Abstract
Pain is a major symptom in cancer patients, and cancer-induced bone pain (CIBP) is the most common type of moderate and severe cancer-related pain. The current available analgesic treatments for CIBP have adverse effects as well as limited therapeutic effects. Acupuncture is proved effective in pain management as a safe alternative therapy. We evaluated the analgesic effect of acupuncture in treatment of cancer pain and try to explore the underlying analgesic mechanisms. Nude mice were inoculated with cancer cells into the left distal femur to establish cancer pain model. Electroacupuncture (EA) treatment was applied for the xenograft animals. Pain behaviors of mice were evaluated, followed by the detections of neuropeptide-related and inflammation-related indicators in peripheral and central levels. EA treatment alleviated cancer-induced pain behaviors covering mechanical allodynia, thermal hyperalgesia and spontaneous pain, and also down-regulated immunofluorescence expressions of neuropeptide CGRP and p75 in the skin of affected plantar area in xenograft mice, and inhibited expressions of overexpressed neuropeptide-related and inflammation-related protein in the lumbar spinal cord of xenograft mice. Overall, our findings suggest that EA treatment ameliorated cancer-induced pain behaviors in the mouse xenograft model of cancer pain, possibly through inhibiting the expressions of neuropeptide-related and inflammation-related protein in central level following tumor cell xenografts.
Introduction
Pain is a major symptom in cancer patients. Most cancer patients with advanced or terminal cancers suffer from chronic pain torment which is related to treatment failure and/or tumor progression.1,2 Cancer-induced bone pain (CIBP) is the most common type of moderate and severe cancer-related pain, which accounts for approximately 75% of all chronic cancer pain. 3 Pain caused by metastasis to the bone is the most common source of moderate and severe pain symptom. Unfortunately, it has been reported that over 50% of patients with advanced or metastatic cancer have inadequate pain relief and control because of the relative limited analgesic efficacy and the side effects of current treatments. 4 The available analgesic treatments for CIBP include opioids, bisphosphonates, radiotherapy, denosumab, nerve block, non-steroidal anti-inflammatory drugs (NSAIDs), but each of them has certain limitations in analgesic efficacy. 5 In addition, the adverse effects and limited therapeutic effects of conventional pharmacological interventions propose critical challenges to pain treatment. Therefore, relatively effective and optimized therapeutic approaches are needed to develop.
Acupuncture therapy is an important part of Traditional Chinese Medicine (TCM), and it has long been applied for pain management as an effective and safe alternative treatment. 6 For decades, many clinical trials have confirmed the effectiveness of acupuncture in controlling cancer pain,6–8 and in reducing the application of analgesics and the accompanying side effects as well,9,10 in order to improve and enhance life qualities of cancer patients. As a milestone in pain management, acupuncture therapy has been recommended in the practice guidelines from European Oncology Nursing Society (EONS), American Society of Clinical Oncology and National Comprehensive Cancer Network (NCCN) as the actively used complementary and alternative medical treatment for managing chronic cancer pain in cancer survivors.6,11–14 In recent decades, there has been an accumulating body of studies dedicate to explore the mechanisms of acupuncture treatment in alleviating cancer pain. However, the underlying analgesic mechanisms of EA’s therapeutic effects are still yet to be fully elucidated.
Previous researches have shown some possible mechanism underlying the analgesic effect of acupuncture against cancer pain. According to available studies, the analgesic actions of acupuncture are correlated to the inhibition of CXCL12, the suppression of the PI3K/Akt/JNK1/2 signaling pathway, downregulation of TRPV1 mRNA, and the control of P2X3R, etc.15–18 In addition, our previous study explored the analgesic effect and intrinsic mechanism of acupuncture therapy against cancer related pain 19 (chemotherapy-induced peripheral pain) suggesting that acupuncture treatment attenuates chemotherapy-induced neuropathic pain in rodents, and the analgesic action of acupuncture might be associated with its inhibitory effect on glial activity and TLR4/NF-κB signals in the spinal cord in rodent. Based on the findings from previous studies, and combined with the results from our previous research, we hypothesize that the analgesic effect of acupuncture on cancer pain might be connected with its control and regulation on nerve growth factor, related enzymes and receptors, as well as the immune inflammatory factors.
The aim of the present study is to observe and evaluate the potential effect of acupuncture on pain and nociceptive behaviors in a mouse xenograft model of cancer pain, and the possible underlying mechanism of the analgesic action induced by acupuncture. Besides, it is noteworthy that the research objectives the present study focus on covers different types of pain, including mechanical hyperalgesia, thermal allodynia and spontaneous pain, thereby contributing a more comprehensive perspective to evaluate the analgesic effect of acupuncture treatment against cancer pain.
Materials and methods
Animals
A total of 34 adult female BALB/c nude mice (20 - 25g) were used in this study. All animals were purchased from China Academy of Military Science (license No. SCKX (JUN) 2007-004) and housed under conditions of controlled temperature (20 °C–25 °C) with a light/dark cycle of 12:12 h light (7:00 a.m. on, 19:00 p.m. off), and allowed access to standard chow and water ad libitum. All procedures complied with the guidelines of ARRIVE (Animal Research: Reporting of In Vivo Experiments) and were conducted in accordance with the National Institutes of Health Guide for the care and use of laboratory animals, and approved by the Institutional Animal Care and Use Committee of China Academy of Chinese Medical Sciences (reference No. 2015010801). All efforts were made to minimize discomfort and the number of animals used. Mice were deeply anaesthetized and euthanized by injection of sodium pentobarbital (60 mg/kg, i.p.) at the end of experiments.
Cell culture
Human breast cancer cell line MDA-MB-231 was purchased from the American Type Culture Collection (Manassas, VA, USA). MDA-MB-231 cells were grown in MEMA medium (Gibco, Grand Island, NY, USA) and supplemented with penicillin, streptomycin, fetal bovine serum (Sigma, St. Louis, MO, USA), and maintained as attached cells at 37°C in 5% carbon dioxide.
Cancer pain model
Animals were inoculated with MDA-MB-231 cells into the left distal femur (Figure 1(a)). The protocol of tumor cell xenografts with MDA-MB-231 cells used has been described in previous studies.20–22 Mice were anaesthetized by sodium pentobarbital (40 mg/kg, i.p.) and an arthrotomy was performed. A superficial incision was made around the skin overlying the patella and the patellar ligament was then cut, exposing the condyles of the distal femur. Mice were posed in a supine position with the ipsilateral stifle joint bent at 90° to provide a clear angle of view to observe the patella. The bone was initially cored with a 30-gauge needle manually inserted at the level of the intercondylar notch. Then a 26-gauge needle was inserted to inject sterile phosphate-buffered saline (PBS) containing 1 × 106 MDA-MB-231 cells (50 μL) into intramedullary space. The injectate was infused slowly over 1 min. To prevent cell reflux outside the bone, the injection site was sealed with dental grade amalgam (generously provide by the department of dentistry in Xiyuan Hospital) followed by irrigation with filtered water. All efforts were made to minimize the suffering and number of animals used. To establish a preclinical model of breast CIBP by intra-femoral injection of MDA-MB-231 cancer cells into the left distal femoral epiphysis of female nude mice. (a) Schematic illustration of the establishment procedure of carcinoma xenograft model. (b) Hematoxylin and Eosin (h) & (e) staining of the bone destruction in carcinoma xenograft mice. Magnification (×200 μm). Visible in the figure are normal (black arrows) and eroded (yellow arrows) trabecular bones.
Bone histology
In the end of the third week after inoculation, some mice from control and tumor cell xenografts groups in random selection were anesthetized with an overdose of sodium pentobarbital and trans-cardially perfused with 200 mL of 0.9% normal saline followed by 400 mL of 4% paraformaldehyde. Tumor-bearing femora and surrounding tissues (left distal femur) were dissected and fixed in 2.5% buffered glutaraldehyde solution for 2 days, postfixed in 10% buffered formalin, and then decalcified in 10% ethyl enediaminetetraacetic acid (EDTA, pH 7.4) over 2 weeks, and embedded in paraffin. Sections 5 µm in thickness were stained with hematoxylin and eosin (HE), and examined by a light microscope (Olympus BX-53).
Experimental design
Thirty-four mice were randomly divided into four groups as follows: Control (n = 10), Tumor (n = 10), Tumor + EA (n = 7), Tumor + sham EA (n = 7).
After baseline behavioral assessments, mice received an intra-femoral injection of carcinoma cells in Tumor, Tumor + EA and Tumor + sham EA groups, or equivalent volume of PBS in Control group. Mice in Tumor + EA received EA treatment at bilateral GB30 for 20 min every other day. Mice in the Tumor + sham EA needled superficially at bilateral GB30 points without electrical stimulation. Behavioral tests were performed at 1, 2, 3, 4, 5, and 6 weeks after inoculation.
Following testing on the third week, 3 mice in Control and 3 mice in Tumor were perfused for histology of HE staining. Following testing on the sixth week, the rest of animals were euthanized for immunofluorescence staining and western blot assays.
Electroacupuncture treatment
Acupoints of GB30 (Huantiao) near bilateral hip joints were used for EA treatment.23,24 The acupoint of GB30 in mice is located at the junction of the lateral 1/3 and medial 2/3 of the distance between the greater trochanter and the hiatus of the sacrum. Acupuncture needles at each of the bilateral hip joints were inserted as follows: one stainless steel needle with 0.25 mm diameter was inserted into the GB30 acupoint and the other one 5 mm below the GB30 acupoint (4 mm in depth). The tips of the electrode cables from an electroacupuncture apparatus (HANS-200A) were connected to a pair of steel acupuncture needles, and constant current pulses (2 Hz, 1 mA) were applied for 20 min. For sham EA, acupuncture needles were inserted into bilateral GB30 points of mice in the same way as in the EA group, but without electrical stimulation. Both EA and sham EA treatments were performed under isoflurane anesthesia and were applied once every other day for 4 weeks following tumor cell xenografts in mice.
Mechanical allodynia
Animals were habituated to the environment for 2 days before behavioral testing. Baseline measurement of mechanical withdrawal thresholds for both hind paws (left and right paws) of mice was performed prior to xenografts operation. Mechanical withdrawal thresholds of both right and left hind paws in mice were measured using calibrated von Frey filaments of binding forces ranging from 0.04 to 2 g (North Coast Medical, Inc., Gilroy, CA, USA), and applied to the plantar surface of mice.
25
A calibrated ascending series of von Frey filaments were applied to the plantar surface of left and right hind paws (Figure 2(d)). The threshold to mechanical stimuli at paw withdrawal was calculated as the force value (in grams). Each trial comprised 4 tests at 5–10 min intervals, and the average was measured. The area under the curve (AUC) of withdrawal thresholds for hind paws in each group was calculated to investigate the overall effect. Electroacupuncture (EA) attenuated mechanical allodynia in the carcinoma xenograft mice model of cancer pain. Paw withdrawal thresholds (PWT) of (a) the ipsilateral hind limb (tumor-injected) and (b) contralateral hind limb (without tumor) in mice were evaluated in a time-course study. (c) Normalized area under the curve (AUC) analysis of Figure 2(a) and (b). (d) Schematic illustration of mechanical von Frey test. **p < .01 versus Control; #p < .05, ##p < .01 versus Tumor. n = 7 ∼ 10 rats/group. One-way or two-way ANOVA followed by Tukey post hoc test was used for statistical analysis. Abbreviations: EA, electroacupuncture; PWT, paw withdrawal thresholds; AUC, area under the curve.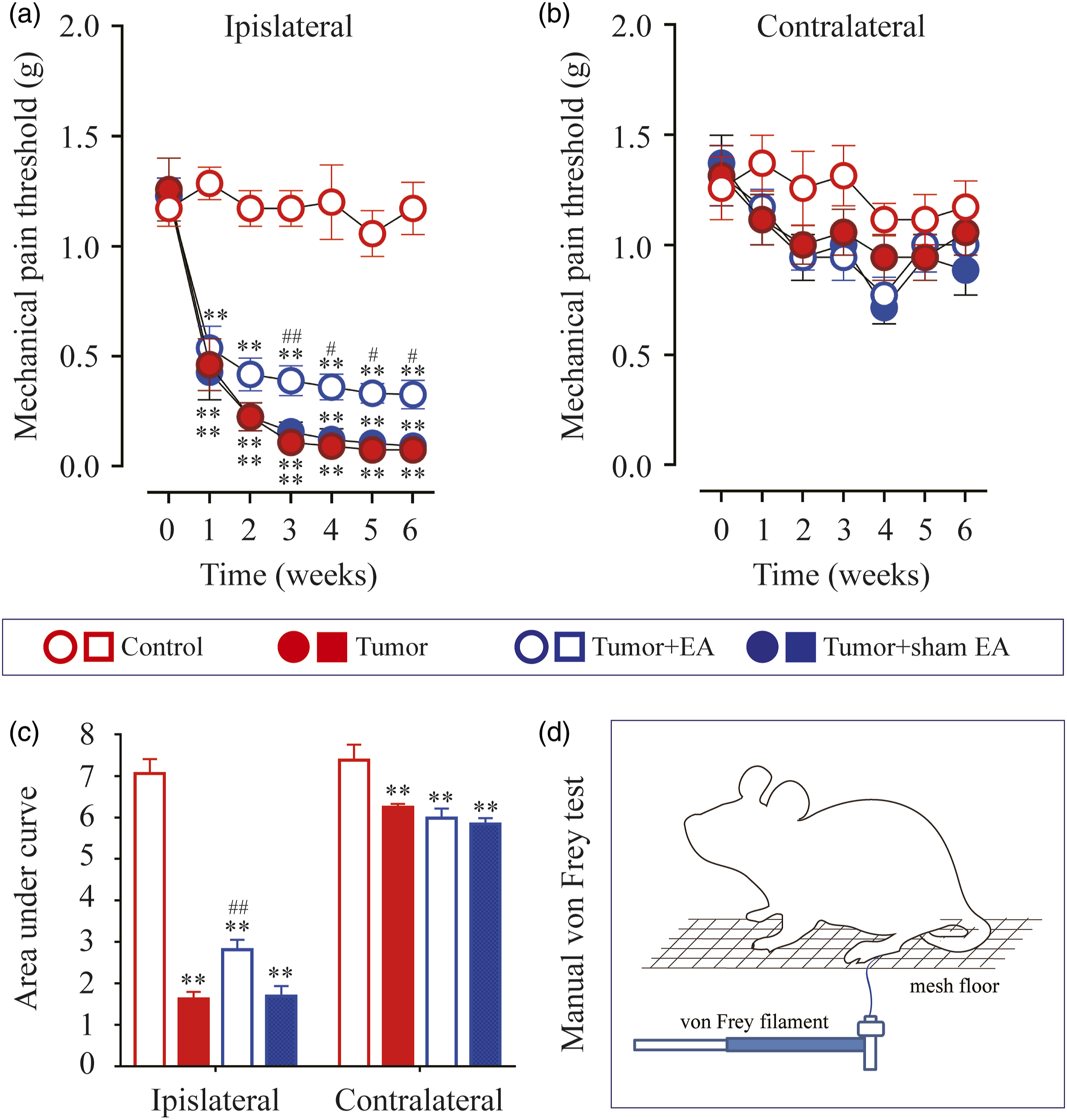
Thermal hyperalgesia
Animals were habituated to the environment for 2 days before behavioral testing. Thermal hyperalgesia was evaluated by measuring paw withdrawal latency to heat stimulation,
26
using a hotplate analgesia meter (Univ. California, San Diego, CA, USA). Each mouse was placed in a Plexiglas chamber containing a smooth, temperature-controlled glass floor and was acclimated to the testing apparatuses prior to the application of thermal stimulus. Paw withdrawal latencies to a radiant source of heat in both right and left hind paws of mice were measured to assess nociceptive thresholds to acute thermal noxious stimulus by using a Paw Thermal Stimulator (Figure 3(d)). The withdrawal latencies of left and right paws to each thermal stimulus were recorded from the start of the radiant heat until the withdrawal reaction occurs. The radiation was automatically shut off at 20 s to avoid the risk of damage to the hind paw. Each trial comprised 4 tests at 5–10 min intervals, and the average was measured. The area under the curve (AUC) of withdrawal latencies for hind paws in each group was calculated to investigate the overall effect. Electroacupuncture (EA) attenuated thermal hyperalgesia in the carcinoma xenograft mice model of cancer pain. Paw withdrawal thresholds (PWT) of (a) the ipsilateral hind limb (tumor-injected) and (b) contralateral hind limb (without tumor) in mice were evaluated in a time-course study. (c) Normalized area under the curve (AUC) analysis of Figure 3(a) and (b). (d) Schematic illustration of thermal withdraw latency test. *p < .05, **p < .01 versus Control; #p < .05, ##p < .01 versus Tumor. n = 7 ∼ 10 rats/group. One-way or two-way ANOVA followed by Tukey post hoc test was used for statistical analysis. Abbreviations: EA, electroacupuncture; PWT, paw withdrawal thresholds; AUC, area under the curve.
Spontaneous pain
Spontaneous foot-lifting number (SFLn) was measured to evaluate the intensity of spontaneous pain in rodents.27,28 Each mouse was placed on a plate with a flat floor in a quiet environment, followed by careful observation and recording of spontaneous pain behavior. After acclimatization, cumulative number of times that mice lift the tumor-bearing limbs (left side) off the floor was recorded, whereas any lift associated with locomotion, grooming, exploratory behavior and body repositioning was excluded from the test. SFLn was recorded for each mouse over 2-min periods with 20-min intervals between 4 tests, and the average was measured. The area under the curve (AUC) of SFLn for left hind paws in each group was calculated to investigate the overall effect.
Immunofluorescence staining
Mice were anesthetized with an overdose of sodium pentobarbital and trans-cardially perfused with 200 mL of 0.9% normal saline followed by 400 mL of 4% paraformaldehyde. The hind paw skin including sub-dermal layers was removed and cryo-protected with 20% sucrose in potassium phosphate buffer saline (KPBS) overnight at 4°C. 29 The collected tissue was sliced into 20-μm thick sections using a freezing microtome (REM-710, Yamato Koki Industrial, Japan). The frozen skin slices were blocked with PBS containing 10% donkey serum, and stained with the primary antibodies to calcitonin gene-related peptide (CGRP) (Rabbit monoclonal, Abcam, diluted 1:500) and p75 (Mouse monoclonal, Abcam, diluted 1:500). Slices were then rinsed in PBS and incubated with secondary antibodies (Donkey anti-rabbit IgG, Alexa Fluor™ 488 and Donkey anti-mouse IgG, Alexa Fluor™ 594, Invitrogen, diluted 1:500). Images were obtained and examined using a confocal fluorescence microscopy (FV1000, Olympus, Tokyo, Japan) and stored on digital media. For each skin sample, the lengths of CGRP and p75 positive nerves per 3–4 high power fields were calculated and assessed in the epidermis.
Western blot assay
After 6 weeks following xenografts operation, mice were killed and the dissected spinal cords (L3-L6) were snap-frozen and stored at −80°C. Whole-cell lysates were prepared from the lumbar region of the spinal cord and protein concentrations were determined using a BCA protein assay kit (Thermo Scientific, USA). Equal amounts of protein extract were resolved by 10% SDS-PAGE gels and transferred to a polyvinylidene difluoride (PVDF) membrane (Merck Millipore, USA). Membranes were then incubated with anti-COX2 (Rabbit monoclonal, Abcam, diluted 1:1000), anti-NF-κB p65 (Rabbit monoclonal, CST, diluted 1:1000), anti-IL-1β (Rabbit monoclonal, Abcam, diluted 1:1000), anti-p75 (Mouse monoclonal, Abcam, diluted 1:1000), anti-TrkA (Rabbit monoclonal, Abcam, diluted 1:1000), anti-VEGFr (Rabbit monoclonal, Abcam, diluted 1:1000) and anti-GAPDH (Rabbit monoclonal, CST, diluted 1:1000). All membranes with antibodies were incubated overnight at 4°C and then incubated with anti-rabbit IgG (1:5000; CST, USA) or anti-mouse IgG (1:5000; CST, USA) for 1 h at room temperature. Protein bands were detected with the use of a chemiluminescent substrate (ECL Prime Western Blotting Detection Reagents; Amersham, UK) and visualized using a luminescent image analyzer (ChemiDoc Imaging System, Bio-Rad, USA).
Statistical analysis
Statistical analysis was performed using GraphPad Prism 8.0 (La Jolla, CA, USA). All data were presented as mean ± SEM and analyzed with repeated-measures one-way ANOVA followed by Dunnett’s post-hoc test, and two-way ANOVA followed by Bonferroni’s multiple comparison test. In all cases, p value of <0.05 was considered a statistical significant.
Results
Histological evaluation of bone destruction
We established a preclinical model of breast CIBP by intra-femoral injection of MDA-MB-231 cancer cells into the left distal femoral epiphysis of female nude mice (Figure 1(a)). HE staining confirmed obvious cancer cell infiltration (within the yellow dotted lines) and significant trabecular bone destruction (yellow arrows) within the distal epiphysis of the ipsilateral femur in the Tumor group at day 21 post tumor cell xenografts (Figure 1(c)). In contrast, neither cancer cell infiltration nor trabecular bone destruction were observed in the distal epiphysis of the nude mice in Control group (Figure 1(b)). Instead, Figure 1(b) showed the normal trabecular bone in Control (black arrows).
Effects of EA on cancer-induced mechanical allodynia
There was no significant difference in the basal paw withdrawal thresholds (PWT) in the ipsilateral hind limb (tumor-injected) among all 4 groups before tumor cell inoculation (p > .05, Figure 2(a)). After inoculation of MDA-MB-231 cells, PWT was assessed on days 7, 14, 21, 28, 35, and 42 (in other words, in the end of week 1, 2, 3, 4, 5 and 6) by von Frey test (Figure 2(d)). To guarantee the reliability of the experiment, PWTs on both ipsilateral and contralateral hind paws were measured. Mice intra-femoral injected with MDA-MB-231 cells in the distal epiphysis of femur showed mechanical allodynia in the ipsilateral hind limb (tumor-injected) and no similar mechanical allodynia in the contralateral hind limb as measured by von Frey test (Figure 2(a) and (b)). The data revealed that PWT of ipsilateral side (tumor-bearing) in Tumor, Tumor + EA and Tumor + sham EA groups presenting significant downward trend on weekends 1−6 after inoculation compared to the Control group. The downward trend can be quantified by AUC evaluation. In comparison with Control group, AUC values of PWT in Tumor, Tumor + EA and Tumor + sham EA groups were significantly decreased respectively (p < .01, Figure 2(c)).
To evaluate the efficacy of EA in alleviating cancer-induced mechanical allodynia, the values of PWTs in ipsilateral hind paw that measured from week 1 to week 6 after inoculation were compared among Tumor, Tumor + EA and Tumor + sham EA groups, respectively. As shown in Figure 2(a), EA treatment significantly attenuated decreasing PWTs induce by tumor at different von Frey testing days (from week 3 to week 6 after inoculation). Compare to Tumor group, PWTs in Tumor + EA group were significant increased at day 21 (week 3, p = .0160), 28 (week 4, p = .0207), 35 (week 5, p = .0140) and 42 (week 6, p = .0302). In addition, AUC analysis during week 1 to week 6 further confirmed the analgesic effect of EA stimulation in tumor-bearing nude mice (p = .0012, Figure 2(c)). Whereas, sham EA treatment showed no analgesic effect, neither in PWT values nor in AUC analysis (p > .05, Figure 2(a) and (c)).
Effects of EA on cancer-induced thermal hyperalgesia
There was no significant difference in the basal paw withdrawal latency (PWL) in the ipsilateral hind limb (tumor-injected) among all 4 groups before tumor cell inoculation (p > .05, Figure 3(a)). After inoculation of MDA-MB-231 cells, PWL was assessed in the end of week 1, 2, 3, 4, 5 and 6 by using a hotplate analgesia meter with heat stimulation (Figure 3(d)). To guarantee the reliability of the experiment, PWL on both ipsilateral and contralateral hind paws were measured. Mice intra-femoral injected with MDA-MB-231 cells in the distal epiphysis of femur showed thermal hyperalgesia in the ipsilateral hind limb (tumor-injected) and no similar thermal hyperalgesia in the contralateral hind limb (Figure 3(a) and (b)). The data demonstrated that PWL of ipsilateral side (tumor-bearing) in Tumor, Tumor + EA and Tumor + sham EA groups presenting significant downward trend on weekends 1−6 after inoculation compared to the Control group. The downward trend can be quantified by AUC evaluation. In comparison with Control group, AUC values of PWL in Tumor, Tumor + EA and Tumor + sham EA groups were significantly decreased respectively (Tumor, p = .0032; Tumor + EA, p = .0102; Tumor + sham EA, p = .0003. Figure 3(c)).
To evaluate the efficacy of EA in alleviating cancer-induced thermal hyperalgesia, the values of PWL in ipsilateral hind paw that measured from week 1 to week 6 after inoculation were compared among Tumor, Tumor + EA and Tumor + sham EA groups, respectively. As shown in Figure 3(a), EA treatment significantly attenuated decreasing PWLs induce by tumor at different testing days (from week 2 to week 6 after inoculation). Compare to Tumor group, PWLs in Tumor + EA group were significant increased at day 14 (week 2, p = .0032), day 21 (week 3, p = .0259), 28 (week 4, p = .0036), 35 (week 5, p = .0071) and 42 (week 6, p = .0181). In addition, AUC analysis during week 1 to week 6 further confirmed the analgesic effect of EA stimulation in tumor-bearing nude mice (p = .0302, Figure 3(c)). Whereas, sham EA treatment showed no analgesic effect, neither in PWL values nor in AUC analysis (p > .05, Figure 3(a) and (c)).
Effects of EA on cancer-induced spontaneous pain
There was no significant difference in the basal value of spontaneous pain in the ipsilateral hind limb among all 4 groups before tumor cell inoculation (p > .05, Figure 4(a)). After inoculation of MDA-MB-231 cells, the spontaneous pain of mice in each group was assessed by observing and calculating the spontaneous foot-lifting (SFL) numbers (SFLn) in the end of week 1, 2, 3, 4, 5 and 6 (Figure 4). No significant SFL was observed in Control group and none of the mice exhibited SFL on the contralateral foot. SFL occurred on the ipsilateral hind limb (tumor-bearing) of mice in Tumor, Tumor + EA and Tumor + sham EA groups. Electroacupuncture (EA) attenuated spontaneous pain in the carcinoma xenograft mice model of cancer pain. (a) The spontaneous foot-lifting (SFL) numbers in ipsilateral hind limb (tumor-injected) in mice were evaluated in a time-course study. (b) Normalized area under the curve (AUC) analysis of Figure 4(a). **p < .01 versus Control; #p < .05, ##p < .01 versus Tumor. n = 7 ∼ 10 rats/group. One-way or two-way ANOVA followed by Tukey post hoc test was used for statistical analysis. Abbreviations: EA, electroacupuncture; SFL, spontaneous foot-lifting; AUC, area under the curve.
In tumor-bearing mice, SFLn of the ipsilateral hind limb (tumor-injected) was significantly elevated after tumor cell inoculation, in comparison with mice in Control group at the corresponding same time points respectively (p < .01, Figure 4(a)). The upward trend can be quantified by AUC evaluation. In comparison with Control group, AUC values of SFLn in Tumor, Tumor + EA and Tumor + sham EA groups were significantly increased respectively (p < .01, Figure 4(b)).
As shown in Figure 4(a), EA treatment significantly reduced the increased SFLn of tumor-bearing mice at different testing days (from week 2 to week 6 after inoculation). Compare to Tumor group, SFLn in Tumor + EA group were significant decreased at day 14 (week 2, p = .0113), day 21 (week 3, p = .0046), 28 (week 4, p = .0014), 35 (week 5, p = .0022) and 42 (week 6, p = .0036). In addition, AUC analysis during week 1 to week 6 further confirmed the analgesic effect of EA stimulation in tumor-bearing nude mice (p < .01, Figure 4(b)). Whereas, sham EA treatment showed no analgesic effect, neither in SFLn values nor in AUC analysis (p > .05, Figure 4).
Effects of EA on expressions of neuropeptide CGRP and p75 in the skin
The main nerve fibers mediating nociception express neuropeptide CGRP and the receptor of p75 neurotrophic factor (p75).30,31 We examined the distribution, expression and intensity of sensory nerve fibers containing CGRP and p75 within ipsilateral hind paw skin in mice at 6 weeks after inoculation by immunofluorescence staining (Figure 5). Electroacupuncture (EA) inhibited expressions of CGRP and p75NTR in ipsilateral hind paw skin in the carcinoma xenograft mice model of cancer pain. Fluorescent micrographs showing that (a) CGRP (green) and p75 (red) staining were significantly enhanced in Tumor group and decreased by EA treatment. Bar graph quantifying the immunolabeled area of (b) CGRP and (c) p75NTR. Scale bar, 100 µm. *p < .05, **p < .01 versus Control; #p < .05 versus Tumor. n = 3 rats/group. One-way or two-way ANOVA followed by Tukey post hoc test was used for statistical analysis. Abbreviations: EA, electroacupuncture.
Compared with Control group, the immunofluorescence intensities (measured by total length of related nerves) of CGRP- and p75-positive nerve fibers were increased within the ipsilateral hind paw skins in tumor-bearing nude mice (CGRP, p < .05, Figure 5(b); p75, p < .05, Figure 5(c)).
As shown in Figure 5(a), EA treatment inhibited the increased expressions of CGRP and p75 within ipsilateral hind paw skin in tumor-bearing mice. Compare to Tumor group, the total length of CGRP-positive nerve fibers in Tumor + EA group were significant decreased (1076 ± 204.5 μm in Tumor vs 540.7 ± 57.51 μm in Tumor + EA, p = .0330, Figure 5(b)) within the ipsilateral hind paw skins in tumor-bearing nude mice. Similarly, in comparison with Tumor group, the total length of p75-positive nerve fibers in Tumor + EA group were decreased (1031 ± 258.5 μm in Tumor vs 578.1 ± 66.67 μm in Tumor + EA, Figure 5(c)) within the ipsilateral hind paw skins in tumor-bearing nude mice as well, but without statistical significance (p = .2051). Whereas, sham EA treatment showed no inhibitory effect on CGRP and p75 expressions.
Effects of EA on expressions of neuropeptide-related proteins in the lumbar spinal cord
The p75, tropomyosin-related kinase A (TrkA) and vascular endothelial growth factor receptor (VEGFr) have been found to play critical roles in nociception of cancer pain.31–33 Western blotting data illustrated cancer pain induces a substantial increase in protein expression levels of p75, TrkA and VEGFr in spinal cord of tumor-bearing nude mice (Figure 6). Protein expressions of p75, TrkA and VEGFr in lumbar spinal cord from experimental mice. Representative (a) Western blots of p75, TrkA and VEGFr protein, and relative levels of (b) p75, (c) TrkA and (d) VEGFr in lumbar spinal cord. *p < .05, **p < .01 versus Control; #p < .05, ##p < .01 versus Tumor. n = 3 rats/group. One-way or two-way ANOVA followed by Tukey post hoc test was used for statistical analysis.
Compared with Control group, the protein expressions of p75, TrkA and VEGFr in spinal cord were increased significantly in mice of Tumor group (p75, p = .0003, Figure 6(b); TrkA, p < .0001, Figure 6(c); VEGFr, p = .0127, Figure 6(d)).
As shown in Figure 6, EA treatment inhibited the increased expressions of p75, TrkA and VEGFr in spinal cord in tumor-bearing mice. Compare to Tumor group, the expression levels of spinal p75, TrkA and VEGFr were significantly decreased from 1.713 ± 0.1308, 2.302 ± 0.04421 and 1.400 ± 0.03355 in Tumor group, to 0.6703 ± 0.04067 (p75, p = .0004, Figure 6(b)), 0.4108 ± 0.01758 (TrkA, p < .0001, Figure 6(c)), and 1.047 ± 0.09110 (VEGFr, p = .0246, Figure 6(d)) in Tumor + EA group, respectively. Whereas, sham EA treatment showed no inhibitory effect on protein expressions of p75, TrkA and VEGFr.
Effects of EA on expressions of inflammation-related proteins in the lumbar spinal cord
Cyclooxygenase 2 (COX2), Nuclear factor-kappa B (NF-κB) and Interleukin-1 beta (IL-1β) contribute to inflammation-related pain perception and nociceptive behavior, including cancer pain.34–36 Western blotting results showed cancer pain induces a significant increase in protein expression levels of COX2, NF-κB and IL-1β in spinal cord of tumor-bearing nude mice (Figure 7). Protein expressions of COX2, NF-κB p65 and IL-1β in lumbar spinal cord from experimental mice. Representative (a) Western blots of COX2, NF-κB p65 and IL-1β protein, and relative levels of (b) COX2, (c) NF-κB p65 and (d) IL-1β in lumbar spinal cord. **p < .01 versus Control; ##p < .01 versus Tumor. n = 3 rats/group. One-way or two-way ANOVA followed by Tukey post hoc test was used for statistical analysis.
Compared with Control group, the protein expressions of COX2, NF-κB and IL-1β in spinal cord were increased significantly in mice of Tumor group (COX2, p < .0001, Figure 7(b); NF-κB, p = .0002, Figure 7(c); IL-1β, p = .0005, Figure 7(d)).
As shown in Figure 7, EA treatment inhibited the increased expressions of COX2, NF-κB and IL-1β in spinal cord in tumor-bearing mice. Compare to Tumor group, the expression levels of spinal COX2, NF-κB and IL-1β were significantly decreased from 2.363 ± 0.07273, 1.416 ± 0.007589 and 1.495 ± 0.1072 in Tumor group, to 0.4051 ± 0.07150 (COX2, p < .0001, Figure 7(b)), 0.8321 ± 0.09930 (NF-κB, p = .0005, Figure 7(c)), and 0.8269 ± 0.05757 (IL-1β, p = .0020, Figure 7(d)) in Tumor + EA group, respectively. Whereas, sham EA treatment showed no inhibitory effect on protein expressions of COX2, NF-κB and IL-1β.
Discussion
The present study provides evidence that inoculation of MDA-MB-231 cells into distal femur of nude mice induced cancer pain mainly in the tumor-bearing hind limb that manifested as mechanical allodynia, thermal hyperalgesia and spontaneous pain. HE staining showed obvious cancer cell infiltration and significant trabecular bone destruction within the distal epiphysis of the ipsilateral femur after tumor cell xenografts. Furthermore, results obtained from measurements of pain perception and nociceptive behavior indicated that cancer-induced pain behavior occurred in the ipsilateral hind limb (tumor-injected) instead of the contralateral hind limb which are demonstrated by the data of PWT, PWL and SFLn that were evaluated respectively by the tests of von Frey, hotplate analgesia and spontaneous pain. The severity of pain in the tumor-bearing ipsilateral hind paw trends to a gradual increase over time simultaneously accompanied by a significant increase of immunofluorescence intensities representing CGRP- and p75-positive nerve fibers within the ipsilateral hind paw skins, as well as a substantial upregulations of protein expression levels of neurological factors (p75, TrkA and VEGFr) and inflammatory factors (COX2, NF-κB and IL-1β) in spinal cord of tumor-bearing nude mice.
EA treatment was performed following tumor cell xenografts, once every other day for consecutive 4 weeks in total. Acupoints of GB30 (Huan-tiao) on both sides near bilateral hip joints were chosen for EA treatment in the present study, due to its analgesic and antinociceptive effects against multiple types of pain including cancer pain, as reported previously.23,24,37 Our present data in this research confirmed the significant analgesic actions of EA treatment on relieving cancer-induced pain behaviors covering mechanical allodynia, thermal hyperalgesia and spontaneous pain. Such antinociceptive effects of EA treatment maintained continuously throughout the 4-weeks entire course of treatment, and extended for another 2 weeks after completion of EA therapy, indicating a remarkable and prolonged analgesic efficacy. However, sham EA treatment showed no antinociceptive effect on cancer-induced pain behaviors, neither in mechanical allodynia, thermal hyperalgesia nor in spontaneous pain, manifesting the specific therapeutic effects of EA intervention.
Cancer-induced bone pain is a complex pain state and its occurrence and development involve components of both neuropathic and inflammatory mechanisms.38,39 Preclinical and clinical researches have demonstrated the important role for the nerve growth factor (NGF) that belongs to the neurotrophin family in the transduction and mediation of both acute and chronic nociceptive signals.40,41 It has also been confirmed that NGF can be activated in the spinal cord following nerve injury and damage, and that its expression is intimate relevant to neuropathic and inflammatory pain.42,43 NGF binds to two types of receptors, TrkA and p75. Interaction of NGF with its two receptors is involved in a variety of physiological and pathological processes including tumorigenesis and cancer pain. 44 Nerve injury and inflammatory infiltration increase the expressions of TrkA and p75 in the spinal dorsal horn and dorsal root ganglion, in turn, the augmentation of TrkA and p75 signals contribute to mechanical and thermal hyperalgesia. Western blot results of the current study showed a significant increase of TrkA and p75 in spinal cord of tumor-bearing animals, indicating a role of spinal NGF receptors involving cancer-induced pain. Whereas such upregulation of spinal TrkA and p75 was inhibited by EA treatment and that might be one of the possible mechanisms underlying the effective action of acupuncture analgesia. Pain mediator calcitonin gene-related peptide (CGRP) plays an important role in central mechanisms of nociceptive signaling, and it is a significant in regulating various pain including migraine, visceral pain, neuropathic pain, and cancer pain.45–48 Our present data in present study showed that EA treatment significantly down-regulated pain-related neuropeptide expressions in ipsilateral hind paw skin and in the lumbar spinal cord, suggesting the possible analgesic mechanism of EA treatment playing a role at both peripheral and central levels.
Clearly, inflammation relates to cancer and that it also relates to pain in general. Inflammation can also be a result of cancer, and on the other hand, cancer pain may also be due to inflammation. 49 Peripheral nerve injuries and diseases often lead to pain persisting beyond the resolution of damage, indicating an active disease-promoting process, which may result in chronic pain. Generally, pain syndrome indicates an active disease-promoting process, which is regarded as a mechanism arising from neuroinflammation. 50 Key players involving in these physiological and pathophysiological process include cytokines and chemokines. Previous studies have shown an important role of inflammatory cytokines in pain states in human and animal models.50–53 Cyclooxygenase-2 (COX-2) is one of the important inflammatory mediators produced in injured nerves. Known as one of the key inducible enzymes, COX-2 is in response to inflammatory stimulation, initiates inflammatory reactions and precipitates inflammatory response inducing tissue injury, and involved in the genesis and progress of neuropathic pain.54,55 Application of COX-2 inhibitors significantly attenuated mechanical allodynia and inhibited expression of inflammatory mediators such as TNF-α, Il-1β and Il-6,54–56 demonstrating the role of COX-2 in neuropathic pain and the control and regulation of COX-2 can be regarded as a therapeutic target to alleviate neuropathic pain. Consistently, COX-2 in the spinal cord of tumor-bearing nude mice was significantly increased in our study. Our present data in present study demonstrated that EA treatment significantly inhibited the protein expression of COX2 in the lumbar spinal cord, suggesting that down-regulation of spinal COX2 might be one of the possible analgesic mechanisms of EA analgesia against cancer pain. The NF-κB family of transcription factors modulate the transcription of angiogenic and tumorigenic chemokines and cytokines that facilitate the growth of tumors. 57 Several tumor systems have been shown to have constitutive activation of NF-κB can be constitutively activated in tumor systems, and that enhance NF-κB activity and then it causes tumor cells to express chemokines, cytokines such as VEGF and IL-1. Moreover, activation of NF-κB is critically involved in controlling neuroinflammation and then evoke and induce the pain caused by it. 58 The release of proinflammatory cytokines (e.g. IL-1β) is an important cause to induce pain, and it results in nociceptor sensitization during the inflammatory process. IL-1β gets involved in hyperalgesia of various pain models, such as neuropathic pain, inflammatory pain, as well as cancer pain.59,60 In the present study, western blot results showed a significant increase of COX2, NF-κB p65 and IL-1β in spinal cord of tumor-bearing animals, suggesting that inflammatory mediators and cytokines play important roles in the nociceptive mechanism of cancer-induced pain. EA treatment significantly inhibited the protein expressions of COX2, NF-κB p65 and IL-1β in the lumbar spinal cord, indicating the possible analgesic mechanism of EA treatment may involves in inhibiting the inflammatory response at central levels.
Current evidence in the present study has confirmed that EA at acupoints of GB30 alleviates pain and nociceptive behaviors (mechanical allodynia, thermal hyperalgesia and spontaneous pain) in the mouse xenograft model of cancer pain. The present study further identified that EA can down-regulate immunofluorescence expressions of neuropeptide CGRP and p75 in the skin of affected plantar area, and inhibit expressions of overexpressed neuropeptide-related (p75, TrkA and VEGFr) and inflammation-related protein (COX2, NF-κB and IL-1β) in the lumbar spinal cord (Figure 8). Schematic diagram of possible mechanism in EA-mediated analgesia of cancer pain. Abbreviations: EA, electroacupuncture.
Conclusion
Taken together, our findings suggest that EA treatment ameliorated cancer-induced pain behaviors in the mouse xenograft model of cancer pain, possibly through inhibiting the expressions of neuropeptide-related and inflammation-related protein in central level following tumor cell xenografts. However, the mechanism exploration of EA treatment in alleviating cancer pain in our present study is still relatively superficial and simple, and the in-depth research on the potential analgesic mechanisms of EA treatment against cancer pain should be paid more attention as well as conduct further in-depth investigation in future studies.
Footnotes
Acknowledgements
We express our appreciation to all our lab. members for fostering a positive and supportive environment that consistently encourages us to explore the potential of our ideas and learn from our setbacks.
Author contributions
Yu-Xue Zhao: Conceptualization, Investigation, Data curation, Formal analysis, Writing-original draft, Writing-review & editing, Funding acquisition, Project administration, Supervision. Ming-Jiang Yao: Investigation, Methodology, Data curation, Formal analysis, Writing-review & editing, Project administration, Supervision. Jian-Wu Shen: Investigation, Methodology, Data curation, Formal analysis, Writing-review & editing, Project administration, Supervision. Wen-Xi Zhang: Investigation, Methodology, Data curation, Formal analysis, Writing-review & editing. Yuan-Xi Zhou: Investigation, Methodology, Data curation, Formal analysis, Writing-review & editing. All authors gave final approval of the version to be published, and agree to be accountable for all aspects of the work.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Natural Science Foundation of Beijing (7202141); National Natural Science Foundation of China Research Grants (81202763 and 82374594); Fundamental Research Funds for the Central public welfare research institutes (201814009, ZZ13-YQ-067 and ZZ-JQ2023005); China Scholarship Council (No. 202005350004).
