgroup) is statistically significant at two hours after surgery (p < .05).
Discussion
The outcomes from this research highlight the impact of post-operative surgical pain on the dorsal horn of the spinal cord and the Rat Grimace Scale (RGS). Importantly, a single dose of preemptive therapy administered prior to surgery effectively mitigated these pain indicators. The importance of assessing ongoing post-operative pain in laboratory rats was identified, and thus, evaluated by two key pain metrics: RGS, which is a pain rating scale,28,29 and the expression of c-fos in the spinal cord. The methodology in this study was rooted in neuroscientific principles, investigating the postoperative changes occurring at molecular, cellular, and behavioral levels within the pain pathways. 28
In this research study, the deep pain group exhibited consistently higher RGS in the saline-treated subgroup across all postsurgical time points. However, the administration of buprenorphine significantly reduced the RGS, corroborating the work by Leung et al., 28 which emphasized the effectiveness of buprenorphine as a postoperative pain reliever. Notably, buprenorphine is a semi-synthetic partial agonist of the µ-opioid receptor and an antagonist of the kappa opioid receptor. Its unique pharmacodynamics associate it with a lower incidence of opioid-related adverse effects and misuse risk when compared to other complete opioid agonists. Furthermore, its extended duration of action stems from its gradual dissociation from the opioid receptor. 30
When buprenorphine was combined with both local and general anesthesia in the current study, a reduction in pain transmission at lower doses was reported. This finding contrasts with the study by Bisgaard et al., 31 which did not find a reduction in visceral pain with subcutaneous instillation of lower dose buprenorphine injection. A similar pattern was observed with carprofen and ketoprofen, two additional non-steroidal anti-inflammatory drugs (NSAIDs), which either required significantly higher dosages or were ineffective at reducing RGS scores at lower doses.19,32 The lower RGS in this study may be attributed to reduce sensitization of dorsal horn neurons, possibly influenced by the peripheral action of lidocaine, which blocks voltage-gated sodium channels, preventing the transmission of pain signals back to the central nervous system. 33 Additionally, the central actions of buprenorphine or tramadol in the presence of isoflurane, which acts on opioid receptors in the central nervous system, 34 may further contribute to these lower scores. The use of different methods for drug administration, when combined (preemptive regimen), accounts for the observed reduction in RGS scores on the Rat Grimace Scale in this study.
In the current study, the tramadol subgroup in both deep and superficial pain groups, in combination with lidocaine, consistently exhibited lower mean Rat Grimace Scale (RGS) at all-time points, extending up to 24 hours post-surgery. Findings of this study resonate with the work of Pratap et al., and Piras et al., who observed a similar sustained decrease in pain scores lasting up to 24 h with intra-peritoneal tramadol administration during laparoscopic appendectomy.35,36 This highlights the enduring effectiveness of tramadol in postoperative pain management.
Continuous observations conducted at predetermined time intervals in this study proved to be more accurate in assessing postoperative pain as compared to single observation points. 37 This approach aligns with the findings of Foroud et al., (2015), suggesting that combining different medication classes to target various points along the pain pathway can yield additive or synergistic effects. Such a strategy minimizes the potential for unfavorable side effects associated with larger doses of specific medications when used in isolation.38,39 Additionally, this study supports the idea that co-administration of tramadol and lidocaine can result in significantly reduced RGS in laboratory rats compared to the saline subgroup. 40 These results demonstrate the potential advantages of combining drugs to achieve better pain management outcomes, as suggested by earlier studies.
Extensive research has delved into the intricate mechanisms of c-fos induction within postoperative pain models, often coupled with behavioral assessments. A plethora of medications have been explored to hinder the activation of c-fos in spinal neurons with some drugs, having demonstrated efficacy, (opioids, neurokinin receptor antagonists, NMDA receptor antagonists, GABAB receptor agonists, EP1 receptor antagonist, non-steroidal anti-inflammatory medicines, anti-convulsants, and nitric oxide synthase inhibitors), the comprehensive suppression of c-fos expression remains a challenge. 40
However, findings reveal a notable departure from this conventional wisdom. Both buprenorphine and tramadol, as part of preemptive regimen, were successful in completely suppressing c-fos expression in the posterior grey column of the spinal cord, including lamina X, within the uterine pain model in current study. This suppression was quantified using Image J software, which allowed to evaluate the optical density and count of c-fos in lamina I-II, III-IV, and X of the rats' spinal cord. This striking reduction in c-fos expression suggests a significant decrease in nociception transmission from the surgical site. Notably, these results are consistent with prior researches that have reported a similar overall reduction in c-fos expression in preemptive treatment groups compared to saline groups in both superficial and deep pain models.41,42
A growing body of evidence points to the potential mechanisms by which tramadol and buprenorphine function to reduce neuronal activity in the spinal cord. They achieve this by diminishing excitatory currents and enhancing inhibitory currents, particularly in the rat dorsal horn laminae I, II, III, and IV. 43 This aligns with the observations in current study, in which reduction in c-fos expression was noted, both in terms of intensity measurement represented by optical density and the number of c-fos counts, particularly in lamina I-II and III-IV. This suggests that these treatments specifically target and reduce neuronal activity within the nociceptive transmission regions. 44
The novel efficacy of buprenorphine and tramadol in suppressing c-fos expression presents promising avenues for the management of postoperative pain, with the potential for broader implications in pain modulation research. These results may pave the way for further investigations into the precise mechanisms by which these medications exert their effects, advancing our understanding of pain pathways and therapeutic interventions.
In the deep pain group, this study noted a subtle difference between tramadol-treated animals and those administered with buprenorphine, with the former displaying slightly lower c-fos levels 2 hours post-surgery. Both tramadol and buprenorphine effectively reduced overall c-fos expression and the transmission of postoperative pain when compared to the saline subgroup, with tramadol exhibiting a slight advantage. This could be attributed to several factors, such as tramadol’s slightly longer half-life or the combined action of lidocaine, tramadol, and isoflurane, or may be due to its mode of action and rout of transmission. The result of this study are comparable to those reported by Kumar et al., who observed transient antinociceptive responses following incisions in postoperative animals that lasted for around 23 and 180 min when plain and liposomal bupivacaine were used, respectively. 45 However, it’s important to note that the difference between tramadol and buprenorphine in this study was statistically insignificant when comparing the two preemptive drugs.
Furthermore, the study revealed an interesting correlation between c-fos expression in laminae I-II, and III-IV and the dorsal central grey area (lamina X) of the spinal cord in the deep pain group. On comparing the saline group with the tramadol or buprenorphine groups in deep pain group, a statistically significant increase in c-fos levels in lamina X was observed, suggesting that visceral pain transmission following uterine surgery was significantly reduced by preemptive treatments. This aligns with earlier findings following other visceral noxious stimulations Qi Xu et al., (2021), 46 contrasting with noxious somatic stimulation, which only caused c-fos activation in the superficial laminae of the spinal cord. In the superficial pain group, buprenorphine, and tramadol, when administered along with lidocaine, led to a reduction in c-fos expression in the lower thoracic and upper lumbar dorsal horn up to lamina IV, except the dorsal central grey area (lamina X). Hence, both analgesic drugs have a consistent effect in suppressing c-fos expression in the same areas in both superficial and deep pain groups, as supported in study conducted by Kovelowski et al., when comparing morphine and tramadol in the hind paws of rats. 47
These findings collectively accentuate the consistent and significant reduction in pain behavior in the preemptive analgesia groups, highlighting the efficacy of preemptive analgesia, particularly buprenorphine + lidocaine, in mitigating pain responses in the uterine pain model. Further, these results hold significant implications for the intensive care setting, where tramadol and buprenorphine can be carefully titrated to provide comprehensive analgesia for postoperative patients. The nuanced differences between these medications and their specific effects on c-fos expression apprise about pain management strategies and offer insights into tailored treatment options.
Further investigation is required to assess dose related effect of the preemptive regime in the postoperative pain model and that only a limited number of analgesia and local anesthesia were used in the current study. Furthermore, only female rats were used. Although previous studies using both the RGS and c-fos expression in the spinal cord did not identify sex differences, it is known that there are sex differences in sensitivity to analgesics. Therefore, the results of this study should be cautiously generalized to males, as their response to the preemptive regime may differ.
Our study is not limited to the expansion of basic knowledge; rather, it has some significant clinical implications. In future studies, it would be valuable to apply the preemptive regime developed in this study to a cohort of humans undergoing gynecological or obstetric surgeries and evaluate its effectiveness using a visual analogue scale for pain assessment. By comparing subjective results with the objective findings from the animal model study, further correlations can be drawn between the preemptive regime and its efficacy in pain suppression. Furthermore, studies on the dorsal root ganglion of the spinal cord and brain level can give a holistic view of different levels of pain suppression.
Conclusion
In conclusion, this research has significantly advanced the comprehension of postoperative pain mechanisms by exploring molecular, cellular, and behavioral dimensions. The combined assessment of RGS and c-fos expression in the spinal cord offers a more comprehensive and reliable approach to pain assessment. In the rat uterine and superficial pain model, the preemptive analgesia regimen administered before surgery effectively mitigated nociceptive pain transmission within distinct spinal cord laminae. This research has unveiled potential targets for refining pain management strategies, particularly in the context of refractory pain unresponsive to conventional treatments. By integrating behavioral and molecular insights, the foundation of evidence-based pain management, enhancing the well-being of research animals, and potentially informing clinical practice has been laid. These findings accentuate the potential of preemptive analgesia as a valuable strategy in the field of pain modulation, stressing the importance of continued research in this vital area.
Footnotes
Acknowledgements
We would like to acknowledge Khyber Medical University Peshawar, Pakistan for funding this project.
Author contributions
Experiment performer and data analysis: Saima Mumtaz, Najma Baseer. Experiment conception and design: Najma Baseer, Saima Mumtaz and Syed Hamid Habib. Manuscript writing: Saima Mumtaz, Najma Baseer and Syed Hamid Habib.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Statement
Data Availability Statement
The data that support the findings of this study are available from the corresponding author upon reasonable request.
