Abstract
Large-diameter myelinated fibers in sciatic nerves are composed of both Aα/β-afferent fibers and Aα-efferent fibers to convey sensory and motor impulses, respectively, via saltatory conduction for rapid leg responses. Saltatory conduction and electrophysiological properties at the nodes of Ranvier (NRs) of these sciatic nerve fibers have not been directly studied. We used ex vivo sciatic nerve preparations from rats and applied patch-clamp recordings at the NRs of both Aα/β-afferent fibers and Aα-efferent fibers in the sciatic nerves to characterize their saltatory conduction and intrinsic electrophysiological properties. The velocity and frequency of saltatory conduction in both types of fibers were similar. Resting membrane potentials (RMPs), input resistance, action potential (AP) threshold, and AP rheobase were also not significantly different at the NRs of the two types of fibers in the sciatic nerves. In comparison with Aα/β-afferent fibers, Aα-efferent fibers in the sciatic nerves show higher amplitude and broader width of APs at their NRs. At the NRs of both types of fibers, depolarizing voltages evoked transient inward currents followed by non-inactivating outward currents, and the inward currents and non-inactivating outward currents at the NRs were not significantly different between the two types of fibers. Using AP-clamp, inward currents during AP upstroke were found to be insignificant difference, but amplitudes of non-inactivating outward currents during AP repolarization were significantly lower at the NRs of Aα-efferent fibers than at the NRs of Aα/β-afferent fibers in the sciatic nerves. Collectively, saltatory conduction, ionic currents, and intrinsic electrophysiological properties at the NRs of Aα/β-afferent fibers and Aα-efferent fibers in the sciatic nerves are generally similar, but some differences were also observed.
Keywords
Introduction
Saltatory conduction of action potentials (APs) through the nodes of Ranvier (NRs) in myelinated nerves1–6 allows rapid sensory responses with high acuity and swift muscle movement with great accuracy. Saltatory conduction enables high-speed and high-frequency AP propagation in myelinated nerves, much more rapid than AP conduction on unmyelinated nerves in mammals. 7 Degenerative impairment occurs at NRs of myelinated nerves in neurological diseases such as inflammatory demyelinating polyneuropathy, multiple sclerosis, and spinal cord injury,8–12 which can result in sensory and motor dysfunction. Saltatory conduction at the NRs is significantly influenced by intrinsic electrophysiological properties of axonal membranes at the NRs. 13 However, only limited electrophysiological studies have so far been performed at NRs of mammalian myelinated nerves. It is unknown whether NRs of different type of myelinated nerves may have similar or different intrinsic electrophysiological properties.
Intrinsic electrophysiological properties of NRs should largely reflect the activity of ion channels on the axonal membranes of NRs. In myelinated nerves of mammals, Nav1.6 voltage-gated Na+ channels are highly clustered at NRs12,14 and are believed to drive nodal membrane depolarization to generate APs at the NRs. 14 Although voltage-gated K+ channels are known to mediate AP repolarization in other parts of neuronal membranes, 15 previous studies have suggested that voltage-gated K+ channels are not or negligibly present at intact NRs of mammals.13,16,17 Interestingly, using immunohistochemical approaches, studies have detected Kv7.2 18 and Kv3.1b 19 voltage-gated K+ channels at mammalian NRs, as well as Kv1.1 and Kv1.2 voltage-gated K+ channels in juxtaparanodal regions. 20 However, the intrinsic electrophysiology properties and ion channel functions at mammalian NRs have been largely unknown for a long time due to the inability to conduct patch-clamp recordings at mammalian NRs. This technical difficulty has now been overcome by the recently developed pressure-patch-clamp recording technique.13,21
By using the pressure-patch-clamp recording technique, we have recently characterized intrinsic electrophysiological properties and ion channels at the NRs of rat trigeminal Aβ-afferent fibers and spinal ventral root motor fibers.13,22,23 We have found that TREK-1 and TRAAK, two-pore domain potassium (K2P) channels that are thermosensitive and mechanosensitive, are the main K+ channels that determine the intrinsic electrophysiological properties of these nerve fibers. These properties include a very negative resting membrane potentials (RMPs), extremely low membrane input resistance, rapid AP repolarization, and narrow AP width.13,22 Furthermore, we demonstrate the presence of these K2P channels at the NRs using the immunochemical approach, which showed strong TREK-1 and TRAAK immunoreactivity clustered at these sites.13,24 Our recent study has also demonstrated that K2P channels play a crucial role in permitting high-speed and high-frequency AP conduction along trigeminal Aβ-afferent fibers, and this function is closely related to their effects on the intrinsic electrophysiological properties at the NRs. 13
Large-diameter myelinated fibers in the sciatic nerves consist of Aα/β-afferent fibers for conveying somatosensory information from the feet and legs and Aα-efferent (motor) fibers for the movement of muscles in these regions. Anatomically, Aα/β-afferent fibers and Aα-efferent fibers in the sciatic nerves are indistinguishable, which is why previous studies did not differentiate between them.17,25–27 As a result, saltatory conduction and intrinsic electrophysiological properties at the NRs of either Aα/β-afferent fibers or Aα-efferent fibers in the sciatic nerves remain unknown. In the present study, we have used a nerve preparation that included the sciatic nerve, L5 dorsal root ganglion (DRG), and dorsal and ventral roots, which allowed us to differentiate between Aα/β-afferent fibers and Aα-efferent fibers in the sciatic nerves using an electrophysiological method. Furthermore, we applied pressure-patch-clamp recordings at the NRs of Aα/β-afferent fibers and Aα-efferent fibers in the sciatic nerves to characterize and compare their intrinsic electrophysiological properties.
Materials and methods
Ex vivo preparation of the sciatic nerve with the dorsal and ventral roots
Male Lewis rats aged between 7–12 weeks were deeply anesthetized with isoflurane and then sacrificed by decapitation. The sciatic nerve, along with L5 DRG, and the attached dorsal and ventral roots were dissected and placed in a petri dish filled with Krebs solution that contained (in mM): 117 NaCl, 3.5 KCl, 2.5 CaCl2, 1.2 MgCl2, 1.2 NaH2PO4, 25 NaHCO3, and 11 glucose. The Krebs solution was saturated with 95% O2 and 5% CO2, had a pH of 7.35, an osmolarity of 324 mOsm, and was maintained at a temperature of approximately 24°C. Connective tissues on the surface of the sciatic nerve bundles were removed with a pair of fine forceps under a dissection microscope. The nerve preparation was affixed in a recording chamber by a tissue anchor and submerged in the Krebs solution. The recording chamber was mounted on the stage of an Olympus BX51 microscope that was equipped with IR-DIC and fluorescent imaging systems. To facilitate the penetration of the patch-clamp electrode through perineural tissues, the ex vivo nerve preparation was briefly exposed to a mixture of 0.07% dispase II (Roche, Indianapolis, IN, USA) and 0.07% collagenase (MilliporeSigma, Billerica, MA, USA) in the Krebs solution for 5–10 min at room temperature, after which the enzymes were washed off with the Krebs solution. The ex vivo nerve preparation was continuously perfused with the Krebs solution at 24°C, which was maintained by a Peltier temperature control system (CL-200A, Warner Instrument, CT, USA).
Pressure-patch-clamp recordings at the nodes of Ranvier
Nodes of Ranvier in the sciatic nerves were visualized under a 40× (NA 0.80) water immersion objective with live images captured by an infrared CCD camera (IR-1000, DAGE-MTI, USA). Patch-clamp recordings were performed at the NRs of large-sized sciatic nerve fibers with diameters from 7 to 12 μm, which fell into the range of Aα- and Aβ-fibers. 28 Recording electrodes were pulled with a Flaming/Brown Micropipette Puller (P-97, Shutter Instruments, CA, USA). The resistance of the recording electrodes ranged from 5 to 8 MΩ after filling recording electrode internal solutions that contained (in mM): 105 K-gluconate, 30 KCl, 0.5 CaCl2, 2.4 MgCl2, 5 EGTA, 10 HEPES, 5 Na2ATP and 0.33 GTP-TRIS salt. The pH of the solution was adjusted to 7.35 with KOH. To access nodal axon membranes by recording electrodes and achieve high quality membrane seals, we used a high-speed pressure-clamp device (HSPC-1, ALA Scientific instruments, NY, USA) that was connected to the patch-clamp recording electrode holder to finely control internal pressures of the patch-clamp recording electrodes while approaching nodal membranes. The pressure-patch-clamp recordings were performed in the same manner as described in our previous studies.13,21,22 Signals of voltage-clamp experiments were recorded and amplified using a multiclamp 700B amplifier, filtered at 10 kHz, and sampled at 10 kHz using the pCLAMP 11 software (Molecular Devices, Sunnyvale, CA, USA). Signals from current-clamp recordings for APs at NRs were low-pass filtered at 10 kHz and sampled at 50 kHz.
To measure ionic currents flowing through nodal membranes following voltage steps, recordings were performed under the whole-cell voltage-clamp configuration with nodal membranes held at −72 mV. Voltage steps were applied from −112 mV to +58 mV (voltage command of −100 to +70 mV) with increments of 10 mV each step and a step duration of 500 ms. Unless otherwise indicated, membrane voltages mentioned in the texts have been corrected for the calculated junction potentials of 12.3 mV for the recording electrode internal solution. In a different set of experiments, pressure-patch-clamp recordings were applied to the NR of sciatic nerves in ex vivo nerve preparations. APs at the NR were first recorded under the current-clamp configuration following electrical stimulation at the distal part of dorsal and ventral roots. AP-clamp recordings29,30 were performed under the voltage-clamp configuration and each NR’s own AP was used as the voltage command waveform. AP-evoked inward currents were quantified by measuring net inward current during the rising phase of AP waveform, and AP-evoked outward currents were quantified by measuring net outward current during the falling phase of AP waveform. To determine the properties of membranes and APs at the NRs, patch-clamp recordings were performed under the whole-cell current-clamp configuration. Step currents were injected into NRs through recording electrodes. Step currents were applied from −100 pA to 1200 pA with increments of 50 pA per step and the duration of each step was 50 ms.
Differentiation between the NRs of Aα/β-afferent fibers and the NRs of Aα-efferent fibers in the sciatic nerves and investigation of their saltatory conduction properties
Sciatic nerves contain both Aα/β-afferent fibers and Aα-efferent fibers which are not visually distinguishable under the microscope. To know whether the NR being recorded was on an Aα/β-afferent fiber or an Aα-efferent fiber in the sciatic nerve, electrical stimulation was applied to either the dorsal root (somatosensory nerve) or the ventral root (motor nerve) to see whether APs were evoked by the stimulation. APs were evoked using a suction stimulation electrode. The distance between the stimulation site and the recording site was approximately 33 mm. The tip of the suction stimulation electrode was approximately 0.5 mm in diameter and was fire-polished. The end of the nerve bundle was aspirated into the suction stimulation electrode to produce a tight fitting by negative pressure. The negative pressure was continuously applied into the suction stimulation electrode to maintain tight fitting during experiments. To initiate APs at the end of the nerve bundle, monophasic square wave pulses were generated by an electronic stimulator (Master-8, A.M.P.I, Israel) and delivered via a stimulation isolator (ISO-Flex, A.M.P.I, Israel) to the suction stimulation electrode. The duration of the stimulation pulse was 50 µs. The stimulation threshold of the nerve, i.e., the minimum stimulation intensity for evoking APs, was first determined and was approximately 100 μA. Then stimulation was applied at the intensity of 2-fold threshold throughout the experiments. Conduction velocity was calculated based on the latency of APs and the length of nerve fibers. The latency of APs was measured from the time of stimulation that was marked by stimulation artifacts to the time when AP was initiated at the recording site. The length of the nerve fiber was the distance between the stimulation site and the recording site. To determine success rates of APs at the NRs following different stimulation frequencies, stimulation pulses were applied to the nerve bundles at frequencies of 1, 10, 50, 100, 200, 500 and 1000 Hz. Stimulation at each frequency was applied for 20 s. Intervals between different tests were 30 s. Success rates of APs conducted through the NRs were defined as the percentage of successfully propagated APs through the nodal recording sites during 20 s period of stimulation.
Data analysis
Electrophysiological data were measured using Clampfit 11 (Molecular Devices). Input resistance was determined with a −10 mV voltage step from the membrane holding voltage of −72 mV. RMP was measured under current-clamp configuration at the zero holding current. AP rheobase was the threshold step current that evoked AP firing. AP threshold was the potential at which AP upstroke started. AP amplitude was measured from RMP to AP peak. AP width was measured as the duration from 50% AP upstroke to 50% AP repolarization. All the above AP parameters were determined with the AP evoked by the rheobase step current at the NR. AP success rates at the NRs were calculated as the number of APs recorded at the NRs divided by the number of electrical stimuli, where the electrical stimuli were applied to the dorsal or ventral roots at different frequencies for 20 s. Curve-fitting was performed using a nonlinear regression fit with the following equation: Y = 100/(1 + 10
Results
Saltatory conduction at the nodes of Ranvier of Aα/β-afferent fibers and Aα-efferent fibers in the sciatic nerves
Figure 1(a) and (b) are the schematic diagram (Figure 1(a)) and anatomical locations (Figure 1(b)) of dorsal roots, L4, L5, and L6 DRGs, their spinal nerves, and the sciatic nerves in a rat. Ventral roots were located on the ventral side and could not be visualized in Figure 1(b). We dissected out these nerve structures along with dorsal and ventral roots (Figure 1(c)). For electrophysiological recording experiments, we further trimmed off the L4 and L6 parts, so that the nerve preparation only contained a sciatic nerve bundle, L5 lumbar spinal nerve, and L5 DRG and their associated dorsal and ventral roots (Figures 1(c) and 2(a)). We made whole-cell patch-clamp recordings at the NRs from large-diameter (7–12 μm) myelinated fibers in the sciatic nerves. Large-diameter myelinated fibers in the sciatic nerves contain both Aα/β-afferent fibers and Aα-efferent fibers
28
which are morphologically indistinguishable. We used an electrophysiological method to differential these two types of sciatic nerve fibers as described below. To determine if a recording was made from an Aα/β-afferent fiber or an Aα-efferent fibers in the sciatic nerve, electrical stimulation was applied at the dorsal or ventral roots, respectively. A recording was made from an afferent fiber in the sciatic nerve when dorsal root stimulation could evoke APs. On the other hand, a recording was made from an efferent fiber in the sciatic nerve when ventral root stimulation could evoke APs (Figure 2(a) and (b)). Anatomy and ex vivo sciatic nerve preparation that contain lumbar dorsal and ventral nerve roots. (a) Schematic diagram illustrates the locations of various nerves in the body of a rat, including the L5 ventral root, L5 dorsal root, L5 DRG, lumbar spinal nerve, and sciatic nerve. (b) Image shows the anatomical locations of lumbar dorsal roots, L4-, L5-, L6-DRGs and their spinal nerves, and sciatic nerves in an adult rat. Lumbar ventral roots cannot be viewed in this image. (c) The ex vivo nerve preparation, freshly dissected from b Recordings from the nodes of Ranvier of Aα/β-afferents and Aα-efferent fibers in the sciatic nerves. (a) Diagram illustrates the experimental setup for patch-clamp recordings from the nodes of Ranvier (NR) of either Aα/β-afferent fibers or Aα-efferent fibers in the ex vivo nerve preparation, which consists of the sciatic nerves, L5-spinal nerve, L5 DRG, and L5 dorsal root as well as ventral root. Two suction stimulation electrodes were used to stimulate either L5 dorsal root or L5 ventral root to evoked action potentials (APs). (b) Two sample traces of APs recorded from two different NRs are presented. Left panel shows that electrical stimulation to dorsal root but not ventral root evoked the AP, and thereby the AP was recorded from the NR of an afferent fiber in the sciatic nerve. The right panel shows electrical stimulation to ventral root but not dorsal root evoked AP, and thereby the AP was recorded from the NR of an Aα-efferent fiber in the sciatic nerve. Arrows indicate stimulation artifacts. (c) Summary data of velocity of saltatory conduction recorded from NRs of either Aα/β-afferent fibers (open bar) or Aα-efferent fibers (filled bar) in the sciatic nerves. (d) Summary data of the threshold intensity for evoking Aα/β-afferent fiber APs (open bar) and Aα-efferent fiber APs (filled bar) by electrical stimulations applied to dorsal roots and ventral roots, respectively. Recordings were made from NRs of Aα/β-afferent fibers and Aα-efferent fibers in the sciatic nerves. (e and f) AP width (e) and AP amplitude (f) recorded from NRs of Aα/β-afferent fibers (open bars) and Aα-efferent fibers (filled bars) in the sciatic nerves. APs were evoked by electrical stimulations applied to either dorsal roots or ventral roots. Data represent Mean ± SEM, *p < 0.05, **p < 0.01, ns, not significantly different, Student’s t test.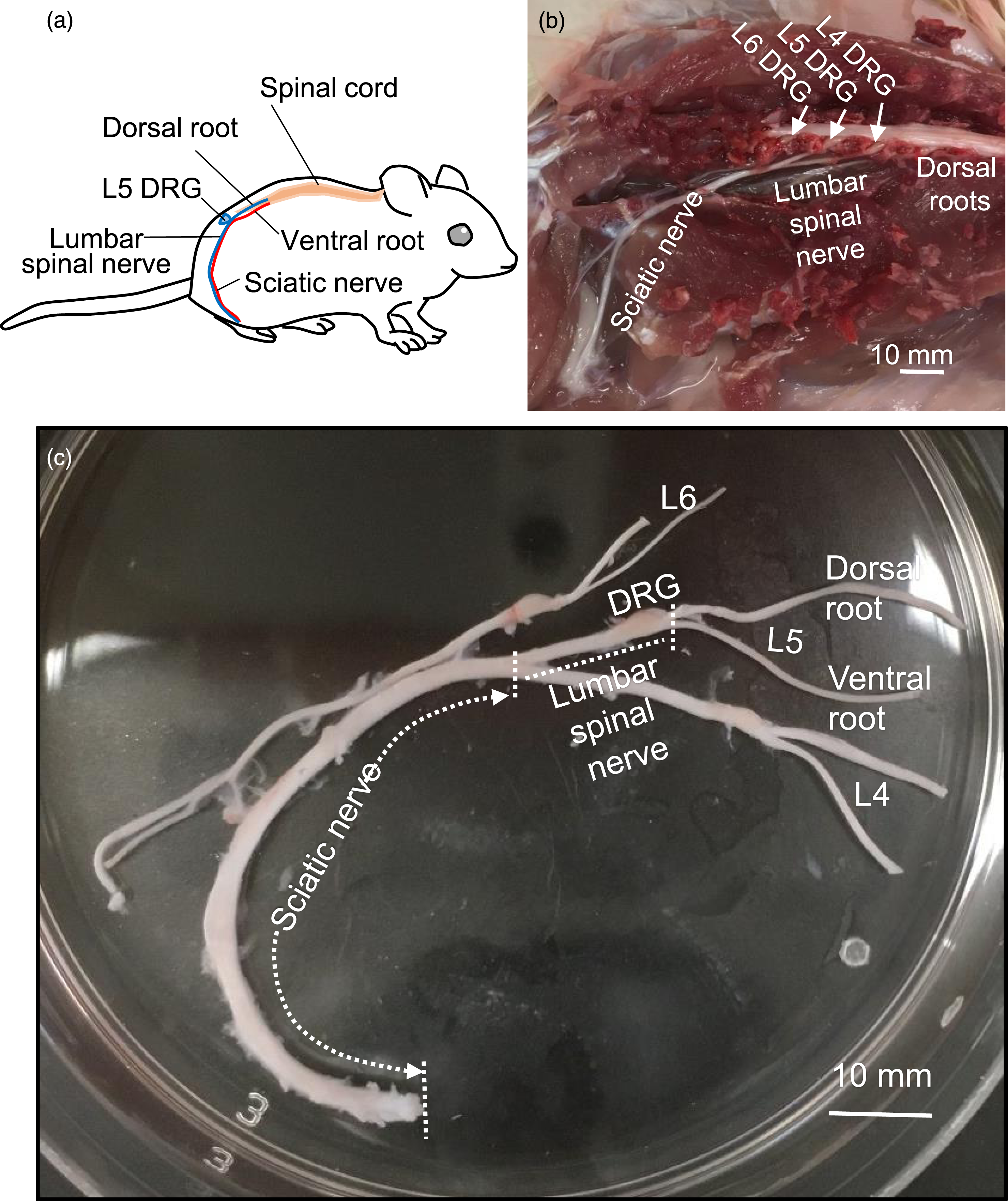

We determined the velocity of saltatory conduction at NRs of Aα/β-afferent fibers and Aα-efferent fibers in the sciatic nerves (Figure 2(b) and (c)). In this set of experiments, APs were elicited by electrical stimulation applied to the dorsal root or the ventral root, and APs was recorded at the NRs of Aα/β-afferent fibers and Aα-efferent fibers in the sciatic nerves, respectively (Figure 2(a) and (b)). AP conduction velocities were 22.9 ± 1.2 m/s (n = 5) at the NRs of Aα/β-afferent fibers and 22.4 ± 1.6 m/s (n = 8) at the NRs of Aα-efferent fibers in the sciatic nerves, which were not significantly different (Figure 2(c)). The thresholds for evoking APs were 104.0 ± 16.3 μA (n = 5) and 103.1 ± 27.7 μA (n = 8) for Aα/β-afferent fibers and Aα-efferent fibers in the sciatic nerves, respectively, which were not significantly different (Figure 2(d)). With stimulation at dorsal or ventral roots, the widths of APs were 0.5 ± 0.01 ms (n = 5) at the NRs of Aα/β-afferent fibers and 0.8 ± 0.09 ms (n = 8) at the NRs of Aα-efferent fibers in the sciatic nerves. The AP widths were significantly broader at the NRs of Aα-efferent fibers than at the NRs of Aα/β-afferent fibers in the sciatic nerves (p < 0.05, Figure 2(e)). AP amplitudes were 99.7 ± 4.0 mV (n = 5) and 115.5 ± 2.5 mV (n = 8) at the NRs of Aα/β-afferent fibers and Aα-efferent fibers in the sciatic nerves, respectively. The AP amplitudes were significantly higher at the NRs of Aα-efferent fibers than at NRs of Aα/β-afferent fibers in the sciatic nerves (p < 0.01, Figure 2(f)).
We determined AP success rate at the NRs of both Aα/β-afferent fibers and Aα-efferent fibers in the sciatic nerves at different stimulation frequencies. In this set of experiments, APs were elicited at dorsal root or ventral root by train stimulation for 20 s at the frequencies of 1, 10, 50, 100, 200, 500, and 1000 Hz while APs were recorded at the NRs of Aα/β-afferent fibers (Figure 3(a)) or Aα-efferent fibers in the sciatic nerves (Figure 3(b)). AP conduction through NRs of Aα/β-afferent fibers in the sciatic nerves had 100% (n = 5) success rate following stimulation at frequency up to 100 Hz (Figure 3(c)), which was reduced to 56.2 ± 5.7% (n = 5) at 200 Hz stimulation, 10.2 ± 2.5% (n = 5) at 500 Hz stimulation, and 1.0 ± 0.5% (n = 5) at 1000 Hz stimulation (Figure 3(c)). AP conduction through NRs of Aα-efferent fibers in the sciatic nerves had nearly 100% (n = 8) success rate following stimulation at frequency up to 50 Hz (Figure 3(c)), which was reduced to 80.8 ± 12.9% (n = 8) at 100 Hz stimulation, 54.4 ± 13.8% (n = 8) at 200 Hz stimulation, 18.3 ± 6% (n = 8) at 500 Hz, and 4.6 ± 1.9% (n = 8) at 1000 Hz stimulation (Figure 3(c)). We used the frequency at which AP success rate was 50% (FS50) to quantitatively describe the success rate of APs (Figure 3(d)). FS50 was 221.6 ± 14.9 Hz (n = 5) in Aα/β-afferent fibers and 261.1 ± 54.8 Hz (n = 8) in Aα-efferent fibers in the sciatic nerves, and were not significantly different between these two types of sciatic nerve fibers (Figure 3(d)). Action potential success rates recorded at the node of Ranvier of Aα/β-afferent fibers and Aα-efferent fibers in the sciatic nerves. (a and b) Two sets of sample traces show AP regenerations at the NRs of Aα/β-afferent fibers (a) and Aα-efferent fibers (b) in the sciatic nerves in response to 200 Hz electrical stimulation for 20 s. Electrical stimulation was applied to either the dorsal root or ventral root, and segmented traces of APs were shown. (c) AP regeneration success rates at the NRs of Aα/β-afferent fibers (dark circles) and Aα-efferent fibers (red circles) in the sciatic nerves in response to electrical stimulation at 1, 10, 50, 100, 200, 500, and 1000 Hz. The recording duration at each frequency was 20 s. Dashed lines indicate the frequency at which AP success rates fell to 50% (FS50). (d) Bar graph shows summary data of FS50 at the NRs in Aα/β-afferent fibers (open bar) and Aα-efferent fibers (filled bar) in the sciatic nerves. Data represent Mean ± SEM, ns, not significantly different, Student’s t test.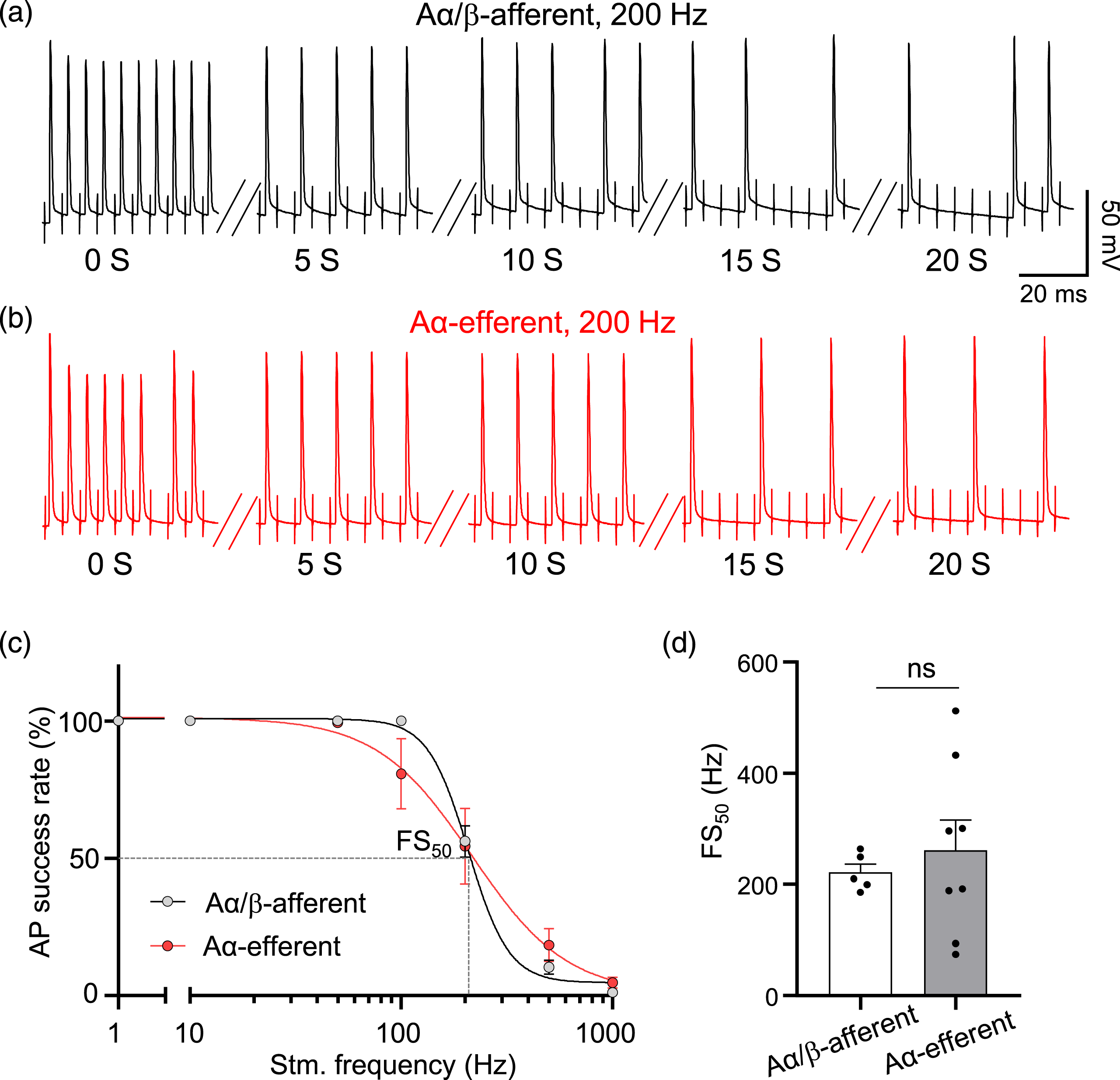
Intrinsic electrophysiological properties at the nodes of Ranvier of Aα/β-afferent fibers and Aα-efferent fibers in the sciatic nerves
We determined intrinsic electrophysiological properties at the NRs of both Aα/β-afferent fibers and Aα-efferent fibers in the sciatic nerves by injecting depolarizing currents to initiate APs at the NRs (Figure 4(a)). Figure 4(a) shows two sample traces of APs, one was recorded from a NR of an Aα/β-afferent fiber, and the other from a NR of an Aα-efferent fiber in the sciatic nerve, in response to depolarizing currents. RMPs were −79.7 ± 1.9 mV (n = 5, Figure 4(b)) at the NRs of Aα/β-afferent fibers and −82.2 ± 1.0 mV (n = 8, Figure 4(b)) at the NRs of Aα-efferent fibers in the sciatic nerves, and were not significantly different. AP amplitudes were 73.7 ± 3.5 mV (n = 5, Figure 4(c)) at the NRs of Aα/β-afferent fiber and 95.5 ± 3.3 mV (n = 8, Figure 4(c)) at the NRs of Aα-efferent fibers in the sciatic nerves, and the AP amplitudes were significantly higher at the NR of Aα-efferent fibers than at NRs of Aα/β-afferent fibers in the sciatic nerves (p < 0.01). AP widths were 0.8 ± 0.02 ms (n = 5, Figure 4(d)) at the NRs of Aα/β-afferent fibers and 1.1 ± 0.1 ms (n = 8, Figure 4(d)) at the NRs of Aα-efferent fibers in the sciatic nerves, and the AP widths were significantly broader at the NRs of Aα-efferent fibers than at the NRs of Aα/β-afferent fibers in the sciatic nerves (p < 0.01). AP thresholds were −51.8 ± 2.9 mV (n = 5, Figure 4(e)) at the NRs of Aα/β-afferent fibers and −55.6 ± 1.7 mV (n = 8, Figure 4(e)) at the NRs of Aα-efferent fibers in the sciatic nerves, and were not significantly different. AP rheobases were 430.0 ± 46.4 pA (n = 5, Figure 4(f)) at the NRs of Aα/β-afferent fibers and 325.0 ± 41.2 pA (n = 8, Figure 4(f)) at the NRs of Aα-efferent fibers in the sciatic nerves, and were not significantly different. Membrane input resistances were 50.8 ± 5.4 MΩ (n = 5, Figure 4(g)) at the NRs of Aα/β-afferent fibers and 56.3 ± 5.5 MΩ (n = 8, Figure 4(g)) at the NRs of Aα-efferent fibers in the sciatic nerves, and were not significantly different. Electrophysiological properties at the NRs of Aα/β-afferent fibers and Aα-efferent fibers in the sciatic nerves. (a) Two sample traces show APs evoked by directly injecting threshold depolarizing currents into a NR of Aα/β-afferent fiber (dark trace) and a NR of Aα-efferent fiber (red trace) in a sciatic nerve. The two APs were aligned at current injection time. Dashed line indicates the resting membrane potential (RMP) at the NR of Aα/β-afferent fiber, which was −80 mV. (b–g) Bar graphs show summary data of electrophysiological parameters of NRs of Aα/β-afferent fiber (open bars) and Aα-efferent fiber (filled bars) in the sciatic nerves, including RMPs (b) AP amplitude (c) AP width (d) AP threshold (e) rheobase for evoking APs (f) and input resistance (g) Data represent Mean ± SEM, **p < 0.01, ns, not significantly different, Student’s t test.
Ionic currents evoked by depolarizing voltages at the nodes of Ranvier of Aα/β-afferent fibers and Aα-efferent fibers in the sciatic nerves
Under the whole-cell voltage-clamp configuration, we examined membrane ionic currents by applying depolarizing voltage steps to NRs of Aα/β-afferent fibers and Aα-efferent fibers in the sciatic nerves (Figure 5). These depolarizing voltage steps evoked transient inward currents, followed by non-inactivating outward currents at the NRs of both Aα/β-afferent fibers (Figure 5(a) left) and Aα-efferent fibers (Figure 5(a) right) in the sciatic nerves. The non-inactivating outward currents displayed a nearly linear current-voltage relationship (I-V curve) (Figure 5(b)) and had a reversal potential at approximately −80 mV for the NRs of both fibers (Figure 5(b)), indicating that these outward currents were mediated by K+ channels. We analyzed non-inactivating outward currents using the area under the I-V curve (AUC) and found that the AUC was not significantly different between the NRs of Aα/β-afferent fibers (31.3 ± 3.3 nA*mV, n = 5) and Aα-efferent fibers (28.3 ± 1.9 nA*mV, n = 8) in the sciatic nerves (Figure 5(c)). Non-inactivating outward currents at the node of Ranvier of Aα/β-afferent fibers and Aα-efferent fibers in the sciatic nerves. (a) Two sets of sample traces show ionic currents recorded at the NR of an Aα/β-afferent fibers (left panel) and an Aα-efferent fiber (right panel) following the application of voltage steps from −72 mV to 58 mV each at an increment of 10 mV. (b) current-voltage (I-V) relationship of the outward currents evoked by the voltage steps applied to NRs of Aα/β-afferent fibers (open circles) and Aα-efferent fibers (closed circles). The non-inactivating outward currents were measured near the end of voltage step as indicated by the open circle and closed circle in a. (c) Summary data of the area under the curve (AUC) of voltage-activated outward currents at the NRs of Aα/β-afferent fibers (open bar) and Aα-efferent fibers (filled bar). Data represent Mean ± SEM, ns, not significantly different, Student’s t test.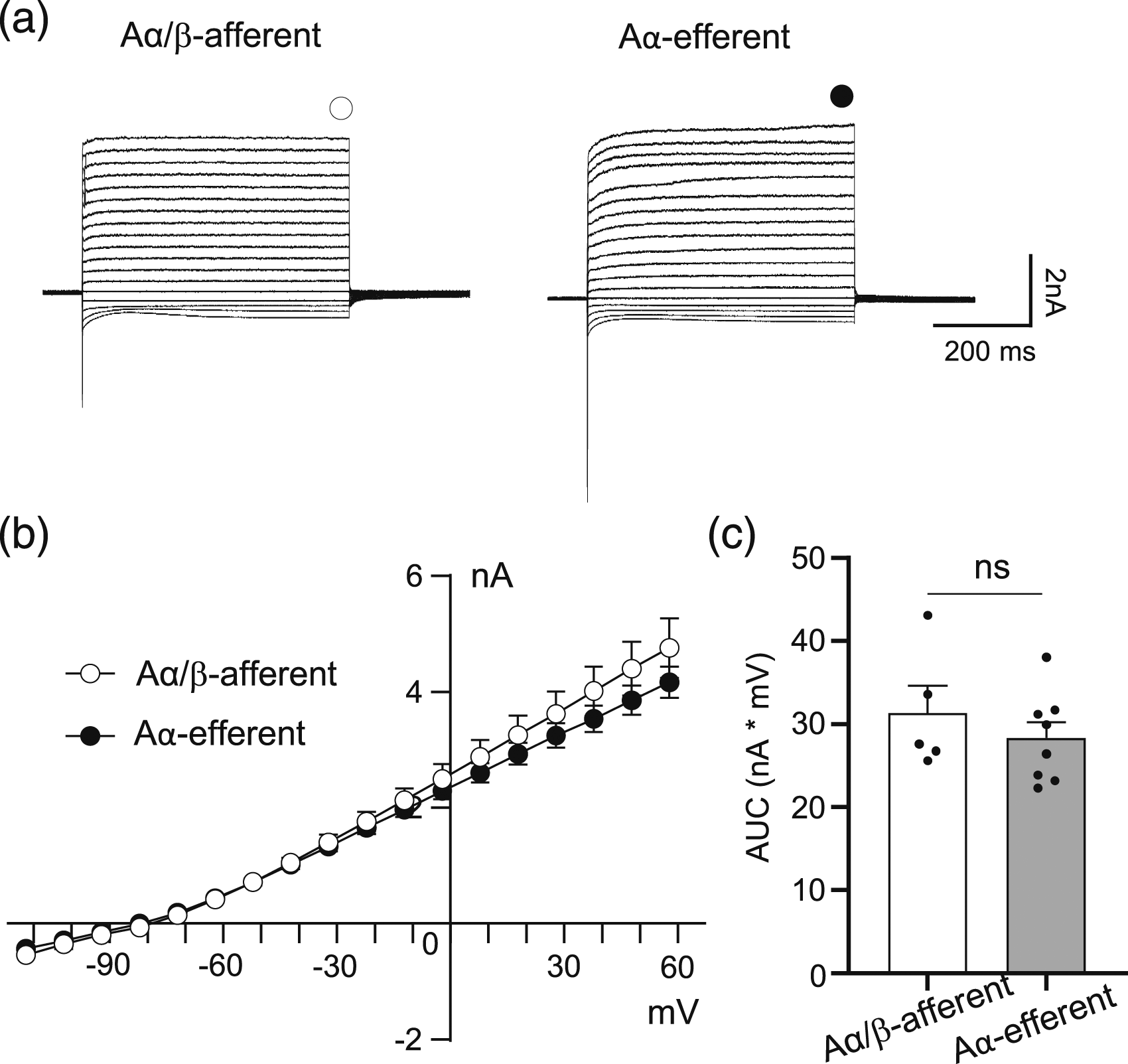
Transient inward currents were evoked by voltage steps at the NRs of both Aα/β-afferent fibers and Aα-efferent fibers in the sciatic nerves (Figures 6(a) and (b)). The I-V curve of the inward currents at the NRs of both Aα/β-afferent fibers (n = 5) and Aα-efferent fibers (n = 8) in the sciatic nerves were consistent with the currents mediated by voltage-gated Na+ channels (Figure 6(c)). It should be noted that there was apparently clamp errors including the escape of space-clamp in these recordings, which was most likely due to too large transient inward currents to be well space-clamped. Quantitatively determined using the area under the I-V curve (AUC) of the inward currents, the AUC was 22.6 ± 3.7 nA*mV (n = 5, Figure 6(d)) for the inward currents recorded from the NRs of Aα/β-afferent fibers, and 31.2 ± 2.5 nA*mV (n = 8, Figure 6(d)) for the inward currents recorded from the NRs of Aα-efferent fibers in the sciatic nerves. The AUCs showed that the inward currents had a strong tendency of higher amplitudes at the NRs of Aα-efferent fibers than at the NRs of Aα/β-afferent fibers in the sciatic nerves (p = 0.07) although not statistically significant. Voltage-activated transient inward currents recorded at the nodes of Ranvier of Aα/β-afferent fibers and Aα-efferent fibers in the sciatic nerves. (a and b) Sample traces show voltage-activated transient inward currents recorded at a NR of an Aα/β-afferent fiber (a) and a NR of an Aα-efferent fiber (b) in the sciatic nerves. The inward currents were evoked by voltage steps applied to the NRs from −72 mV to 58 mV each at a 10-mV increment as indicated under the current traces. (c) Current-voltage relationship of voltage-activated inward currents recorded at NRs of Aα/β-afferent fibers (open circles) and NRs of Aα-efferent fibers (closed circles) in the sciatic nerves. (d) Summary data of the AUC of voltage-activated inward currents at the NRs of Aα/β-afferent fibers (open bar) and Aα-efferent fibers (filled bar) in the sciatic nerves. Data represent Mean ± SEM, Student’s t test.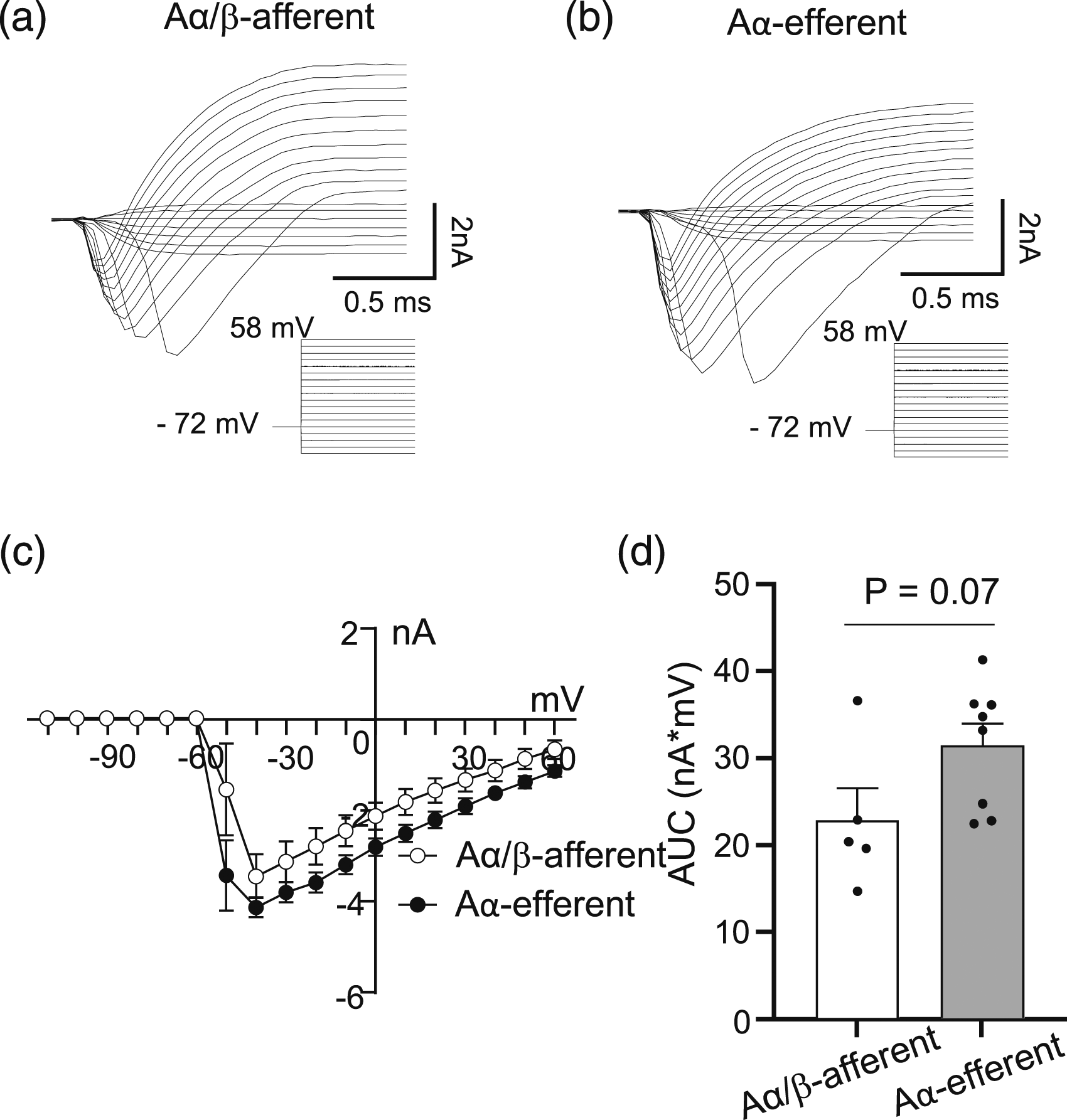
AP-clamp recording technique
29
was applied to NRs in order to investigate the ionic currents during an AP (Figure 7). In AP-clamp recordings, AP waveforms obtained from current-clamp recordings at NRs were applied as command voltages to the NRs to quantitatively determine the ionic currents during AP firing at the NRs. As shown in Figure 7(a) and (b), AP waveforms evoked inward currents followed by outward currents at NRs of both Aα/β-afferent fibers (Figure 7(a)) and Aα-efferent fibers (Figure 7(b)) in the sciatic nerves. The inward currents were mediated by voltage-gated Na+ channels, while the outward currents were mainly mediated by thermal K2P channels.
13
The amplitudes of AP-evoked inward currents were 6. ± 0.6 nA (n = 5) at the NRs of Aα/β-afferent fibers and 9.5 ± 1.0 nA (n = 8) at the NRs of Aα-efferent fibers in the sciatic nerves (Figure 7(c)), showing a strong tendency of higher amplitudes at the NRs of Aα-efferent fibers (p = 0.07, Figure 7(c)). The amplitude of AP-evoked outward currents was 1.9 ± 0.2 nA (n = 5, Figure 7(d)) at the NRs of Aα/β-afferent fibers, which was significantly larger than that of Aα-efferent fibers (0.8 ± 0.2 nA, n = 8, p < 0.01, Figure 7(d)) in the sciatic nerves. Inward and outward currents during action potentials at the nodes of Ranvier of Aα/β-afferent fibers and Aα-efferent fibers in the sciatic nerves. (a) Sample trace on top illustrates currents recorded at a NR of an Aα/β-afferent fiber in the sciatic nerve under AP-clamp mode. The trace on the bottom was the AP waveform (dashed line) for the AP-clamp recording. (b) Similar to a except the recording was made from a NR of an Aα-efferent fiber in the sciatic nerve. (c) Summary data of inward currents recorded at NRs of Aα/β-afferent fibers (open bar) and Aα-efferent fibers (filled bar) in the sciatic nerves during AP depolarization phase. (d) Summary data of outward currents recorded at NRs of Aα/β-afferent fibers (open bar) and Aα-efferent fibers (filled bar) in the sciatic nerves during AP repolarization phase. Data represent mean ± SEM. **p < 0.01, Student’s t test.
Discussion
The present study applied the pressure-patch-clamp recording technique to the NRs of both Aα/β-afferent fibers and Aα-efferent fibers in the sciatic nerves to characterize their saltatory conduction, intrinsic electrophysiological properties, and main ionic currents at their NRs. We have shown that saltatory conduction, most of intrinsic electrophysiological properties, and main ionic currents are similar at the NRs of these two types of sciatic nerves. Significant differences have been observed between Aα/β-afferent fibers and Aα-efferent fibers of the sciatic nerves in the amplitude and width of APs and the outward currents during APs. The saltatory conduction and intrinsic electrophysiological properties at the NRs of Aα/β-afferent fibers and Aα-efferent fibers in the sciatic nerves are consistent with those in trigeminal Aβ-afferent fibers and ventral root motor fibers described in our previous studies.13,22,23,31 Thus, in general, large-diameter fast conducting myelinated fibers examined so far show high similarity in saltatory conduction and most intrinsic electrophysiological properties at their NRs, even though these nerve fibers anatomically locate at different sites in the body and have different physiological functions.
Since Aα/β-afferent fibers and Aα-efferent fibers in the sciatic nerves are visually indistinguishable, it was not possible to know directly whether large-diameter myelinated fibers recorded in sciatic nerves were Aα/β-afferent fibers or Aα-efferent fibers in previous electrophysiological studies.17,25–27 In the present study, we have made sciatic nerve preparation with both dorsal and ventral roots. By applying electrical stimulation to either the dorsal roots or ventral roots, we can identify the NRs of Aα/β-afferent fibers or Aα-efferent fibers in the sciatic nerves. To the best of our knowledge, this is the first electrophysiological recordings from identified Aα/β-afferent fibers and Aα-efferent fibers in the sciatic nerves.
We have shown in our previous studies that saltatory conduction and intrinsic electrophysiological properties at the NRs of trigeminal Aβ-afferent fibers13,21,22 and ventral root motor fibers 23 are mainly determined by thermal and mechanical sensitive K2P channels. These K2P channels are the principal type of K+ channels clustered at the NRs of large-diameter myelinated nerves in previous studies.13,24 Although not directly determined in the present study, the NRs of Aα/β-afferent fibers and Aα-efferent fibers in the sciatic nerves may also use the K2P channels as the main type of K+ channels to control saltatory conduction and intrinsic electrophysiological properties at their NRs. This is supported by the intrinsic electrophysiological properties which show very low input resistance and very negative RMP at the NRs of these sciatic nerve fibers, consistent with the intrinsic electrophysiological properties of NRs of other large-diameter myelinated fibers in our previous studies.13,22,23 Under the voltage-clamp configuration, strong non-inactivating outward currents are recorded at the NRs of both Aα/β-afferent fibers and Aα-efferent fibers in the sciatic nerves. Strong non-inactivating outward currents have been first observed at the NRs of trigeminal Aβ-afferent fibers and NRs of motor fibers in the ventral roots in our previous study, and the outward currents are largely mediated by K2P channels.13,23 The strong non-inactivating outward currents recorded at the NRs of both Aα/β-afferent fibers and Aα-efferent fibers in the sciatic nerves may also be mainly mediated by K2P channels. In the present study, the amplitudes of non-inactivating outward currents at the NRs are not significantly different between Aα/β-afferent fibers and Aα-efferent fibers in the sciatic nerves. However, in AP-clamp recordings, the outward currents at the NRs have higher amplitudes in Aα/β-afferent fibers than in Aα-efferent fibers in the sciatic nerves. This raises the question of whether NRs of Aβ-afferent fibers and Aα-efferent fibers in the sciatic nerves may express different subtypes of K2P channels which may have different activation kinetics, although their outward currents at steady state show the same amplitude. Previous studies have shown that both TREK1 and TRAAK are co-expressed at the NRs of trigeminal Aβ-afferent fibers but only TREK1 channel immunoreactivity is detectable at the NRs of ventral root motor fibers. 13 Future studies are needed to investigate types of K2P channels expressed at the NRs of Aα/β-afferent fibers and Aα-efferent fibers in the sciatic nerves.
In the present study, we have found that APs at the NRs of Aα-efferent fibers in the sciatic nerves have relatively larger amplitudes and broader width compared to those at the NRs of Aα/β-afferent fibers in the sciatic nerves. Using AP-clamp recordings, we have observed a strong tendency of larger inward currents during AP upstroke and significantly smaller outward currents during AP repolarization at the NRs of Aα-efferent fibers compared to those at the NRs of Aα/β-afferent fibers in the sciatic nerves. These larger inward currents, mediated by voltage-gated Na+ channels, may contribute to the higher amplitude of AP upstroke, while smaller outward currents may cause slower AP repolarization or broader AP width at the NRs of Aα-efferent fibers. Our recent study on the NRs of rat lumbar spinal ventral roots showed that voltage-activated inward currents could be completely blocked by TTX, indicating these inward currents are mediated by TTX-sensitive voltage-gated Na+ channels. 23 Although not tested in the present study, the inward currents at the NRs may be also mediated by TTX-sensitive voltage-gated Na+ channels, most likely Nav1.6 channels, since strong immunoreactivity of Nav1.6 has been shown to be clustered at the NRs of myelinated nerves.12,14
Previously we have shown that saltatory conduction and intrinsic electrophysiological properties at the NRs of trigeminal Aβ-afferent fibers are not significantly affected by voltage-gate K+ channel blockers, indicating that the role of voltage-gated K+ channels is negligible at the NRs in trigeminal Aβ-afferent fibers. However, in our more recent studies, we have found that saltatory conduction and intrinsic electrophysiological properties are affected by voltage-gated K+ channel blockers and enhancers in a small but significant degree at the NRs of spinal ventral root fibers.23,31 Immunohistochemical studies have revealed the presence of Kv7.2 immunoreactivity at the NRs of myelinated nerve fibers. 18 In addition to Kv7.2, immunoreactivity to Kv1.1 and Kv1.2 channels has been observed in juxtaparanodal regions of myelinated nerve fibers. 20 The expression level of these voltage-gated K+ channels at the NRs may vary across different types of nerves. It will be interesting to investigate in future whether voltage-gated K+ channel blockers and enhancers may have varying effects on saltatory conduction and intrinsic electrophysiological properties at the NRs of Aα/β-afferent fibers and Aα-efferent fibers in the sciatic nerves. It will also be interesting to examine whether other ion channels, such as hyperpolarization-activated cyclic-nucleotide-gated channels (HCN), 32 may play roles in intrinsic electrophysiological properties and saltatory conduction at the NRs of these afferent fibers and efferent fibers.
Footnotes
Acknowledgements
We thank Dr. Hirosato Kanda for his scientific advice and technical support.
Authors’ contributions
JGG conceived the experiments and wrote the paper. ST performed experiments, analyzed data, and wrote the paper. All authors read and approved the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by NIH grants DE018661, DE023090 and NS109059 to J.G.G.
Ethical approval
All experimental procedures were approved by the appropriate institutional animal care and use committees of the University of Alabama at Birmingham.
Consent
Consent to participate is not applicable for this study.
Availability of data and materials
All data generated or analyzed during this study are available from corresponding author on reasonable request.
