Abstract
Acupuncture, as a traditional treatment, has been extensively used in China for thousands of years. According to the World Health Organization (WHO), acupuncture is recommended for the treatment of 77 diseases. And 16 of these diseases are related to inflammatory pain. As a combination of traditional acupuncture and modern electrotherapy, electroacupuncture (EA) has satisfactory analgesic effects on various acute and chronic pain. Because of its good analgesic effects and no side effects, acupuncture has been widely accepted all over the world. Despite the increase in the number of studies, the mechanisms via which acupuncture exerts its analgesic effects have not been conclusively established. A literature review of related research is of great significance to elaborate on its mechanisms and to inform on further research directions. We elucidated on its mechanisms of action on inflammatory pain from two levels: peripheral and central. It includes the mechanisms of acupuncture in the periphery (immune cells and neurons, purinergic pathway, nociceptive ion channel, cannabinoid receptor and endogenous opioid peptide system) and central nervous system (TPRV1, glutamate and its receptors, glial cells, GABAergic interneurons and signaling molecules). In this review, we collected relevant recent studies to systematically explain the mechanisms of acupuncture in treating inflammatory pain, with a view to providing direction for future applications of acupuncture in inflammatory pain and promoting clinical development.
Introduction
Inflammatory pain, a common clinical symptom of various acute and chronic diseases, has marked effects on the quality of life for patients.1,2 Moreover, it has serious social and economic consequences, 3 especially chronic inflammatory pain. The incidence rate is high in developing countries, especially in middle-aged and elderly people. 4 Despite the wide variety of available painkillers, their efficacy is low and they are associated with various serious side effects. 5 Redness, swelling, increased heat, pain, and loss of function are the five main cardinal symptoms of inflammation. Its also characterized by decreased pain thresholds, enhanced pain responses, and spontaneous pain. 6 Inflammatory mediators (substance P (SP), histamine and prostaglandin E9) are locally released due to tissue damage. These mediators activate immune cells and release various proinflammatory factors. Peripheral nociceptor cells are induced by proinflammatory factors to produce action potentials, which eventually lead to pain. 7 There are several approaches for alleviating inflammatory pain. The first involves the use of central nervous system drugs such as opioid receptor agonists to inhibit pain. However, these drugs are associated with several side effects, such as respiratory depression, drug resistance, gastrointestinal motility decline and drug addiction. The second option involves the use of non-steroidal anti-inflammatory drugs (NSAIDs), which have less ideal analgesic effects.8,9 Due to its good analgesic effects and minimal side effects, acupuncture is a viable option for treatment of inflammatory pain. 10
From sharpened stones to stainless steel needles, acupuncture therapy has been widely used in East Asia since 6000 BC.11,12 The WHO reports that acupuncture is recommended for treatment of 77 diseases, 16 of which are associated with inflammatory pain. As a combination of traditional acupuncture and modern electrotherapy, electroacupuncture (EA) can have satisfactory analgesic effect on various acute and chronic pain.13,14 Controllability of stimulation makes it very popular in basic research and clinical practice. 15 Due to wide clinical applications of acupuncture, more and more studies are now aimed at elucidating on its mechanisms. In studies on effects and mechanisms of acupuncture on inflammatory pain, various animal models have been used.The mechanism of acupuncture analgesia has been gradually discovered and confirmed. We reviewed relevant studies published in the past 5 years. First, we summarized the effects of acupuncture parameters on analgesia in inflammatory pain models. Second, we elucidated on how acupuncture mediates analgesia at the peripheral and central levels through the many bioactive chemicals. Based on this review, the roles and mechanisms of acupuncture in anti-inflammation can be further explored to provide a new direction for clinical acupuncture applications in inflammatory pain.
Influence of parameters
The analgesic effects of EA are affected by various parameters, including the different stimulation times and frequencies. Different frequencies of EA have different therapeutic effects on pain. Fang et al. 16 reported that EA has parameter-specific effects on CFA induction of chronic inflammatory pain relief, these effects are mainly determined by stimulation frequencies rather than stimulation time at a defined stimulation period. Whether stimulation lasted for 20 min, 30 min or 45 min, 100 Hz EA had the greatest analgesic effects and the weakest analgesic effects were established at 2 Hz. The effect of EA’s analgesia were not dependent on the period of stimulation. 17 Moreover, high-frequency EA stimulation is more effective than low-frequency stimulation.18,19 Acupuncture also follows that (e.g. 120–180 rotations per minute).20,21 In other Inflammatory pain models 2/100 Hz or 2 Hz EA was selected to achieve the best analgesic effect. Chai et al. found the 2/100 Hz EA has better effect on acute gout arthritis. 22 Lin et al. Revealed that EA with 2/100 Hz at 1.3 mA can better relieve muscle inflammatory pain. 23 In the study, they found that EA with intensity of A fiber threshold (EA-Ta:EA with 2/100 Hz at 1.3 mA) is more effective for inflammatory muscle pain. EA-Ta was considered to be the minimal stimulation intensity required to evoke EMG of A-activities with a conductive velocity of 4–36 m/s. In the study, the spontaneous EMG activities and imbalances of weight bearing caused by CFA were alleviated by EA. Furthermore, EA-Ta stimulation improved the activities of low-threshold mechanoreceptive (LTM) neurons and inhibited the wide-dynamic range (WDR) neurons in spinal dorsal horns. EA-Ta is an appropriate intervention for relieving inflammatory muscle pain. Mechanistically, EA-Ta activates LTM neurons and inhibits WDR neuron activities in the spinal dorsal horn, resulting in negative correlation between the firing rates of the two types of neurons.
Bai et al. reported that the therapeutic effect of acupuncture has been improved through the transformation of needle. 24 They found that micro/nano porous acupuncture needles (PN) with a large surface area are superior to traditional acupuncture needles (CN) for treatment of CFA-induced hyperalgesia. This is because, PN exhibits a greater needle grasping power, resulting in greater morphological changes in subcutaneous and muscle tissues. Moreover, the analgesic effects of PN treatment increase with increasing time. Therefore, PN is an effective tool for improving the efficacy of acupuncture in clinical medicine.
Therefore, the stimulation frequency of acupuncture is particularly important for its analgesic effects, and there is an optimal frequency preference for different inflammatory pain models. For instance, in CFA-induced chronic inflammatory pain, 100 Hz electroacupuncture and high-frequency acupuncture have the best analgesic effects, while in acute gout arthritis and muscle inflammatory pain, 2/100 Hz electroacupuncture can achieve better effects. Different needles may exert profound analgesic effects of acupuncture, which need to be further elucidated.
Peripheral mechanisms
Peripheral mechanism of acupuncture in treating inflammatory pain.

CFA: complete Freund’s adjuvant; MIA: Monoiodoacetic acid; KOA: knee osteoarthritis MSU:monosodium urate; EA: Electroacupuncture; MA: Manual acupuncture; DRG: dorsal root ganglia; TRPV1: transient receptor potential vanilloid 1; PKC: protein kinase C; PI3K: phosphatidylinositide 3-kinase; SP: substance P; CGRP: calcitonin gene-related peptide; IBA1: ionized calcium binding adaptor molecule 1; CB2R: cannabinoid type 2 receptors; GFAP: glial fibrillary acidic protein; CREB: cyclic response-element binding protein; cAMP: Cyclic adenosine monophosphate; NF-κB: nuclear factor kappa-B; CaMKII: Ca2+/calmodulin-dependent protein kinase II; iNOS: inducible nitric oxide synthase; mTOR: mammalian target of rapamycin; PKA: protein kinase A; ERK: extracellular regulated protein kinases; JNK: c-Jun N-terminal kinase; AKT: protein kinase B; Nav1.7: Voltage gated sodium ion channel 1.7; Nav1.8:Voltage gated sodium ion channel 1.8; NK-1R: Neurokinin-1 receptor; CD68: Macrophage sialoprotein; TNF-α: Tumor necrosis factor-α; IL-1β:Interleukin-1β; IL-2: Interleukin-2; IL-4: Interleukin-4; IL-5: Interleukin-5; IL-6: Interleukin-6; IL-10: Interleukin-10; IL-13: Interleukin-13; IL-17A: Interleukin-17A; IL-17F: Interleukin-17F; IL-18: Interleukin-18; IL-22: Interleukin-22; S100B:S100 calcium binding protein B; RAGE:Advanced glycosylation end product receptor; COX-2: Cyclooxygenase; Iba-1: Ionized calcium binding adapter molecule 1; NLRP3: NOD-like receptor thermal protein domain associated protein 3; FOXP3:Forkhead box protein P3; ANXA1:annexin1; FPR2/ALX receptor:Formyl peptide receptor 2; ASC: apoptosis-associated speck-like protein containing a CARD; Caspase-1: cysteinyl aspartate specific proteinase-1; Caspase-2: cysteinyl aspartate specific proteinase-2; p38:mitogen-activated protein kinase-p38; eATP: extracellular adenosine-triphosphate; TH-IR neurons: tyrosine hydroxylase immunoreactive neurons; NTPDase-2: nucleotidases; NTPDase-3: nucleotidases; IFN-γ: Interferon γ.
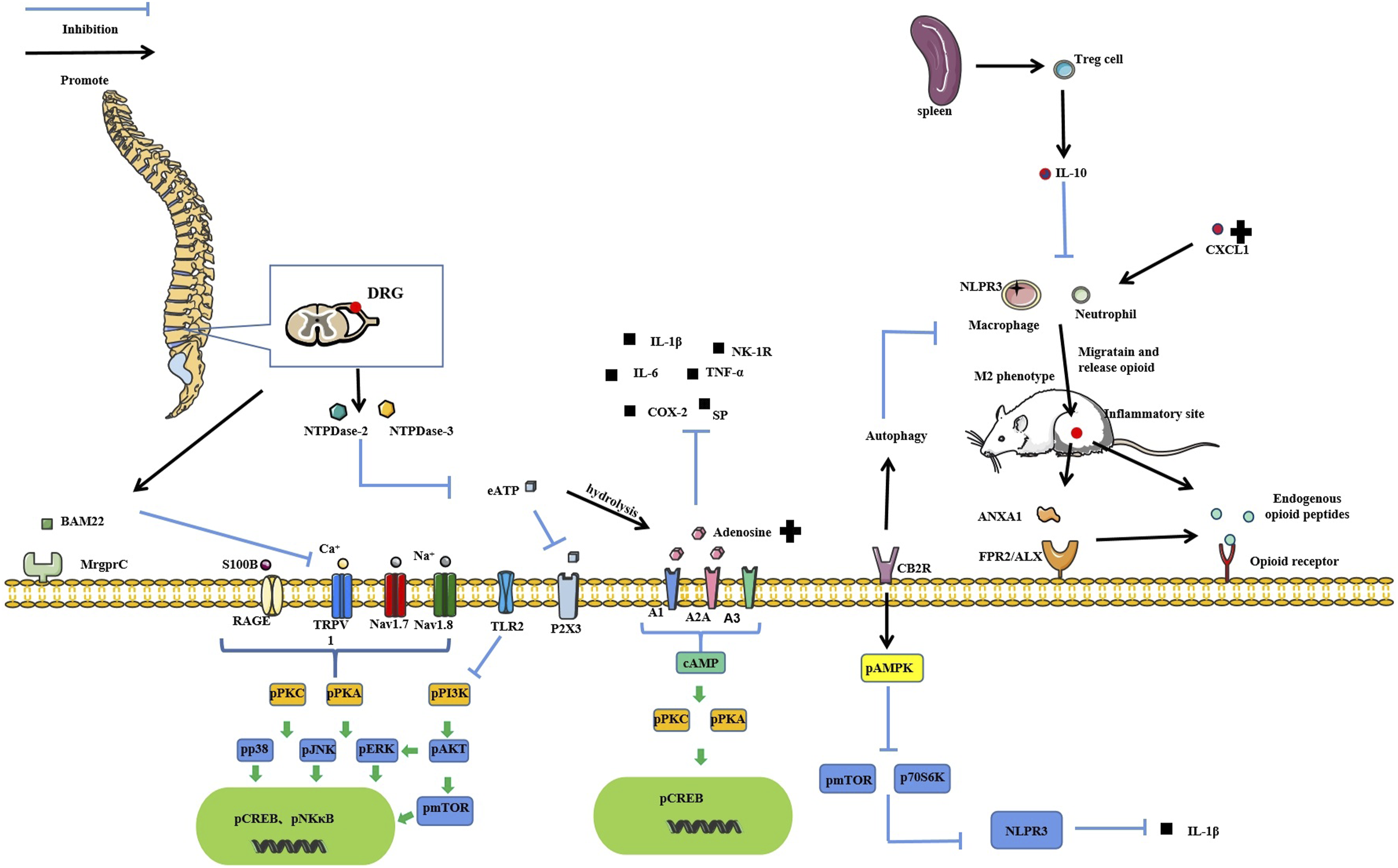
The peripheral mechanisms of acupuncture in treating inflammatory inflammatory pain. DRG: dorsal root ganglia; TRPV1: transient receptor potential vanilloid 1; PKC: protein kinase C; PI3K: phosphatidylinositide 3-kinase; SP: substance P; CB2R: cannabinoid type 2 receptors; CREB: cyclic response-element binding protein; cAMP: Cyclic adenosine monophosphate; NF-κB: nuclear factor kappa-B; mTOR: mammalian target of rapamycin; PKA: protein kinase A; ERK: extracellular regulated protein kinases; JNK: c-Jun N-terminal kinase; AKT: protein kinase B; Nav1.7: Voltage gated sodium ion channel 1.7; Nav1.8:Voltage gated sodium ion channel 1.8; NK-1R: Neurokinin-1 receptor; CD68: Macrophage sialoprotein; TNF-α: Tumor necrosis factor-α; IL-1β:Interleukin-1β; IL-6: Interleukin-6; IL-10: Interleukin-10; S100B:S100 calcium binding protein B; RAGE:Advanced glycosylation end product receptor; COX-2: Cyclooxygenase; Iba-1: Ionized calcium binding adapter molecule 1; NLRP3: pyrin domain containing 3; FOXP3:Forkhead box protein P3; ANXA1:annexin1; FPR2/ALX receptor:Formyl peptide receptor 2; eATP: extracellular adenosine-triphosphate; NTPDase-2: nucleotidases; NTPDase-3: nucleotidases; MrgprC: Mas-related G protein-coupled receptor C; BAM22: bovine adrenal medulla 22 peptide; P70S6K: phosphoprotein 70 ribosomal protein S6 kinase.
Immune cells and neurons
At the peripheral level, inflammatory pain is mainly mediated by inflammatory reactions, which mediates interactions between immune responses and nociceptor pain sensitization. Tissue injury and inflammatory reactions mediate neural immune responses and release the inflammatory medium that stimulates and corresponds to the receptor at the end of nociceptor. Continuous inflammatory stimulation enhances the transmission of nociceptor hyperalgesia pain signals to the dorsal horn of the spinal cord via the dorsal root ganglion. Long term inflammatory pain can produce central sensitization. 25 Neuroimmune interactions are essential for peripheral and central sensitization. Immune cells, including mast cells, macrophages, neutrophils and lymphocytes among others participate in inflammation through different ways. They either release inflammatory mediators to promote inflammatory pain, or release endogenous analgesic substances such as opioid peptides to inhibit inflammatory pain. 26
Physiologically, IL-10, a key anti-inflammatory cytokine produced by activated immune cells, plays a key role in controlling immune responses. 27 Immune cells, especially regulatory T cells (Treg cells), induce IL-10 production to maintain immune homeostasis.28,29 Mechanistically, EA stimulates the activation of Treg cells to induce the production of higher levels of IL-10. The number of macrophages and neutrophils is reduced, thereby suppressing the expressions of pro-inflammatory mediators (IL-1 β, NLRP3 and TNF- α) and ultimately inhibiting inflammation and pain. 30 Moreover, MA at bilateral ST36 was shown to dynamically regulate local cellular communication networks of inflammatory joints. The key cells that mediate the effects of joint acupuncture are monocytes/macrophages, which can inhibit macrophage maturation towards the M1 phenotypes, implying that acupuncture promotes macrophage differentiation at the inflammatory site into the M2 phenotypes, so as to achieve the anti-inflammatory and analgesic effects. 21 Anti-inflammatory cytokines such as TGF- β and IL-10 expression, arginine is metabolized into ornithine and polyamine intermediates to promote healing, which is the characteristic of macrophage polarization to “M2” phenotype. 31 These findings have been verified in other models. For instance, in spinal cord injury rat models, the proportion of M1 macrophages as well as TNF-α, IL-1β and IL-6 expressions were suppressed by EA, while the abundances of IL-10 and M2 macrophages were increased. This increase was confirmed by up-regulated expressions of M2 markers (CD206 and NT-3). The fire needling applied to KOA mice confirmed this process.32–34
Wang et al. 35 found that after trinitrobenzene sulfonic acid (TNBS) stimulation, tyrosine hydroxylase immunoreactive (TH-IR) neurons in the L6 dorsal root ganglion of colitis rats increased and overgrew to sensory neurons, resulting in colitis and secondary hindfoot hypersensitivity. Applications of EA at ST36 and ST37 can reduce colitis and allergic reaction, which may be mediated by inhibiting TH-IR germination in L6 Dorsal root ganglion (DRG). The pivotal roles of sympathetic activities in pathologic pain development and persistence have been documented. Research reported that mechanical pain behaviors was found in rats with ligated L5 spinal nerve and L5 DRG. And both two models lead to neuroinflammatory changes in the sympathetic ganglia, such as macrophage response, activation of satellite glial cells, and increased T cell count, as well as increased excitability of sympathetic neurons. However, local sympathectomy significantly inhibited the pain behavior of the two models. 36 This implies that the sympathetic activity aggravates pain and inflammation in innervated areas. Both EA (at acupoints ST36 and ST37) and guanethidine (a sympathetic blocker) administration relieved the pain and attenuated inflammatory injuries of the distal colon, and secondary hindpaw hyperalgesia by inhibiting tyrosine hydroxylase(TH) expressions in L6 DRG.
Acupuncture induces IL-10 expressions by promoting Treg cell activations or polarization of M1 type macrophages towards the M2 type. In addition, the increase of M2 type macrophages also increases TGF- β to achieve anti-inflammatory effect. Acupuncture also plays an important role in plasticity of neurons for inflammatory pain. It inhibits excess germination of TH-IR to the sympathetic nerve, thereby reducing the secondary hindpaw pain caused by colitis.
The purinergic pathway
Extracellular nucleotides, nucleosides and their receptors, P1 and P2 purinergic receptors (including P1 adenosine receptor, P2 ionic receptor P2X and metabolic receptor P2Y), catabolic enzymes and nucleoside transporters constitute the main regulatory systems in the body. 37 The purinergic pathway plays different roles in pain processes. Studies on this aspect have mainly focused on nucleotide induced cellular signals, adenosine triphosphate (ATP) and P2 ion receptor (P2X3), as well as on adenosine and its receptor.
Adenosine (ADO), a downstream product of adenosine triphosphate (ATP), plays an anti-nociceptive role by binding to A1, A2A and A3 receptors.38,39 Electroacupuncture relieves inflammatory pain by increasing adenosine levels and activating adenosine receptors. Research report, pain is related to SP released by dorsal root ganglion, but adenosine can inhibit its expression. Mechanistically, SP and other inflammatory mediators activate immune cells to release pro-inflammatory factors and induce peripheral nociceptor cells to produce action potentials, resulting in pain. In a previous study, 40 they found that EA stimulation reduced CFA induced pain in rats by inhibiting the expression of inflammatory pain related factors, such as SP, NK-1R, CD68, TNF-α, IL-1β, and IL-6 in rat DRG by increase adenosine level. Direct injection of AG (A1 receptor agonist, N6-cyclopentyladenosine) at ST36 exerts comparable effects. Furthermore, Dorsal Nerve Root Transection (DNRR) and SP receptor antagonist (CP96345) injection blocked EA-induced analgesia in CFA rats, also increased the expressions of SP, NK-1R and their downstream inflammatory molecules. This proves that electroacupuncture relieves inflammatory pain by increasing adenosine levels at inflammatory sites and inhibiting SP secretion. Liao et al. also reported the important role of adenosine in acupuncture analgesia.41,42This effect is achieved by activating local adenosine signal pathways of acupoints and inhibiting PKC-ε and cyclooxygenase-2 (COX-2).
Tang et al. reported that neurons in the peripheral sensory ganglia, which are at the front in reception of nociceptive inputs, are the primary targets of acupuncture regulation. The ATP neurotransmitter system is an important pathway from the periphery to the spinal cord. In this system, extracellular ATP (eATP) and its hydrolysates play different roles in pain transmission by binding their respective receptors. During peripheral neuropathic pain, inflammatory pain or visceral pain, expressions of P2X3 (P2X2/3), P2X7 and P2Y1, 2 and 6 receptors, especially the P2X3 receptor, have been shown to be markedly elevated. However, adenosine, the downstream product of eATP, exerts anti-nociceptive effect by binding to its A1, A2A and A3 receptors.38,39 Acupuncture plays an analgesic role by reversing the elevated expressions of P2X3 in different pain types.15,43 It has been reported that P2X3 is a purinoceptor with ion channel activities in response to eATP. 44 It is mainly expressed in small- and medium-diameter DRG neurons (diameter <35 µm). Studies have confirmed that EA inhibits P2X3 to alleviate inflammatory pain.16,17 Xiang et al. 17 used CFA-induced inflammation pain models and found that protein expressions of P2X3 were elevated in DRG, 100 Hz EA stimulation attenuated CFA-induced mechanical allodynia and reversed P2X3 elevation in L4-6 DRG after CFA injection. Overall, there is conclusive evidence that eATP is an endogenous ligand of P2X3 (P2X2/3), P2X4 and P2X7 receptors, which contribute to pain transmission, and its hydrolysate ADP is an agonist of P2Y1, P2Y12 and P2Y13 receptors, which can also enhance pain perception. 15 Shen et al. reported that unilateral acupuncture can relieve bilateral hyperalgesia, and in this process, eATP metabolism in peripheral sensory nervous systems is simultaneously regulated. 45 In this study, CFA induced bilateral thermal hyperalgesia and ipsilateral allodynia, accompanied by increased mechanical sensitivities and eATP levels of bilateral DRG, but decreased eATP levels of bilateral sciatic nerves.Acupuncture has anti-nociceptive effects on bilateral hind paws and reverses the above changes of eATP in DRG and sciatic nerves. The mRNA expressions of nucleotidases (NTPDase-2 and -3) are also modulated during acupuncture analgesia. Furthermore, regulations of NTPDase-2 and - 3 in the bilateral ganglia and sciatic nerves are inconsistent. In the CFA group, the mRNA expressions of NTPDase-2 in the sciatic nerve were higher than those in the ganglion. In contrast, in the CFA + acupuncture treatment group, the contralateral DRG expressed higher levels of NTPDase-2 than the nerve. Changes in NTPDase-3 expressions were consistent between the two sides. The mRNA expressions of NTPDase-3 in treated nerves were significantly elevated. This explains how acupuncture regulates eATP hydrolysis.
Nociceptive ion channel
Inflammation is a pathological result of tissue damage. The damage releases endogenous biochemical substances, including transient receptor vanilla like member 1 (TRPV1), which can activate nociceptors and transmit signals from the periphery to the neurons and to the brain via dorsal root neurons and the spinal cord. 46 TRPV1 is a non-selective calcium-permeable cation channel, and the first unit of the six members in the TRPV subfamily to be specified. It is mainly expressed in small diameter DRG neurons (areas of less than 200 pixels) of L4-L6 DRG. Due to its low PH (<6.0) and high temperature (>43° C), its significance in inflammation, and its tendency to activate receptors by binding specific molecular sites and increasing membrane depolarization are well known. It can result in a surge of neurotransmitters and further cascade to subsequent neurons or nuclei via the intercellular kinase pathway, protein kinase C, protein kinase A dependent and MAPK pathways.47–50 These irritants are important components of secreted inflammatory mediators that are responsible for the five typical symptoms (redness, swelling, increased heat, pain, and loss of functions). 51
TRPV1 has increased expression and function in DRG of various types of pain models, such as inflammatory pain (CFA model, 52 carrageenan model, capsaicinmodel 53 ), neuropathic pain (CCI model 54 ), visceral pain, 55 among these CFA induced inflammatory expression is further enhanced. This shows that TRPV1 plays a significant role in inflammatory pain. 56 A previous study reported that CFA-induced inflammatory pain in mice activated TRPV1 and triggered the signal pathway to activate the nociceptive Nav channel, simultaneously activating astrocytes and glial cells, and releasing RAGE and its ligand, S100B. The EA and TRPV1 gene deletions were shown to eliminate these changes. 41 Lu et al. found that after CFA injections, the expressions of TRPV1 and associated signaling pathways were elevated. However, these effects were suppressed by EA treatment and TRPV1 gene deletions. 57 The EA treatment (1 mA for 15 min at 2 Hz) at the bilateral ST36 reduced CFA-mediated mechanical hyperalgesia as well as thermal hyperalgesia and reversed the increase in TRPV1 and associated downstream molecules in DRG and the spinal cord (SC). Such as rotein kinases (pPKA, pPI3K, pPKC, pERK, pp38, pJNK, and pAkt), transcription factors (pCREB and pNKκB), nociceptive channels (Nav1. 7 and Nav1. 8). In the TRPV1-/- mice the hyperalgesia and the increase not induced. It has been reported that EA reduces chronic inflammatory pain by down-regulating TRPV1 pathway signaling from peripheral DRG to central SC. The TRPV1 and downstream molecules were also altered in SC, implying that they are involved in central sensitization. These findings have been confirmed in previous studies.16,58 Liu et al. found that Mas-related G protein-coupled receptor C (MrgprC) alleviates CFA-induced thermal hyperalgesia and is involved in EA analgesia by inhibiting the TRPV1 pathway. 59 The MrgprC is an atypical opioid receptor that is activated by opioids. The degradation product of proenkephalin (bovine adrenal medulla 22 peptide (BAM22)) is its unique endogenous ligand. In acute inflammatory pain, TRPV1 is a major downstream target in MrgprC-activated signaling pathways. 60 Moreover, EA and MrgprC regulate chronic inflammatory pain by inhibiting the expressions, translocation and PKC-mediated phosphorylation of TRPV1 in DRG. 59 It was revealed that EA at ST36 alleviated the CFA-induced thermal hyperalgesia. Treatments with EA suppressed the CFA-induced increase in TRPV1 and PKC expressions, TRPV1 translocation and p-TRPV1 levels in DRG and spinal cord dorsal horns (SCDH). And BAM8-22 (a selective agonist of MrgprC) can achieve the same effect. Intrathecal administration of MrgprC small interfering RNA (MrgprC siRNA) blocked the analgesic effects while EA reduced TRPV1 translocations, p-TRPV1 levels and PKC□ in DRG. Therefore, Mrgprc participates in EA analgesia in different ways via the PKC/TRPV1 pathways in DRG and SCDH.
In summary, peripheral sensitization is a key component in pain formation, which involves multiple ion channels (receptors). Among them, P2X3 and TRPV1 are the two most important receptors that are widely expressed in peripheral sensory conduction pathways.17,61,62 In vivo,P2X3 and TRPV1 regulate the occurrence and development of various pathological pain through different ways.52,53,63
The analgesic effects of EA are also associated with inhibition of co-expressions of P2X3 and TRPV1. Chronic inflammatory pain activates the TRPV1 receptor, which promotes the secretion of neurotransmitters such as SP and CGRP among others 64 to activate mast cell-secreted protease and tryptase, 65 thereby activating TRPV1. 66 When the sensitivity of TRPV1 is up-regulated by neurotransmitters, in vivo activation of the P2X3 receptor is also increased, 63 which is evidence of interactions between peripheral TRPV1 and P2X3 under pathological conditions. Liu et al. 67 found that EA could not completely alleviate the physical interactions between TRPV1 and P2X3 in CFA-induced hyperalgesia, thus, the analgesic effects of EA may partly be attributed to inhibition of co-expressions and indirect interactions between TRPV1 and P2X3 in DRG. In this study, the bilateral ST36 and BL60 of CFA rats were treated with 100 Hz EA. Treatment with EA markedly up-regulated the mechanical paw withdrawal thresholds (mPWTs) and inhibited CFA-mediated elevations in co-expressions of TRPV1 and P2X3 in L4-L5 DRG as well as the spontaneous pain behavior (SPB) induced by the increase. They also found that in CFA and SNI rats treated with electroacupuncture (100 Hz or 2 Hz) for 3 days, enhanced effects of the TRPV1 agonist on SPB induced by P2X3 agonist were weakened, which may be attributed to indirect interactions between TRPV1 and P2X3.
Cannabinoid receptors
Cannabinoid receptors belong to the G-protein coupled receptor (GPCR) superfamily with two major subtypes: cannabinoid type 1 receptors (CB1Rs) and cannabinoid type 2 receptors (CB2Rs), which couple to inhibitory Gi/o proteins. 68 The CB1Rs are mainly expressed in the central nervous system (CNS) and the peripheral nervous system (PNS) while the CB2Rs are mainly distributed in the peripheral nervous system, immune cells (e.g., lymphocytes, neutrophils, and macrophages), hematopoietic cells, myocardial cells, epithelial cells, fibroblasts, osteoblasts, and skin keratinocytes. 69 In lower levels of the CNS, the CB2Rs are primarily found on the microglia, but have also been identified on some neurons (e.g., in the hippocampus, cortex, and substantia nigra).70,71 The CB2Rs-associated signal transductions are highly involved in anti-inflammatory actions, 72 and without the negative effects observed with CB1Rs activation (e.g., cognitive impairment, catalepsy, as well as the negative impacts on learning and memory) 73 Activation of CB2Rs enhances IL-10 expressions and suppresses the expressions of pro-inflammatory mediators such as TNF-α, CCL2 and nitric oxide. 72 Therefore, CB2Rs is an important target for analgesia research.
In inflammatory pain, acupuncture affects the NLRP3 inflammasome via CB2Rs to achieve the therapeutic effects. The NLRP3 (NLR family, Pyrin domain containing 3, also known as NALP3 or Cryopyrin) inflammasome is an intra-cytoplasmic protein complex. It is composed by NLRP3, ASC (apoptosis-associated speck-like protein containing a CARD) and pro-Caspase-1. It can accelerate the maturation of inflammatory cytokines and is closely associated with inflammatory pain. In a previous study, 74 EA relieved inflammatory pain by suppressing activation of the NLRP3 inflammasome through cannabinoid CB2Rss, mainly in the macrophage system of inflammatory tissues, and delaying the maturation of IL-1β. The CB2Rs is an important regulator of the NLRP3 inflammasome in macrophages. In another study, 75 activation of CB2Rs alleviated colitis by enhancing autophagy and inhibiting NLRP3 inflammasome activation in mice macrophages. This is because activated CB2Rs increases the levels of phosphorylated AMPK and reduces the levels of phosphorylated mTOR and P70S6K, promoting the inhibition of macrophage autophagy and NLRP3 inflammasome. Applications of EA at “Dachangshu” (BL25) were also shown to alleviate pain and inflammation in colitis mice models via CB2R activation of macrophages and inhibition IL-1β iNOS expressions. 76 In a knee osteoarthritis (KOA) model established by injection of iodophor monosodium into the knee, EA at “Neixiyan” (Ex-LE4) and “Dubi” (ST35) increased the mechanical threshold, thermal latency, and weight borne after KOA modeling. Moreover, EA enhanced CB2Rs expressions in fibroblasts and suppressed IL-1β expressions in the menisci. However, the EA-associated effects were eliminated by CB2Rs-knockout. Thus, EA inhibited chronic pain in KOA mice models by activating CB2Rs to suppress IL-1 β expressions. 77
Endogenous opioid peptides system
It has been documented that EA can drive monocytes/macrophages containing opioid peptides to migrate to inflammatory sites, and locally release opioid peptides to bind receptors on peripheral sensory neurons, thereby exerting peripheral analgesic effects.78,79 Endogenous opioid peptides play an indispensable role in acupuncture analgesia. 41 Some analgesic mechanisms are more or less associated with them.
Acute gout arthritis is one of the most painful inflammatory conditions. In a previous study, 22 rat models of acute gout arthritis were established via injection of viamonosodium urate (MSU) into ankle joints. Treatment of the rats with EA effectively alleviated pain responses as well as inflammation and significantly upregulated the expressions of the endogenous opioid receptor agonist (β-endorphin) in local ankle skin tissues. In addition, EA achieved analgesic effects that are equivalent to those of the widely used NSAID (indomethacin), and these effects were peripheral k- and μ-opioid receptor-mediated.
The mechanisms underlying the analgesic effects of acupuncture and recombinant chemokine ligand 1 (CXCL1) may both be associated with a peripheral opioid-mediated pathway. 20 Chemokines are a family of small secreted cytokines that are closely related to inflammatory pain. 80 They can regulate inflammatory pain in different peripheral and central regions.81,82 As a member of the CXC chemokine family, CXCL1 is a well-known neutrophil attractant. During inflammation, neutrophils infiltrate infected sites and inhibit pain through opioid peptides. 83 Moreover, CXCL1 induces the release of endogenous opioid peptides in inflammation. 84 In a previous study on rheumatoid arthritis, Xu et al. used multiple enzyme-linked immunosorbent assays (ELISAs) to determine the cytokines/chemokines that are involved in anti-injury effects of acupuncture and moxibustion. Only CXCL1 was found to be markedly elevated at multiple time points as acupuncture effectively alleviated inflammatory pain. 20 This finding was later confirmed.20,85 They further investigated the role of CXCL1 in EA analgesia and revealed that serum CXCL1 has analgesic effects and may be a key anti-nociceptive medium in acupuncture-mediated treatment of inflammatory pain. The analgesic effects of acupuncture and recombinant CXCL1 were alleviated by an i.pl. injection of a non-selective opioid receptor antagonist (naloxone). Therefore, acupuncture may induce neutrophils to infiltrate the infection sites and release opioids by up-regulating CXCL1 expressions to achieve analgesia.
The pro-resolving mechanism is a recently discovered endogenous process that regulates inflammation. Persistence of inflammation has been attributed to the inability of organisms to produce enough pro-resolving mediators, or the secreted mediators cannot bind their receptors. 86 In a previous study, 87 the antihyperalgesic effects of EA on inflammatory pain were found to involve annexin1 (ANXA1) and the Formyl peptide receptor 2/ALX(FPR2/ALX) pro-resolution pathway. These effects were induced by activation of FPR2/ALX receptors and crosstalk communication with opioid systems. Activation of human neutrophils by ANXA1 enhances the secretion of endogenous opioids such as Met-enkephalin. 88 Activation of FPR2/ALX is involved in molecular pathways that are associated with tonic opioid release from neutrophils in inflammatory pain. 89 ANXA1 activates the FPR2/ALX receptor precede release of opioids induced by EA. This shows that EA promotes ANXA1 secretion, activates FPR2/ALX receptor and interacts with it, then induces the release of opioid peptides and binds with their receptors to establish analgesic effects.
The above findings show that acupuncture regulates pain through multiple complex systems in the periphery, involving interactions between immune cells and nociceptors, the peripheral purinergic pathway, the endogenous cannabinoid system, opioid peptide systems and nociceptive ion channels. Moreover, immune cells play an indispensable role in peripheral analgesic mechanisms of acupuncture.By expressing various pro-inflammatory substances (such as SP, IL-1β, and NLRP3 among others), they are involved in pain generation, and they also secrete anti-inflammatory and analgesic substances (such as IL-10 and opioid peptides, etc.) to relieve inflammation and pain. Therefore, immune cells are involved in almost all peripheral analgesic mechanisms of acupuncture. From the perspective of the purinergic pathway, acupuncture can directly regulate eATP levels in peripheral nerves, inhibit the expressions of its receptor (P2X3). Through nucleotidase to promote eATP hydrolysis, and prevent the combination of eATP and P2X3 to relieve pain. Acupuncture can also increase the local levels of adenosine, and activate its receptors to inhibit the DRG-secreted SP. Thus, SP cannot activate immune cells to release pro-inflammatory factors, achieving the purpose of analgesia.
From a cannabinoid receptor perspective, CB2R is widely distributed in various immune cells. Acupuncture activates CB2Rs in macrophages to enhance autophagy, inhibit NLRP3 inflammasome activation and delay IL-1β maturation to regulate pain. The AMPK-mTOR-P70S6K signaling pathway is involved in this process. From an endogenous opioid peptide system perspective, mononuclear cells/macrophages containing opioid peptides are the basis of this system, and acupuncture can promote these cells to migrate to inflammatory sites and release opioid peptides to bind its receptors. Moreover, as a neutrophil attractant, CXCL1 may participate in analgesia through the peripheral opioid peptide system. In the recently discovered pro-resolving mechanism, ANAX1 and FPR2/ALX are involved in the process by which neutrophils secrete opioids.
The relieving effect of acupuncture on inflammatory pain is not a function of certain immune cells but involves interactions between various immune cells. For instance, acupuncture promotes Treg cell activation, thereby elevating IL-10 expressions while inhibiting the number of macrophages and neutrophils. In addition, acupuncture improves pain by regulating the expressions and secretion of various substances in immune cells as well as by dynamically regulating the local cellular communication network of inflammatory sites. It mainly inhibits M1 macrophages and promotes macrophage polarization towards the M2 phenotype, and improves expressions of anti-inflammatory factors (TGF- β and IL-10). Moreover, it enhances the metabolism of arginine into ornithine and polyamine intermediates to promote healing.
In addition, TPRV1 and its related molecules (e.g. downstream molecules pPI3K, pAkt, pmTOR, members of MAPK subfamily pERK, pp38, pJNK, transcription factors pCREB, PNKκB and nociceptive channels Nav1.7 and Nav1.8) were inhibited during acupuncture analgesia. And the expression, translocation and PKC mediated phosphorylation of TPRV1were also inhibited by acupuncture through MrgprC. What can further prove the complexity of acupuncture analgesia mechanism is that acupuncture can achieve the goal by inhibiting the co expression and indirect interaction between TRPV1 and P2X3.
Central mechanisms
Central mechanism of acupuncture in treating inflammatory pain.
SC: spinal cord; SCDH: spinal cord dorsal horn; PFC: prefrontal cortex; PAG: periaqueductal gray; ACC: anterior cingulate cortex; SSC: somatosensory cortex; TG: trigeminal ganglion; TNC: trigeminal nucleus caudalis;TRPV1: transient receptor potential vanilloid 1; PKC: protein kinase C; PI3K: phosphatidylinositide 3-kinase; GFAP: glial fibrillary acidic protein; CREB: cyclic response-element binding protein; cAMP: Cyclic adenosine monophosphate; NF-κB: nuclear factor kappa-B; CaMKII: Ca2+/calmodulin-dependent protein kinase II;GluR1:glutamate receptor 1; GluR2: glutamate receptor 2; ICA69: Islet cells Antigen 69; PICK1: protein interacting with Cα kinase 1; NRG1: Neuromodulin 1; ErbB4: Receptor protein tyrosine kinase ErbB4; S100B:S100 calcium binding protein B; RAGE:Advanced glycosylation end product receptor; mTOR: mammalian target of rapamycin; PKA: protein kinase A; ERK: extracellular regulated protein kinases; JNK: c-Jun N-terminal kinase; AKT: protein kinase B; Nav1.7: Voltage gated sodium ion channel 1.7; Nav1.8:Voltage gated sodium ion channel 1.8;SP: substance P; IBa1: ionized calcium binding adaptor molecule 1;GRK2: G protein coupled receptor kinase 2; p38MAPK: p38 mitogen-activated protein kinases; TNF-α: Tumor necrosis factor-α; IL-4:Interleukin-4; IL-6:Interleukin-6; RIP3:receptor-interactingprotein3; NLRP3: NOD-like receptor thermal protein domain associated protein 3.
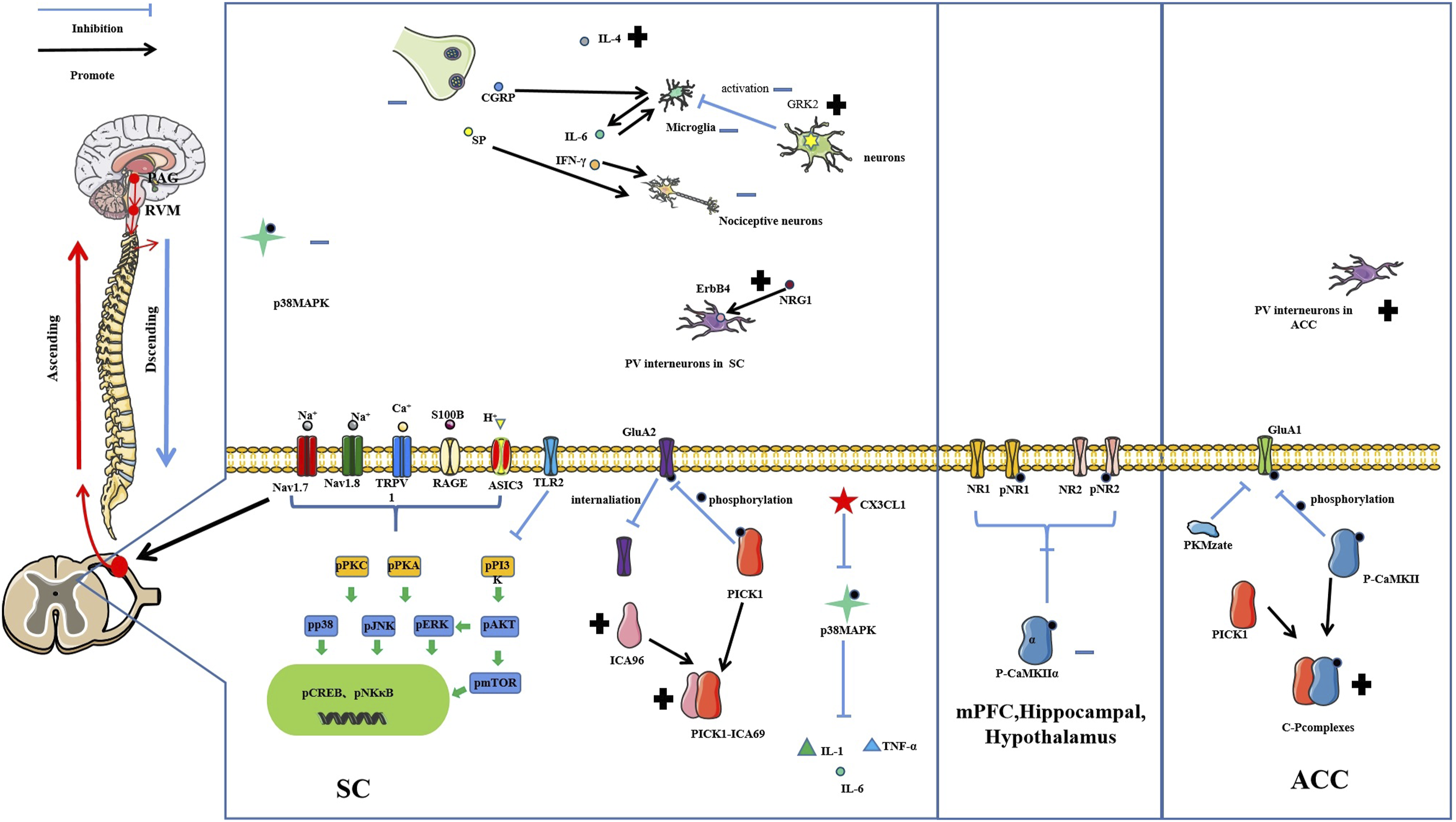
The central mechanisms of acupuncture in treating inflammatory inflammatory pain. SC: spinal cord; PFC: prefrontal cortex; PAG: periaqueductal gray; RVM: rostral ventromedial medulla; ACC: anterior cingulate cortex; TRPV1: transient receptor potential vanilloid 1; PKC: protein kinase C; PI3K: phosphatidylinositide 3-kinase; CREB: cyclic response-element binding protein; cAMP: Cyclic adenosine monophosphate; NF-κB: nuclear factor kappa-B; mTOR: mammalian target of rapamycin; PKA: protein kinase A; ERK: extracellular regulated protein kinases; JNK: c-Jun N-terminal kinase; AKT: protein kinase B; Nav1.7: Voltage gated sodium ion channel 1.7; Nav1.8: Voltage gated sodium ion channel 1.8; ASIC3: acid sensing ion channel subunit 3; GluA1:glutamate receptor 1; GluA2: glutamate receptor 2; ICA69: Islet cells Antigen 69; PICK1: protein interacting with Cα kinase 1; NRG1: Neuromodulin 1; ErbB4: Receptor protein tyrosine kinase ErbB4; TLR2:Toll like receptor 2; GRK2: G protein coupled receptor kinase 2; p38MAPK: p38 mitogen-activated protein kinases; AIC3:acid-sensing ion channel 3; NR1: N-methyl-D-aspartate receptors NR1; NR2: N-methyl-D-aspartate receptors NR2; IFN-γ: Interferon γ; IL-4: Interleukin-4; IL-6: Interleukin-6.
Nociceptive ion channel
The TRPV1 distributed in the periphery 61 and in the central nervous system. 100 It’s where they participate in regulation of inflammatory pain and harmful stimuli. 111 Mechanistically, EA reduces pain by downregulating TRPV1 and downstream molecules supporting central sensitization. Moreover, EA regulates chronic inflammatory pain by inhibiting the expressions, translocations and PKC-mediated phosphorylation of TRPV1 in SCDH via MrgprC. 58 It has been documented, 100 that after CFA injection, expressions of TRPV1 and related molecules, such as downstream molecules (pPI3K, pAkt and pmTOR), members of the MAPK subfamily (pERK, pp3, pJNK), transcription factors (pCREB, PNKκB), nociceptive channels (Nav1. 7 and Nav1. 8), were elevated in the PFC and hypothalamus, but decreased in the PAG region. Treatment with EA alleviated thermal and mechanical hyperalgesia in mice by reversing this trend. Moreover, EA on Hegu (LI4) proved that electroacupuncture at the distal point of the pain site has the same anti-inflammatory effects as that at the local point. This proves that TRPV1 and related molecules are involved in inflammatory pain, and that distal EA reduces inflammatory pain by regulating TRPV1 and related molecules in PFC, hypothalamus and PAG. In another study, 101 EA resulted in inflammatory pain regulation by down regulating the protein levels of TRPV1, ASIC3, Nav1. 7, Nav1. 8, RAGE, S100B, pPI3K, pmTOR, pAkt, pERK, pPKAIIα, pPKCε, pCREB and pNFkB in V, VIa and VII of mice cerebellar lobules. These results indicate that EA at ST36 can treat CFA-induced inflammatory pain by regulating TRPV1 and related molecular pathways in cerebellar lobules V, VIa and VII.
TPRV1 is involved in inflammatory pain sensations at the brain level and also affects pain-related emotions. Lin et al. reported that EA can relieve pain and control depression through TPRV1 and related factors. 112 Most TRPV1 proteins and related molecules were significantly reduced in mPFC, hippocampus and PAG of chronic pain and depression comorbidity (CPDC) mice models, but increased in the amygdala of mice models. These trends were reversed by EA. Moreover, EA suppressed the expressions of pro-inflammatory cytokines and mediators. The therapeutic effects of acupuncture on both depressive and chronic pain symptoms were alleviated in part through their anti-inflammatory effects.92,93
Glutamate receptors
Glutamate receptors can be grouped into N-methyl-D-aspartate receptors (NMDAR) and α-Amino-3-hydroxy-5-methyl-4-isoxazole propionic acid receptors (AMPAR). The NMDAR contains five subunits, NR1 and NR2 (A-D). The AMPAR receptor is a heteropolymer that is assembled by several subunits, including GluA1, GluA2, GluA3 and GluA4. These receptors play important roles in pain regulation.
When CFA was injected into the hind paw of mice, it promoted ser880 phosphorylation in GluA2 and subsequent internalization of GluA2, resulting in elevated AMPAR on the neuronal membrane. Ultimately, the improved permeability promotes Ca2+ influx, leading to inflammatory pain hypersensitivity. 113 Moreover, interactions of spinal proteins with C kinase 1 (PICK1) may maintain inflammatory pain by promoting PKCa-mediated GluA2 phosphorylation at Ser880 and GluA2 internalization in SCDH. However, CFA did not cause this phenomenon in PICK1 knockout mice. Thus, PICK1 plays an important role in pain maintenance. It has been reported 102 that EA promotes ICA69 expressions in SCDH and regulates GluA2 phosphorylation through PICK1, so as to achieve the anti-hyperalgesic effects on CFA-induced inflammatory pain. Prior to EA, ICA69 protein levels in SCDH were relatively low, and the binding with PICK1 proteins in the cytoplasm were not abundant. Therefore, the intermediate PICK1 protein can be freely transported to the cell membrane, resulting in GluA2 phosphorylation. Once GluR2 is phosphorylated to GluA2-p, endogenous GluA2 is transferred from the cell membrane to the cytoplasm. The AMPA receptors lacking GluA2 can increase the Ca2+ influx, leading to cellular inflammation and hyperalgesia. Besides, EA treatment elevated ICA69 protein expressions and formed a rich complex with PICK1, so that PICK1 was retained in the cytoplasm. This prevented GluR2 phosphorylation so that AMPARs could maintain the impermeability of cell membranes to Ca2+, and ultimately produce anti-inflammatory and anti-pain effects.
The the anterior cingulate cortex (ACC) is a major brain area for pain and pain-related emotions. 114 As a multifunctional serine/threonine kinase, Ca2+/calmodulin-dependent protein kinase II (CaMKII) is widely distributed in the central and peripheral nervous systems, its phosphorylated protein (P-CaMKII) is involved in memory, emotion and pain regulation in ACC. Treatment with EA alleviates inflammatory pain by inhibiting CaMKII-GluA1 phosphorylation in ACC. 94 In the study, EA was shown to inhibit P-CaMKII and GluA1 expressions in ACC. P-CaMKII involved in EA analgesia by forming pCaMKII-PICK1(C-P) complexes. That is because the amounts of C-P complexes were increased by EA, and achieved the analgesic effects by the inhibitory effects of C-P complexes on GluA1 phosphorylation. However, as earlier mentioned, at the SC level, the analgesic effects are achieved by inhibiting GluA2 phosphorylation through ICA69 and PICK1 complexes. 102 This shows that even the same molecule can form different complexes at different levels, affecting different receptors.
As an isoform of protein kinase C with persistent activities, Protein kinase Mzeta (PKMzeta) is involved in neural plasticity that is associated with pain and anxiety disorders. 115 It has been found 90 that EA inhibits the activation of PKMzeta in ACC, improves paw withdrawal thresholds (PWT) and reduces pain related anxiety-like behaviors. In this study, EA at ST36 reduced CFA-induced allodynia and anxiety-like behaviors. Moreover, EA reversed the increase in p-PKMzeta (T560) and GluA1 protein expressions in rat ACC after CFA injection. Injection of zeta-interacting protein (ZIPan inhibitor of PKMzeta) into rats ACC resulted in comparable effects. Therefore, the PKCzeta/PKMzeta-GluA1 pathway is involved in EA regulation of pain and pain-induced anxiety-like behaviors.
In addition, in a study on chronic pain and depression comorbidity, 92 it was found that NMDARs (pNR1, NR1, pNR2B, NR2B) and calcium/calmodulin-dependent protein kinase type II alpha isoforms (pCaMKIIα) in the prefrontal cortex (PFC), hippocampus and hypothalamus of mice were markedly suppressed after CFA injection. EA reversed this decrease and reduced the depressive behaviors. This proves that EA treats the comorbidities by upregulating NMDA signaling pathways in PFC, hippocampus and hypothalamus.
Microglia
Glial cells in the CNS are grouped into three: astrocytes, microglia and oligodendrocytes. 116 They play important roles in pain generation and maintenance. For instance, microglia can secrete TNF-α, 117 while astrocytes can secrete IL-1β. 118 Recent studies have majorly focused on microglia, therefore, we elucidated on the roles of microglia in acupuncture analgesia. Microglia are distributed in the spinal cord and brain and play a role in local environment maintenance. 119 After pathophysiological stimulation, the microglia become activated and their morphology changes, the number of cells increases, and gene expressions of proinflammatory cytokines such as IL-6 and IL-1β in the spinal cord also been upregulated. 120 These proinflammatory cytokines act as mediators to participate in the crosstalk process with nociceptive neurons, and ultimately promote pain and maintenance. 121 In formalin-induced acute inflammatory pain models, 108 pre-EA (rats received EA treatment at the same side Zusanli (ST36) and Zhongwei (BL40) acupoints (2 Hz, 1 mA) for 30 min before formalin injection) significantly alleviated the pain. This is because, pre-EA significantly suppresses the expressions of IFN- γ, IL-6, SP and CGRP as well as the activation of microglia and neurons, thereby interrupting the crosstalk between the microglia and spinal sensory neurons. In addition, it suppressed spinal neuroinflammation and central nociceptive sensitization. Even though both pre-EA and post-EA (rats received the same treatment immediately after formalin injection) significant suppressed the formalin-induced increase of Iba-1 and c-fos, pro-inflammatory cytokines (IL-6 and IFN- γ), pain neurotransmitters (SP and CGRP). And the decrease of anti-inflammatory cytokine IL-4 were reversed. However, regulation of pre-EA on these factors is better than those of post-EA.
Toll like receptors (TLRs) are an evolutionarily conserved pattern recognition receptors that are widely expressed in neurons, microglia and astrocytes of the CNS. They can regulate innate and adaptive immunity against exogenous or endogenous dangerous ligands and risk-related molecular patterns after tissue injury.122–125There are 10 different subtypes and different specific ligands of TLR. 126 Activation of TLR in glial cells can produce a wide range of proinflammatory cytokines, including IL-1β, IL-6 and TNF-α. Moreover, its expressions in the central nervous system contributes to glial cell activation. 125 In CFA mice models, it was confirmed that, 95 TLR2 and downstream molecule (pPI3K, pAkt, pmTOR), MAPK subfamiliy members(pERK, pp38, pJNK), transcription factors(pCREB, pNKκB)nociceptive channels (Nav1.7 and Nav1.8) were significantly increased in DRG, SC and thalamus. Treatment with EA suppressed CFA-induced mechanical and thermal hyperalgesia and downregulated the increased signals of TLR2 pathway members from peripheral DRG to the CNS as well. Treatment with Cu-cpt22 (TLR2 antagonist) achieved the same effects. Thus, EA can relieve inflammatory pain by downregulating the increased signaling of TLR2 pathway-related molecules, from peripheral DRG to the CNS.
The G protein coupled receptor kinase 2 (GRK2) is a serine/threonine kinase that can protect cells from excess stimulation. 127 Inflammatory pain in rodent models has been associated with reduced GRK2 expressions in the spinal cord. 128 Electroacupuncture therapy increasing the abundance of GRK2 neuron to restore GRK2 expressions, then inhibiting microglial activation and suppressing neuroinflammation in the spinal cord, thereby promoting inflammatory pain relief. 106
Signaling molecules
Recent studies have majorly focused on p38 mitogen-activated protein kinases (p38MAPK), therefore, we elucidated on the significance of p38MAPK in acupuncture treatment of inflammatory pain. Activation and phosphorylation of p38MAPK enhances the expressions of transcription factors (TNF-α and ATF2) as well as inflammatory mediators (IL-1 and COX-2) in the spinal cord, thereby promoting pain.129,130 It has been revealed, 97 that p38MAPK expressions were elevated in SCDH of CFA-injected rats. In addition, EA suppressed CFA-induced mechanical and thermal hyperalgesia and edema by reversing the increase in p38MAPK expressions in SCDH. Ni et al. 131 found that chronic constrictive nerve injury leads to mechanical hyperalgesia and increases the expressions of phosphorylated p38MAPK (p-p38MAPK) in the ventrolateral periaqueductal gray (vlPAG). In another study, 97 EA treatment can significantly increase PWT and relieve paw edema. In addition, EA reversed the increase of p-p38MAPK-IR cells in PAG and RVM after CFA injection. These suggested that EA could disrupt descending facilitation in PAG, RVM, and SCDH and exhibits anti-hyperalgesic effect by inhibiting p38MAPK activation in descending facilitatory pathway.
The p38MAPK also plays important roles upstream or downstream of different pathways in SC to mediate the analgesic effects of EA. Li et al. 82 proved that p38MAPK phosphorylation and release of cytokines (IL-1β, IL-6, TNF-α) was increased in the spinal cord after CFA injection. Treatment with EA alleviated CFA-induced hyperalgesia by inhibiting CX3CL1 and p38MAPK expressions. Furthermore, this analgesic effect was CX3CL1 inhibition dependent. This shows that in the spinal cord, EA therapy plays an analgesic role by suppressing signal transduction of the CX3CL1-p38MAPK cytokine axis and the subsequent cytokine release. Jin et al. found that in pain sensitive priming (HP) models established by carrageenan and PEG2 (the transition model from acute pain to chronic pain), EA alleviated the transition from acute pain to chronic pain via the p38 MAPK/TNF-α signaling pathway in SCDH. 109 They found that EA at ST36 and Bl60 reduced the mechanical claw withdrawal threshold and p38 MAPK/TNF-α levels in the spinal dorsal horn of HP rats. Administration of U-46619 (selective p38 MAPK agonist) attenuated this effect. This shows that EA relieves pain and inhibits the transformation from acute inflammatory pain to chronic inflammatory pain. This effect is achieved by inhibiting the p38 MAPK/TNF-α signaling pathway in the dorsal horn of the spinal cord.
GABAergic interneuron
Although the abundance of GABAergic interneurons in the brain is limited, they are particularly important in controlling inhibition. 132 Moreover, GABAergic interneurons in the CNS are indispensable in pain regulation. GABAergic interneurons in the ACC and basolateral amygdala (BLA) are associated with chronic inflammation.133,134 Transplantation of GABAergic cells into ACC reduced neuropathic pain and pain aversion, 135 implying that GABAergic interneurons in ACC are crucial in regulation of pain and associated emotions. Parvalbumin (PV) is a subtype of GABAergic interneurons. The PV neurons in the hippocampus and amygdala are closely associated with negative emotions such as anxiety or depression.136,137 The PV-positive cells were found to be decreased in neuropathic pain mice models, proving its association with pain. 138 Shao et al. 99 found that EA ameliorates chronic inflammatory pain and pain-related anxiety by upregulating PV interneurons in ACC after CFA injection. Moreover, they established that EA for ST36 and BL60 reduced mechanical pain sensitivity as well as anxiety-like behaviors and increased PV interneurons in ACC, that had been reduced by CFA injection. The same effects were found in rats with chemogenetic activated PV. The effects of EA were impaired by chemogenetic inhibition of PV interneurons in ACC.
Neuromodulin 1 (NRG1) belongs to the epidermal growth factor family and can produce six different types of proteins (I-VI). 139 Receptor protein tyrosine kinase (ErbB4) is a receptor tyrosine kinase of the EGF receptor family that binds NRG1 and is therefore, activated by NRG1. ErbB4 is expressed in GABAergic interneurons and is indispensable in GABAergic signal transduction and neuronal plasticity. 140 NRG1-ErbB4 signaling is involved in GABAergic conduction in the CNS. 141 Some studies showed that EA can increase NRG1 expressions in muscles, proving that there is a potential relationship between EA and NRG1.142,143 Wan et al. 98 found that EA alleviated pain hypersensitivity because it increased NRG1 expressions in SC of CFA-induced inflammatory pain mice models. This analgesic effect was partly mediated by NRG1-ErbB4 signaling. In addition, co-applications of EA and NRG1 could not enhance EA-mediated analgesia. In mice injected with CFA or carrageenan, inhibition and neutralization of the ErbB4 receptor reduced the mechanical threshold, but did not affect the heat latency. At the same time, they found a decrease in baseline levels of the mechanical threshold in two kinds of mice with ErbB4 knockout from PV+ neurons, which proved that PV+ interneurons are very important in maintaining normal mechanical sensation and analgesic effects of NRG1-mediated mechanical hyperalgesia.
In conclusion, among the different analgesic pathways in the CNS, TPRV1 has attracted much attention in recent years due to its wide distributions in peripheral and central nervous systems. As earlier mentioned, acupuncture inhibits the expressions of TRPV1 and downstream factors in SC, PFC hypothalamus can cerebellar lobules, but increased in the PAG. Its translocation and PKC-mediated phosphorylation can also be inhibited in SCDH through MrgprC. Thus, acupuncture can relieve pain and related depression by regulating TPRV1 expressions in different brain regions.
Glutamate receptors are widely distributed in neurons and are indispensable in regulation of inflammatory pain during acupuncture. In the CNS, acupuncture regulates pain and its related emotions via different pathways that are mediated by different glutamate receptors. These mechanisms involve inhibition of glutamate receptor phosphorylation. Even the same factors can inhibit the phosphorylation of different receptors by forming different complexes at different levels. For instance, PICK1 forms a complex with ICA96 in SC to inhibit GluA2 phosphorylation, while in ACC, it forms a C-P complex with P-CaMKII to inhibit GluA1 phosphorylation-mediated acupuncture analgesia. In addition, acupuncture inhibited the PKCzeta/PKMzeta-GluR1 pathway in ACC and up-regulated the NMDAR pathway in PFC, hippocampus and hypothalamus, relieving inflammatory pain-related adverse emotions.
From an intermediate neuron perspective, acupuncture relieves pain by up regulating GABAergic intermediate neurons in ACC and also relieves pain-related anxiety as well as depression. In addition, EA increased NRG1 expressions in SC and activated ErbB4 analgesia in GABAergic interneurons in inflammatory pain mice models.
The P38MAPK is activated and phosphorylated in SCDH of CFA rats, and increases the production and expressions of transcription factors as well as inflammatory mediators in the spinal cord. This process was also found in PAG. Moreover, EA can destroy the descending facilitation of PAG, RVM as well as SCDH, and exerts anti-hyperalgesic effects by inhibiting the activation of p38MAPK and its downstream factors (TNF- α, ATF2, IL-1, and COX-2) in the descending facilitation pathway. This effect is probably mediated by the CX3CL1-p38MAPK/TNF-α pathway in SC. Acupuncture can also treat inflammatory pain by inhibiting microglial activation and TLR2 expressions in the central nervous system. The microglia in the CNS and their associated proinflammatory cytokines are also regulated during acupuncture analgesia, and in this process, they interact with each other. The TLR2 and its downstream molecules, which are widely distributed in glial cells, also play an important role in this process. Activation of TLR2 promotes proinflammatory cytokine production and glial cell activation. This effect can also be reflected in microglia. In addition, acupuncture can inhibit microglial activation, reduce neuro-inflammation and relieve pain by restoring the number of GRK2 in neurons.
In summary, the mechanisms of acupuncture in central analgesia are very complex, and they not only involve different mechanisms at different levels of the spinal cord and brain, but also relates to interactions between ascending pathway of the spinothalamic tract and the descending pathway of the brainstem. The rich neuronal types (such as excitatory neurons, and inhibitory interneurons among others) and glial cells in the CNS participate in pain regulation via production of neurotransmitters and a series of inflammatory factors. The roles of various signaling pathways are assignable. Pain is often accompanied by different levels of pain emotions, and acupuncture can relieve pain and these bad emotions at the same time, which is also a feature of the mechanisms involved in acupuncture analgesia at the brain level.
Discussion and prospects
Pain is a distressing experience associated with actual or potential tissue damage that has sensory, emotional, cognitive and social components. As a symptom, inflammatory pain often appears alone or with some chronic diseases in clinical practice. It seriously affects the quality of life for individuals, and causes damage to physical and mental health. Its development is mostly caused by local tissue damage due to chemical drug stimulation, mechanical stimulation, thermal stimulation, trauma, infection or postoperation, which cause inflammatory reactions. Local tissues secrete various neurotransmitters and mediators, and causes peripheral as well as central sensitization, resulting in pain. As a treatment approach for pain, acupuncture has been gradually accepted by people in recent years Studies on acupuncture analgesia in various types of pain have been increasing. Different animal models have also been used to study the therapeutic effects of acupuncture on these different pain types. In this review, we summarized recent evidence on mechanisms of acupuncture in treatment of inflammatory pain. The analgesic and anti-inflammatory effects of acupuncture on inflammatory pain are complex processes involving multi-levels, multi-channels and multi-targets. They involve the peripheral and central levels, opioid peptide system, cannabinoid system, Purinergic signal, monocyte macrophage system and glial cells distributed therein. Various inflammatory factors and mediators are also closely associated with inflammatory pain. It is not difficult to found that in recent years, there are more and more studies focused on the mechanism of the central system, especially the brain, not only for pain itself, but also for pain related adverse emotions.
In studies of various acupuncture analgesia-related factors, opioid peptides, ephedrine and its receptors, as well as Purinergic signal are still the research hotspots. The TRPV1 and its downstream molecules have attracted much attention in recent years due to their wide distributions in peripheral and central nervous systems. Apart from the above widely investigated molecules, new mechanisms are also being discovered and studied, including the role of pro-resolving mediators in inflammatory pain. The apelin/APJ system has also been recently proven to be related to acupuncture. These studies are of great significance as they inform clinical drug research. In addition, the current research is not only the study of a single pathway, but also began to pay attention to the correlation and interaction between the two pathways (such as TRPV1 and P2X3). New technologies such as genetics and proteomics have also been used for related research, which may reveal the mechanism of acupuncture in treating inflammatory pain from a deeper place.
Although the mechanism of acupuncture in treating inflammatory pain has been studied to some extent, there are still some deficiencies. First, acupuncture treatment often targets ST36 acupoints in inflammatory pain, with other acupoints such as BL60, SP6, and GB34 also selected for experimentation. While some studies have confirmed the specificity of acupoint selection in the analgesic effect of acupuncture, there is limited research exploring the therapeutic differences and mechanisms between various acupoints. This may present a potential area for future investigation. Second, studies on adenosine and its receptors in the spinal cord and brain are limited. Further more interrelationships between various mechanisms are still unclear. For instance, the mechanisms of acupuncture analgesia in brain regions involves different brain regions and nuclei, and relationships between these brain regions and nuclei should be researched further. Correlations and interactions among the various pathways also require further research. Pain is closely accompanied by bad emotions. Can all pain relief mechanisms in the brain region also regulate these bad emotions? Finally, studies have proven the analgesic effects of pre-EA, which has a better curative effect than EA. How does this difference come about? We reviewed relevant studies in recent years and identified research gaps thereby providing new directions and ideas for future research.
Footnotes
Author contributions
Yuxin-Fang and Di-Zhang were responsible for the conception and design of the study. Qingxiang-Zhang and Mengmeng-Zhou drafted the article. Mingzhu-Huo were responsible for review and editing. Yuxin Si and Youlin Zhang reviewed and critically revised it.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (NSFC) No. 81973944, the Natural Science Foundation of Tianjin No.20JCYBJC00200 and No.22JCYBJC00990, Youth Talent Promotion Project of the Tianjin Association for Science and Technology No. TJSQNTJ-2020-15.
