Abstract
Pain involves both sensory and affective dimensions. The amygdala is a key player in linking nociceptive stimuli to negative emotional behaviors or affective states. Relief of pain is rewarding and activates brain reward circuits. Whether the reward circuit from the ventral tegmental area (VTA) to the central amygdala (CeA) is involved in pain relief remains unexplored. Using a model of experimental postsurgical pain, we found that pain relief elicited conditioned place preference (CPP), activated CeA-projecting dopaminergic cells in the VTA, and decreased dopaminergic D2 receptor expression in the CeA. Activation of the VTA–CeA neural pathway using optogenetic approaches relieved incisional pain. Administration of a D2 receptor agonist reversed the pain relief elicited by light-induced activation of the VTA-CeA pathway. These findings indicate that the VTA-CeA circuit is involved in pain relief in mice via dopamine receptor D2 in the CeA.
Introduction
Pain is a multidimensional conscious experience that involves sensory and negative affective components. 1 Relief of pain is rewarding and activates brain reward/motivation circuits, via which dopaminergic inputs from the ventral tegmental area (VTA) innervate brain regions, including nucleus accumbens (NAc), amygdala and so on.2,3 The amygdala is a forebrain structure composed of several distinct nuclei, and is thought to be a key neural substrate for integrating diverse emotional states with pain. 4 Emerging studies have shown the central amygdala (CeA) can play a role in driving reward-related behaviors. 5 However, the precise functional connectivity between VTA dopaminergic projection neurons and their postsynaptic targets in the CeA and the role of this circuit in pain relief have not been described. Knowledge of the brain reward circuit (from the VTA to the CeA) could lead to the development of new therapeutics for the treatment of the sensory aspects of pain and comorbid emotional symptoms.
Ongoing pain is difficult to demonstrate in animals. However, in the presence of ongoing pain, pairing of manipulations that are not rewarding but relieve pain, such as peripheral nerve block (PNB), elicits conditioned place preference (CPP).6–8 Thus, CPP resulting from pain relief indicates reward and is also an important measure of ongoing pain. Dopamine receptors are generally classified into two major subclasses, i.e., D1-like (D1, D5) and D2-like (D2, D3, D4) receptors, according to their pharmacological characteristics and associated signaling pathways. 9 In the rodent central nervous system, the most abundant dopamine receptors are D1 receptors and D2 receptors. Despite extensive studies on the role of dopamine receptors in nociception processing, how different types of dopamine receptors contribute to pain relief remains elusive.
In this study, we used dopamine transporter (DAT)-Cre transgenic mice, optogenetic stimulation and behavioral pharmacology approaches to determine the role of dopaminergic projections from the VTA to the CeA in the relief of incisional pain and to clarify the effects of D1 and D2 receptors in the VTA-CeA circuit in the modulation of pain.
Materials and methods
Animals
Male wild-type C57BL/6 mice (8–10 weeks) and DAT-Cre bacterial artificial chromosome transgenic mice (8–10 weeks) were used in this study. Dopamine transporter-Cre mice were purchased from Shanghai NanFang Model Biotechnology Co. The Cre mice were on the C57BL/6 genetic background. The mice were housed under standard conditions on a 12-hour light-dark cycle with ad libitum access to water and food pellets. All behavioral tests were conducted by an investigator blinded to the treatment groups. All experiments were conducted in accordance with the ethical guidelines of the National Institutes of Health and the International Association for the Study of Pain.
Animal model of plantar incision pain
All mice were anesthetized with 2% halothane delivered via a nose cone. A 0.5-cm longitudinal incision was made in the skin and fascia of the left plantar aspect of the foot starting 0.5 cm from the proximal edge of the heel and toward the toes with a number 11 blade. In most animals, the plantaris muscle was elevated and incised longitudinally. Sham-operated animals were anesthetized, and the left hind paw was cleaned, but no incision was made. Peripheral nerve block was achieved by injection of 100 μL of lidocaine or saline (1%, wt/vol) into the PF under isoflurane anesthesia. The muscle origin and insertion were kept intact. After hemostasis with gentle pressure, the skin was closed with 2 5-0 nylon sutures and an FS-2 needle. The wound site was covered with neomycin, and the animals were allowed to recover in their cages.
Nociception test
Mechanical allodynia was assayed using von Frey filaments as described by Chaplan et al. 10 The experiment was performed by two authors who were blinded to the experimental treatments. The mice were placed on wire mesh platforms in clear cylindrical plastic enclosures, and filaments were applied to the center of the plantar surface of the unincised hind paw or to the wound edge of the incised hind paw. Withdrawal of the hind paw from the floor was scored as a response. When no response was obtained, the filament of the next highest force in the series was applied to the same paw; if a response was obtained, the with the next lowest force was applied. The paw withdrawal threshold was estimated based on this testing protocol. The “up-down” approach was used to calculate mechanical sensitivity. 10 The mechanical pain threshold of the mice was measured before light stimulation, and then a blue light (465 nm) with a frequency of 20 Hz and intensity of 10 mW was applied for 2 minutes, and turned off for 2 minutes. Afterwards, the mechanical pain threshold was measured again.
The thermal paw withdrawal latency (PWL) was determined using the plantar test (Ugo Basile, Varese, Italy). Each mouse was placed on a transparent glass plate over a radiant heat generator for 30 min for acclimatization. The intensity of the light source was adjusted so that normal animals exhibited a retraction latency of 12–15 s, and a cut-off time was set in advance to prevent tissue damage. The latency to withdrawal from the heat stimulus was measured three times at an interval of ≥5 min, and the average latency in the three tests was calculated as the PWL.
Conditioned place preference (CPP)
A single-trial conditioning protocol was used to assess CPP as previously described.7,11 All mice were handled by the experimenter before the preconditioning phase. On the preconditioning day, the mice were placed into the CPP boxes and given access to all chambers; the time spent in each chamber over 15 minutes was determined by an automated system to verify that the mice did not show a chamber preference during preconditioning. Following preconditioning, the mice underwent incision or a sham procedure and were placed in their home cages overnight. On the conditioning day (24 h postincision), the mice received saline (100 μL) injection into the PF and were immediately (within 2 min) placed in the appropriate pairing chamber. Four hours later, the mice received lidocaine injection (1%, wt/vol; 100 μL) into the PF and were placed in the opposite chamber. Chamber pairings were counterbalanced. On the test day, 20 h following the afternoon pairing, the mice were placed in the CPP box and given access to all chambers, and their behavior was recorded for 15 min for analysis of chamber preference.
Open field test (OFT)
The open field test (OFT), a classical test for measuring anxiety-like behaviors in rodents, 12 was conducted in an illuminated chamber (45*45*30 cm) divided into a central zone and an outer zone. In the real-time OFT, the mice were allowed to explore freely for 10 min, and the locomotor activity of each animal in the two zones was video-recorded and analyzed with an automated video-tracking system (EthoVision XT, Noldus Information Technology). The 10 min of the trial was divided into two consecutive 5 min periods: the light was kept off in the first period (control), while light stimulation was applied during the second 5 min period. A reduced time spent in the unprotected central zone (central time) was regarded as an indicator of anxiety-related behavior. The total distance traveled in the entire chamber during the test was recorded and used as a measure of general locomotor activity.
Elevated plus maze (EPM) test
The EPM consisted of a central platform (6 × 6 cm2), two closed arms (30 × 6 × 20 cm3) and two opposing open arms (30 × 6 cm2). The maze was placed 100 cm above the floor. A mouse was placed in the central platform facing a closed arm and was allowed to explore the maze for 5 min. The time spent in the open arms and the number of entries into the open arms were analyzed using Smart3.0 software. The area was cleaned between tests using 75% ethanol. The percentage of time spent in the open arms and the percentage of time spent in the open arms were measured to assess anxiety-like behaviors.
Retrograde tracing
Mice were anesthetized with 2% isoflurane and placed in a stereotaxic frame. Then, 0.2 µl of the retrograde tracer fluorogold (FG) (4%; Thermo Fisher) was injected into the CeA (AP: 1.22 mm; ML: 2.8 mm; DV: 4.8 mm). 13 as described previously. 14 The needle was left in place for an additional 10 minutes before withdrawn from the brain. At 7 days after injection, the mice were deeply anesthetized with isoflurane and perfused with 4% paraformaldehyde for imaging or immunohistochemistry.
Drug administration
A D2 receptor antagonist (1 mg/mL; Sigma) was injected with a 30-gauge needle into the CeA (AP: 1.22 mm; ML: 2.8 mm; DV: 4.8 mm) to regulate the activity of D2 receptor. The injection volume was 1 µL.
Optical stimulation
Four weeks after virus injection, a fiber optic cannula (Doric Lenses) was stereotaxically implanted in the brains of anesthetized mice just above the CeA (AP: 1.22 mm; ML: 2.8 mm; DV: 4.8 mm). After implantation, the animals were singly housed and allowed to recover for 7 d before behavioral tests. For optical stimulation, we used a power supply (Plexon 08-06-A-37) with an output power of 10 mW and a light wavelength of 465 nm. The laser properties were modulated by a waveform generator (Plexon Inc.; 6500 Greenville Avenue, Suite 700, USA) that allowed us to control the frequency, pulse square, and pulse width. Blue light pulses of 20 Hz, 15 ms, and 10 mW were delivered, and the laser intensity was set at 10 mW, with continuous light exposure for optical stimulation. Optical modulation was performed prior to (pre), during (laser-on), and after (post) optical stimulation for 5 minutes each.
Stereotaxic surgeries
All mice were anesthetized with 1%–3% isoflurane and placed in a stereotaxic frame (David Kopf Instruments). Viruses (AAV8-EF1a-DIO-ChR2-mCherry; OBIO Technology; Shanghai) (0.5 µL) were injected into the VTA (AP: 3.1 mm; ML: 0.8 mm; DV: 4.25 mm) with a 1 µL Hamilton syringe at a speed of 0.05 µL/min. The needle was left in place for an additional 10 min and then slowly withdrawn. Immunohistochemistry was used to confirm that the stereotaxically injected viruses adequately transduced the VTA without significant spread to other regions. Optical fibers (200 μm inner diameter, N.A. 0.66; Doric Lenses) were implanted into the CeA in all mice. All injections and implantation procedures were performed in 7- to 8-week-old male DAT-cre animals.
Western blotting
Mouse brain tissues were fully ground in lysis buffer A (10 mM Tris, 1 mM phenylmethylsulfonyl fluoride, 5 mM MgCl2, 5 mM EGTA, 1 mM DTT, 100 mM NP-40, 40 mM leupeptin, and 250 mM sucrose) and stored on ice for 30 min before centrifugation at 4°C and 1000 × g for 15 min. The protein concentration was then measured using a bicinchoninic acid protein assay (Solarbio, Beijing, China). Samples (30 μg total protein) were mixed with protein loading buffer (Solarbio, Beijing, China) and heated at 99°C for 5 min before being loaded onto 10% sodium dodecyl sulfate-polyacrylamide gels (CWBIO, China) for electrophoresis. The proteins were transferred onto polyvinylidene fluoride membranes (Immobilon®-P, Merck KGaA, Darmstadt, Germany) at 30 V and 4°C overnight. After being blocked with 3% bovine serum albumin (BSA) (Solarbio, Beijing, China) and incubated overnight at 4°C with primary antibodies against D1R (1:1000; catalog No. MAB5290, Millipore), D2R (1:1000 catalog No. AB5084P, Millipore), and GAPDH (catalog No. RM2002 L, Beijing Ray), the membranes were washed six times for 10 min each in Tris-buffered saline and 1% Tween. The membranes were then incubated with horseradish peroxidase-conjugated anti-rabbit secondary antibody (1:1000, Jackson laboratories) for 2 h at room temperature. The signals were visualized after processing with an enhanced chemiluminescence (ECL) prime kit and FluorChem E (AlphaImager ProteinSimple, San Jose, CA, USA). The average band density was determined using ImageJ software.
Immunohistochemistry

Statistical analysis
All data were analyzed using Prism software, version 9.0 (GraphPad, La Jolla, CA). The data are presented as the means ± SEMs. One-way analyses of variance (ANOVA) followed by Tukey’s tests for multiple comparisons was used to compare the Western blot results among three or more groups. Two-way ANOVA followed by Bonferroni’s multiple comparison test was used to analyze the behavioral data. Student’s t tests were used for comparisons between two groups. Main and interaction effects were considered significant at p < 0.05.
Results
Experimental plantar incision produced significant mechanical allodynia, thermal hyperalgesia and anxiety–like behaviors in mice
Our study showed that compared with those in the sham group, mice that underwent incision exhibited significant mechanical allodynia and thermal hyperalgesia. Compared with those in the sham group, mice in the incision group displayed a significant reduction in the mechanical paw withdrawal threshold (PWT) and thermal paw withdrawal latency (PWL) from 6 h to 3 d after surgery (Figure 1(a), (c)). There was no significant between-group difference in the PWT and PWL on the contralateral side (Figure 1(b), (d)). Effects of experimental surgical incision on mechanical and thermal nociceptive thresholds and anxiety-like behaviors in mice. (a–d) The ipsilateral mechanical paw withdrawal threshold (PWT) and thermal paw withdrawal latency (PWL) were decreased in mice that underwent incision from 6 h to 3 d after surgery (sham,n = 8; incision,n = 8,***p < 0.001 vs.the sham group; two-way ANOVA, mechanical:F(2,26) = 11.05,***p < 0.001,thermal:F(2,30) = 9.576, ***p < 0.001). (e–h) Experimental plantar incision induced obvious negative emotions on day 3 after surgery, as indicated by the open field test (OFT) (e, f, n = 6 per group; t test,**p < 0.01, ,t = 3.575) and EPM test (g, h) (n = 6 per group; t test,**p < 0.01,t = 4.064, ***p < 0.001 vs the sham group; t = 5.823).
Pain, as a strong stressor, is known to cause comorbid emotional disorders, including anxiety, stress, and depression in pain patients.15–17 Our study showed that hind paw incision induced anxiety-like behaviors, as determined by the OFT and EPM test. Compared with the sham group, the incision group showed a significant reduction in the percentage of time spent in the center zone in the OFT (Figure 1(e)) and a significant reduction in the percentage of OA entries and time spent in the OAs in the EPM test (Figure 1(g), (h)). No significant difference in total travel distance in the OFT was observed between the incision group and the sham operation (Figure 1(f)). These results indicated that mice exhibited obvious sensory pain symptoms and negative emotions after surgical incisions.
Pain relief elicited CPP and activated VTA neurons
The sensation of pain generates an aversive state that demands a behavioral response (for pain, a motivation to seek relief). In contrast, relief of pain and return to homeostatic balance are rewarding.3,18,19 The specific brain circuits that process pain relief, however, remain elusive. As demonstrated in Figure 1, evoked pain hypersensitivity was still obvious at 6 h. Peripheral nerve block (PNB) with popliteal fossa (PF) lidocaine injection 6 h postincision resulted in a strong preference for the chamber paired with PNB, indicating pain relief. In contrast, in sham-operated animals, CPP for the PNB-paired chamber was not observed (Figure 2(b)). PNB on the side contralateral to the injured hind paw did not result in CPP, confirming that lidocaine at the dose injected into the PF injection did not induce systemic pain relief, as demonstrated previously.
20
These results showed that relief of ongoing incisional pain produced CPP and pain relief-elicited reward. Pain relief elicited CPP and activated VTA neurons. (a–b) The time spent in the chambers paired with popliteal fossa (PF) injection of lidocaine (n = 6; ***p < 0.001,Interaction:F (2, 30) = 15.05; vs the saline group; two-way ANOVA). (c) A coronal section view of the ventral tegmental area (VTA) labeled with AAV8-EF1α-DIO-mCherry in a DAT-Cre mouse. (d–e) Lidocaine block increased the number of Fos-positive cells in the VTA in injured mice (n = 6; **p < 0.01,Interaction; F (1, 20) = 14.51; two-way ANOVA; scale bars, 500 μm and 50 µm).
Emerging preclinical data have shown that offset of an acute noxious stimulus activates brain regions, particularly the ventral tegmental area (VTA)21,22 Activation of brain regions in injured mice following PNB was investigated using immunohistochemistry for FOS, a marker of neuronal activity. The results showed that PNB significantly increased the number of Fos-positive cells in the VTA in injured mice. Incision injury itself or PNB in sham mice did not change FOS expression (Figure 2(c)–(e)). The results indicated that activation of VTA neurons mediated the reward of pain relief. However, the c-Fos + cell count in the NAc or amygdala did not change significantly after pain relief although findings have demonstrated that VTA-NAc circuit participated in pain relief.23,24
DAergic projections from the VTA to the CeA in mice
To identify VTA projections to the CeA, the retrograde tracer FG was injected into the CeA. This resulted in retrograde labeling of many CeA-projecting neurons in the VTA (Figure 3(a)). These projections were bilateral, with obvious predominance on the ipsilateral side. DAergic projections from the VTA to the CeA in mice. (a) Photomicrographs showing FG injection sites in the CeA and the resultant FG-labeled neurons in the VTA. Scale bars 500 µm and 50 µm. (b) CeA-projecting neurons in the VTA (FG-labeled neurons) were dopaminergic (positive for TH). Scale bar, 500 µm. The lower panel shows the expression of FG and TH in the respective areas indicated in the upper panel at high magnification. Scale bar, 50 µm. (c) Local dopaminergic neurons (positive for TH) in the VTA were successfully transfected with AAV8-EF1α-DIO-ChR2-mCherry (AAV-ChR2-mCherry). Scale bars, 500 µm and 50 µm. (d) Photomicrographs showing AAV8-EF1α-DIO-ChR2-mCherry or AAV8-EF1α-DIO-mCherry injection sites in the VTA and the resultant mCherry-labeled axonal fibers and terminals in the CeA. Scale bar, 500 µm and 50 µm.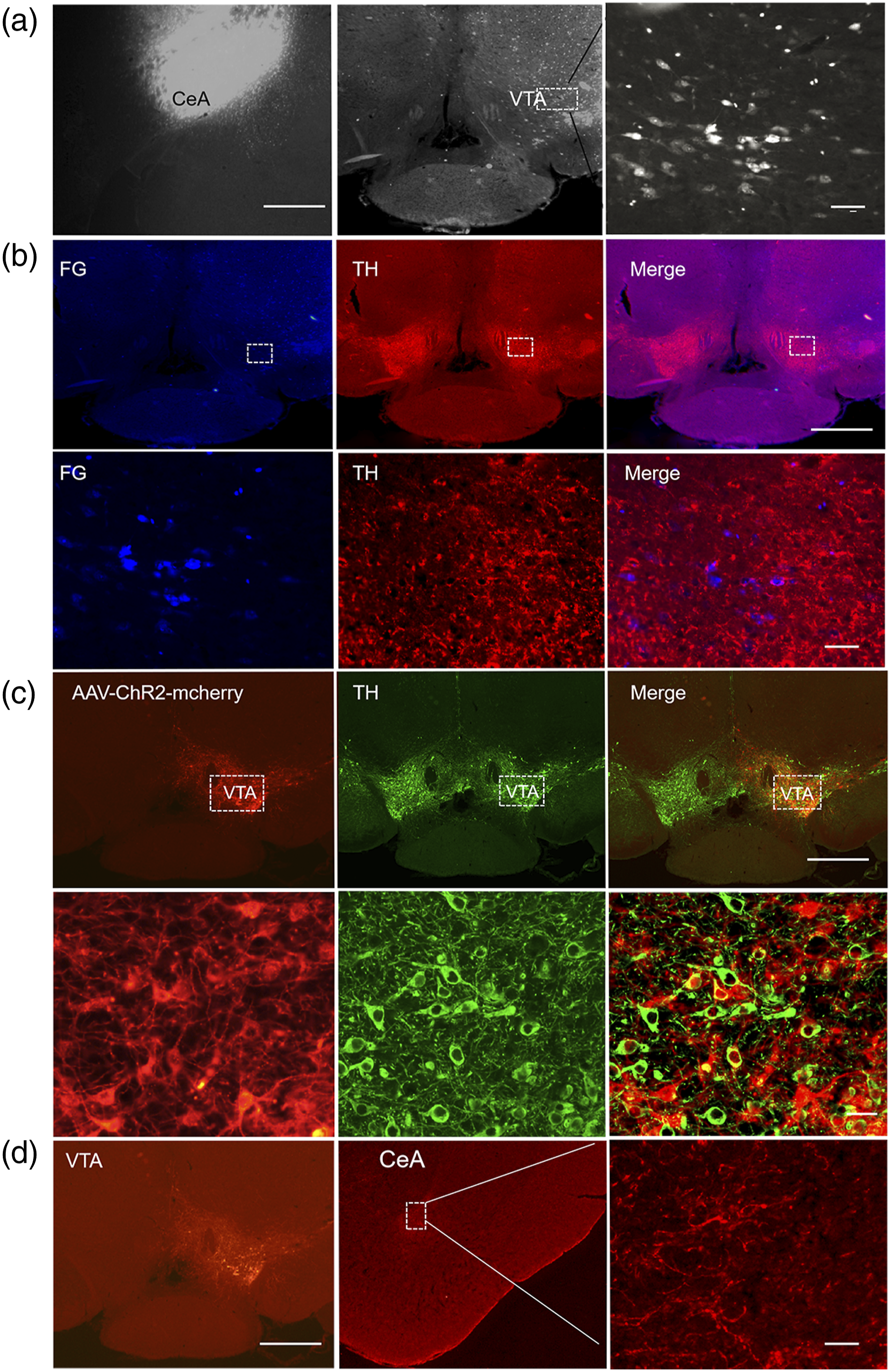
To determine whether the observed retrogradely labeled neurons in the VTA were catecholaminergic (dopaminergic), we performed immunohistochemistry for tyrosine hydroxylase (TH), the rate-limiting enzyme in dopamine synthesis and a marker of dopaminergic neurons. Our double staining data showed that CeA-projecting neurons in the VTA were dopaminergic (Figure 3(b)), suggesting that the CeA received projections from the VTA and may have been under the control of dopamine.
To characterize the functional connections of the VTA-CeA circuit, AAV8-EF1α-DIO-ChR2-mCherry was injected into the VTA of DAT-Cre mice (Figure 3(c)). Our double staining data showed that ChR2-mCherry-labeled neurons in the VTA were positive for TH, suggesting that the virus successfully transfected local dopaminergic neurons through the DAT promoter (Figure 3(c)). Four weeks later, we examined mCherry expression in the CeA. Abundant mCherry-labeled fibers were observed in the CeA (Figure 3(d)). These results illustrated the existence of strong, target-specific dopaminergic projections from the VTA to the CeA, suggesting that the VTA-CeA circuit might contribute to pain relief.
Activation of the VTA–CeA neural pathway relieved incisional pain
To explore the potential role of dopaminergic projection from the VTA to the CeA in incisional pain, we used an optogenetic approach and DAT-Cre mice injected with AAV8-EF1α-DIO-ChR2-mCherry (AAV-ChR2) or the control AAV8-EF1α-DIO-mCherry (AAV-mCherry) into the VTA. During specific activation of the CeA-projecting fibers and the terminals originating from the VTA, the mechanical thresholds of the bilateral hind paws were tested, and behavior in the open field test and CPP test were assessed (Figure 4(a)). Activation of the VTA–CeA neural pathway relieved incisional pain. (a) Experimental schedule. Behavioral tests, cannula implantation, microinjection, and incision were conducted as indicated in the schedule. (b) The mechanical threshold of the ipsilateral hind paw was significantly increased in incised mice that received AAV-ChR2 virus injection during blue light stimulation. (n = 6; **p < 0.01,Interaction:F (1, 24) = 8.197, vs the no light group; two-way ANOVA). (c) The percentage of time spent in the center zone in the OFT was significantly increased in the AAV-ChR2-injected group during light stimulation. However, optogenetic stimulation had no obvious effects on the total distance traveled in either the AAV-ChR2-injected group or the AAV-mCherry-injected group. (n = 6; **p < 0.01,Interaction:F (1, 21) = 12.17,vs the no light group; two-way ANOVA) (d) In the CPP test, the time spent in the chamber paired with light stimulation was significantly increased in the AAV-ChR2-injected group, whereas the time spent in the chamber paired with light stimulation was not significantly changed in the AAV-mCherry-injected group (n = 6; **p < 0.01,F (1, 20) = 14.25 vs the no light group).
We found that specific excitation of the excitatory opsin ChR2 in dopaminergic projections from the VTA to the CeA with light significantly increased the mechanical threshold of the ipsilateral hind paw in incised mice. Injection of AAV-ChR2 during blue light stimulation exerted obvious analgesic effects on the ipsilateral hind paw in incised mice (Figure 4(b)). In incised mice infected with AAV-mCherry, light stimulation had no effects on the mechanical threshold of the ipsilateral hind paw. Optogenetic stimulation had no effect on the mechanical threshold of the contralateral hind paw in either the AAV-ChR2-injected group or the AAV-mCherry-injected group (Figure 4(b)).
The percentage of time that AAV-ChR2-injected incised mice spent in the center zone in the OFT was significantly increased during light stimulation. However, optogenetic stimulation had no obvious effect on the total distance traveled in either the AAV-ChR2-injected group or the AAV-mCherry-injected group (Figure 4(c)).
According to the CPP test, the time spent in the chamber paired with light stimulation was significantly increased in the AAV-ChR2-injected group, whereas the time spent in the chamber paired with light stimulation was not significantly changed in the AAV-mCherry-injected group (Figure 4(d)). This directly demonstrated that activation of the VTA–CeA pathway contributed to reward seeking resulting from pain relief.
These results of the optogenetic experiments suggested that specific activation of the VTA–CeA neural pathway produced analgesic (pain relief) and anxiolytic effects in incised mice. The VTA–CeA neural pathway was involved in the modulation of nociception and comorbid emotional behavior deficits.
Pain relief decreased dopaminergic D2 receptor expression in injured mice
The above results provide functional evidence that specific activation of VTA-projecting dopaminergic neurons in the CeA via stimulation of fibers from the VTA produce analgesic and anxiolytic effects in incised mice. However, the potential underlying mechanism remains unknown.
The expression of the D1 receptor and D2 receptor in the CeA was assessed by using Western blotting. The expression of D2 receptor was significantly decreased in the CeA in incised mice treated with PNB compared with incised mice treated with saline. However, there was no significant difference in dopamine D1 receptor expression between groups (Figure 5(a)). Pain relief increased dopaminergic D2 receptor expression in injured mice. D2 receptor expression in the CeA was significantly decreased in incised mice treated with PNB compared with incised mice treated with saline (n = 6; *p < 0.05 vs the incision+saline group; one-way ANOVA,Interaction::F (3, 20) = 6.274). The expression of D1 receptor was not significantly changed. (b) In the CeA, D1 receptor was mainly expressed in glutamatergic excitatory neurons (positive for glutaminase). In the CeA, D2 receptor was predominantly expressed in GABAergic inhibitory neurons (positive for GABA). Scale bar, 50 μm.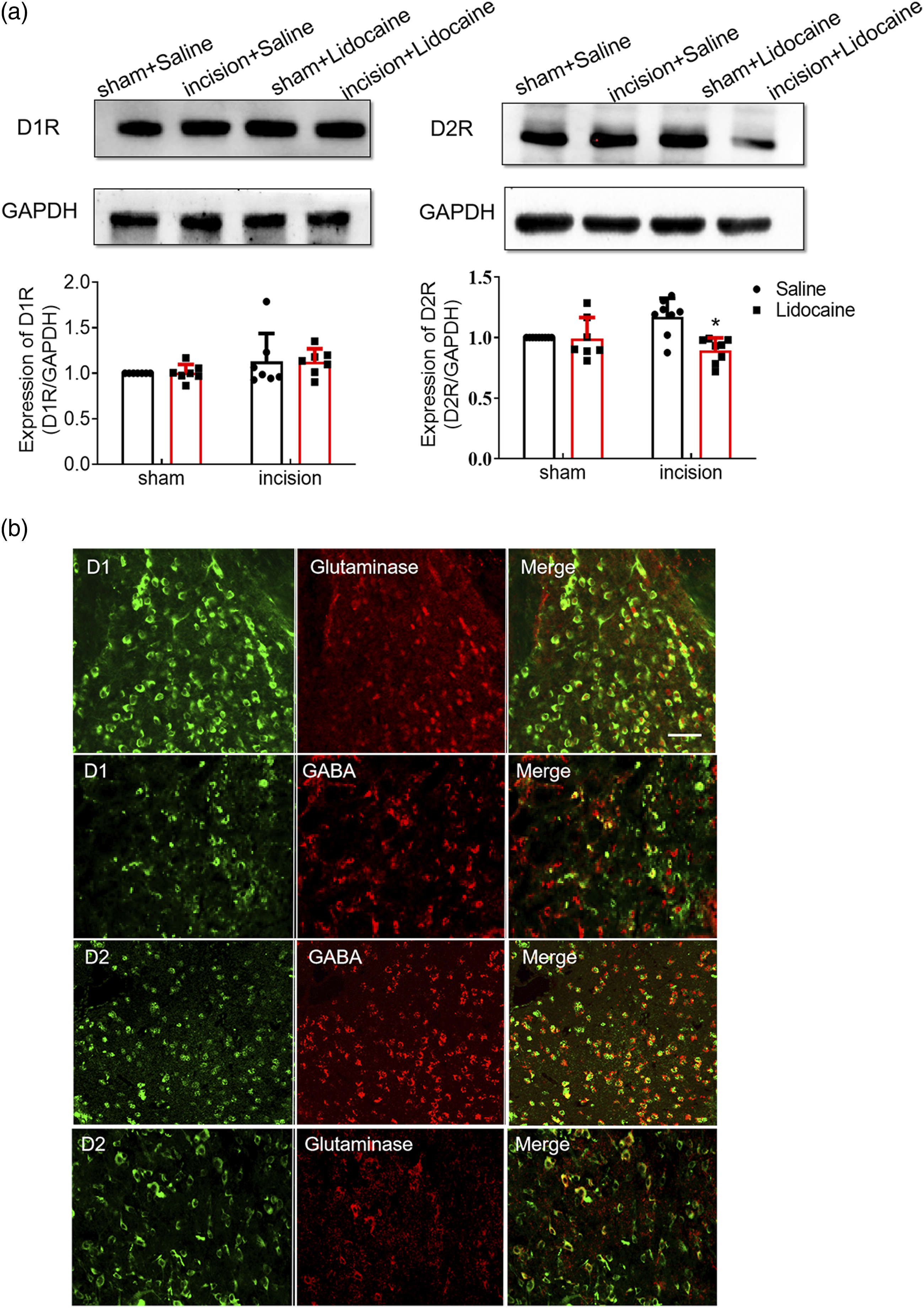
Furthermore, we examined the neuronal subtypes expressing the two dopamine receptors using double immunofluorescence. Glutaminase and GABA (γ-aminobutyric acid) are markers of glutamatergic excitatory neurons and GABAergic inhibitory neurons, respectively. We observed that in the CeA, D1R-positive neurons colabeled with glutaminase were more abundant than those colabeled with GABA, that D2R-positive neurons were primarily colabeled with GABA, and that only a few D2R-positive neurons were colabeled with glutaminase. Our data indicated that most D1 receptors were expressed in excitatory neurons in the CeA but that D2 receptor was predominantly expressed in inhibitory neurons in the CeA (Figure 5(b)).
D2 receptor in the CeA is involved in pain relief
According to the above results, we found that specific activation of CeA-projecting dopaminergic neurons in the VTA produced obvious analgesic effects. The expression of D2 receptor in the CeA was decreased after pain relief, and the expression of D1 receptor did not change significantly. To assess the role of D2 receptor in the VTA-CeA pathway, we combined optogenetic approaches with behavioral pharmacologic manipulations. Before specifically activating the VTA-CeA pathway, a D2 receptor agonist (quinpirole, 5 µg/µl) was injected intraperitoneally, and then mechanical threshold measurements, the OFT, and the CPP test were performed.
The results showed that compared with DMSO injection, quinpirole injection significantly reversed the pain relief caused by activation of the VTA–CeA neural pathway in incised mice. The mechanical threshold of the ipsilateral hind paw during light stimulation was significantly decreased in the quinpirole injection group compared with the DMSO injection group (Figure 6(a)). The percentage of time spent in the central area in the OFT during light stimulation was significantly decreased by quinpirole injection (Figure 6(b)). However, quinpirole injection had no obvious influence on the mechanical threshold of the ipsilateral hind paw or the percentage of time spent in the central area in the OFT in the absence of no light stimulation. Administration of a D2 receptor agonist (quinpirole) reversed the pain relief caused by light-induced activation of the VTA-CeA pathway. (a) The mechanical threshold of the ipsilateral hind paw during light stimulation was significantly decreased in the quinpirole injection group compared with DMSO injection group. (b) The percentage of time spent in the central area in the OFT during light stimulation was significantly decreased by quinpirole injection. (c) The time spent in the chamber paired with quinpirole injection was significantly reduced compared with that spent in the chamber paired with DMSO injection following light stimulation (A:Interaction:F (1, 21) = 42.81,B:Interaction:F (1, 20) = 12.10,***p < 0.001, (c)Interaction:F (1, 26) = 5.666,n = 6; *p < 0.05 vs the DMSO injection group with light stimulation; two-way ANOVA).
In the CPP test, the time spent in the chamber paired with quinpirole injection was significantly reduced compared with that spent in the chamber paired with DMSO injection following light stimulation (Figure 6(c)). There was no significant difference between the quinpirole injection group and the DMSO injection group in the absence of light stimulation. These results demonstrated that administration of a D2 receptor agonist reversed the pain relief caused by light-induced activation of the VTA-CeA pathway. Dopamine receptor D2, but not D1, mediates the circuit from the ventral tegmental area to the central amygdala, which is involved in pain relief.
Discussion
This study provides evidence that activation of the VTA-CeA pathway is sufficient for pain relief, preliminarily suggesting that D2 receptor is involved in the process of analgesia.
Pain is a subjective and multidimensional experience with sensory, affective, and cognitive components. 25 The subjective unpleasantness of pain is essential to the human experience but difficult to evaluate in laboratory animals. In agreement with previous findings, our current behavioral data demonstrated that removal of the aversive state resulting from ongoing postsurgical nociceptive input by PNB elicited CPP. Importantly, CPP was not observed in sham-treated mice following PNB, which likely greatly diminished injury-induced ongoing pain. Additionally, consistent with previous reports showing that tissue or nerve injury can induce anxiety/depression-like behaviors in rodents,26,27 our results demonstrated that experimental surgical incision could produce pain-related negative emotional behaviors such as anxiety, as revealed by the OFT and EPM test.
The finding that relief of ongoing incisional, neuropathic, osteoarthritic, and inflammatory pain6,8,11 produces CPP confirms that pain relief elicits reward. Accumulating evidence suggests that midbrain dopamine neurons modulate acute and chronic pain, as well as the process of analgesia/pain relief.28–35 However, the underlying mechanisms that regulate pain and reward during pain relief in the midbrain dopamine system have not previously established directly. The CeA has been intensely investigated for its role in pain-related behaviors. Emerging studies have shown that CeA can also play a role in driving reward-related behaviors.36,37 The precise functional connectivity between VTA dopaminergic projection neurons and the CeA and the role of this circuit in pain relief have not been described. Here, using retrograde tract tracing methods, we clearly demonstrated that the VTA sent direct projections to the CeA. Double staining showed that CeA-projecting neurons in the VTA were dopaminergic. Furthermore, we found that PNB increased the number of Fos-expressing cells in the VTA in injured mice. These findings suggest the existence of strong, target-specific dopaminergic projection from the VTA to the CeA and indicate that the VTA-CeA circuit might contribute to pain relief and the affective dimension of pain.
As a subcortical structure and part of the limbic system in the medial temporal lobe, the amygdaloid complex, which comprises several anatomically and functionally distinct nuclei, is a key player in linking nociceptive stimuli to negative emotional behaviors or affective states.38–42 The CeA is the major output nucleus for the amygdaloid complex. The CeA receives large amounts of nociceptive information through the spinoparabrachioamygdaloid pathway41–44 and various sensory information, including nociceptive information, from thalamic and cortical regions through the BLA.41,42,45–48 Specific nociceptive inputs and various pieces of information can be integrated into the CeA, which is believed to underlie the generation of the negative emotional or affective dimension of pain.40,49 The mesolimbic reward circuitry, which is composed of dopaminergic neurons of the VTA and their projections to several brain regions, is critical for pain sensation and pain-related emotional experiences.28–31,33–35,50 Previous studies have characterized VTA projections to the CeA, but not extensively. Much focus has been on VTA dopamine neurons, as well as local modulation of dopamine signaling in the CeA. 51 More studies showed that the NAc is a main target of VTA dopaminergic neurons. Findings have demonstrated that pain relief increased c-Fos expression in the NAc and induced dopamine release in the NAc under acute inflammatory pain conditions.52,53 In this study, anterograde and retrograde tracing studies demonstrated that VTA dopaminergic neurons were also found to directly project to the CeA. Although there were fewer of these projections than of those projecting to the NAc, we could not exclude a key role for amygdala-projected dopaminergic signaling in pain relief and the affective dimension of pain. In this study, optogenetic stimulation and DAT-Cre transgenic mice were employed to confirm the role of amygdala-targeted dopaminergic signaling in both pain relief/reward and the negative emotion of pain. The results of the optogenetic experiments suggest that specific activation of the VTA–CeA neural pathway produces analgesic (pain relief) and anxiolytic effects in incised mice. The VTA–CeA neural pathway was found to be involved in the modulation of nociception and comorbid emotional behaviors.
As an important neurotransmitter in the midbrain reward system, dopamine acts on metabotropic D1-and D2-like receptors. D2-like (D2, D3, D4) receptors can inhibit adenylyl cyclases, the enzymes that catalyze the generation of cAMP, through activation of the inhibitory G-protein. Our results showed that D2 receptors were predominantly expressed in inhibitory neurons in the CeA. D2 receptor expression was significantly decreased in the CeA after pain relief. These findings raised the possibility that downregulation in D2 receptor-mediated inhibition induced activation of CeA inhibitory neurons in the CeA after pain relief. However, in our study the c-Fos + cell count in the CeA did not change significantly after pain relief. It is possible that c-Fos is only useful for measuring activation, not inhibition.
Since the CeA consists of around 95% GABA neurons, the role of GABAergic neurons would be a determinant contributor for excitability of CeA nucleus. The previous study has confirmed that brief electrical stimulation applied to the CeA evokes antinociception in the rat and cat. 54 Consequently we postulate that disinhibition in GABACeA (caused by decreased level of D2 receptor) may facilitate analgesic (pain relief) and anxiolytic effects in incised mice. It seemed that analgesia might be mediated by decreased D2 receptor expressing and activation of CeA inhibitory neurons in the CeA in our study. Additionally, there is an endogenous descending antinociceptive system composing from periaqueductal gray (PAG) - rostral ventromedial medulla-spinal cord. 55 Our findings were also consistent with previous reports showing that the increased activity of CeA - PAG circuit alleviated nociception in depressive states. 56
The dopaminergic system is involved in the modulation of nociception. Nevertheless, the role that different dopaminergic receptors play in nociceptive modulation has not been fully understood. Conflicting results have emerged regarding the differential role of dopaminergic receptors in nociception processing. D2 receptor antagonists have been reported to exert analgesic effects and reduce anxiety-like behavior. 57 Paradoxically, D1 receptors and D2 receptors agonist produced dose-dependent antinociceptive effects in formalin-induced nociception, neuropathic pain, and postoperative nociception.58,59 There were reports out there, saying that the agonist and antagonist both reduced neural activity despite having the desired opposite effect on dopamine signalling. 60 In this study, administration of a D2 receptor agonist reversed the pain relief elicited by light-induced activation of the VTA-CeA pathway. However, the activation of D2 or the blockade of D1 receptors within the insular cortex (IC) elicited antinociception. Optogenetic excitation of A11 dopaminergic neurons markedly attenuates the CCI-ION-induced neuropathic pain and this effect is completely inhibited by intra-Vc injection of D2 receptor antagonist. 9 It was likely that the dopaminergic system in the CeA did not play the same role in the modulation of pain as it did in the IC and the spinal trigeminal nucleus caudalis (Vc). Thus, our study also has some limitations. D2 receptor antagonist treatment should be further utilized to validate the analgesic and anxiolytic effects in incised mice. Further experiments should be done to elucidate the role of dopaminergic receptors in the CeA in relation to different pain model.
The VTA comprises a heterogeneous population of dopamine-, GABA-, and glutamate-releasing neurons. Dopaminergic neurons are the most abundant, while GABAergic and glutamatergic neurons account for ∼30% and 2% to 5% of the total population, respectively. In our study, retrograde labeling with FG indicated that VTA dopaminergic neurons projected to the amygdala, that most VTA neurons that projected to the CeA were dopaminergic neurons, and that these neurons were predominantly responsible for the effects we observed in the present study. Nonetheless, we cannot exclude the contributions of other populations or projecting neurons.
In summary, we report that alterations in dopaminergic circuits between the VTA and CeA play an important role in pain relief. Future investigations addressing the specific mechanism underlying dopamine signaling through D2 receptor are required to elucidate the role of the VTA-CeA circuit in pain relief.
Footnotes
Author contributions
Huang and Wang contributed equally to this manuscript as first authors. Li planned the experiments. Lin generated the animal model and performed cannula implantation. Huang and Wang carried out behavioral tests and Western blot assays. Cao and Ren carried out the immunostaining experiments and data analysis. Huang and Wang wrote the manuscript. Li and Zang discussed the manuscript. All authors read and approved the final manuscript.
Conflict of interest
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (grant number 81600973 to Z-H L).
Ethical approval
The animal study was reviewed and approved by the Animal Care and Use Committee of Zhengzhou University.
