Abstract
Chronic pain is one of the most common, costly, and potentially debilitating health issues facing older adults, with attributable costs exceeding $600 billion annually. The prevalence of pain in humans increases with advancing age. Yet, the contributions of sex differences, age-related chronic inflammation, and changes in neuroplasticity to the overall experience of pain are less clear, given that opposing processes in aging interact. This review article examines and summarizes pre-clinical research and clinical data on chronic pain among older adults to identify knowledge gaps and provide the base for future research and clinical practice. We provide evidence to suggest that neurodegenerative conditions engender a loss of neural plasticity involved in pain response, whereas low-grade inflammation in aging increases CNS sensitization but decreases PNS sensitivity. Insights from preclinical studies are needed to answer mechanistic questions. However, the selection of appropriate aging models presents a challenge that has resulted in conflicting data regarding pain processing and behavioral outcomes that are difficult to translate to humans.
Introduction
With a predicted 30% increase of individuals 65 years and older in the U.S. in the next 10 years, understanding the prevalence of chronic pain, comorbidities, mechanisms, and therapeutic strategies in age-related pain conditions is more important than ever.1,2 Older adults are particularly vulnerable because of the increased prevalence of degenerative musculoskeletal disorders, frailty, and multimorbidity placing them at increased risk for pain states.3–5 Moreover, behavioral complications during aging, such as cognitive impairments could lead to decreased self-care, self-awareness, and worsening of health conditions, such as prolonged pain and increased hospitalization.6–8
The International Association for the Study of Pain (IASP) recently updated its definition of pain as “an aversive sensory and emotional experience typically caused by, or resembling that caused by, actual or potential tissue injury”. 9 Chronic pain is defined as pain that persists past the normal healing time, usually lasting more than 12 weeks, making it distinct from acute pain.10,11 Chronic pain is a complex condition that involves sensory, affective, cognitive, and behavioral components. 10 It has a heterogeneous etiology such as tissue injury, inflammation, autoimmunity, chemotherapeutics, and stress referred to as functional pain conditions. In addition, chronic pain is often comorbid with medical conditions like depression, anxiety, sleep disturbances, and fatigue. 12 Recovery from chronic pain is often bleak, with the most recent estimates by Li et al. at 30%. 13 Duration of previous chronic pain, age, chronic diseases, personality traits, and differences in cortico-limbic structural and functional connectivity are some factors that have been associated with a lower probability of recovery.13,14 Chronic pain syndromes are common in older adults and are associated with social isolation, functional disability, significant suffering, and increased healthcare resource utilization and costs. Chronic pain is also a risk factor for accelerated cognitive decline and premature death, suggesting a mechanism between pain and cognition. 15 As such, chronic pain has been increasingly recognized as a condition/disease of its own, particularly in older adults.16–18
This review begins with a brief overview of the epidemiology of pain, pain assessment, and pain management in older adults, as these have been reviewed excellently elsewhere. The main focus is on a thorough review of both pre-clinical research and clinical data on chronic pain among older adults to present a unifying concept, identify knowledge gaps, and provide the basis for future research and clinical practice.
Epidemiology of pain and aging
Chronic pain is an epidemic. In the U.S., one in five adults (20.4%, i.e., 50 million) report suffering from pain almost daily for at least 6 months, according to data from the 2016 National Health Interview Survey.19–21 The prevalence increases with advancing age: 27.8% of 45–64 year-olds, 27.6% of 65–84 year-olds, and 33.6% of 85 years and older have reported chronic pain. 20 By severity, 8% of U.S. adults (19.6 million) reported high-impact chronic pain, that is, pain that restricted life or major work activities on most days or every day during the past 6 months. 20 By sub-type, the broad prevalence of chronic neuropathic pain is 10% of U.S. adults, with significant variation based on age and ethnicity.22,23
Population-based pain prevalence estimates are often broad. Pain prevalence estimates from the National Health and Aging Trends Study (NHATS), a survey of a nationally representative sample of Medicare beneficiaries aged 65 and older in the U.S., employ conservative definitions of pain and activity-limiting pain and range between 13% and 49%. Several patient-level factors including comprehension and memory, attitude/behavioral problems, and language factors contribute to the wide variability in the reporting of chronic pain in older adults. In a sample of 7601 adults enrolled in NHATS, over half of the participants (52.9%) reported bothersome pain in the previous month, whereby back pain was most commonly reported (30.3%), followed by knee pain (24.8%), and shoulder pain (19.9%). 24 Moreover, a large British longitudinal cohort and meta-analysis found that chronic pain is a risk factor (57% greater risk) for early death. 25
Important to consider is the contribution of pain in dementia and how it is reported in Alzheimer’s patients. 26 Indeed, chronic pain can be a contributing factor to premature death 27 and has also been linked to increased cognitive decline and dementia (Whitlock EI et al., 2017). Therefore, it is important to assess the relationship and role of chronic pain in Alzheimer’s disease and dementia. 28 These findings suggest that shared mechanisms such as environmental exposures, genetics, or molecular targets could play a role in both conditions.28,29 Moreover, degeneration in brainstem regions modulating descending pain inhibition such as periaqueductal gray matter has been found in Alzheimer’s disease. 30 Cortico-limbic regions that have been implicated in pain modulation are severely affected by Alzheimer’s pathology and pain is associated with cognitive deficits that are more pronounced with aging.31–39 Furthermore, an NHATS study focused on older adults with pain and dementia, reported that out of 804 participants, nearly two-thirds (63.5%) reported bothersome pain in the last month (vs 54.5% among the matched cohort without dementia), and 43.3% reported activity-limiting pain (vs 27.2% among the matched cohort without dementia). 40 These studies in patients with dementia suggest potentially shared mechanisms between pain (or its treatment) and cognitive impairment. It may also suggest that patients with dementia are able to recognize and report pain, which is not necessarily to be expected with cognitive impairment, and therefore becomes a clinical concern.
Regardless, the burden of chronic pain and other comorbid conditions related to aging is expected to continue to increase with the growing proportion of older adults (≥65 years of age) representing 16.0% in 2018 to almost 20.6% by 2030. 41 The economic negative impact of chronic pain on the health care system and the economy is staggering. In the United States alone, estimated costs attributable to chronic pain, including direct medical costs, disability, and loss of productive time, exceeds $600 billion annually. 19 Along with the chronic pain crisis, the current opioid epidemic has resulted in a rising number of drug overdose deaths associated with prescription and synthetic opioid abuse/misuse. 42
Chronic pain assessment in older adults
A thorough assessment of chronic pain is an integral step toward developing an appropriate management plan for patients. Older adults are likely to underreport their pain for numerous reasons, including, but not limited to, assumptions that pain is a normal part of aging, fear of being labeled weak or identified as a complainer, fear of opioid addiction, and previous dismissal of pain report by healthcare providers.43–45
A comprehensive chronic pain assessment for older adults integrates a complete medical history, physical examination, relevant diagnostic tests, as well as an evaluation of sensory deficits, affective changes, and cognitive impairment.45,46 Patients with chronic pain should be assessed at each visit. An array of assessment tools are available to assess pain intensity among patients. Simple and easy-to-understand tools are the most effective for use with older adults, who are more likely to face cognitive impairments. 47 The most widely used pain assessment tools for older adults are the Numeric Rating Scale (NRS), Patient-Reported Outcomes Measurement Information System (PROMIS), Oswestry Disability Index (ODI), Verbal Descriptor Scale (VDS), Iowa Pain Thermometer (IPS), and Faces Pain Scale-Revised (FPS-R), all of which have been demonstrated to be valid and reliable. 43 NRS asks the patients to rate their pain from 0 to 10 with 0 indicating no pain and 10 representing the worst possible pain. 48 PROMIS is an NIH-funded rigorously tested patient-reported outcome (PRO) measurement tool. 49 The survey utilizes a computer adaptive testing system to target quality of life comprehensively and includes a pain assessment component. ODI assesses the functional limitations of low back pain in everyday life through a ten-question survey with each question targeting a specific activity of daily living (ADL). 50 Questions are scored to a maximum of five points and summed, with higher scores indicating a more severe disability. VDS asks the patient to describe his/her pain from “no pain,” to “mild,” “moderate,” “severe,” or “pain as bad as it could be”. 51 IPS is a modified VDS with seven pain descriptors of increasing pain intensity. 52 FPS-R asks patients to select one of six facial expressions that correspond with their pain, for a score between 0 to 10. 53 Additional comprehensive standardized pain assessment tools that assess the intensity and functional impact of pain are also available: Brief Pain Inventory, Geriatric Pain Measure, and Pain Disability Index. 44
When evaluating older adults with cognitive decline, observational methods along with self-report are used.24,54,55 Caregivers are also enlisted to provide their assessments of the patient’s pain. Reviews of various measures to assess pain in older adults have been summarized elsewhere.10,46,48
Managing chronic pain in older adults
U.S. Guidelines for standardized care of chronic pain in the elderly.

Non-pharmacologic pain management strategies
Non-pharmacological interventions include psychosocial interventions, complementary and integrative health therapies, rehabilitation therapies, and exercise. 10 A recent meta-analysis of the efficacy of psychological interventions on chronic pain analyzed 22 studies on the impact of cognitive behavioral therapy (CBT)-based interventions on chronic pain outcomes. 60 The interventions produced small yet statistically significant benefits for pain relief, catastrophizing beliefs, and self-efficacy. Moreover, interventions were strongest when delivered using group-based approaches. The authors concluded that there is a need for psychological interventions that generate greater treatment effects for older adults.
Caregiver support is important in successful pain management programs for older adults, especially among those with cognitive decline. 61 Health professionals should partner with caregivers on proper medication management for patients. 62
Pharmacological treatments for chronic pain
Age-associated physiological changes can affect the way the body responds to pain medication. 63 Additionally, older adults tend to have more comorbidities, which can complicate pharmacologic pain management. 64 Therefore, in the elderly, pain medications need to be monitored carefully by a clinician to avoid side effects, adverse drug-drug interactions, and over- or under-use.63–66 Treatment plans vary according to origin and pain intensity. Paracetamol (acetaminophen) is the preferred treatment for older adults with mild-to-moderate pain, with minimal side effects. 10 NSAIDs are commonly used to treat pain and can be prescribed in combination with acetaminophen. However, NSAIDs pose increased risks with age and should therefore be prescribed for the shortest duration possible at the lowest effective dose, due to potential adverse effects, including gastrointestinal toxicity, nephrotoxicity, and cardiovascular risk. 67
Opioids are usually considered when pain is moderate-to-severe and other treatments have been unsuccessful. Opioid use needs to be monitored carefully due to the increased risk of adverse effects among older adults, which include dependence, ileus, and respiratory depression. In 2016, the Centers for Disease Control and Prevention issued 12 guidelines for prescribing opioids for chronic pain, which specify: (1) non-opioid therapies are preferred over opioids, (2) when opioids are used, prescribing the lowest possible effective dosage to reduce risks of opioid misuse and overdose, (3) and, monitoring all patients closely when prescribing opioids. 68 Precautions when using opiates in the elderly population have been reviewed elsewhere. 69
Chronic pain mechanisms and aging
Pain processing is not static but plastic, with the pain becoming chronic through long-term changes in the neural structures.15,70,71 The comprehensive understanding of the complexity of persistent pain and underlying mechanisms remains a challenge because studies are often focused on individual compartments of the nervous systems, such as nociceptors in the PNS, spinal circuits in the CNS, and to a lesser extent, different brain regions. There is still a lack of a system-wide understanding that integrates pain-related changes from nociceptor populations to higher-order processing of pain signals accounting for affective and cognitive aspects of behavior. 72 Peripheral sensory neurons undergo alterations with aging, leading to a decreased response to evoked stimuli73,74 and subsequent loss of pain sensation. CNS pain pathways such as the spinothalamic, spino-parabrachio-amygdaloid, and other pathways relay signals from the spinal cord to the brain. 15 The brain regions implicated in pain perception, cognitive processing, and aversive experiences include the posterior thalamus, sensorimotor cortex, limbic regions (medial thalamus, amygdala, ventral striatum/nucleus accumbens, hippocampus, medial prefrontal cortex), paralimbic regions (insular cortex), and pain modulatory centers (periaqueductal gray, PAG, and rostral ventromedial medulla, RVM).32,39,75–77
Compared to physiological pain, neuroplastic changes in the peripheral and central nervous system such as “central sensitization” play a prominent role in chronic pain development and persistence.78–80 Failure of descending inhibition or a switch to descending facilitation are believed to be mechanisms that allow the persistence of pain.81–84 The descending pain modulatory system converges on the PAG-RVM system that connects brain regions to spinal nociceptive processing. 84 Neuroplasticity in the corticolimbic circuitry that interconnects brain regions such as the medial prefrontal cortex and sub-cortical limbic areas such as the amygdala, contributes to the complexity of pain, its affective and cognitive dimensions, pain modulation, and comorbidities, and may predict pain persistence and resilience.14,32,33,77,85–88 Peripheral and central/spinal sensitization contributes to increased nociceptive signals to the brain to engage sensory (thalamo-cortical) and affective (cortico-limbic) systems. 89 Pain conditions are associated with functional and structural changes in these brain regions31–34,71 and differences in connectivity between prefrontal cortical and limbic regions such as the hippocampus and amygdala play a critical role in the prediction and amplification of chronic pain. Cortico-limbic reorganization amplifies nociceptive signals to the brain and drives chronification through emotional learning. 14
A better understanding of the mechanisms of pain transitioning from acute to chronic is critically important for pain management. Brain scans of patients with chronic pain have shown structural changes in these brain regions, including the reduced amygdala, hippocampus, and medial prefrontal cortex volumes.14,33,85,86 Studies using positron emission tomography (PET)-magnetic resonance in combination with radioligand tracer have been used to show glial activation in clinical studies in low back pain 90 and fibromyalgia 91 patients. These findings point towards neural and non-neuronal mechanisms of chronic pain.71,85
As we age, our immune system is primed, such that a low level of inflammation is prevalent throughout the body chronically in the absence of injury (inflammaging).92,93 Inflammatory mediators in the periphery, such as cytokines, chemokines, bradykinins, and other algogens can prolong the firing of nociceptors and reduce their threshold for activation.70,94 It has been well documented that systemic pro-inflammatory modulators are consistently upregulated in the elderly.92,93,95,96 Cytokines such as tumor necrosis factor alpha (TNFα), interleukins, and signaling molecules like inducible nitric oxide synthase (iNOS) are known activators of nociceptors for mediating pain responses.92,97 Aging also alters cell adhesion molecules via inflammation, which can affect wound healing.
98
Another major factor in age-related inflammation is the perturbance of anti-inflammatory and redox pathways. It has been shown that antioxidants are decreased with advanced age, leading to an upregulation of pro-inflammatory pathways.92,99 Currently, gerontologists and nutritionists recommend caloric restriction in the elderly, to help combat the activation of pro-inflammatory signaling and redox-responsive transcription factors like those of the peroxisome-proliferator-activated receptor (PPAR) family.
100
Downstream activation of relevant pathways such as prostaglandin E2 (PGE2), cyclic adenosine monophosphate (cAMP), and cyclooxygenase 2 (COX-2) can be prevented and pain hypersensitivity can be avoided through calorie restriction.70,101 Thus, it is likely that aging-associated chronic inflammation drives the development and persistence of chronic pain in geriatric individuals. Some unique aspects of inflammation and pain hypersensitivity in the PNS and CNS during aging are presented in Figure 1. Unique aspects of pain in aging. (a) Persistent low-grade inflammation (pink patches) in the aged is characterized by activated immune cells, namely microglia in the CNS and macrophages in the periphery. Secretion of pro-inflammatory factors affects nerves in the periphery as well as responses in the CNS. (b) Differences in PNS inflammatory state, neuronal activity, and pain response in aged peripheral tissues, compared to young. Chronic secretion of pro-inflammatory mediators damages nerves and alters pain sensitivity in the aged. (c) CNS immune sensitization or microglia become “primed”, leading to behavioral deficits in the aged.
Additionally, an increase in the frequency of injuries/disease/physiological degeneration is associated with increased persistent pain. In the elderly, chronic pain is mainly caused by musculoskeletal conditions; neuropathic pain, ischemic pain, and pain associated with cancer and its treatment are also major contributors. 64 Also, elderly women experience a high incidence of pain due to vertebral compression fractures. 64 The geriatric population is particularly vulnerable to these comorbidities and is consistently moving towards more independent/unassisted living. These contributing factors can lead to decreased care and increased painful outcomes. 102
The elderly have constant low-grade inflammation 103 and often aging-associated neurodegeneration, 104 which suggests that changes in brain structure play an important role in neuroplasticity associated with chronic pain. Age-associated comorbidities show decreased CNS plasticity and tissue degeneration.6,105,106 However, dynamic PNS plasticity with altered pain sensitization during aging and in response to inflammatory stimuli107–109 makes it especially challenging to develop a unifying hypothesis that captures these complex and seemingly opposing interactions (Figure 1). It is only logical that the research focus is now shifting toward investigations into aging-associated degeneration and inflammation and how these factors may facilitate chronic pain development.
Sex differences in pain
Epidemiological and clinical studies provide strong evidence for a greater prevalence of chronic pain syndromes such as fibromyalgia, osteoarthritis, and musculoskeletal pain, in females compared to males. 110 Also, it has been well-documented that males and females experience pain differently. 111 However, preclinical and clinical evidence for sex differences in pain sensitivity and tolerance is less clear.112–114 A meta-analysis of 122 articles published between 1998 and 2008 investigated sex differences in the perception of various experimental pain modalities in healthy subjects. 115 The study found that pressure pain thresholds were lower in females than in males, while cold and ischemic pain thresholds were similar in both sexes. Similarly, tolerance to thermal and pressure pain was lower in females than in males, but there was no sex difference in ischemic pain tolerance. Additionally, pain intensity did not show any sex differences in many of the pain modalities studied. Based on these findings, the authors proposed that sex differences in pain sensitivity show inconsistent patterns. However, a reanalysis of the same set of articles, which considered the direction of the sex difference reported by each article, showed that women were consistently more sensitive to pain than men, irrespective of pain modality or outcome measure. 116 Depression was not shown to mediate sex differences whereas the evidence for involvement of anxiety is further complicated in aged individuals as their hormonal levels are changed over time. Preclinical studies have shown that sex differences in pain processing and modulation exist at the molecular, cellular, and systems levels and involve genetic, hormonal, and neuroimmune factors. 113
The contribution of hormones to differences in pain sensitivity between sexes is unclear. Estrogen receptors are distributed throughout the peripheral and central nervous system in both males and females. Estrogen alpha and beta subtype receptors have been shown to be present in regions of the brain responsible for pain processing and modulation such as the amygdala, hypothalamus, periaqueductal gray, and regions of the dorsal spinal cord.117,118 In the peripheral nervous system, estrogen receptors punctate the cell surface of all nociceptors.
Preclinical studies in ovariectomized rodents suggest a role for estrogen in pain chronification that is yet to be well-defined. Some studies demonstrated increased mechanical and thermal hyperalgesia in ovariectomized rodents that were alleviated with estrogen administration.119,120 Other studies found estrogen-dependent hyperalgesic priming in female rodents but increased pain after knocking out or inhibiting estrogen receptors.121–123 This lack of clarity in the preclinical setting and a need to improve our understanding of hormonal influence in pain development offers exciting opportunities for further research. It should be noted that the role of other sex hormones, specifically androgens, remains to be clarified. Given this ambiguity, it is not clear if and how physiological or psychological factors could mediate sex differences in pain perception. However, cognitive and social factors appear to partially affect these differences, with individual history influencing pain responses in the female population. 124
Clinical studies found that sex differences in pain perception are subtle. A study conducted on both sexes of reproductive age determined that noxious laser-evoked potentials were lower in men than women, but there were no differences in subjective pain ratings. 125 Another study showed that behaviorally defined parameters had clear sex-based differences. 126 Measures of two such parameters, acceptance of pain and the fear of movement (kinesiophobia), showed that when men and women are subjected to the same magnitude of pain, women tend to be more active, accept the pain, and draw on social support while men have a lower activity level, kinesiophobia, and mood disturbances. 126
Thus, it becomes imperative to study sex differences with regard to different aspects of pain (sensorimotor, affective, cognitive, social) and at different ages in clinical pain conditions as well as in pre-clinical models to determine underlying mechanisms. Animal models of pain are well suited for mechanistic and preclinical studies, whereas experimental human pain models have limitations due to ethical concerns about invasive stimuli and because the prediction for clinical pain would be based on psychophysical readouts, 127 which would be particularly difficult in advanced age populations due to factors like cognitive impairment. Robust pain assessments need to be conducted to accurately classify chronic pain and detect sex- and age-related differences. This would include assessing the sensory and affective parameters of pain, the temporal aspect or location of pain, and the distribution of pain in a patient’s body. 128 Again, advanced-age individuals may have difficulties in accurately expressing such aspects, and therefore, more objective measures of pain mechanisms need to be included such as brain imaging, biomarkers, genetic factors, pharmacological phenotypes, and others to understand sex differences in pain and aging and to design effective strategies targeting the burden of pain. 128 Additional challenges include multiple comorbidities and ongoing medical treatments.
Preclinical pain research on aging
Animal models used for studying pain processing and testing therapeutic efficacy rely on non-report measures such as evoked responses (mechanical and thermal sensitivity tests or vocalizations) and non-evoked pain behaviors (grimace scales, conditioned place preference, and other operant assays) in models of inflammatory, neuropathic, or functional pain. A major challenge in the field is the need for pain-related parameters beyond nociceptive reflex measurements. 129
Preclinical studies assessing age-related differences in pain processing and behavior (2000 – present).
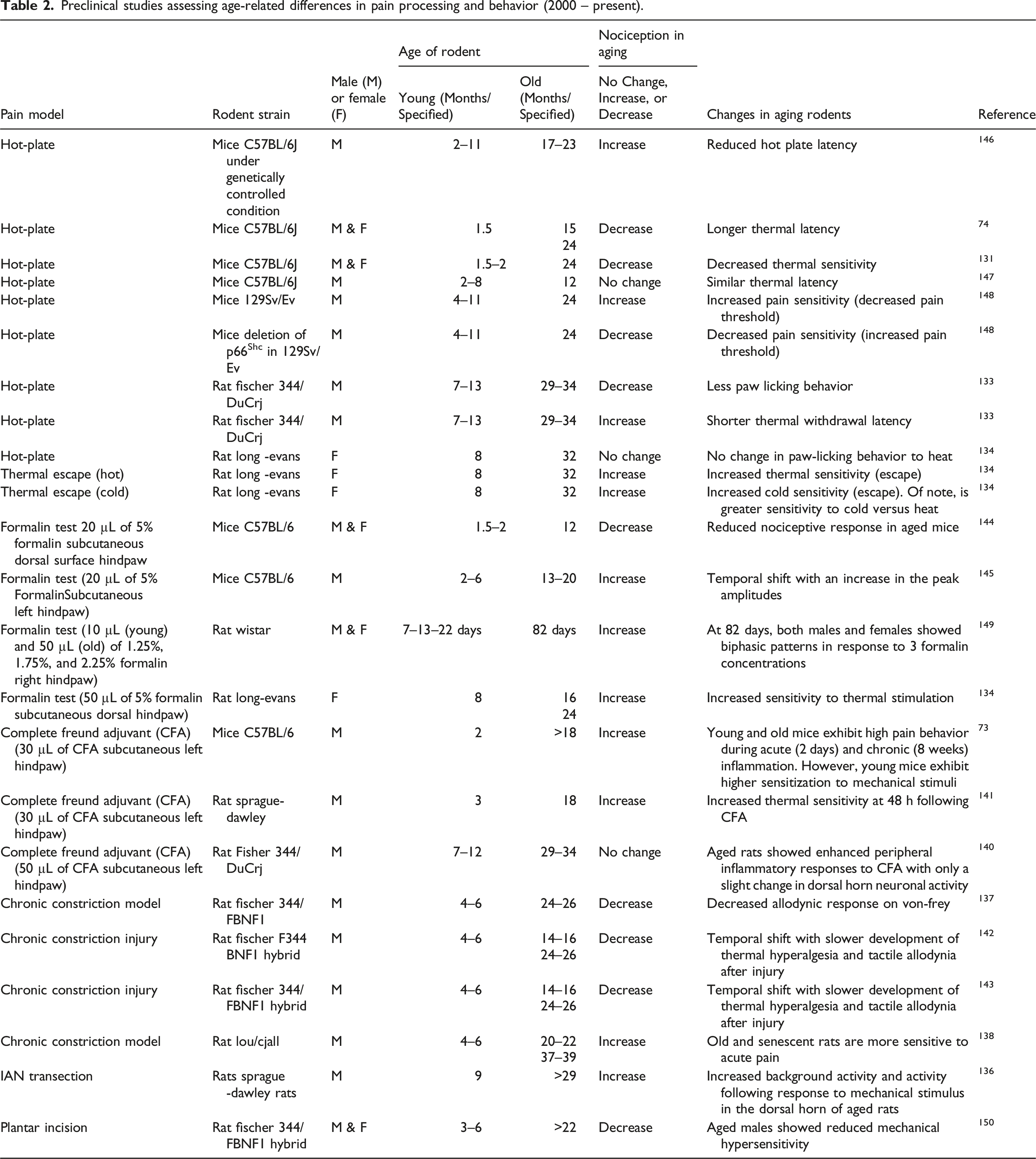
Aged rodents demonstrate altered neuronal activity in the spinal cord. 109 As compared to young adult dorsal horn neurons, aged dorsal horn neurons showed increased excitability but reduced excitatory synaptic input and increased GABAergic inhibitory synaptic input. 109 Studies on dorsal horn neural function in pain models have also shown increased background and evoked activity in aged (>29 months old) compared to young rodents.135,136 To that effect, temporal aspects of treatments or duration of testing on animal pain models become important for protocol relevance.
Numerous studies have shown increased neuroinflammation in aged rodents.106,155,162–164 It is known that chronic activation of immune cells, both in the periphery and the CNS, can lead to increased sensitization.105,106,164–166 However, opposing effects of inflammation and immunosenescence are reported for pain. Some preclinical studies have shown that inflammation and neuroinflammation are higher in the aged than young and decreased pain sensitivity was observed.130,154,156,167 Other preclinical studies have reported increased pain with inflammatory conditions.73,103 Peripheral infusion of lipopolysaccharide (LPS) into young and aged mice caused higher levels of neuroinflammation in the aged females compared to young females, as well as young and aged males, indicating sex and age differences. 168 In contrast, mechanical hypersensitivity responses to complete Freund’s adjuvant (CFA) are reduced in aged male and female mice, while pro-inflammatory cytokine levels in the spinal cord are higher, despite no changes in the dorsal root ganglia. 154 These data suggest that sex-specific differences may also be influenced by age in rodent models, which adds a layer of complexity and yet opens up opportunities for new research avenues. It has been shown that spinal microglia and astrocytes in aged rodents are in a relatively higher primed or reactive state and secrete cytokines that affect the PNS; however, their pain behavior does not necessarily reflect that change.152,156,168–170 This highlights an important gap in characterizing the responses of aging rodents to pain tests. Reduced pain sensitivity despite increased aged-induced inflammation with advanced age, may suggest that the response to pain-relieving therapies based on anti-inflammatory actions may be quite subtle in these animals. Thus, there is a need to re-evaluate methods used to assess pain behaviors and outcomes in aged rodent pain models.
Based on current literature, aged rodents display, in most studies, decreased pain behavior, but more activation of neurons and immune cells and higher levels of pro-inflammatory mediators.146,170–172 This suggests that there is a disconnect in age-induced inflammation, nociceptor signaling, and ascending facilitation to the brain, which may result in decreased pain prevalence in the aged. Interestingly, there are no studies assessing age differences in pain behaviors, neuronal function, and inflammation in the brain, and therefore studies addressing this knowledge gap are needed.
Clinical research on pain and aging models
Clinical studies that highlight age-related differences in pain perception and assessment (2000–Present).

When devising a clinical study to address aging-related changes in general, there are important considerations. As study participants are enrolled voluntarily, there is an inherent participation bias towards healthier older patients who often have fewer comorbidities, less physical health decline, improved cognitive abilities, and better mental health than the average older adult. This is important in clinical practice and may affect the results of studies on pain perception and pain intensity in the elderly. Older patients have been found to report less pain from similar pathology regarding pain frequency and severity, further exacerbated by female gender and specific ethnicity differences. 181 As such, caution should be taken in generalizing the results of such studies.
Implications and future directions for clinical practice and pre-clinical and clinical research
To address chronic pain management among older adults in the clinical setting, the U.S. Pain Management Best Practices Inter-Agency Task Force has called for the development of pain management guidelines for older adults that address their unique risk factors; consideration of multidisciplinary approaches combining pharmacological and non-pharmacological approaches; and the establishment of appropriate pain management education for health care providers who treat older adults. 57
At present, there is a paucity of studies on pain during aging in the diagnostics and therapeutics field, with most published studies reporting outcomes of pain-relieving medications or therapy after various surgeries or disease modalities.40,67,191,192 While these are important areas of investigation required to address the need for novel treatments and their administration, the mechanistic underpinnings of pain in older adults remain unclear. Furthermore, the preponderance of published studies focused primarily on men, limiting our understanding of chronic pain syndromes in women.126,146,156 While using human experimental models for pain research has ethical considerations for inflicting or evoking pain to study pain responses, animal models offer a wider array of tools to examine different aspects of pain and underlying mechanisms in different pain conditions but rely on surrogate measures of pain.127,129,161 A unifying concept about altered pain sensitivity and neuroinflammation in aging has yet to emerge from preclinical studies. Moreover, studies assessing pain thresholds over the lifespan have been inconsistent.108,193 The understanding of the two inevitable conditions of the human, age and pain, has been complicated in a cloud of conflicting data from preclinical and translational studies, due to inconsistencies in pain modality testing (site, testing paradigm, and tools used), age of subjects, and lack of utilization of both sexes.175,193 A more robust characterization of aging pain models is necessary to document behavioral responses to pain, correlate them with neurobiological changes, and determine if these changes are age and/or sex-dependent. With the knowledge gap regarding age-related changes in pain processing in the brain, there are numerous advanced-age animal models for various neurodegenerative diseases 194 that could be repurposed to study various aspects of pain in aged animals and conditions of cognitive decline.
Footnotes
Acknowledgements
The authors would like to thank all present and previous lab. members of the Burton, Neugebauer, and Guindon labs.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by NIH grants R35GM147094 and R21DK130015 (M.D.B), Rita Foundation Award in Pain (M.D.B), and the University of Texas Rising STARS program research support grant (M.D.B). NIH grants R01 NS038261, R01 NS106902, R01NS118731, R01 NS120395, R01 NS129552 (V.N.) NIH grant R01 DA044999, Rita Allen Foundation, Division of Diabetes, Endocrinology, and Metabolic Diseases (DK130015), National Institute of General Medical Sciences (GM147094-01).
