Abstract
During adolescence, a second period of central nervous system (CNS) plasticity that follows the fetal period, which involves sleep deprivation (SD), becomes apparent. SD during adolescence may result in abnormal development of neural circuits, causing imbalance in neuronal excitation and inhibition, which not only results in pain, but increases the chances of developing emotion disorders in adulthood, such as anxiety and depression. The quantity of surgeries during adolescence is also consistently on the rise, yet the impact and underlying mechanism of preoperative SD on postoperative pain remain unexplored. This study demonstrates that preoperative SD induces upregulation of the P2Y12 receptor, which is exclusively expressed on spinal microglia, and phosphorylation of its downstream signaling pathway p38Mitogen-activated protein/Nuclear transcription factor-κB (p38MAPK/NF-κB)in spinal microglia, thereby promoting microglia activation and microglial transformation into the proinflammatory M1 phenotype, resulting in increased expression of proinflammatory cytokines that exacerbate persisting postoperative incisional pain in adolescent mice. Both intrathecal minocycline (a microglia activation inhibitor) and MRS2395 (a P2Y12 receptor blocker) effectively suppressed microglial activation and proinflammatory cytokine expression. Interestingly, supplementation with dehydrocorydaline (DHC), an extract of Rhizoma Corydalis, inhibited the P2Y12/p38MAPK/NF-κB signaling pathway, microglia activation, and expression of pro-inflammatory cytokines in the model mice. Taken together, the results indicate that the P2Y12 receptor and microglial activation are important factors in persistent postoperative pain caused by preoperative SD in adolescent mice and that DHC has analgesic effects by acting on these targets.
Introduction
The development of the mammalian central nervous system (CNS), including the brain and spinal cord, is a progressive process of maturity. Adolescence, regarded as a transitional phase between childhood and adulthood, is characterized by significant functional and structural changes in the CNS, mainly marked by a reduction in gray matter and an increase in white matter tissue. 1 During adolescence, the CNS is particularly vulnerable to environmental changes, stress, and other a multitude of other factors. These factor in turn impact the process of neurodevelopment through various mediators, including the crosstalk between the immune system and neurons, which can have profound effects on cognition, sensation, behavior, and emotions that maybe persist into adulthood and throughout life. 2
Correspondingly, significant changes also occur in sleep patterns during adolescence, such as delayed sleep and a reduction in total sleep time. Worryingly, sleep deprivation (SD) among adolescents is a prevalent public health concern, with 57.8% of junior students and 72.7% of senior students in the United States experiencing sleep problems. 3 Poor sleep is significantly associated with reduced gray matter volume in the medial precuneus cortex, anterior cingulate cortex, and hippocampus,4,5 which render adolescents more susceptible to emotional disorders, impaired memory, and exacerbation of pain. 6 Moreover, in comparison to excitatory afferent neurons, inhibitory afferent neurons (such as GABAergic neuron) undergo a delayed developmental process during adolescent CNS, which is the physiological basis for imbalance of excitatory and inhibitory neuronal activity(E/I). These changes ultimately lead to irreversible modifications in CNS function and the occurrence of a series of molecular event sequences.7,8
Annually, more than 320 million individuals worldwide undergo surgical procedures, resulting in the patients suffer from different levels of postoperative pain. 9 Postoperative pain usually does not exceed one week and results in little or no delay in the patient's recovery. However, in some cases, acute postoperative pain persists beyond the normal tissue healing period and progresses to chronic pain. 10 Postoperative pain mainly results from inflammation triggered by tissue injury or direct neuronal damage. Local inflammatory mediators can cause an increase in neuronal excitability in the CNS, leading to hyperalgesia or serious pain. The spinal cord plays a dominant role in the transmission and regulation of pain information. Spinal central sensitization, as a form of neuroplasticity, represents an imbalance of E/I in the spinal dorsal horn, which results in a reduction in the pain threshold following nociceptive and innocuous stimuli. 11 The mechanisms underlying the transition from postoperative acute pain to chronic pain have so far remained unclear.
SD and pain are highly comorbid, with a bidirectional interaction between them. SD serves as a risk factor for the onset and development of chronic pain by lowering the pain threshold, inducing hyperalgesia and spontaneous pain. 12 During periods of both poor sleep and pain, the CNS immune system becomes activated, leading to a proinflammatory environment, which may trigger spontaneous pain or contribute to existing pain. Additionally, poor sleep has been shown to amplify the transmission of pain information in the spinal cord, and the harmful effects of insufficient sleep on pain sensitivity have a cumulative effect. 13 The combination of pain and sleep dysfunction during adolescence may lead to imbalance of neuronal excitation and inhibition, ultimately impacting the adult neuronal function. However, the underlying mechanism remains unclear at present. Few studies had utilized the adolescent mice planter incision model, which mimics the impact of SD during adolescence on surgical pain and changes on the spinal cord level were not documented.
As the resident immune cells, microglia engage in crosstalk with other cells to maintain a stable microenvironment in the CNS. Microglia within the adolescent CNS are characterized by a low activation threshold and remarkable functional plasticity in response to environmental stress signals, enabling them to perform monitoring and engulfing functions effectively. 14 Interestingly, the phenotype and function of adolescent microglia with the CNS are slightly different from those of adult and elderly microglia.15-17 Nevertheless, the precise contribution of microglial activation to the development of acute pain induced by SD in adolescence remains ambiguous.
The purinergic receptor P2Y12 is exclusively expressed on microglia in the spinal cord and are the main mediator of spinal cord microglial activation. 18 The P2Y12 receptor plays a pivotal role in mediating the crosstalk of microglia and neurons, promoting microglial activation and synaptic plasticity of primary sensory neurons. Activation of the P2Y12 receptor triggers the release of proinflammatory cytokines by microglia, ultimately culminating in a cascade reaction of neuroinflammation that results in pain. 19 However, until now, there has been no existing research that has investigated the involvement of microglial P2Y12 receptors in the spinal cord regarding the impact of SD-induced persistent postoperative pain in an adolescent mouse model.
Dehydrocorydaline (DHC) is an alkaloid extracted from Rhizoma Corydalis, which exhibits antidepressant, anti-inflammatory, and analgesic properties. It is highly bioavailable upon oral administration and can penetrate the blood-brain barrier to exert its pharmacological actions on the CNS. Our previous studies have demonstrated that the analgesic effect of DHC is associated with the inhibition of the microglial inflammatory response in the spinal cord.20,21 The effects and mechanisms of DHC on preoperative SD-induced persistent postoperative pain in adolescent mice have not been previously reported. In this study, we further postulated that preoperative SD enhances the activity of microglial P2Y12 and triggers microglial activation, thereby adjusting the transition from acute surgical pain to persistent pain in adolescent mice. Importantly, the analgesic effects of DHC may be exerted through the above mechanism.
Materials and methods
Animals
C57BL/6J mice (3-week-old males purchased from Vital River, China) were housed for one week under SPF conditions with a controlled 12-hour light/dark cycle. All experiments were conducted in accordance with the approval of the Laboratory Animal of the Ethics Committee of Nanjing Drum Tower Hospital. The mice were randomly assigned into control group (group C), incision group (group I), SD group (group S), and SD+incision group (group SI). In further experiments, mice in group SI were injected intrathecally with minocycline 30 μg/dose (microglia activation inhibitor, group SI+Mino) or MRS2395 30 μg/dose (P2Y12 receptor antagonist, group SI+MRS) or volume equivalent 2% DMSO (group SI+V). Treatments were administered once daily for five times during SD. Furthermore, the mice in group SI were administered intraperitoneal injections of DHC at a dosage of 20 mg/kg once daily for five times following SD until day 5 after surgery (group SI+DHC). The control group received an equivalent volume dose of 2% DMSO (group SI+V).
Sleep deprivation process
The mouse SD model was induced in an SD chamber, as previously described. 22 In brief, mice subjected to SD were placed in an SD chamber (Xinruan, China) for 4.5 days and kept awake by a continuously rotating metal bar, which was set to the same speed, force, and direction of rotation to minimize experimental error. Food and water were placed on the walls of the chamber for the mice to take freely. Mice in the non-SD group were housed under normal feeding conditions.
Plantar incision model preparation
The plantar incision model was generated as previously reported. 22 In brief, the mice were anesthetized with sevoflurane, immobilized in the supine position, and the right hind paw was routinely disinfected with sterile iodophor and covered with cavity wipes. Next, an incision was made approximately 0.5cm incision from the heel of the right hind paw towards the toe, after which the muscle was gently and carefully elevated at the incision site. Subsequently, the wounds were sutured with 5-0 nylon thread and antimicrobial ointment was applied to prevent infection. Finally, mice were allowed to recovery in their cages postoperatively.
Drug Preparation
Minocycline (Meilunbio, China), MRS2395(Sigma, USA), and DHC (Vicmed, China) were individually dissolved in 2% DMSO and subsequently diluted with normal saline. Minocycline and MRS2395 were administered initially at 30 min prior to SD induction, once daily during SD, for a total of five times. The first injection of DHC was given at 30 min prior to incision and continued once daily until postoperative day 5 for a total of five times.
Paw Withdrawal mechanical threshold
The mechanical pain of mice was performed prior to SD (Baseline, BL) and on day 1, 3, 5, 7, 10, and 14 after incision. Behavioral assessment of the DHC intervention mice assessed at BL and day 6, 7, 8, 9, 10, and 14 after incision.
According to the standard procedure, mice were stimulated with Von Frey (Stoelting, USA) of different stimulation strengths on the palmar skin of the right foot, and the mice were considered positive when they showed a rapid retraction response. Mice were acclimated in a quiet environment for 30 min prior to testing. Tests were measured with five stimuli of differing intensity, and this intensity was the PWMT value for that mouse when there were ≥ 3 positive responses. The minimum stimulus force that caused rapid withdrawal of the paw was regarded as the PWMT. All behavioral tests were performed by experimenters who were blinded to the groups.
Immunofluorescence
Mice were anesthetized with sevoflurane and sequentially perfused with saline and 4% paraformaldehyde via the heart, and the entire lumbosacral enlargement was removed and fixed in 4% paraformaldehyde for 24 h, followed by gradient dehydration with 15% and 30% sucrose at 4°C. The L3–L5 spinal cords were sliced into 20-μm-thick sections and placed on glass slides. Sections were washed three times with phosphate buffered saline (PBS) and then permeabilized with 0.3% Triton for 15 min, followed by blocking with serum for 1 h at room temperature. After blocking, the sections were incubated overnight at 4°C with primary antibody: rabbit anti-IBA1(1:500, Wako, Japan). The sections were then washed three times with PBS and incubated with secondary Alexa 488-conjugated goat anti-rabbit antibody (1:2000, Invitrogen, USA) for 1 h at room temperature. Finally, the sections were stained with DAPI (Abcam, UK), dried, and imaged under a laser confocal microscope (Olympus, Japan).
Western blotting
Mice were anesthetized and sacrificed at day 1 and 7. The spinal cord was removed and quickly stored at −80°C. Toal protein from the spinal cord tissue was extracted according to standard laboratory procedures. Proteins were separated by electrophoresis using 10% SDS‒PAGE gels and transferred to PVDF membranes. The membranes were then blocked with 5% BSA for 1 h at room temperature and incubated with the following antibodies: rabbit anti-P2Y12 (1:1000, Abcam, UK), rabbit anti-p-p38MAPK (1:1000, CST, USA), rabbit anti-p38MAPK (1:1000, Aclinal, China), rabbit anti-p-P65 (1:1000, CST, USA), mouse anti-P65 (1:500, Affinity, China), rabbit anti-IL-6 (1:1000, Abcam, UK), rabbit anti-IL-1β (1:1000, Abcam, UK), rabbit anti-iNOS (1:1000, Proteintech, China), mouse anti-Arg1 (1:1000, Proteintech, China), and rabbit anti-β-actin (1:50,000, Proteintech, China) overnight at 4°C. After, the membranes were incubated with the secondary antibody for 1 h at room temperature, after which the protein bands were visualized using enhanced chemiluminescence solution. Images were captured and quantitative densitometry analysis was realize using Image J.
Statistical analysis
All data were expressed using mean ± SD and the data were analyzed and graphed using GraphPad Prism (Version 8.3.0). Analysis of behavioral changes between the different groups at each time point were performed using two-way repeated-measures ANOVA. The differences among groups were analyzed by using the t test or one-way ANOVA. All ANOVAs were followed by the Bonferroni’s post hoc test. p < 0.05 indicated a statistical difference.
Results
Preoperative SD aggravates and prolongs postoperative pain in adolescent mice, and promotes microglial activation and release of proinflammatory cytokines in the spinal cord
Preoperative SD exacerbates and prolongs postoperative incisional pain in adolescent mice
To assess the effect of SD on postoperative incisional pain, we measured the PWMT at different time in adolescent mice. Results indicated that the lowest PWMT in group I was at postoperative day 1, and gradually recovered to BL at postoperative day 7. Compared with group I, the PWMT in group SI was significantly lower at postoperative day 1(group I vs group SI, p < 0.05) to day10, and returned to BL at day14. The results indicated that SD exacerbated postoperative pain and prolonged the duration of pain. In addition, compared with group C, the PWMT decreased in group S after SD, which returned to baseline on postoperative day 10, although there was no statistical difference between the groups (group C vs group S, p > 0.05), a trend that was also found in group SI (Figure 1). The data suggest that SD exacerbated postoperative pain and prolonged pain duration in adolescent mice. Time-dependent changes in the PWMT after continuous 4.5-day-SD in mice was tested by Von Frey. n = 8 per group. Group C vs group I, &&&P < 0.001; group C vs group SI, ###P < 0.001; group I vs group SI, $$P < 0.01, $$$P < 0.001.
SD accelerates microglial activation and the inflammatory response in the spinal cord
On postoperative day 1, compared with group C, microglia in spinal dorsal horn were activated in group S, group I, and group SI, and particularly in group SI, which showed a significant increase in the number of microglia and enlarged cell body (p < 0.05; Figure 2(a), (c), (d)). At the same time, expression of the microglia M1 phenotype marker, iNOS, significantly increased, while at of the M2 phenotype marker, Arg1, significantly decreased (p < 0.05 for both; Figure 3(a), (c), (d)). Additionally, the microglia activation was accompanied by increased expression of proinflammatory cytokines. western blotting results showed that the expression of IL-1β and IL-6 were significantly increased in group S, group I, and group SI on postoperative day 1 compared with group C. Among the groups, expression of IL-1β and IL-6 in group SI was the most significant (p < 0.05; Figure 3(a), (e), (f)). The expression of microglia in the spinal dorsal horn was measured by immunochemical staining with IBA1 antibody on postoperative day 1 and 7. The number and body area of microglia in each group. Scale bar = 50 μm, n = 3 per group. (a) Microglial activation on postoperative day 1. (b) Microglial activation on postoperative day 7. (c, d) The number and body area of microglia in each group on postoperative day 1 and 7, ###P < 0.001. The protein levels in the spinal cord were detected with western blotting, n = 4 per group. The results were considered significant at #p < 0.05, ##p < 0.01, and ###p < 0.001. (a) Protein expression in the spinal cord on postoperative day 1. (b) The protein expression in spinal cord on postoperative day 7. C-F iNOS, Arg1, IL-1β, and IL-6 levels in the spinal cord of each group postoperative day 1 and day 7.
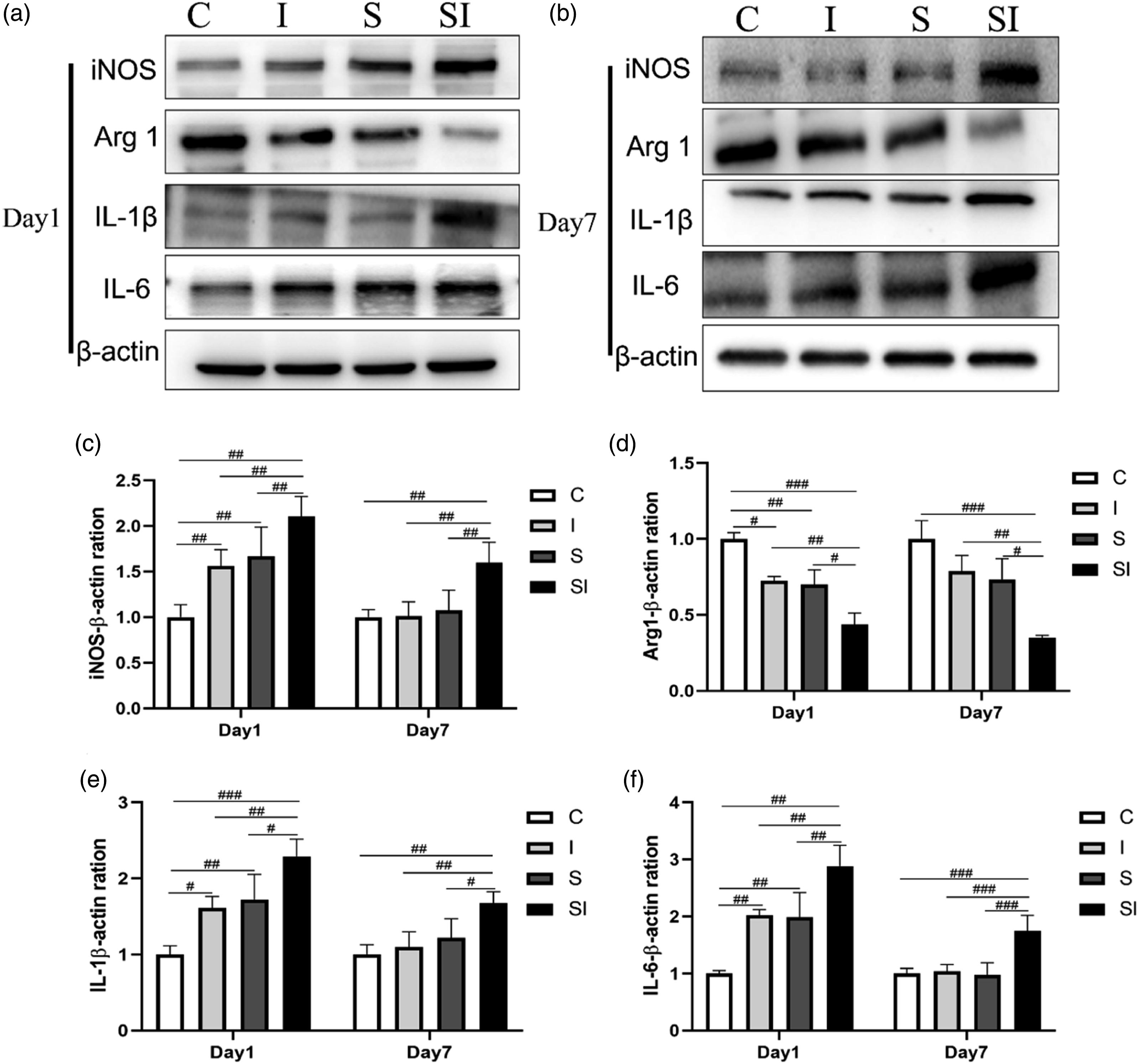
On postoperative day 7, compared with group C, the expression levels of IL-1β and IL-6 in group S and group I were no longer significant (p > 0.05). However, microglial activation and expression levels of the two inflammatory cytokines remained significant in group SI (p < 0.05; Figure 2B–(d); Figure 3B–(f)).
Intrathecal administration of minocycline suppresses microglial activation and downregulates the release of proinflammatory cytokines in the spinal cord, thereby ameliorating and reducing the time of postoperative incisional pain in adolescent mice
Mice were randomly divided into two groups, namely group SI+V and group SI+Mino. Compared with group SI+V, group SI+Mino had a significantly increased PWMT on postoperative day 1, 3, 5, 7, and 10, (p < 0.05 for all days). The PWMT of group SI+V returned to BL on postoperative day 14, whereas that of group SI+Mino returned to BL on postoperative day 10 (Figure 4). The results showed that intrathecal injection of minocycline ameliorated and reduced the time of postoperative incisional pain. Time-dependent changes in the PWMT after intrathecal injection of minocycline in mice was tested by Von Frey, n = 8 per group, #P < 0.05, ##P < 0.01, ###P < 0.001.
The immunofluorescence results revealed a significant decrease in both the number and body area of microglia within the dorsal horn of the spinal cord in group SI+Mino compared with group SI+V (p < 0.05; Figure 5(a)–(c)). Similarly, group SI+Mino demonstrated a significant decrease in iNOS expression and a significant increase in Arg1 expression compared with group SI+V (p < 0.05). Finally, compared with group SI+V, the expression of proinflammatory cytokines, including IL-6 and IL-1β, was significantly reduced in the dorsal horn of the spinal cord in group SI+Mino on postoperative day 7 following intrathecal injection of minocycline (Figure 6(a)–(e)). These findings suggest that intrathecal administration of minocycline effectively suppressed microglial activation, facilitated the transition of microglia from M1 to the M2 phenotype, and reduced the level of inflammatory cytokines. Effects of minocycline on microglia activation in mice on postoperative day 7, n = 3 per group,#p < 0.05, ###p < 0.001. (a) microglial activation detected by immunofluorescence. (b,c) The number and body area of microglia in each group on postoperative day 7. The protein levels in the spinal cord after minocycline injection, n = 4 per group. (a) The protein expression in the spinal cord on postoperative day 7. (B-E) iNOS, Arg1, IL-1β, and IL-6 levels in the spinal cord of each group on postoperative day 7, ##P < 0.01, and ###P < 0.001.

Intrathecal administration of MRS2395 suppresses microglial activation and attenuates the expression of proinflammatory cytokines in the spinal cord, thereby ameliorating preoperative SD-induced persistent postoperative pain in adolescent mice
Preoperative SD induces activation of the P2Y12/p38MAPK/NF-κB signaling pathway in the spinal cord of adolescent mice following surgery
On postoperative day 1, compared with group C, there were no significant differences in expression of p38MAPK or P65 in group I, group S, or group SI (p > 0.05). However, significant increases were observed in the expression of the P2Y12 receptor, p-p38MAPK, and p-P65 in group I, group S, and group SI when compared with group C (p < 0.05 for all; Figure 7(a), (c), (e)-(h); Figure 8(a)). The effects of SD on the P2Y12/p38MAPK/NF-κB signaling pathway in each group, n = 4 per group. (a-d) The protein expression in the spinal cord on postoperative days 1 and 7. 7. (e-h) p38MAPK, p-p38MAPK, P65, and p-P65 levels in the spinal cord of each group on postoperative day 1 and day 7, #P < 0.05, ##P < 0.01, and ###P < 0.001. The P2Y12 receptor expression in spinal cord in each group and pain behavior after intrathecal injection of MRS2395. (a) The P2Y12 receptor expression on Day1 and Day7, n = 4. (b) PWMT was tested by Von Frey after intrathecal injection of MRS2395, n = 8. #P < 0.05, ##P < 0.01, and ###P < 0.001.

On postoperative day 7, compared with group C, there were no significant differences in expression of p38MAPK or P65 in group I, group S, or group SI (p > 0.05 for all). Additionally, there were no significant differences in the expression of the P2Y12 receptor, p-p38MAPK, or p-P65 in group I and group S (p > 0.05 for both). Compared with group I, the expression of the P2Y12 receptor, p-p38MAPK, and p-P65 in group SI exhibited a significant increase(p < 0.05 for both; Figure 7(b), (d)-(h); Figure 8(a)).The above findings suggest that preoperative SD enhances the activation of the P2Y12/p38MAPK/NF-κB signaling pathway on postoperative day 1 in groups S, I, and SI. Interestingly, group S and group I recovered to BL on postoperative day 7, whereas the signaling pathway in group SI of remained activated.
Intrathecal injection of MRS2395 inhibits activation of the P2Y12/p38MAPK/NF-κB signaling pathway, suppresses microglial activation, downregulates proinflammatory cytokine release, and alleviates postoperative pain caused by SD
Compared with group SI+V, the PWMT of mice in group SI+MRS was significantly increased on postoperative day 1, 3, 5, and 7 (p < 0.05 for all), but returned to baseline on postoperative day 10 (Figure 8(b)).
The immunofluorescence results revealed a significant reduction in the number and cell body area of microglia in the dorsal horn of the spinal cord in mice from group SI+MRS compared with that of group SI+V (p < 0.05; Figure 9(a)–(c)). Furthermore, compared with group SI+V, mice in group SI+MRS exhibited a significant decrease in iNOS expression and significant increased Arg1 expression (p < 0.05; Figure 11(a)-(c)). These findings suggest that intrathecal administration of MRS2395 impeded microglial activation and facilitated microglial polarization from the M1 to the M2 phenotype in the spinal cord on postoperative day 7. Effects of MRS2395 on microglia activation on Day7, n = 3 per group,#P < 0.05. (a) microglia activation detected by immunofluorescence. (b, c) The number and body area of microglia in each group on Day7.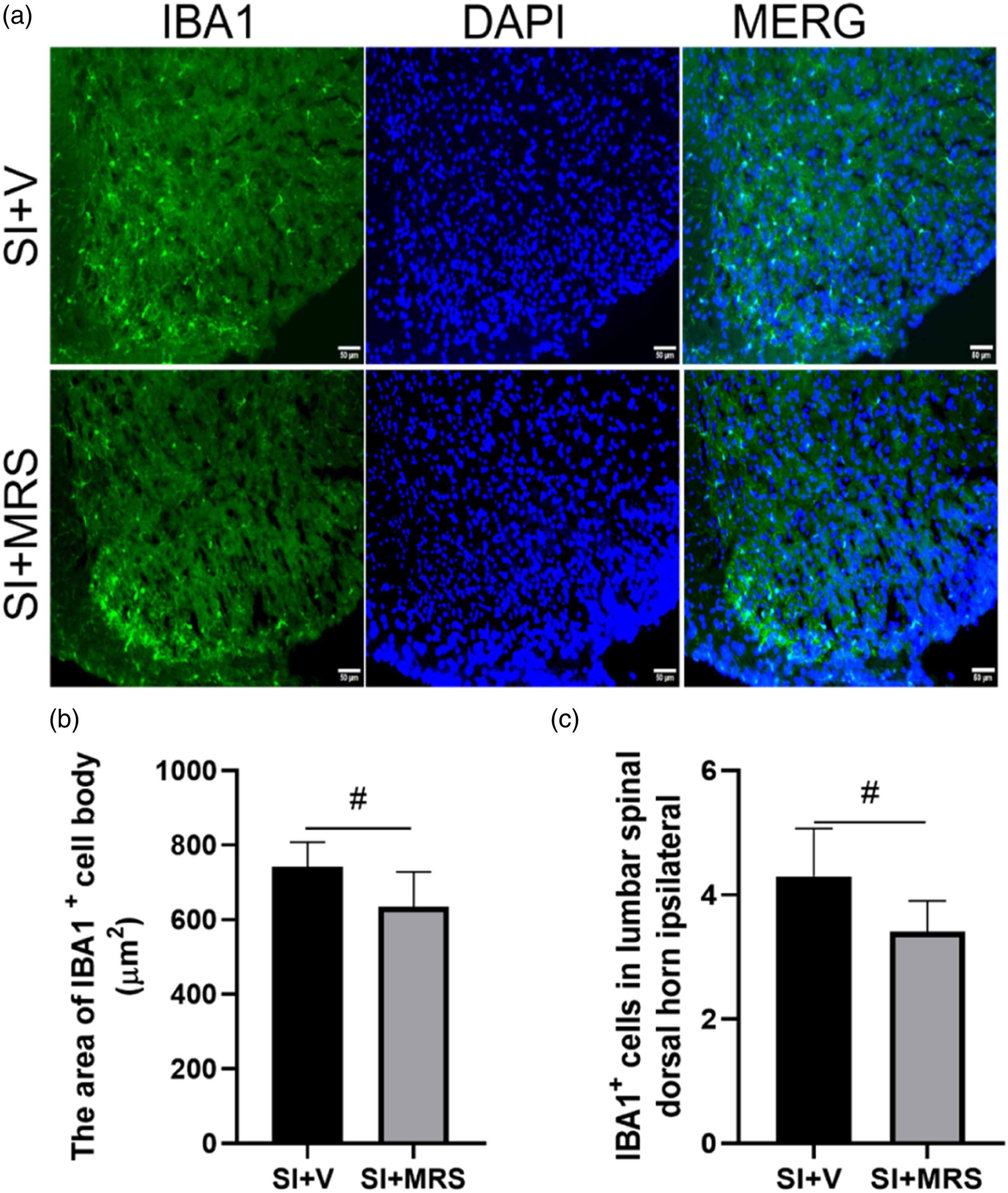
On postoperative day 7, compared with group SI+V, there were no significant differences in the expression of p38MAPK and P65 in group SI+MRS (p > 0.05 for both). However, expression of the P2Y12 receptor, p-p38MAPK, and p-P65 were significantly reduced, (p < 0.05 for all; Figure 10(a)-(g)). Furthermore, the expression of IL-6 and IL-1β were significantly reduced (p < 0.05; Figure 11(a), (d), (e)).The expression of iNOS and Arg1 in group SI+MRS was reversal compared with group SI (p < 0.05;Figure 11(a)-(c)). The inhibitory effects of MRS2395 on P2Y12/p38MAPK/NF-κB signaling pathway examined by immunoblotting, n = 4 per group,#P < 0.05. (a,d) The protein levels in spinal cord on Day 7. (b-c,e-g) p38MAPK, p-p38MAPK, P65, p-P65 and P2Y12 receptor levels in spinal cord of Day7. The inflammation related proteins expression after MRS2395 intervention on Day7, n = 4 per group,#P < 0.05,##P < 0.05. (a) The proteins expression after MRS2395 injection.(b-d) iNOS, Arg1, IL-1β and IL-6 levels in spinal cord on Day7.
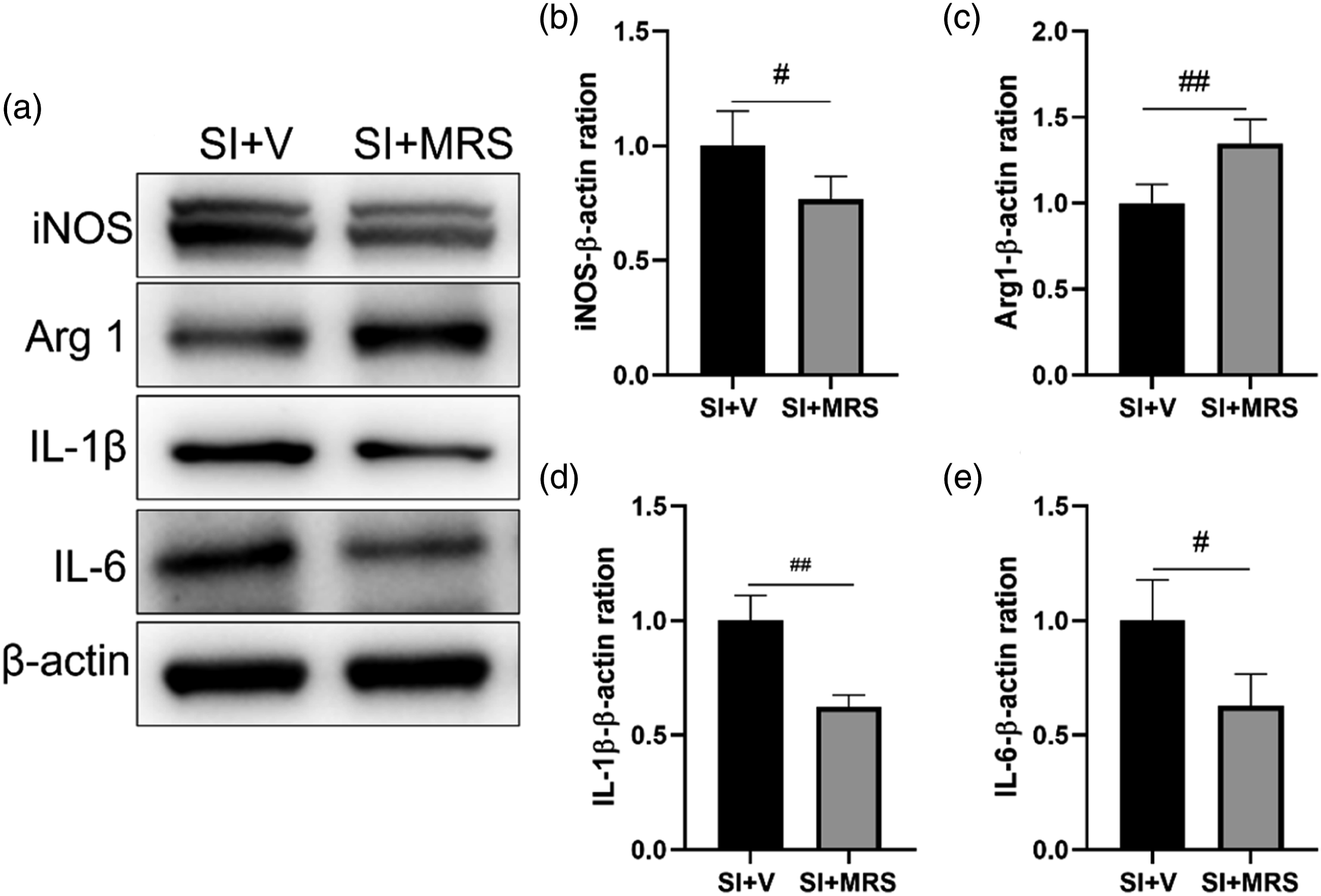
The above results indicate that intrathecal injection of MRS2395 inhibited the P2Y12/p38MAPK/NF-κB signaling pathway, attenuated microglial activation, facilitated the transition of microglia from the M1 to the M2 phenotype, and reduced the release of proinflammatory cytokines in the spinal cord of adolescent mice.
Intraperitoneal injection of DHC inhibits the activation of the P2Y12 signaling pathway and microglia, reduces the expression of proinflammatory cytokines in the spinal cord, and alleviates postoperative incisional pain in adolescent mice
Compared with group SI+V, group SI+DHC exhibited a significant increase in the PWMT from postoperative day 6 until before returning to BL on day 10 (p < 0.05), whereas that of group SI+V returned to BL on day 14(Figure 12). Pain behavior was exhibited by PWMT after injection of DHC, n = 8. ###P < 0.001.
On postoperative day 7, the number and cell body area of microglia in the dorsal horn of the spinal cord were significantly reduced in group SI+DHC compared with group SI+V (p < 0.05 for both; Figure 13(a)-(c)). Furthermore, iNOS expression was significantly reduced while that of Arg1 was significantly increased in the spinal cord of group SI+DHC compared with group SI+V (p < 0.05 for both; figure 15(a)-(c)), indicating that DHC inhibits microglial activation and promotes their phenotypic transformation from M1 to M2. The effect of DHC on microglial activation on Day7, n = 3 per group,##P < 0.01, ###P < 0.001. (a) microglia activation detected by immunofluorescence with IBA1. (b, c) The number and body area of microglia in each group on Day7.
Compared with group SI+V, there was no significant difference in the expression of p38MAPK and P65 in group SI+DHC (p > 0.05), while expression of P2Y12 receptor, p-p38MAPK, and p-P65 were significantly decreased (p < 0.05 for all; Figure 14(a)-(g)). In addition, compared with group SI+V, proinflammatory cytokine expression including IL-6 and IL-1β was significantly in group SI+DHC reduced with statistical significance in spinal cord (p < 0.05; Figure 15(a), (d), (e)). Besides, the expression of iNOS and Arg1 was overturned after DHC adminstratin(p < 0.05; Figure 15(a)-(c)) The suppression of DHC on P2Y12/p38MAPK/NF-κB signaling pathway detected by immunoblotting, n = 4 per group,#P < 0.05. (a,d) The protein levels in spinal cord on Day 7. (b-c,e-g) p38MAPK, p-p38MAPK, P65, p-P65 and P2Y12 receptor levels in spinal cord of Day7. The inflammation related proteins expression after DHC injection on Day7, n = 4 per group, #P < 0.05,##P < 0.05. (a) The proteins expression after DHC injection. (b-d) iNOS, Arg1, IL-1β and IL-6 levels in spinal cord on Day7.

The results above demonstrate that intraperitoneal injection of DHC effectively inhibited the activation of the P2Y12/p38MAPK/NF-κB signaling pathway and microglia, promoted the transformation of microglia from the M1 to the M2 phenotype in the spinal cord, and reduced release of inflammatory cytokines to alleviate postoperative pain in adolescent mice.
Discussion
This study demonstrate that preoperative SD facilitates the activation of the P2Y12/p38MAPK/NF-κB signaling pathway in spinal cord microglia, leading to enhanced microglial activation and release of inflammatory cytokines, which in turn exacerbate postoperative incisional pain in adolescent mice. Furthermore, we found that DHC effectively suppressed microglial activation by inhibiting the P2Y12/p38MAPK/NF-κB signaling pathway, promoted the polarization of microglia towards the M2 phenotype, reduced the expression of proinflammatory cytokines, and alleviated preoperative SD-induced persistent postoperative pain. Overall, the result indicate spinal microglial activation via the P2Y12 receptor in an adolescent postoperative pain model. Furthermore, the novel findings indicate that P2Y12 is a new target of DHC in persistent postoperative pain induced by SD.
Research has shown that SD not only increases pain sensitivity and lowers pain thresholds in individuals, but also induces spontaneous pain.23-27Although previous studies have demonstrated that preoperative SD can exacerbate postoperative pain and promote nociceptive sensitization, the subjects were all adulthood and did not specifically investigate the effects of SD on pain during adolescence. In our study, we induced SD in adolescent mice prior to surgery to mimic the impact of SD on postoperative pain. Our results indicate that preoperative SD exacerbated and prolonged postoperative pain, causing persistent postsurgical pain. Using the PWMT as an indicator pain, we found that SD decreased the pain threshold in mice, while spontaneous pain had not yet transpired, as result that persisted for approximately 10 days. This finding slightly deviates from the research results of Huang et al, 22 and may be attributed to differences in the duration of SD induction and the age of mice. More importantly, SD significantly increased and prolonged postoperative incisional pain. Hence, preoperative SD heightened the susceptibility of adolescent mice to surgical pain, leading to an increased pain sensitivity and prolonged duration of pain.
The role of sleep is particularly crucial for teenagers in the maturation stage of the CNS. The prevalence of SD, a common sleep disorder among surgical patients, is observed in more than half of patients undergoing surgery. 28 In addition to triggering inflammation and slowing wound healing, SD is likely involved in the transition from acute to chronic pain. Indeed, a study indicated that preoperative SD significantly exacerbated postoperative pain following breast surgery. 29 A preclinical study also demonstrated that exposure to SD prior to surgery resulted in a delayed recovery of mechanical pain and thermal hyperalgesia induced by incision in rats. 30 SD in adults is associated with reduction in pain thresholds, and can be ameliorated by short increases in sleep. However, it should be noted that adolescents do not exhibit the same restorative effects.31,32 Furthermore, compared with adults, adolescents suffering from SD have an increased risk of developing chronic pain after an acute injury, increasing the probability of chronic pain. 33 Existing evidence suggests that neuroinflammation functions as the principal mechanism underlying the transition from acute to chronic pain. Microglial activation and subsequent release of pro-inflammatory cytokines play an important role in spinal cord neuroinflammation and persistent pain. Indeed, even a slight reduction in sleep duration has been shown to be accompanied by an increase in inflammatory factors, which is more evident among adolescents. 34 The “double-hit hypothesis” of microglia provides a useful explanation for the persistent postoperative pain resulting from SD in adolescent mice, whereby in the adolescent CNS, SD induces microglial activation and enhances the inflammatory response to subsequent surgical intervention, 35 resulting in chronic pain. 36 Moreover, the deleterious impact of chronically activated microglia during adolescence may persist into adulthood in some cases.16,37,38 As shown in our experiments, significant microglial activation occurred in both the SD and incision mouse models on postoperative day 1. However, SD induced mild microglial activation, whereas in the incision model microglial activation was significantly increased, with microglia exhibiting an amoeboid morphology, which was concentrated in lamina Ⅰ–Ⅱ of the spinal dorsal horn. Microglia in group SI exhibited more pronounced activation than that of group I. However, microglial activation persisted exceeding postoperative day 7 only in group SI. The results suggest that SD and incision-induced activation of spinal cord microglia in adolescent mice is self-healing. It is believed that adolescent stress leads to long-term activation of spinal microglia, 39 including persistent alterations in gene expression. 40 During SD and pain, activated microglia in the spinal cord release proinflammatory cytokines, including IL-1β and IL-6, which play an important role in pain progression. Overall, the results of the current study indicate that the prevention and treatment of chronic pain should focus on reducing neuroinflammation and microglial activation.
When resting microglia detect any potential chemical substances or abnormal signals from neurons and other glia, they become activated, which alters both their morphology and function. 41 Specifically, during microglial activation, besides an increase in numbers, a morphological transformation from a bifurcated to an ameboid shape occurs, as does increased secretion of cytokines. 42 Typically, activated microglia can be classified into the pro-inflammatory M1 phenotype and anti-inflammatory M1 phenotype. M1 phenotype microglia may disrupt the balance between excitatory and inhibitory synaptic transmission in the CNS, inducing hypersensitivity and chronic pain. The secretion of proinflammatory cytokines by M1 microglia, such as TNF-α, IL-6, and IL-1β, predominantly contributes to the sustained inflammatory response, 43 with iNOS expression being the most common marker of M1 microglia. 44 M2 phenotype microglia suppress the microglial inflammatory response, facilitating the recovery of the microglial resting state and homeostasis, with Arg1 being the most common marker of M2 microglia. 41 Following exposure to adverse stimuli or immune challenge during adolescence, there is an increase in M2 microglia within the CNS, 45 which contributes beneficially to optimize neural circuitry to the demands of the environment. Therefore, M1/M2 microglia imbalance is a significant hallmark of neuroinflammation and a driver of acute and chronic pain.46,47Targeting the activation and polarization of spinal cord microglia likely leads to anti-inflammatory and analgesic effects, and is thus a promising treatment for pain.21,48 Our results demonstrated that microglial activation was associated with an increase in iNOS expression and a corresponding decrease in Arg1, indicating that the activation of spinal microglia induced by SD was predominantly of the M1 phenotype, which in led to increased expression of proinflammatory cytokines, thereby promoting neuroinflammation and pain. Interestingly, the inhibition of microglial activation by minocycline alleviated SD-induced postoperative pain to further confirm these findings.
The P2Y12 receptor is known to be widely expressed in the CNS, primarily on microglia with low expression on astrocytes, neurons, and oligodendrocytes. 49 Moreover, the P2Y12 receptor is exclusively expressed on microglia in the spinal cord and serves as a specific marker for this population. 50 The P2Y12 receptor plays a crucial role in microglial activation under pathological conditions and triggers downstream activation of the p38MAPK/NF-κB signaling pathway, thereby facilitating the expression and release of pro-inflammatory cytokines.51,52 Neurons release ATP in an activity-dependent manner, leading to the upregulation of microglial P2Y12 and subsequent microglial activation, to induce diverse phenotypic responses involved in pain regulation. 53 The P2Y12 receptor on microglia transmits inflammatory signals by activating MAPK, which is a key signaling cascade molecule in immune cells. 54 In particular, p38MAPK is primarily activated in spinal cord microglia, while exhibiting minimal activity in neurons.55,56 Phosphorylated p38MAPK further activates intracellular NF-κB, which upregulates proinflammatory gene transcription and amplification of inflammatory signals57,58 and induces microglial polarization towards M1 phenotype, 59 to promote the expression and secretion of proinflammatory cytokines. Importantly, the microglial inflammation facilitated by increased P2Y12 receptor expression is closely associated with the p38MAPK/NF-κB signaling pathway. If adverse stimuli persist, microglial activation may become chronic, which in turn increases the risk of chronic inflammation and pain.48,58,60 Here, we showed that phosphorylation of p38MAPK and NF-κB was inhibited by the blockade of the P2Y12 receptor using MRS2395, whereas an antagonist of p38MAPK and NF-κB did not affect P2Y12 expression, indicating that the P2Y12 receptor acts upstream to activate the p38MAPK/NF-κB signaling pathway.61-63 In the current study, we also showed that both SD and planter incision led to increased expression of P2Y12, p-p38MAPK and p-P65 on postoperative day 1, levels of which returned to BL on postoperative day 7. Due to the exclusive expression of the P2Y12 receptor on microglia in spinal cord of mice, 64 it is evident that SD and plantar incision induced upregulation of the P2Y12 receptor on microglia, leading to p38MAPK/NF-κB phosphorylation in microglia and subsequent microglial activation, changes that significantly increased and prolonged pain in the SD and plantar incisional pain models. We also showed that intrathecal injection of MRS2395 significantly reversed these changes in the spinal cord on postoperative day 7, indicating that SD can activate microglia to a certain extent through the P2Y12/p38MAPK/NF-κB signaling pathway, causing prolonged microglial activation in response to subsequent incision. In future research, the P2Y12 receptor maybe represent a promising target for clinical analgesic treatment.
Rhizoma Corydalis, a traditional Chinese medicine, has been utilized for thousands of years as an analgesic to alleviate abdominal, traumatic, pelvic inflammatory, and other types of pain. The herb has been demonstrated to possess significant analgesic properties, as well as sedative and hypnotic effects. 65 In recent years, many studies have investigated the pharmacological effects of Rhizoma Corydalis extract and elucidate its mechanism of action. DHC is a principal alkaloid extracted from Rhizoma Corydalis, which possesses analgesic properties. Here, we showed that intraperitoneal injection of DHC significantly inhibited microglial activation in the spinal dorsal horn and reduced expression of the P2Y12 receptor, p-p38MAPK, p-NF-κB, and inflammatory cytokines. Furthermore, DHC shortened postoperative pain. These findings suggest that DHC has the ability to inhibit the P2Y12/p38MAPK/NF-κB signaling pathway in spinal cord microglia, thereby suppressing microglial activation and proinflammatory cytokines release, ultimately alleviating postoperative pain. Previous studies have demonstrated that DHC participates in the regulation of inflammation and analgesia through diverse mechanisms. DHC possesses the ability to inhibit cholinesterase activity and modulate the function of the cholinergic anti-inflammatory response. 66 Furthermore, DHC effectively inhibits Nav1.7, attenuating formalin-induced inflammatory pain with low cardiotoxicity. 67 In addition, the expression of TNF-α, IL-1β, and IL-6 in the spinal cord decreased following administration of DHC, thereby mitigating edema, effects that were reversed following administration of naloxone reversed these changes. 20 Intraperitoneal administration of DHC has also been shown to mitigate the production of proinflammatory cytokines in both blood and the spinal cord, induce microglial polarization towards the M2 phenotype in the spinal cord, and alleviate bone cancer-induced pain. 68 DHC also regulates the key enzymes 2-AG and MAGL, which are involved in cannabinoid metabolism, thereby activating the endogenous cannabinoid system to exert analgesic effects. 69
Based on above findings, it is evident that DHC can exert analgesic effects through multiple mechanisms, primarily by inhibiting the inflammatory response, which is consistent with our current findings. Interestingly, DHC also inhibits platelet activity by binding to the P2Y12 receptor,70-72 suggesting that it maybe also mediate microglial activation through the P2Y12 signaling pathway, leading to inhibition of proinflammatory cytokine expression and secretion, thus resulting in pain relief.
The current study indicates that the anti-inflammatory effect of DHC is associated inhibition of the P2Y12/p38MAPK/NF-κB signaling pathway, which mitigates microglial activation, promotes M2 polarization, reduces inflammatory cytokine expression, and alleviates preoperative SD-induced postoperative persistent pain in adolescent mice.
Our study has some limitations. First, to avoid the impact of estrogen on experimental results, we only examined male adolescent mice. Nevertheless, gender differences in microglia on pain have been documented, and endocrine development during adolescence differs from that of adulthood. Hence, the potential impact of preoperative SD on female adolescent mice should not be overlooked. Moreover, further exploration is required to investigate the impact of SD on brain related pain signaling pathways and other types of glial cells. Finally, it is imperative to establish a standardized SD model.
Footnotes
Author contributions
Haikou Yang , Yufeng Zhang and Qingling Duan conceived the idea, performed the experiments and wrote the manuscript. Kun Ni and Yang Jiao designed the experiments. Jixiang Zhu and Jian Sun analyzed the data. Jian Sun, Wei Zhang and Zhengliang Ma supervised the study.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (Grant Numbers 82171225 and 81971044).
