Abstract
Development of chronic pain has been attributed to dysfunctional GABA signaling in the spinal cord. Direct pharmacological interventions on GABA signaling are usually not very efficient and often accompanied by side effects due to the widespread distribution of GABA receptors in CNS. Transplantation of GABAergic neuronal cells may restore the inhibitory potential in the spinal cord. Grafted cells may also release additional analgesic peptides by means of genetic engineering to further enhance the benefits of this approach. Conopeptides are ideal candidates for recombinant expression using cell-based strategies. The omega-conopeptide MVIIA is in clinical use for severe pain marketed as FDA approved Prialt in the form of intrathecal injections. The goal of this study was to develop transplantable recombinant GABAergic cells releasing conopeptide MVIIA and to evaluate the analgesic effect of the grafts in a model of peripheral nerve injury-induced pain. We have engineered and characterized the GABAergic progenitors expressing MVIIA. Recombinant and nonrecombinant cells were intraspinally injected into animals after the nerve injury. Animals were tested weekly up to 12 weeks for the presence of hypersensitivity, followed by histochemical and biochemical analysis of the tissue. We observed beneficial effects of the grafted cells in reducing hypersensitivity in all grafted animals, especially potent in the recombinant group. The level of pain-related cytokines was reduced in the grafted animals and correlation between these pain markers and actual behavior was indicated. This study demonstrated the feasibility of recombinant cell transplantation in the management of chronic pain.
Introduction
Mechanisms underlying development and maintenance of chronic pain are complex and involve pathophysiological changes in several pain signaling pathways. 1 Effective management of chronic pain must reflect this complexity; multitargeting yet local approaches are therefore needed. Although several approaches are used in the clinical setting to provide local delivery of analgesic compounds, long term delivery is still a challenging problem. 2 Recombinant cell-based therapy of chronic pain can provide sustained, targeted, and customized delivery of analgesic compounds. A key event underlying development of chronic pain is reduced inhibition in the spinal cord caused by dysfunctional signaling of inhibitory neurotransmitter.3–11 Although the exact mechanisms of spinal GABAergic dysfunction are still matter of debate, it is possible that a combination of several pathophysiological events, such as injury-induced dysfunction of GABA receptors, reduced production of GABA by injured spinal neurons or downregulation of chloride channels and reversal of GABAergic signaling, contribute to the development of neuropathic pain.12–16
Pharmacological targeting of GABA receptors is infeasible due to widespread location of GABAergic receptors throughout the CNS. Transplantation of GABAergic cells showed reduction of chronic pain and partial restoration of the inhibitory balance in the spinal cord.17–23
To improve the analgesic outcome of this approach, cells may be engineered to produce additional analgesic peptides. In this project, as a recombinant peptide produced by GABAergic cells, conotoxin MVIIA is investigated in an animal model of peripheral neuropathic pain. The omega-conopeptide, MVIIA, an N-type Ca2+ channel blocker, was one of the earliest tested in animal models and is in clinical use for severe pain marketed as FDA approved Prialt (Ziconotide).24–26 However, broad clinical use of analgesic peptides is limited by poor CNS penetration and side effects at high analgesic doses due to diffusion to higher CNS centers.27,28 Conopeptides are strong candidates for recombinant expression at target sites to spinal cord pain processing centers using cell-based strategies, which can provide more circumscribed delivery at lower doses. A previous study showed that combined intrathecal ziconotide and GABA agonist baclofen improved neuropathic pain scores in animal models and in some case studies. 29 Therefore, MVIIA delivery via GABAergic cells may provide an ideal combination to alleviate chronic pain without side effects resulting from repeated high dose drug administration. Preliminary data were presented previously.30,31
Material and methods
Engineering of recombinant GABAergic neuronal progenitor cells (NPCs)
The Miami Project based Viral Vector Core (VVC) generated the required lentiviral particles. The MVIIA plasmid and the viral particles were designed based on our previously generated vector with secretable peptide. 32 MVIIA cDNAs, obtained from Dr Baldomero Olivera (University of Utah), were amplified by PCR to introduce restriction sites that allow sub-cloning into viral vectors. Since MVIIA is amidated, to facilitate proper post-translational modification in mammalian cells, we have amplified the MVIIA cDNA by PCR and subcloned in frame with the signal peptide of peptidylglycine α-amidating monooxygenase (PAM), the enzyme responsible for amidation of neuropeptides. 33 The signal sequence directs transit of the recombinant MVIIA via the PAM vesicles and keeps it in close proximity to the amidating enzyme complex. Briefly, the plasmid with PAM signal sequence for amidation and targeting of the recombinant gene into the secretory pathway32,34,35 was flanked by BglII and XbaI restriction enzymes. BglII and XbaI restriction sites were added by PCR to the 5′ and 3′ end of the cDNA of MVIIA respectively and subcloned into the frame with PAM sequence. The BamHI-XbaI fragment containing PAM-MVIIA was subcloned into lentiviral vector pRRLsinPPT provided by VVC. Vectors were amplified in E. Coli bacterial cultures (Stbl10), plasmids were insolated and purified by Maxiprep (Qiagen) and verified sequences submitted to VVC to generate viral particles. Lentiviral concentrations were determined by ELISA for the virus p24 protein. Typical preps yield > ∼1.0 × 107 pg/mL of p24, corresponding to ∼1.0 × 1011 viral particles/ml.
E14.5 fetal neocortical tissue from Sprague-Dawley rats was microdissected into Hank’s balanced salt solution (HBSS) and a cell suspension created via mechanical trituration. Cells were plated at an initial concentration of 1 × 106 cells/mL of the culture media containing 10 ng/mL of human recombinant basic fibroblast growth factor (FGF-2; Sigma) in 75 cm2 treated cell culture flasks (Corning). 24 h post-harvest cells were collected, centrifuged at 800 r/min/3 min, the pellet was gently resuspended in a small volume of media and viral particles were added at 1 × 1011 viral particles/mL for 4 h. Cells were then resuspended in culture media (1 × 106 cells/mL) and cultured for 3–4 days. Twenty-four h prior to transplantation, FGF was withdrawn from media in order to initiate pre-differentiation to GABAergic progenitors, as we have found this environment to be favorable for GABAergic differentiation. 36
Cell viability
Cell viability was evaluated after lentiviral transduction before each transplantation. Cells were resuspended with HBSS. 10 μL of cell suspension was mixed with 10 μL of 0.4% Trypan blue and evaluated on hemocytometer. Non-viable cells stain blue. Cells were observed under ×10 magnification with bright light microscope. Viability was calculated as % of viable cell (pale) out of all cells (pale and blue).
Immunocytochemistry
Cells were plated into 12 well plates or eight well chambers coated with poly-L-ornithine/fibronectin at concentration 5 × 105/well and incubated at 37°C for 2–3 days. Cells were fixed with 4% paraformaldehyde, washed and incubated in 5% normal goat serum for 2 h and overnight in primary antibodies (γ-ammino butyric acid (GABA), 1:200, Sigma; vesicular GABA transporter (VGAT), 1:200, Abcam; β-tubulin 1:1000, Sigma; doublecortin, 1:1000, Sigma; glial fibrillary acidic protein (GFAP), 1:1000, Abcam; MVIIA, 1:50, 21st Century Biochemicals) followed by incubation with appropriate secondary antibodies (Alexa Fluor 488, 594, anti-rabbit, anti-mouse, 1:250, Invitrogen). A negative control staining was run for each antibody with the omission of primary antibody. After final wash the upper structure of the chamber was carefully removed and cells were coverslipped (VectaShield, Vector).
Transduction efficiency and cell phenotype
The % of transduction efficiency was estimated based on number of MVIIA+ cells out of DAPI labeled cells. Phenotypes of the transduced cells were quantified based on double labeling with MVIIA and β-tubulin, GABA or GFAP antibodies. DAPI was used as nuclear staining in all procedures.
Slides with cells stained with the combination of MVIIA with β-tubulin/GABA/GFAP antibodies were evaluated under epifluorescence microscope with digital camera at ×20 magnification in five wells. Colocalization of markers was evaluated using Image J software (NIH). For transduction efficiency, all visible DAPI labeled cells were marked and % of MVIIA colocalization was calculated. For evaluation of the phenotype, MVIIA+ cells were marked and % of colocalization with β-tubulin, GABA or GFAP was calculated. At least 80–100 cells were used in the analysis.
FLISA analysis of cell supernatants
FLISA analysis was used to detect recombinant MVIIA peptide and GABA in the cell culture samples. Cells at 3–4 days post viral transduction were centrifuged at 800 r/min/3 min, supernatants were collected, treated with N-Per extraction buffer (Thermo Fisher) and processed for BCA analysis. Samples and positive controls (GABA and MVIIA) were loaded in 96 well plate in the same protein concentration established by the BCA method. Samples and controls were diluted by 1/10 in each row. Capture buffer was used for the initial coating according to the manufacturer protocol (Odyssey FLISA, Li-Cor). Samples were incubated overnight at 4°C followed by incubation in the blocking buffer for 3 h at room temperature and primary antibodies (GABA, 1:100, Sigma; MVIIA, 1:50, 21st Century Biochemicals) overnight at 4°C. Plates were then washed and incubated in secondary antibody (IRDye goat anti-rabbit) for 2 h, washed and absorbance measured by Odyssey Infrared Imager (Li-Cor). Data were analyzed by Image J (NIH). Optical density of FLISA spots for each antibody and treatment were averaged out of five measurements.
Animals
Male Sprague-Dawley rats (140–160 g at the time of the first surgery, Envigo, IN) were used in the experiments; pregnant female Sprague-Dawley rats were used for E14 embryo harvesting (Envigo, IN). Animals were housed two per cage with free access to food and water in 12 h light/dark cycle. Experimental procedures were reviewed and approved by the University of Miami Animal Care and Use Committee and followed the recommendations of the “Guide for the Care and Use of Laboratory Animals” (National Research Council). All surgical procedures were conducted under 2.5% Isoflurane/O2 anesthesia using aseptic conditions. For all experiments, animals were randomly assigned to the experimental groups and all tests and evaluations were done in a blinded manner. The number of animals per group was estimated at the beginning of the study by power analysis using SigmaStat with the input data based on our previous studies and desired power set at 0.8 with alpha 0.050.
Surgeries
Animals were anesthetized with 2%–3% isoflurane in O2, and the common sciatic nerve exposed on one side at the mid-thigh level using aseptic surgical techniques. Four 4-0 chromic gut ligatures spaced about 1 mm apart were loosely tied around the sciatic nerve proximal to the trifurcation. Following surgery, the musculature was sutured in layers, and the skin closed with wound clips. In sham animals, nerve was exposed but no ligatures were placed. 37
All animals (n = 42 for CCI, n = 8 for sham) were monitored for the presence of pain related behavior using behavioral tests (see below). Only CCI animals displaying pain related behavior in at least one pain test at 1 week post injury were used for the subsequent experiments (n = 39, three animals were excluded from the experiment due to the lack of injury-induced hypersensitivity at 1 week post injury). Sham animals were used as age-controls for histochemical and biochemical analyses, no pain-related phenotype was observed in this group (data not shown).
For transplantation, cells were pelleted (1500 r/min/3 min) and resuspended in HBSS media at a concentration of 50,000 cells/μL. Cells were transplanted at 1 week post CCI to target early stages of chronic pain development in this model. A laminectomy was performed to expose the L4 spinal level. 38 Cells were injected into the ipsilateral dorsal horn with a 10 μL Hamilton syringe attached to a pulled glass pipet (diameter ∼ 50 μm). A small puncture was made in the meninges and 1.0 μL of cell suspension (∼5 × 104 cells) was stereotactically injected on the injury side (5 mm lateral from the central artery, 1 mm ventral from the surface of the dorsal horn). Following transplantation, the area was covered with elastic sheathing, the overlying musculature sutured, and the skin closed with wound clips. Control animals underwent the identical surgical procedures but received saline injection instead of cell suspensions. Animals with grafted cells received daily cyclosporine injections (IP, 10 mg/kg; Bedford Labs, OH) starting 2 days prior the surgery up to 2 weeks post grafting and then remained on oral administration (45 mg/kg) for the duration of the study. 19
In a preliminary screening study, small groups of CCI animals (n = 3/group) were used to confirm the analgesic activity of recombinant peptide released from the recombinant cells. At 10 days post CCI, an intrathecal catheter (7.5–8 cm; ReCathCo, PA) was inserted into these animals via slit in the atlanto-occipital membrane and threaded down the intrathecal space to reach the lumbar region. The catheter was secured to the neck muscles with sutures.39,40 Rats were allowed to recover at least 3 days following intrathecal surgery prior to use in experiments.
Behavioral tests
Animals were observed daily for the presence of any health issues. Behavioral tests were conducted weekly up to 12 weeks post injury. Tests for tactile and cold allodynia were performed on the same day, followed by thermal hyperalgesia test on the following day. Before behavioral testing, animals were observed for at least 20 min for the presence of any side effects induced by MVIIA, such as movement-evoked tremors and tail writhing, observed previously in a study with certain concentration of intrathecally delivered peptide MVIIA. 41 No visible adverse effects were observed in any animals with recombinant grafts.
Tactile hypersensitivity
The threshold level to an innocuous mechanical stimulus was measured with calibrated von Frey hairs ranging from 0.25 to 15 g, following the Dixon’s 42 Up-Down test. Animals were placed into clear plastic cages on an elevated wire mesh floor. Calibrated von Frey filaments were applied to the plantar skin of the hind paw with increasing force, until a response was observed. The response consisted of a brisk hind paw withdrawal together with at least one other sign of cognitive response to the stimulus, such as vocalization, head turns towards the stimulus or moving away from the stimulus, in order to avoid recording of responses evoked by spinal reflexes only. A total of six responses were recorded and the withdrawal threshold was calculated using the Dixon calculator.
Cold hypersensitivity
Sensitivity to a non-noxious cooling stimulus was evaluated using acetone. Animals were placed into clear plastic cages on an elevated wire mesh floor. 100 μL of acetone was dropped onto the lateral margin on the hind paw from a blunted 22 ga needle attached to a syringe. Acetone was applied to the hind paw five times, with about 1–2 min between applications. Positive responses in the form of paw withdrawal and shaking/licking the paw were recorded out of five trials and converted to a percent response frequency. 43
Hargreaves test (heat hypersensitivity)
Rats were placed into an inverted clear plastic cage on an elevated glass floor and a radiant heat source beneath the glass was aimed at the plantar hind paw. Infrared heat source was activated manually together with the timer. The timer was stopped when an animal withdraws the paw, and the latencies were recorded. Positive response was considered as paw withdrawal in concert with licking the paw and moving away from the stimulus. To avoid tissue damage in the absence of a withdrawal, the cutoff was set at 20 s. The average latency was calculated from three trials with 30 s apart. 44
In the preliminary screening set, heat hypersensitivity was used to evaluate responses to the intrathecal injection of supernatant from nonrecombinant and recombinant NPCs cultures at 2 weeks post CCI. Both supernatants were injected in a volume of 5 μL, followed by 5 μL saline flush. The concentration of protein in supernatants was determined by BCA method (Thermo Fisher), and used at 50 μg/5 μL/injection. Saline (10 μL) was injected as a control. Animals were evaluated before the injection and 30 min post injection.
Immunohistochemistry
All animals were perfused or sacrificed, and tissue was harvested for immunohistochemical (n = 5/group) and biochemical evaluation (n = 5/group) following conclusion of behavioral evaluations. Animals were deeply anesthetized and intracardially perfused with 0.9% saline followed by 4% paraformaldehyde in 0.1 M phosphate buffer. Spinal cords were removed and post-fixed overnight, followed by incubation in 30% sucrose for 48 h. Serial sections cut on cryostat at 40 μm were collected either as slide mounted or free floating.
Immunohistochemical staining was used to detect the grafts and to evaluate levels of cellular phenotypes and some pain-related markers. Sections were incubated in 5% normal goat serum, followed by overnight incubation in primary antibodies in 4°C (recombinant conopeptide MVIIA 1:200 (21st Century Biochemicals); γ-ammino-butyric acid (GABA) 1:200 (Sigma); Neuronal N (NeuN) 1:1000 (Genetex); glial fibrillary acidic protein (GFAP) 1:1000 (Millipore); Iba-1 1:1000 (Wako), followed by incubations in secondary antibodies (Alexa Fluor 488, 594, anti-rabbit, anti-mouse, anti-guinea pig, 1:200, Invitrogen) in 5% NGS. A negative control staining was run for each antibody with the omission of primary antibody. Sections are glass mounted, air dried and coverslipped by VectaShield (Vector Labs). Analyses were done with a Zeiss Axiovert 200M research microscope (Ludl Electronic Products), DVC cooled camera and multi-band fluorescent filters allowing for viewing single, double or triple fluorophores at the same time.
Immunostaining for GABA and Iba-1 was analyzed by ImageJ Integrated Density tool in 8–10 serial sections (distance between sections 160 μm) per animal (n = 3–4/group) of the lumbar spinal cord. Density of the immunostaining was analyzed in manually drawn borders of LI-LIII. All values were normalized for the background signal and average integrated density values were compared between groups.
Analysis of the amount of MVIIA grafted cells was conducted in 8–10 serial sections of the lumbar tissue per animal from three grafted animals. Immunostained sections of lumbar spinal cord were analyzed with Neurolucida (MicroBrightField Inc, Williston, VT) under ×60 magnification. MVIIA+ cells were counted within 0.015 mm2 region and stereological estimation of cells count was calculated by Neurolucida. Results are presented as average ±SEM.
FLISA and ELISA analysis of tissue
For biochemical analysis, ipsilateral and contralateral lumbar spinal cord tissue samples from the areas with visually identified injection sites (L4-L5 segments, cca 5 mm long) were collected from the anesthetized animals after decapitation, frozen on dry ice and stored at −80°C. The presence of MVIIA in the grafted animals was also evaluated by FLISA. Spinal cord samples were homogenized in RIPA lysis buffer (Santa Cruz) and protein concentration was estimated by the BCA method (Thermo Fisher). Positive samples (MVIIA protein, Bachem) and samples from grafted animals were then processed for FLISA evaluation as described above.
Biochemical evaluation of pain related cytokines in the spinal cord homogenates was conducted by ELISA analysis. We evaluated the levels of IL-1β, TNFα and IL-10 in the homogenates of the lumbar spinal cord from CCI animals from each treatment group, as these cytokines has been previously hypothesized to play a role in the development and maintenance of neuropathic pain.45–48 Spinal cord frozen samples were homogenized in RIPA buffer (Santa Cruz) and the protein level was estimated by the BCA method (ThermoFisher). For ELISA detection of cytokines, kits for IL-1β, TNFα and IL-10 (Abcam) were used following the manufacturer protocol. Briefly, samples diluted to the same concentration of proteins were loaded onto 96 well plate in triplicates, together with standards, incubated with the blocking serum, followed by primary and secondary antibodies. Plates were read by a microplate reader (Molecular Devices) and results were analyzed by SoftMax Pro.
To examine more closely the potential relationship between levels of pain-related cytokines and the behavior, correlation analysis tests (GraphPad Prism) were run using data from ELISAs in combination with pain scores for individual animals used in the ELISA evaluations.
Statistical analysis
All statistical analyses were done in Graph Pad Prism 8.4.2. Data are expressed as mean ± SEM with statistical significance taken at p < 0.05. Relative optical density in FLISA was analyzed by t-test. All behavioral tests were compared using two-way RM ANOVA with Holm-Sidak post-hoc test. Immunodensity and ELISA values were evaluated by one-way ANOVA with Tukey multiple comparison test. Correlation analysis was performed by simple linear regression module.
Results
Cell culture analysis
The average viability at 3–4 days post lentiviral transduction was 82.5% which is suitable for grafting procedures. 49
The transduction efficiency was estimated at 73.6% of DAPI cells. The majority of MVIIA positive cells were expressing β-tubulin (92.6 ± 11.6%) and GABA (66.3 ± 6.8%), indicating the desirable neuronal phenotype (Figure 1(a)–(d), (i)). Colocalization with astroglial marker GFAP was detected in 8.5 ± 1.9% of MVIIA positive cells (Figure 1(i)). Visual comparison of nonrecombinant and recombinant cells using β-tubulin (Figure 1(a) and (b)), GABA (C–D), doublecortin (E–F) and VGAT (G–H) staining did not show any significant morphological changes in response to the presence of MVIIA. Immunocytochemical detection of proneuronal markers and recombinant peptide MVIIA derived from E14.5 neuronal progenitor cells (NPCs). DAPI (blue) as nuclear staining. (a) β-tubulin indicates proneuronal phenotype of cells, with no green signal detected from MVIIA in nonrecombinant cells. (b) Overlap between red β-tubulin and green MVIIA creates yellow color in co-expressing cells in recombinant group. (c, d) GABAergic phenotype was confirmed in both non-recombinant and recombinant cultures, with yellow color indication overlapping expression of GABA (red) and MVIIA (green) markers in the recombinant group. Morphological differences between non-recombinant and recombinant cells were not observed using additional proneuronal markers: doublecortin (e–f) and VGAT (g–h). (i) Quantitation of phenotypes of recombinant cells. Scale mark = 30 μm.
Results of FLISA analysis of cell cultures shows the presence of GABA in both the non-recombinant and recombinant cells as expected, and the presence of MVIIA peptide in the supernatant of recombinant cells only (p < 0.001 NPCs vs rNPCs, Figure 2). (a) FLISA analysis of cell culture supernatant for the presence of GABA and MVIIA. GABA was detected in both non-recombinant and recombinant cells cultures, MVIIA signal was significantly stronger from the background in recombinant cells supernatant samples. (b) Quantification of relative optical density of FLISA dots. ***p < 0.001 between the indicated groups.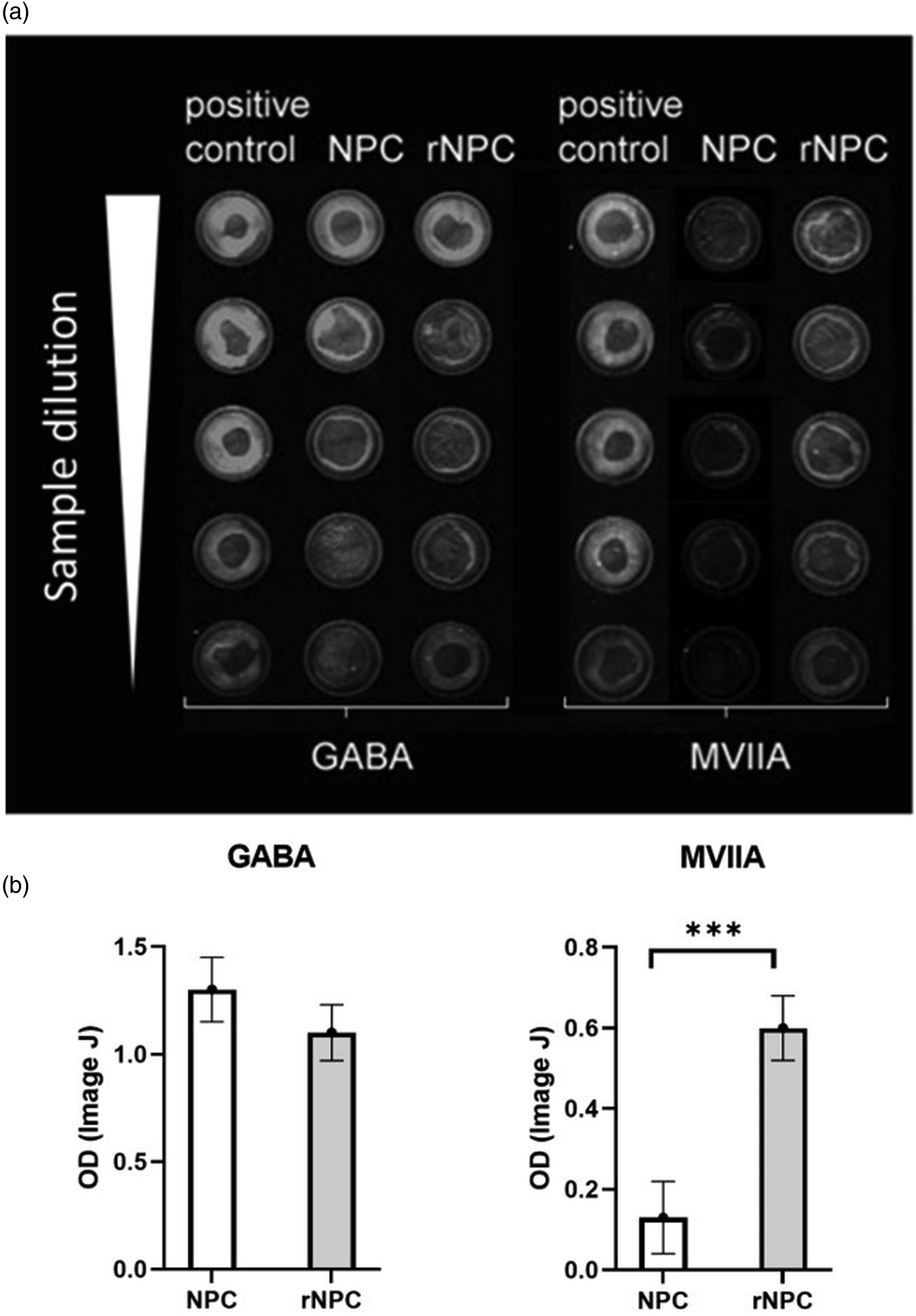
Behavior
To confirm proper expression of the recombinant peptide and the preservation of its analgesic effect, we ran a preliminary screening study with a subgroup of CCI animals. At 2 weeks post CCI, when hypersensitivity was fully developed, animals were intrathecally injected with supernatant of either NPCs or recombinant NPCs; saline injection was used as a control. Animals receiving supernatant from recombinant GABAergic NPCs showed attenuation of heat hypersensitivity at 30 min post injection (p < 0.05, Supplement Figure S1). Animals injected with the supernatant from nonrecombinant GABAergic cells also showed a trend toward attenuated hypersensitivity, but the effect was not significantly different.
In the main group of animals, CCI induced development of hypersensitivity to mechanical and thermal stimulation within the first week post injury in all animals. Cells were grafted at 1 week post CCI when the hypersensitivity was already developed. Alleviation of pain related behavior was observed in animals with cell grafts.
Saline injected animals (controls) developed signs of tactile hypersensitivity that persisted throughout the 12 weeks of experiment (p < 0.001 vs pre-injury baseline). Animals with non-recombinant grafts showed mild attenuation of hypersensitivity, with significant differences from the control group starting at 3 weeks post grafting (week 4) till week 9 (p < 0.05). Animals with recombinant cells showed more robust outcomes overall throughout the progression of the study. From weeks 7–12, the effect was significantly different from the non-recombinant graft (p < 0.05). The withdrawal threshold of animals in this group reached almost the pre-injury baseline levels towards the end the experiment (p > 0.05 weeks 9–12 vs baseline). Overall F (2, 19) = 28.19, p < 0.001 (Figure 3(a)). Behavioral evaluation of the cell therapy in the CCI model. BL responses are pre-CCI, followed by weekly testing after CCI. Data are shown as mean ± SEM *p < 0.05 NPC versus control; +p < 0.05, ++p<0.01, +++p < 0.001 rNPC versus control; #p < 0.05, ##p < 0.01 between grafted groups; n = 10/group.
Saline injected animals developed cold hypersensitivity with a peak between weeks 4–7 post injury. Non-recombinant grafts prevented development of peak cold hypersensitivity, with significantly lower response rate compared to control animals starting at 4 weeks post CCI (3 weeks post grafting) and lasting till the end of experiment (p < 0.05). Recombinant grafts further attenuated ongoing hypersensitivity, reaching almost the pre-injury baseline level with minimal to no responses to acetone droplets at 8 weeks post grafting (p > 0.05 vs baseline). The overall cold responses were lower than in control group starting at 2 weeks post grafting and were lower compared to the non-recombinant group between weeks 5 and 7 (p < 0.05). Overall F (2, 19) = 54.59, p < 0.001. (Figure 3(b)).
Saline injected animals progressively developed hypersensitivity to noxious heat stimulation that remained stable throughout the rest of experiment. Non-recombinant grafts attenuated heat hypersensitivity starting at 2 weeks post grafting with statistically higher latency compared to the control animals throughout the remainder of the experiment (p < 0.05). Recombinant grafts attenuated heat hypersensitivity starting at 2 weeks post grafting with significantly better outcome compared to both control (p < 0.01) and non-recombinant (p < 0.05) grafted groups starting at week seven and lasting through the remainder of the study. Values at weeks 9–12 in the recombinant group were comparable to pre-injury baseline (p > 0.05). Overall F (2, 19) = 23.31, p < 0.001 (Figure 3(c)).
In general, recombinant graft showed stronger and longer lasting pain attenuation effect compared to non-recombinant graft.
Immunohistochemistry
To assess for potential pain-reducing mechanisms of the grafts, immunocytochemical and biochemical analyses were conducted. We observed reduction in GABA immunodensity in the spinal dorsal horn in the control untreated CCI animals, particularly in the lateral superficial region (Figure 4(a)–(d)), that was partially restored by grafting of either recombinant or non-recombinant cells (white arrows in Figure 4(a)–(d)). Image analysis of stained tissue sections showed differences in the optical density of GABA staining between sham animals and CCI animals (Figure 4(i), *p < 0.05, **p < 0.01)) and between CCI animals with and without cell treatment (##p < 0.01 between indicated groups). Immunohistochemical detection and evaluation of optical density of GABA (a–d, i), and Iba-1 (e–h, j) in sham, CCI and CCI with grafted cells animals. (a–d, i): reduced immunoreactivity observed for GABA in superficial dorsal horn after CCI, with partially restored immunoreactivity after cell grafts. (e–h, j): Increase in Iba-1 immunoreactivity observed in all CCI animals compared to sham group, with slight reduction in animals grafted with rNPCs. *p < 0.05, **p < 0.01 versus sham; ##p < 0.01 between groups.
Activation of microglial cells detected by Iba-1 immunostaining was observed in the CCI model. Although there was a trend toward decreased microglia in the transplanted groups, particularly the recombinant NPCs, these did not reach statistical significance compared with control animals at the 12 weeks post CCI (Figure 4(e)–(h)). Image analysis of Iba-1 staining showed increased Iba-1 immunodensity in CCI animals compared to sham (Figure 4(j), *p < 0.05, **p < 0.01).
Recombinant grafted cells were detected in the lumbar spinal cord with minimal spreading from the injection site (about 0.8 mm rostral and caudal). Grafted cells show predominantly a neuronal phenotype (NeuN, Figure 5(a) and (b)) and were located mostly alongside the injection route in the dorsal horn. No MVIIA+ cells were detected in the ventral horn. The MVIIA+ processes were detected in the medial part of the dorsal horn and alongside the injection route. We did not find any overlap of the MVIIA signal with the glial marker GFAP. Stereological analysis showed 1283 ± 125 MVIIA+ cells in the tissue at 11 weeks post grafting. That is approximately 2.5% of total grafted cells, similar to previous reports of 2%–5% NPC transplant survival.21,23,50–54 The presence of MVIIA in the spinal tissue was also analyzed by FLISA. MVIIA was detected in the ipsilateral lumbar spinal cord dissected from the area where intraspinal injection was visually identified (L4-L5), from animals with recombinant graft (Figure 5(c) and (d), p < 0.01). No MVIIA was detected in contralateral sides of the dissected spinal tissue nor in the ipsilateral L4-L5 lumbar spinal cord of animals grafted with non-recombinant NPCs. (a) Recombinant cells detected in the spinal dorsal horn. NeuN (red) label for neuronal cells, GFAP (green) for astrocytes and MVIIA (blue) labeling cells producing MVIIA protein. Overlap between NeuN and MVIIA is shown in magenta (some examples are indicated in b, at arrows). No overlap between GFAP and MVIIA was observed. (b) Enlarged area of the box in a, within the graft epicenter. (c) FLISA analysis of the MVIIA in the spinal cord homogenate (d) Comparison of FLISA OD values, **p < 0.01 versus CCI rNPC ipsi (ipsilateral to CCI and transplants).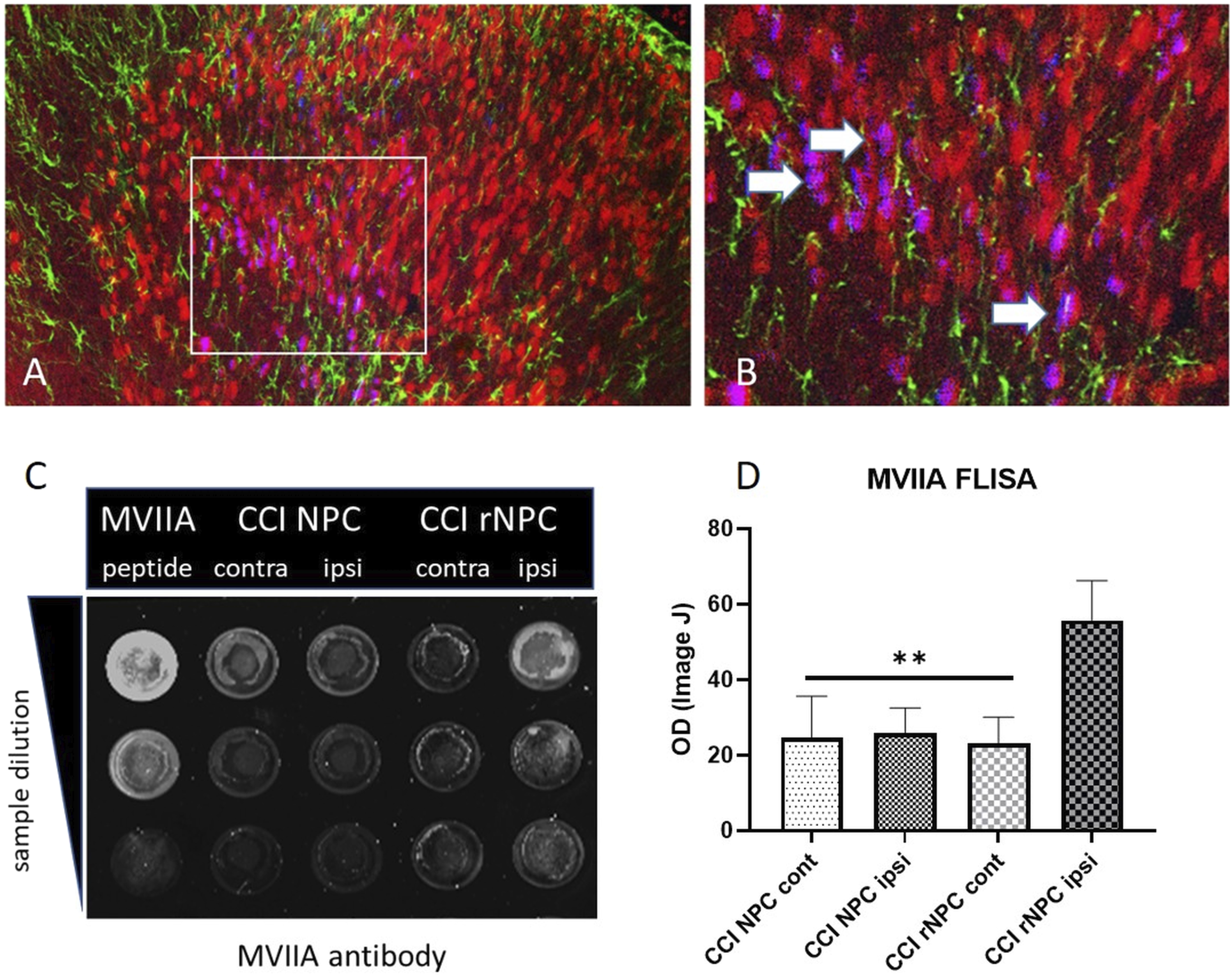
Biochemical analysis of spinal tissue
The levels of cytokines in lumbar spinal homogenates were evaluated by ELISA kits. At 12 weeks post-CCI, elevated proinflammatory cytokines IL-1β and TNFα were found in the lumbar spinal cords of all CCI animals compared to sham controls (Figure 6(a) and (b), p < 0.001). The levels of IL-1β and TNFα were reduced in CCI animals treated with either NPC or rNPC compared to CCI groups (p < 0.05). A more robust reduction of spinal IL-1β levels was observed in rNPC groups compared with non-recombinant NPC (p < 0.05). Anti-inflammatory IL-10 showed similar levels between groups all groups, with no significant changes after either CCI or transplantation (Figure 6(c)). ELISA quantification of (a) IL-1β, (b) TNFα, and (c) IL-10 in lumbar spinal cord homogenates in CCI animals with different treatments. +++p < 0.001 versus sham, *p < 0.05 versus CCI control, #p < 0.05 between grafted groups.
In correlation analysis, values from behavioral tests and the level of each of the monitored cytokine were compared (Figure 7). Significant correlations between behavioral responses and the proinflammatory cytokines were detected for heat hypersensitivity and the levels of IL-1β and TNFα (Figure 7(c) and (f) respectively). There was a trend towards positive correlation for the tactile and cold hypersensitivity tests, but the values did not reach statistical significance (Figure 7(a), (b), (d), (e)). No correlation was detected between IL-10 values and behavioral outcome (data not shown). Correlation analysis of the level of cytokines (a–c) IL-1β and (d–f) TNFα and behavior scores for tactile, cold and heat hypersensitivity in CCI animals treated with saline (open circles), NPC (full squares) or rNPC (full triangles). p and R2 values are indicated.
Discussion
Previous studies in our lab demonstrated that recombinant GABAergic NPCs have the potential to alleviate chronic pain.18,55 The current study further confirmed and extended our hypothesis, that local targeting of pain-signaling pathways using recombinant cells can induce significant pain relief in peripheral nerve-injured animals. In this study, animals grafted with recombinant GABAergic cells producing MVIIA peptide showed reduced tactile, heat and cold hypersensitivity as well as reduced levels of IL-1β in spinal homogenates.
Our previous findings suggested that grafted GABAergic cells significantly attenuate hyperexcitability of spinal dorsal horn neurons and contribute to the reduction of injury-induced hypersensitivity in grafted animals.18,20–22 The analgesic effect observed in these and other studies could be due to the direct release of GABA from the grafted cells and subsequent modulation of ascending pronociceptive signals. 56 Indirectly, neuroprotective effects of GABA on intrinsic interneurons, suggested by studies evaluating ischemic models and cell death,57,58 could also contribute to the restoration of inhibitory potential in spinal pain processing sites.
Functional integration of the grafted cells within the host tissue also appears to contribute to the alleviation of mechanical hypersensitivity induced by peripheral injury as demonstrated by Braz at al. 23 However, participation of the endogenous GABAergic cells cannot be ruled out. Several studies demonstrated the beneficial effect of grafted stem cells on the survival and regeneration of cells in the surrounding areas, indicating the graft contribution to this effect via trophic factors release.59–61 The contribution of the grafted GABAergic cells to the observed analgesic effect was indirectly evaluated in our previous study where GABA receptor agonist and antagonists changed the behavioral and electrophysiological responses in animals with CCI, with stronger response in the animals grafted with GABA progenitors. 22
Pharmacological studies have suggested that the most effective approach to control neuropathic pain is to simultaneously target several pain modulatory systems. The synergistic action of inhibitory GABA agonists and molecules that interfere with excitatory signaling, such as glutamate receptor antagonist or calcium channel blockers has been reported by several studies.62–67 Although a marked improvement in pain related behavior is reported from these studies, the disadvantage is the need for repeated administration of the substances, usually by intrathecal catheters.
Recombinant cells can provide a feasible alternative to this approach, when cells are engineered to target several signaling pathways that are known to be involved in the development of chronic pain. We have evaluated this approach in our previous studies using NMDA antagonist peptide serine histogranin as an adjunct analgesic peptide released by GABAergic grafts. Grafted animals with peripheral and spinal cord injury-induced pain showed more potent and longer lasting attenuation of hypersensitivity compared to animals treated with non-recombinant cells.18,20
An issue related to cell therapy is the overall survival capacity of the graft. It has been demonstrated by several studies including our own that as low as 2%–5% of grafted cells are present within the tissue at 1-month post grafting, although some therapeutic effects are still observed.21,23,50–54 Long term immunosuppression can increase the risk of infection and malignancy, as reported by clinical studies.68–70 To enhance the beneficial effect of the grafts despite the limited survival rate, equipping the cells with cDNA of additional therapeutic substance that can be released from the graft is a viable approach.
Conopeptides, small peptides produced by marine cone snails, with unique affinity to various receptors within the CNS, have become a major focus for new drug development. Conotoxin MVIIA is marketed as FDA approved drug Prialt (ziconotide) for clinical management of severe pain. Ziconotide is a nonopioid analgesic agent binding to N-type voltage-sensitive calcium channels and blocking pronociceptive neurotransmitter release from nociceptive afferent nerves in the dorsal horn of the spinal cord. 71 Due to its limited CNS penetration, intrathecal pumps are used to deliver it to the site of action. Several clinical studies demonstrated analgesic effect of intrathecally delivered ziconotide in patients with refractory pain.72–74 However, due to side effects elicited by higher concentration of the drug, the individual levels need to be carefully titrated for each patient. On the other hand, the combination of low concentration of ziconotide and GABA agonist baclofen was reported to be beneficial in the reducing of pain related symptoms in preclinical and clinical studies.29,75,76 Our previous studies showed beneficial effects of GABAergic cells after their intraspinal injection in the CCI model of neuropathic pain, although the effect was only partial.22,77 The insertion of MVIIA cDNA into GABAergic cells was therefore intended to further increase the analgesic effect of our cell-based approach.
In the current study, enhanced effects of recombinant graft producing MVIIA compared to nonrecombinant graft were observed for tactile and heat hypersensitivity, when recombinant NPCs showed stronger and more stable effect in attenuation of hypersensitivity compared to nonrecombinant cells. Less differences between grafted cell types were found for cold hypersensitivity, however, both treatments led to very strong attenuation of cold hypersensitivity, reaching almost the pre-injury levels. Our results indicate that recombinant MVIIA peptide plays a role in the observed analgesic effect. In our hypothesis, indirectly supported by our results and discussed below, MVIIA probably contribute to reduced release of proinflammatory cytokines that are involved in pronociceptive signaling. This is supported by the observation of reduced immunoreactivity of glial cells and significant reduction of IL-1β in spinal cord tissue of animals with recombinant graft. Reduced levels of TNFα in the spinal tissue of treated animals were observed as well. In our study, immunodensity of Iba-1 microglial cells were upregulated in all CCI animals compared to sham, with moderate reduction observed in the animals with recombinant graft. The involvement of glial cells in the development of chronic pain has been reported by several studies. Activated glial cells release proinflammatory cytokines, and stimulate nociceptive neurons.78–84 In line with this observation, biochemical analysis of the spinal cord tissue showed increased pro-inflammatory cytokine levels at 12 weeks following CCI, and this was substantially reduced by both types of NPC grafts, with levels of IL-1β normalized to near pre-injury levels in the recombinant groups. This prolonged spinal inflammatory cytokine expression in non-transplanted CCI animals and its reduction by NPC transplants correlated in particular with heat hypersensitivity. Prolonged hypersensitivity in this model has also been observed in previous reports.85–87
As the microglia and macrophages are the major source of inflammatory mediators produced post injury, the reduced levels of IL-1β and TNFα in our experiments might be related to the attenuated activity of glial cells in animals treated by the recombinant cells. A hypothesized possibility is a direct contribution of recombinant cells via the N-type calcium channels expressed on microglia as reported by Sageusa et al. 88 In their studies, reduced levels of tactile allodynia after peripheral nerve injury was observed in knockout mice lacking the expression of these channels on microglia/macrophages. It is possible that MVIIA construct release from the recombinant cells might inhibit glial cell activity via calcium channels. Further pharmacologic and electrophysiological experiments are needed to clarify mechanisms of reduced inflammation by recombinant MVIIA NPC transplants.
Furthermore, histological analysis revealed an interesting observation, when grafted cells, detected by MVIIA antibody in recombinant grafts, were detected within the transplant area, but an increased immunodensity of the GABA in the superficial spinal dorsal horn of these animals was also observed in more remote areas from the grafted cells. As the observed GABA immunodensity was detected in the superficial laminae where those cells are naturally present, and in areas where MVIIA cells were not observed (in recombinant groups), it is reasonable to assume that increased GABA immunodensity was generated by endogenous GABAergic neurons and their processes. This might further indicate that trophic support from grafted cells is distributed in the area around the graft and in its close vicinity but can also reach more remote areas. Also, the reduced levels of inflammatory mediators as we observed in the treated animals might contribute to a less hostile environment thus supporting increased viability of the endogenous GABA cells following CCI.
Together, these findings indicated strengthened antinociceptive effects of transplanted GABAergic NPCs when equipped with pain-reducing conopeptide MVIIA. The overall beneficial effect of this strategy might be further increased by using multiple injection sites, thus increasing the total amount of grafted engineered cells. The feasibility and safety of this approach for this and other chronic pain indications will be evaluated in future studies.
Conclusion
Our study demonstrated the feasibility of recombinant cell transplantation in the management of chronic pain. The proposed concept may be useful in the therapy of other disorders of the nervous system as well. Several clinical trials using stem cell therapies are currently ongoing. 89 Equipping these cells with additional features allowing them to locally deliver therapeutic peptides using recombinant cell strategies may enhance the therapeutic potential of cell-based treatments.
Supplemental Material
Supplemental Material - Analgesic effect of recombinant GABAergic precursors releasing ω-conotoxin MVIIA in a model of peripheral nerve injury in rats
Supplemental Material for Analgesic effect of recombinant GABAergic precursors releasing ω-conotoxin MVIIA in a model of peripheral nerve injury in rats by Stanislava Jergova, Melissa Hernandez and Jacqueline Sagen in Molecular Pain
Footnotes
Acknowledgements
We thank Amanda Pacheco-Spiewak and Anjalika Eeswara for their technical assistance in behavioral testing and data analysis, and Kevin Liebmann and Neal Chandnani technical assistance in tissue preparation and assays.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The University of Miami and J.S. and S.J. hold rights to intellectual property used in the study and may financially benefit from the commercialization of the intellectual property.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Department of Defense Discovery Award PR182408.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
