Abstract
Important neurotrophic factors that are potentially involved in degenerative intervertebral disc (IVD) disease of the spine’s lumbosacral (L/S) region include glial cell-derived neurotrophic factor (GDNF) and growth associated protein 43 (GAP-43). The aim of this study was to determine and compare the concentrations of GAP-43 and GDNF in degenerated and healthy IVDs and to quantify and compare the GAP-43-positive and GDNF-positive nerve fibers. The study group consisted of 113 Caucasian patients with symptomatic lumbosacral discopathy (confirmed by a specialist surgeon), an indication for surgical treatment. The control group included 81 people who underwent postmortem examination. GAP-43 and GDNF concentrations were significantly higher in IVD samples from the study group compared with the control group, and the highest concentrations were observed in the degenerated IVDs that were graded 4 on the Pfirrmann scale. In the case of GAP-43, it was found that as the degree of IVD degeneration increased, the number of GAP-43-positive nerve fibers decreased. In the case of GDNF, the greatest number of fibers per mm2 of surface area was found in the IVD samples graded 3 on the Pfirrmann scale, and the number was found to be lower in samples graded 4 and 5. Hence, GAP-43 and GDNF are promising targets for analgesic treatment of degenerative IVD disease of the lumbosacral region of the spine.
Introduction
Pain in the lumbar region of the spine is one of the most common causes of disability in people living in developed countries. 1 Such pain is often associated with intervertebral disc (IVD) degeneration, which is a multi-stage process. Initial micro-injuries can cause painful damage to the external structure, which consists of a well-innervated fibrous ring.2–4 Multiple minor injuries can lead to damage to the structure of the nucleus pulposus, which leads to protrusion. 2 These changes result in a loss of the ability to store water in the disc and strengthen the nociceptive effect via an ingrowth of free nerve endings and vessels into the newly formed cracks.2,3 Inflammatory conditions and neoinnervation within the degenerated disc may significantly influence the pain experienced by patients and limit their fitness.5–7
The development of discogenic pain is a complex process that involves many proteins and factors at the cellular level,5–7 such as growth associated protein 43 (GAP-43) and glial cell-derived neurotrophic factor (GDNF).5–7
GAP-43, which is also known as neuromodulin, F1, and B-50, is a cytoplasmic protein specific to nerve tissue and one of the principal members of the protein kinase C (PKC) family.8,9 The GAP-43 gene contains three exons. The first exon encodes the membrane-targeting domain, the second exon encodes a calmodulin-binding domain and a PKC phosphorylation site, and the 5′-flanking sequence directs initiation of RNA transcription from several sites.10,11 Once transcription is complete, the resulting GAP-43 mRNA is stabilized by a neuron-specific RNA-binding protein (HuD). 12 There is evidence that HuD expression increases during brain development, neural regeneration, and learning, indicating that it has a stabilizing role in the maintenance of GAP-43 mRNA overexpression during neurodevelopmental processes in hippocampal dentate granule cells, neurons in the lateral amygdala, and layer V of the neocortex. 13 In contrast, the amino acid sequence of GAP-43 lacks membrane-spanning domains and glycosylation sites.14,15 GAP-43 is highly hydrophilic; however, it contains a short (43 amino acids) hydrophobic fragment that facilitates the anchoring of this neurotrophic factor to the cytoplasmic side of synaptic plasma membranes. 16 The overriding process affecting the formation of the biologically active form of GAP-43 is phosphorylation. GAP-43 is only expressed in neurons, with higher concentrations found in axon terminals and growth cones; it is absent in dendrites and myelinated axons. 17
GAP-43 plays a key role in the activation of signal transduction. Specifically, it regulates the organization of the cytoskeleton at the end of the neurite in the neuron growth cone during development. 18 It is also involved in neurite growth and conduction. 18 Differential expression of GAP-43 and neurofilament proteins has been observed during peripheral nerve regeneration using bioartificial conduits. 19 In adults, GAP-43 expression is generally low, except in nerve structures, which are found in areas of intensified plasticity or synaptic reconstruction. 19 Thus, GAP-43 is associated with nerve growth and plays an important role in neuron development and regeneration. 19 As a result of structural impairments in fully developed neurons, their growth cones, and some glial cells, an increase in the concentration of GAP-43 has been observed. 20
GDNF, the second protein of interest in this study, is characterized by its low molecular mass and belongs to a family of neurotrophic factors that significantly impact the survival rate of numerous types of neurons, synaptic plasticity differentiation, modulation of axonal and dendritic elaboration, and cell proliferation. 20 GDNF is a member of the transforming growth factor beta (TGFβ) superfamily due to its characteristic localization and the presence of seven cysteine residues; however, its nucleotide sequence is only slightly similar to that of TGFβ2. 21
GDNF is strongly expressed in both the peripheral and central nervous systems. It may be released by Schwann cells, oligodendrocytes, astrocytes, motor neurons, and skeletal muscle cells during the development and growth of neurons and other peripheral cells. 22 GDNF is synthesized in mammalian cells as a precursor protein (211 amino acids). Within the endoplasmic reticulum, it undergoes folding, dimerization, modification by N-linked glycosylation, and proteolytic cleavage by endoproteinase, and the protein convertases (PACE4, PC5A, PC5B, and PC7).24–26 These modifications result in the generation of the mature form of GDNF (134 amino acids).24–26 The active form of GDNF is a homodimer. 21
Unlike the other members of the TGFβ superfamily, GDNF family members do not exhibit affinity for serine/threonine kinase receptors; however, like the other neurotrophins, they interact with receptors with tyrosine kinase enzymatic activity (RTKs).22,23 The GDNF receptor (RET) is encoded by a protooncogene.24–26 It is also important to note that the signal transduction mediated by GDNF is a multistep process.24–26 First, GDNF binds via glycosylphosphatidylinositol (GPI) to membrane-anchored proteins called α-receptors for GDNF, namely glial cell line-derived neurotrophic receptor GFR α1 and GFR α2. GFR α1, in both the monomeric and dimeric forms, has high substrate specificity and affinity for the dimeric form of GDNF, and the resulting complex activates the RET receptor through its homodimerization and autophosphorylation.24–26
GDNF, like other proteins in the neurotrophin family, causes nerve renervation and regeneration. 27 Neurotrophins support and help restore numerous neuron populations, including motor, sensory, dopaminergic, sympathetic, and parasympathetic neuron populations. 26 GDNF has the most prominent effects on enteric, sympathetic, and dopamine neurons; however, it is also a potent regulator of motor neuron survival in the peripheral nervous system. 22 The most important features of GDNF are its ability to prevent apoptosis in motor neurons during their development, reduce the loss of neurons during development, save cells from death induced by axotomy, and prevent chronic degeneration. 22
Considering the significant roles that GAP-43 and GDNF play in the development of discogenic pain, the aim of this study was to determine and compare the concentrations of GAP-43 and GDNF in degenerated and healthy IVDs and to quantify and compare the GAP-43-positive and GDNF-positive nerve fibers.
Materials and methods
Ethics
The present research adhered to the tenets of the Declaration of Helsinki. Data confidentiality and patient anonymity were maintained at all times. Informed consent was obtained from all patients. Approval for this study was obtained from the Bioethical Committee operating at the Regional Medical Chamber in Krakow (no. 162/KBL/OIL/2021). The collection of postmortem material for research is regulated by the Act of 1 July 2005, of the Government of Poland, on the collection, storage, and transplantation of cells, tissues, and organs (Journal of Laws of 2020, Item 2134). Art. Five of this Act is based on the construct of opting out and states the following: (1) “If a deceased person did not express objection, when alive, it is allowed to recover cells, tissues, or organs from such person human cadaver for transplantation purposes. (2) A legal representative of a living minor or other person, which doesn’t have a full legal capacity, may state an objection for such individual. (3) Minors above 16 years old may state an objection themselves. (4) The regulations of paragraph 1-3 shall not be observed in case of recovery of cells, tissues and organs performed in order to diagnose a cause of death and assess treatment management during postmortem examinations.”
This means that, as a rule, it is permitted to collect tissues, cells, or organs from a human corpse unless the person from whom the tissues, cells, or organs are to be removed objected to this action during their lifetime. Information was obtained from the deceased’s family about the type of work the person performed, their physical activity, and their use of non-steroidal anti-inflammatory drugs.
Study group
The inclusion and exclusion criteria for the study group participants.

Magnetic resonance imaging (MRI) analysis
Magnetic resonance imaging (MRI; Signa Hde 1.5T General Electric Medical System, Poland) was used to assess the radiological advancement of degenerative changes in the lumbosacral spine. MRI was performed in the SE. T1, SE. T1 FLuid-Attenuated Inversion Recovery (FLAIR), FSE T2, and Short Tau Inversion Recovery (STIR) sequences in transverse and sagittal sections with 3 mm and 4 mm thick layers. Degeneration was quantified using the Pfirrmann scale. 29 Two expert neurosurgeons independently assessed the images and assigned Pfirrmann grades for the observed IVD degeneration.
Surgical procedure
A microdiscectomy procedure was performed on each patient in the study group. First, a skin incision was made in the lumbosacral region, and the paraspinal muscles were dissected. Next, the IVD sequester was removed, and the IVD was removed (discectomy). A drain was placed at the wound site for 24 h to drain the accumulating blood.
Control group
The control group consisted of 81 people (43 women, 53%; 38 men, 47%) with mean age 31.5 ± 1.5 years old. The biological material was obtained from Caucasian human cadavers during organ harvesting. Each postmortem examination was conducted within 48 h from the confirmed moment of death and after the patient’s death certificate was obtained.
The inclusion and exclusion criteria for the control group participants.

Intervertebral disc (IVD) collection from cadavers
After donor qualification and during the postmortem examination, the front surface of the lumbosacral spine section was exposed in a manner typical for a postmortem examination. A flat sectional knife was then used to completely to excise the appropriate IVD of the lumbosacral spine. The IVD was then placed in a disposable zip-lock bag and labeled with the date of collection, section number, and donor’s initials. The excellent quality of the IVD samples collected allowed for the separation of fragments from the nucleus pulposus, making it possible to perform additional analysis of the degeneration process.
There was no alternative method or source available for obtaining IVDs for the control group, and other researchers have used and described this method.30,31
Securing clinical material for molecular analysis
The IVDs of the lumbosacral spine obtained from the study group and the cadavers were carefully rinsed to remove any blood, preserved in RNAlater (Thermo Fisher Scientific, Waltham, MA, USA; catalog number AM7020), and stored at −80°C until used in the enzyme-linked immunosorbent assay (ELISA) and western blot analysis.
Hematoxylin-eosin (H&E) staining
The IVDs collected from the study group and the cadavers were assessed histologically via hematoxylin-eosin (H&E) staining using an H&E staining kit (Abcam, Cambridge, MA, USA; catalog number ab245880). Paraffin sections 5 μm thick were stained according to the manufacturer’s protocol. Two independent investigators evaluated each slide. A representative image of an H&E stained slide can be found in one of our previous publications. 28
Enzyme-linked immunosorbent assay (ELISA) for the determination of the concentration of GDNF and GAP-43 in human lumbosacral IVDs
Each collected IVD was cut into fragments approximately 1 mm in size, and the fragments were placed in a new tube and weighed. The samples were incubated in a solution of 4 M guanidine hydrochloride (Sigma Aldrich, St Louis, MO, USA; catalog number G3272), 1 M sodium acetate (Sigma Aldrich, St Louis, MO, USA; catalog number S2889-250G), Triton 2% (Sigma Aldrich, St Louis, MO, USA; catalog number X100), supplemented with protease inhibitor cocktail (Sigma Aldrich, St Louis, MO, USA; catalog number P8340) for 12 h at 4°C. The samples were then centrifuged (3000 rpm, 10 min), and the resulting supernatant was stored at −20°C.
A combination of polyclonal anti-GDNF antibody (STI, Poznan, Poland, catalog number bs-1024R) and polyclonal anti-GAP-43 antibody (STI, Poznan, Poland, catalog number bs-0154R) were used according to the manufacturers’ protocols.
The anti-GDNF and anti-GAP-43 antibodies were diluted in phosphate-buffered saline (PBS; pH 7.6; Merck, Sigma Aldrich, St Louis, MO, USA; catalog number P3744) and applied to Pierce NeutrAvidin plates (Thermo Fisher Scientific, Waltham, MA, USA; catalog number 15,509) and incubated at room temperature for 2 h. The plates were then washed three times with blocking solution (1% bovine serum albumin [BSA] in PBS; Sigma Aldrich, St Louis, MO, USA; catalog number A2153). Then 50 μL of standards or 1:20 diluted samples were added to individual wells. The plates were incubated for 3 h at room temperature, after which time the wells were washed three times with PBS. After this step, specific anti-GDNF or anti-GAP-43 antibodies conjugated with horseradish peroxidase (HRP) were added; the plates were then incubated for 3 h at room temperature. In the final step, each plate was washed three times with phosphate buffer and 100 μL of BM Chemiluminescence ELISA Substrate (buffered solution containing luminol/4-iodophenol and buffered solution containing a stabilized form of H2O2; Sigma Aldrich, St Louis, MO, USA; catalog number 11,582,950,001) was added. An M200PRO plate reader (Tecan, Männedorf, Switzerland) was used to evaluate absorbance at 540 nm.
Positive controls (cells from the HeLa human cervical cancer cell line) and negative controls (samples without primary antibody) were included in the assay. All samples were analyzed in duplicate. Mean values were used for analysis.
Western blot analysis
IVD samples placed in RNAlater solution (Thermo Fisher Scientific, Waltham, MA, USA; catalog number AM7020) were rinsed with PBS and placed in a 2 mL tube, to which 500 μL of radioimmunoprecipitation assay (RIPA) buffer (Sigma Aldrich, St Louis, MO, USA; catalog number R0278) supplemented with a cocktail of protease and phosphatase inhibitors (Sigma Aldrich, St Louis, MO, USA; catalog number PP1010) was added. A homogenizer (T18 Digital Ultra-Turrax, IKA Poland Sp. z o.o., Warsaw, Poland) was used to completely homogenize the samples, and they were then placed horizontally on ice and stirred on a rocking plate for 1 h. The samples were then centrifuged (12,000 g, 4°C, 10 min), and the supernatant was collected from each sample and stored at −80°C until further analysis.
After the samples were thawed, the total protein concentration was determined using a bicinchoninic acid (BCA) kit (Thermo Fisher, Waltham, MA, USA; catalog number 23,225) according to the product’s guidelines. All protein concentration measurements were calculated using a standard curve generated using BSA standard solutions (Sigma Aldrich, St Louis, MO, USA; catalog number A9576).
A combination of polyclonal anti-GDNF antibody (STI, Poznan, Poland, catalog number bs-1024R; 1:1000 dilution; molecular weight 15 kDa) and polyclonal anti-GAP-43 antibody (STI, Poznan, Poland, catalog number bs-0154R; 1:1000 dilution; molecular weight 43 kDa) were used, according to the manufacturers’ protocols. An HRP-conjugated goat anti-rabbit IgG (BioRad, Milan, Italy; catalog number 1,706,515; dilution 1:3000) was used as the secondary antibody. Glyceraldehyde 3-phosphate dehydrogenase (GAPDH; Santa Cruz Biotech, Dallas, Texas, USA; molecular weight 37 kDa; catalog number sc-25,778; 1:500 dilution) was used as an endogenous control protein.
An equal amount of protein (20 μg) was loaded into each well of a 12% sodium dodecyl sulfate polyacrylamide gel (POL-AURA, Dywity, Poland; catalog number PA-03-2962-D#25G), and electrophoretic separation was performed at 170V for 1.5 hours. The next step involved an overnight electrotransferring the proteins to a polyvinylidene difluoride (PVDF) membrane (pore size 0.45 μm; Thermo Fisher, Waltham, MA, USA; catalog number 88,518). After the protein transfer, membrane was blocked with 1X Tris-buffered saline (TBS) containing 0.1% Tween-20 (Sigma Aldrich, St Louis, MO, USA; catalog number 91,414) supplemented with 5% non-fat dry milk powder (Merck, Sigma Aldrich, St Louis, MO, USA; catalog number NIST1549A). An anti-GDNF or anti-GAP-43 primary antibody was then added, and the membrane was incubated for 12 h at 40°C). The membrane was then washed, and the secondary HRP- coniugated antibody was added. The optical density of each blot lane was measured using Kodak MI 4.5SE software (Kodak, Rochester, NY, USA). IVD samples to which no primary mouse monoclonal IgG1 κ anti-GDNF antibody was added were used as negative controls. Cells from the HeLa human cervical cancer cell line were used as positive controls.
Quantification of GDNF-positive and GAP-43-positive nerve fibers in human lumbosacral spine IVDs
IVDs dehydrated in a sucrose gradient were placed in the CRYOMATRIX reagent (Thermo Fisher Scientific, Waltham, MA, USA; catalog number 6,769,006) and frozen. The resultant blocks were cut into 8–10 μm thick sections using a freezing microtome (Leica Biosystems RM2235, Sydney, Australia). The sections were then placed on a Polysine® Menzel-Glaser glass slide (Thermo Fisher Scientific, Waltham, MA, USA) and stored at −20°C.
A combination of polyclonal anti-GDNF antibody (Abcam, Cambridge, MA; catalog number 18,956; 1:200 dilution) and anti-GAP-43 antibody (Novus Biologicals, USA; catalog number NBP1-92,714S; 1:200 dilution) was used, according to the manufacturers’ protocols.
The slides containing the IVD sections were thawed and washed with TBS supplemented with 0.1% Tween 20 (TBST; Avantor International Ltd., Gdansk, Poland; catalog number J77500.K8), and then non-specific binding sites were blocked by incubating the samples in blocking solution (10% goat serum in TBST) for 30 min at room temperature (Thermo Fisher Scientific, Waltham, MA, USA; catalog number 50,197Z). The slides were incubated for 24 h with either anti-GDNF or anti-GAP-43 primary antibody and then washed to remove any unbound primary antibody. Subsequently, the samples were incubated with anti-rabbit secondary antibody labeled with AlexaFluor 568 fluorescent dye (Thermo Fisher Scientific, Waltham, MA, USA; 1:500 dilution) for 2 h at room temperature in a dark chamber. After any excess secondary antibody was removed, the slides were sealed with Vectashield antifade mounting medium (Vector Laboratories, Newark, CA, USA; catalog number H-1000). Positive and negative controls were included.
The GDNF-positive and GAP-43-positive nerve fibers were quantified using fluorescence and confocal microscopy (Olympus, Tokyo, Japan). Sections containing the highest numbers of nerve fibers and/or immunopositive cells were visualized at the site of their highest concentration in two to four consecutive sections, for a total of 10–20 fields of view. To avoid double counting, only cells with a nucleus were counted. The area of one field of view was 0.0449 mm2. The results are expressed as arithmetic means per mm2 area.
Statistical analyses
The statistical analyses were performed using the STATISTICA 13 PL program (Krakow, Poland) at a statistical significance threshold of p < 0.05.
The results were first assessed in terms of normal distribution using the Shapiro–Wilk test. Due to the fulfillment of the test assumptions, further statistical analyses were conducted using parametric methods.
To determine whether the observed differences in the number of either the GAP-43-positive or GDNF-positive fibers and in the GAP-43 or GDNF concentration between the study and control groups were statistically significant, a Student’s t-test was performed for independent groups. However, when comparing the number of GAP-43-positive or GDNF-positive fibers and GAP-43 or GDNF concentration in the study group according to the Pfirrmann scale scores, the one-way ANOVA and Tukey’s post hoc test were used. The data are presented as means and standard deviations, and include a 95% confidence interval (95% CI). Before conducting the ANOVA, homogeneity of variance was checked using Leven’s test.
Results
The GAP-43 and GDNF concentrations varied in the degenerated IVDs with Pfirrmann grade
The GAP-43 and GDNF concentrations found in the degenerated and healthy intervertebral discs and the corresponding Pfirrmann grades.

aStudy vs. Control.
bPfirrmann 2 vs. Pfirrmann 3
cPfirrmann 3 vs. Pfirrmann 4
dPfirrmann 4 vs. Pfirrmann 5
ePfirrmann 2 vs. Pfirrmann 4
fPfirrmann 2 vs. Pfirrmann 5
gPfirrmann 3 vs. Pfirrmann 5; GDNF, glial cell-derived neurotrophic factor; GAP-43, growth associated protein 43; 95%Cl, 95% confidence interval.
Confirmation by western blot that the GAP-43 and GDNF concentrations varied in the degenerated IVDs with Pfirrmann grade
Next, we evaluated the concentration of GAP-43 and GDNF in the degenerated and healthy IVD samples using the western blot technique. The positive-control protein, GAPDH, was expressed in each sample at similar levels, demonstrating that the technique was performing as required. The highest levels of GAP-43 expression were noted in samples graded Pfirrmann 4, and GAP-43 expression was higher in the study samples than in the control samples. Similarly, the highest levels of GDNF expression were noted in samples graded Pfirrmann 4, and lower protein expression was observed in the grade 5 lesions in the study group than in the control group. An example electropherogram is shown in Figure 1. Expression of GAP-43 and GDNF in IVD samples from the control group and from the study group. The expression levels were determined using the western blot technique. GDNF, glial cell-derived neurotrophic factor; GAP-43, growth associated protein 43; P2, P3, P4, and P5, Pfirrmann grade 2, 3, 4, and 5, respectively.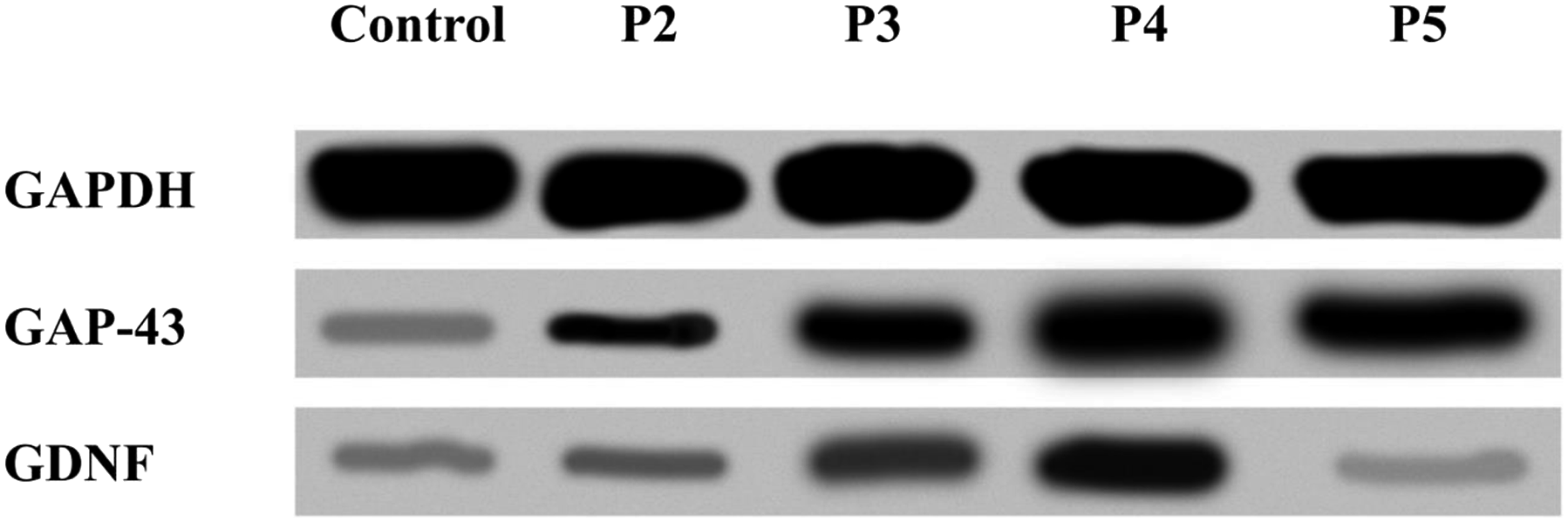
The number of GAP-43-positive and GDNF-positive nerve fibers varied in the degenerated IVDs with the Pfirrmann grade
Number of GAP-43-positive and GDNF-positive nerve fibers in the examined degenerated and healthy intervertebral discs.

aStudy vs. Control.
bPfirrmann 2 vs. Pfirrmann 3
cPfirrmann 3 vs. Pfirrmann 4
dPfirrmann 4 vs. Pfirrmann 5
ePfirrmann 2 vs. Pfirrmann 4
fPfirrmann 2 vs. Pfirrmann 5
gPfirrmann 3 vs. Pfirrmann 5; GDNF, glial cell-derived neurotrophic factor; GAP-43, growth associated protein 43; 95%Cl, 95% confidence interval.
Discussion
It is difficult to imagine what medicine would look like today without the recent significant developments in molecular biology that have produced vital tools, techniques, and treatments, and furthered our understanding of conditions.32,33 In our work, we have taken a molecular biology-based approach to evaluate the role of GAP-43 and GDNF in the degeneration of human IVDs of the lumbosacral region of the spine and the development of discogenic pain. The findings of this study show that there is a significant association between IVD degeneration and GDNF and GAP-43 concentration, as well as the number of GDNF-positive and GAP-43-positive nerve fibers.
However, there is limited information available in the literature about the role of GDNF and GAP-43 in the IVD degenerative process and the development of discogenic pain that accompanies degenerative spine disease.5–7,34,35 Yamada et al. also observed that a significantly higher concentration of GDNF was associated with more severe degenerative changes in IVDs. 36 It must also be noted that they established a chondrocyte cell culture from obtained IVD samples and exposed the cells to pro-inflammatory factors, such as interleukin 1 beta (IL-1β). 36 Yamada et al. also emphasized that GDNF is an important factor in the generation and generation and transmission of discogenic pain. 36 Thus, it seems that in a microenvironment in which advanced degeneration occurs, the expression of GDNF increases under the influence of pro-inflammatory factors.37,38
Our findings indicate that there is a significant increase in GDNF concentration and an invasion of GDNF-positive nerve fibers in degenerated IVDs. However, it is interesting to note that when the degree of radiological degeneration was graded 5, there was a significant decrease in the concentration of GDNF and the number and concentration of GDNF-positive nerve fibers. Therefore, it is possible that the number of nerve fibers and the concentration of neurotrophic factors increase as the degree of IVD degenerative changes increases only until a certain stage, at which point it seems that the intercellular matrix still enables the occurrence of biochemical processes. 39
Although, Jung et al., in their work on the mechanism of discogenic pain, have proven that administration (by injection) of Freund’s adjuvant into the structures of disc L5–L6 in rats results in the overexpression of GDNF in the dorsal root thalamus and the dorsal root ganglia. 40 Based on their findings, it may be concluded that an increase in the concentration of GDNF in degenerated IVDs may the influence the transmission and nociceptive pain signals. 40 In turn, the findings of Boucher et al. suggest that GDNF may be a promising therapeutic target in combating neuropathic pain, the etiopathogenesis of which has not been studied to a sufficient degree; current treatment modalities are often insufficient because GDNF causes a decrease in ectopic release in the damaged sensory neurons. 41 Similar observations were made by Fang et al., who used an anti-GDNF antibody to silence the expression of GDNF in rats that were first administered Freund’s adjuvant intrathecally. 42 The pain did not immediately subside; however, there was a noticeable reduction in the painful sensations during the observation period.43,44 This suggests that GDNF may suppress mechanic hyperalgesia in a model of delayed inflammatory pain.43,44 In addition, Amaya et al. have shown that inhibiting the activity of GDNF alleviates thermal hyperesthesia connected with the acute phase of inflammation, and as a result, it may be assumed that restoring the physiological concentration of GDNF constitutes a promising therapeutic approach to treating hyperalgesia induced by a chronic inflammatory condition. 45 In the context of our study, the measurement of the concentration of GAP-43 has added significance, as it constitutes a marker for the newly created nerve fibers that contribute to the creation of nociceptive stimuli during IVD degeneration.18–20
Furthermore, our findings suggest that in the most advanced cases of IVD degeneration, there is no mass penetration of new free nerve endings into the IVD.6,46–48 This conclusion is based on the observation that the number of GAP-43-positive fibers and the concentration of GAP-43 were both significantly lower in the samples obtained from patients with degenerative changes graded 5 than in samples from patients with less severe degenerative changes.
During the progressive degeneration of human IVDs, GAP-43-positive nerve fibers from the neurons of dorsal root ganglia penetrate the IVDs to various depths. These are essentially nociceptive and proprioceptive sensory fibers.10,16 As shown earlier, greater innervation of the degenerating IVDs is associated with, among other activities, changes in the concentrations of various unidentified and identified factors, including neurotrophic and growth factors (e.g. GDNF) and many lesser-known pro-inflammatory cytokines. 49 Buma et al. conducted an experiment using an animal model to evaluate innervation of the knee joint. 50 They induced arthrosis in knee joints of mice that were several weeks old by administering a solution of bacterial collagenase, and administered a physiological salt solution to other knee joints that served as controls. 50 They observed a statistically significant higher number of GAP-43-positive nerve fibers in the area of the arthropathic joints compared with the healthy joints.17,50–52 Similarly, Jiang et al. examined the association between GAP-43 expression and nerve quantities in rats with degenerated IVDs and found that the GAP-43 mRNA and protein levels in degenerated IVDs were significantly higher than those in the IVDs of the control group. 53 Critically, this study showed that high expression of GAP-43 can occur in degenerated areas of IVDs, along with an accompanying inflammatory condition in the area. 53 In addition, Inoue et al. studied degenerated IVDs in rats and used GAP-43 as a marker to evaluate the regeneration of axons, which they assumed was induced after damage to the disc. 54 Our results confirmed that disc herniation outside the fibrous ring induces damage to the nerves and an increase in the regeneration of axons in the discs.55,56 Results obtained by us suggest that GAP-43 may be an effective marker in the identification of neoinnervation in the area of degenerated IVDs.57,58
The current study has both strengths and weaknesses. In terms of the strengths, human IVDs were used, a control group was included, and relatively high numbers of participants were included in both the study and control groups. These factors make it possible to identify the actual patterns of expression of evaluated factors in a given pathology. It must also be taken into account that it is challenging to prepare IVD material for molecular studies. Further studies are required to determine the expression of other neurotrophins and pro-inflammatory factors in both degenerated and healthy IVDs, to identify the correlation between results obtained from IVDs and peripheral blood, to establish a cell culture of chondrocytes from the collected IVDs, and to examine the effects of selected pro-inflammatory factors, drugs, and siRNA vectors on the cells. “Such experiments will provide critical information on the role neurotrophins play in IVD degeneration and discogenic pain and on potential therapies.
In conclusion, we have confirmed that during the degeneration of IVDs in the lumbosacral region of the spine, newly created nerve endings invade the degenerating IVDs. Our findings indicate that it is necessary to undertake further research to understand the factors and mechanisms involved, especially the roles of GAP-43 and GDNF in the generation and maintenance of discogenic pain. This study is important because it provides valuable knowledge about the roles of GDNF and GAP-43 in degenerative IVD disease, which is still insufficient.
Footnotes
Author Contributions
Conceptualization: RS, DG, and WM; methodology: RS, TF, WG, and; software: BOG and KB; investigation: RS, M. Gralewski, and DG; resources: M. Garczarek, and M. Gadzieliński; data curation: RS; writing—original draft preparation: RS, DG, M. Gralewski, and KB; writing—review and editing: RS; supervision: RS and BOG; project administration: RS, WM, and BOG. All authors have read and agreed to the published version of the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
