Abstract
Can mice recognize themselves in a mirror? The answer is unclear. Previous studies have reported that adult mice – when shown itch-like videos - demonstrated itch empathy. However, this was proven to be unreproducible in other studies. In the present study, we wanted to examine whether adult mice were able to recognize their mirror image. In our testing, we found that mice spent more time in the central area in an open field with mirrors surrounding the chamber than those in a normal open field. In a similar open field test with four mice placed in four directions, mice showed similar behavioral responses to those with mirrors. These results indicate that mice are able to recognize images in the mirror, however, they cannot distinguish their own mirror images from the mirror images of other mice. To repeat the experiments of itch empathy, we compared the itch responses of mice in the mirrored environment, to those without. No significant difference in itching responses was detected. Differently, in the case of chemical pain (formalin injection), animals’ nociceptive responses to formalin during Phase II were significantly enhanced in the mirrored open field. A new format of heat map was developed to help the analysis of the trace of mice in the open field. Our results suggest that mice do recognize the presence of mice in the mirror, and their nociceptive - but not itch - responses are enhanced.
Introduction
Mirror self-recognition is the ability to understand the appearance of oneself. Some animals, including chimpanzees, orangutans, and even dolphins, also have a primitive sense of self. 1 The mirror test has been used to determine whether an animal possessed the capacity for visual self-recognition. 2 It has been reported that animals could only pass the mirror test after training,3,4 suggesting that the mirror test is not related to self-consciousness. 5 However, other studies showed that mice that had regular exposure to mirrors could distinguish photographs of their own caged partners from unfamiliar mice. It remains unclear whether the mice could recognize their own image in the mirror.
The self-recognition of mice could be affected by other factors, whether the somatosensory affect the self-recognition has been not reported. Itch and pain are two common somatosensory. Previous studies have reported that adult mice demonstrated itch-like responses after watching itch-like videos or seeing other mice having itching responses, which means that the mice are able to recognize others and experience empathy. 6 However, it has been reported that the mice failed to pass the mirror test due to their poor vision. 7 Dr. M. Catherine Bushnell group reported that they failed to be reproduced contagious itch behaviors for normal mice observing the well-established histamine model of acute itch in demonstrator mice by employing multiple behavioral analysis approaches. 8 In addition, our group also found that mice showed no significant itch-like responses after watching itching video of mice by using a four-iPad paradigm. 9
Pain, like itch, is also an unpleasant somatic sensation. A previous study has shown that pain sensitivity in mice were altered by exposure to their cage-mates, but not to strangers. 10 Recently, our group reported that a long-term observational pain in mice were found in siblings but not strangers. 11 However, it remains to be determined if mice are able to recognize themselves, and if sensory behavioral responses such as pain and itch may be affected by observing their mirror images. In the present study, we combined the mirror test and open field test to study the mice behavior. Open field tests are known to cause anxiety to animals due to social isolation, fear of unknown environments, and open space.12,13 Mice were allowed to move in a rectangular open field surrounded either by mirrors on all four sides, mirror on one side and the rest by walls, or by cages, with their companions on all four sides. We also examined the mirrors effects under itch behavior of histamine injected mice and pain behavior of formalin injected mice in the open field. At the same time, we developed a new system that generates a heatmap based on an animal’s trajectory in the open field.
Materials and methods
Animals
All behavior studies were done on male and female C57BL/6 mice purchased from the Experimental Animal Center of Xi’an Jiaotong University (6–8 weeks old). All mice were randomly housed by three to four per cage under standard laboratory conditions (12 h light/12 h dark, temperature 22–26°C, air humidity 55–60%). Food and water were available ad libitum. All research protocols performed in this experiment have been approved by the Ethics Committee of Xi’an Jiaotong University.
Behavioral tests in the open field
Mice were handled and habituated for 30 min in 3 days before the experiments. Each mouse was tested in the open field chamber. For mice behavior in the open field with mirrors, please refer to our previous work. 9 For the mirror condition test, one or four rectangular mirrors leaned against the walls of the open field to observe whether mice can recognize the images in the mirror.
Mice were placed in an open field (43.2 × 43.2 × 30.5 cm3) inside a dimly lit, with constant temperature room (< 50 lx in the center of the open field). An activity monitoring system (Smart 3.0, Panlab, USA) and a new system were used to record horizontal locomotor activity. Briefly, Smart 3.0 uses paired sets of photo beams to detect movement (number of photo beams: 16; space between the beams: 2.5 cm; number of zones: X: 17, Y: 17). Our proposed system records the video of the mice behavior and gets all information from it. Each mouse was placed in the center of the open field, and activity was measured for 60 min. Central zone was defined by zones from (4, 4) to (13, 13). 14
For companion tests, mice were treated in the same way as in the previous open field experiment. The companions, not like the cage-mates or the littermates, were randomly chosen from the other unfamiliar C57BL/6 mice. We just put one transparent and sealed chamber (5 × 43.2 × 30.5 cm3) besides each side. Mice were placed in each chamber. In this device, the mice in the open field could see, but not smell, its companions. The behavior of the mice in the open field were recorded for 60 min.
Itching behavior of mice
The itching demonstrator mouse was chosen randomly, and 500 μg histamine (Sigma, USA) dissolved in 50 μL saline was injected subcutaneously (s.c.) to the nape of its neck.9,15 The total number of itching bouts of the observers was recorded for 60 min after injection and then obtained by manual counting from the video. The scratching bout was defined as lifting of either hind limb to scratch at the nape of neck and replacing the paw onto the floor, regardless of the number of scratching strokes that occurred between the first lift to final lowering of the hind limb. The mice were randomly put in the open field with or without mirror.
Pain behavior of mice
Each mouse was injected s.c. of left hind paw with 10 μL 5% formalin. Immediately after injection, the mouse was placed in the open field, and the activity of the mouse was recorded by a video camera from underneath the chamber, linked to a computer placed outside the chamber for observation and recording. Phase I of the pain behavior (i.e., paw licking) is caused by the direct effect of formalin on nociceptors, which peaked for 0–5 min to evoke intense but short-duration pain behavior. Phase Ⅱ of the behavior is induced by inflammatory responses caused by formalin which occurred at 30–45 min after formalin injection.16,17
Pain-related responses, or nocifensive behavior, flinching number and the time of pain-related behaviors were recorded immediately after formalin injection and for up to 60 min post-injection. 18 The pain-behavior time is defined as the cumulative amount of time the animal spent on licking, scratching, flicking, or biting the injected hind paw. 19
A novel system for behavior video analysis
In order to understand how mice respond to different environments, it is necessary to use a computer vision system to analyze the behavior of mice because we need to be able to record the mice behavior from the top or the bottom. There are several existing systems, such as Smart 3.0, that can be utilized for observation from the top. In our work, we developed a new system called “Moustat” to extract the trajectory of mouse when inside the open field. The Moustat system is used to detect and trace the mouse, which then generates different statistics such as heatmap, average travel distance, ratio in ROIs, etc. Prior to this system, these statistics could not be derived easily. The Moustat system performs a pre-processing step which rectifies the obtained trajectory coordinate to compensate for the distortion from imperfect camera angles.
From the video, detecting objects, such as a mouse, is a standard task in the computer vision community. There exist many metrics for detecting an object, such as color difference, background subtraction,20,21 or more complicated deep neural network based method such as Yolo 22 or CenterNet. 23 In the Moustat system, we resort to a background subtraction-based method to locate image regions (i.e., the mouse) that do not belong to the static background (i.e., the box). Since the video is recorded at a constant frame rate (10 fps in our experiments), each location in the picture represents 0.1 seconds of stay at that location. By accumulating the dwell time for each pixel, a heatmap can be obtained. In pixel locations with longer dwell time, there will be higher heat, and vice versa.
Statistical analysis
All data are expressed as Mean ± SEM. Statistical comparisons between two groups were performed to identify significant differences. When the data was normally distributed, and the variance was homogeneous, two-tail unpaired Student’s t-test was used. p < 0.05 was considered statistically significant.
Results
Mirror Open field behavior in mice
To verify whether mice recognize the image in the mirror, we put the mice in an open-field with four mirrors on each side (Figure 1(a)), and used the video to record the activity of mice (Figure 1(b) and (c)). We observed that mice spent more time in the central area compared with those in a normal open field. We took the time of mice staying in the central area in the first 10 min as the base 1, and counted the ratio of mice spending time in the central area every 10 min for 60 min. We found that the ratios were significantly changed during 20–30 min and 30–40 min in both the male and the female groups (Figures 1(d) and (e); mirror vs. control, 2.80 ± 0.33 vs. 0.69 ± 0.08 for 20 – 30 min, t = 0.015, and 3.50 ± 0.52 vs. 0.94 ± 0.15 for 30–40 min, t = 0.046, in the male mice group; 3.17 ± 0.19 vs. 1.00 ± 0.27 for 20–30 min, t = 0.031, 2.86 ± 0.15 vs. 1.04 ± 0.27 for 30–40 min, t = 0.043, in female mice group, *p < 0.05, n = 8 mice for each group, Student’s t-test). Interestingly, there was no behavior difference between male and female mice in the open-field with four mirrors on each side. Next, we attempted to study if the mice spending more time in the central area was due to fear of the objects in the mirror or just less anxiety, and thus we performed a side mirror open field test. We found that the mice in the one mirror open field still spent more time in the central area as compared with those in the normal open field (Figures 1(g) and (h); n = 6 mice). These results suggest that mice can see the objects in the mirror and are influenced by those images. Since mice spent more time in the central area, it is believed that the mice have less anxiety in the open field with mirror. Mirror induced behavior changes of mice in the open filed tests. 
Heatmap generation of mice trajectory
Specifically, for better visualization, we adopted the Moustat system to track the mice behavior. We defined a small heat kernel for each pixel in the frame, such that for each pixel (x, y) on the obtained trajectory (Figure 2(a)), we added the kernel to a small neighboring region centering at (x, y). The heat kernel has a regular Gaussian shape, and the value at the centering position is 1.0 as shown in Figure 2(b). Note that the width (covariance) of the Gaussian kernel is not that important except that larger Gaussian kernel size makes the obtained heatmap smoother. Once we aggregate the heat from all positions in a trajectory, we can obtain a heatmap as shown in Figure 2(c), which more intuitively reflects the residence time, and position, of mice in the open field. We used the heatmaps which were generated from the video to show the mice position and dwell time (Figure 2(d)). Heatmap generation. 
Companion-around Open field behavior in mice
Our next step was to investigate whether less anxiety is due to the recognition of objects in the mirror, or the recognition of themselves, and we put four companions around the open field. The four companions were put into the boxes beside the open field (Figure 3(a)), and these boxes are transparent and sealed, such that the test mice in the open field could see the outside companions but could not smell them. Similarly, the video tracers were used to record the activity of the mice in the open field. We found that again the tested mice spent more time in the central area. We compared the central time ratio in three conditions and found that, there was no significant difference between companions or mirror surrounding open field (Figure 3(b); companion vs. control, 2.64 ± 0.14 vs. 0.69 ± 0.08 for 20–30 min, t = 0.020, and 2.49 ± 0.07 vs. 0.94 ± 0.15 for 30–40 min, t = 0.014, 3.00 ± 0.18 vs. 1.05 ± 0.14 for 40–50 min, t = 0.021, #p < 0.05, n = 6 mice for companion group, Student’s t-test). We used heatmap to describe the track of the mice (Figure 3(c)) where it is clearly seen that the mice spent more time in the central area. Therefore, we concluded that the mice behavior in the companion-around open field is similar to that in the mirror surrounding open field. The mice did not distinguish the image in the mirror from the companions. Companions induced behavior changes in mice. 
Mirror Open field behavior in mice with histamine-induced itch model
From the above experiments, we found that mice behavior is affected by the mirror in the open-filed. Since it’s not clear to what extent mirrors affect the mice, we preformed difference disease models in the mice and tested the behavior changes in the open field. We injected the histamine subcutaneously to the nape of mice neck and then randomly put the mice in the normal open field or mirror surrounding open field. After we analyzed the track of mice and counted the number of scratching bouts, we found that the numbers of scratching bouts showed no difference in the normal group and the mirror group, nor between the male and female groups (Figure 4(b); control vs. mirror, 45.80 ± 7.68 vs. 34 ± 3.84 for male group, 53.80 ± 11.93 vs. 52.90 ± 7.85 for female group, n = 5 mice for each group, Student’s t-test). We still used the heatmap to compare the trajectory and dwell time in the open field (Figure 4(c)) and found that the mice spent more time in the corners no matter whether mirrors were present or not. The dwell time in the center was not increased in itch behaviors mice in the presence of mirrors (Figure 4(d)). These results suggest that the effect of the mirror to reduce anxiety is weakened or lost completely after histamine is injected. Itch behavior changes under mirror condition in mice. 
Mirror Open field behavior in mice with formalin -induced pain model
Next, to investigate the effect of mirrors on the pain behavior of mice, formalin (5%, 10 μL) was injected into the left hind paw of the mice, and then the mice were put in the normal or mirror surround open filed (Figure 5(a)). Flinching number in phase I and phase Ⅱ were recorded immediately after formalin injection for up to 60 min post-injection and the licking time of pain behaviors was also recorded. In phase I (0–5 min), the mice flinch and lick the hind paw quickly, almost all the flinch bouts were less than one second. We considered it as one time flinching. In phase Ⅱ, the mice always kept licking or biting their hind paw in one flinching. We count the number of flinching and the time of pain behavior. There was no difference in the number of flinching either in phase I or phase II with or without mirror (Figure 5(c); control vs. mirror, 26 ± 0.7874 vs. 25.4 ± 0.23 for phase I, 27.6 ± 1.13 vs. 34.2 ± 0.88 for phase II, n = 5 mice for each group, Student’s t-test). However, in phase II, the time of pain behavior including licking, scratching, flicking, or biting was greatly increased in the mirror surround open field compared with those in normal open field (Figure 5(d); control vs. mirror, 80.20 ± 10.82 vs. 247.60 ± 56.06, t = 0.019, *p < 0.05, n = 5 for each group, Student’s t-test). This difference in phase I and phase II may be due to the different pathways of various pain types. The heatmap (Figure 5(e)) and central time ratio statistical data (Figure 5(f)) also showed that the mice spent more time in the corners even the mirror is on, indicating that the effect of the mirror on the trajectory of the mice is weakened after formalin injection. Pain behavior changes under mirror condition in mice. 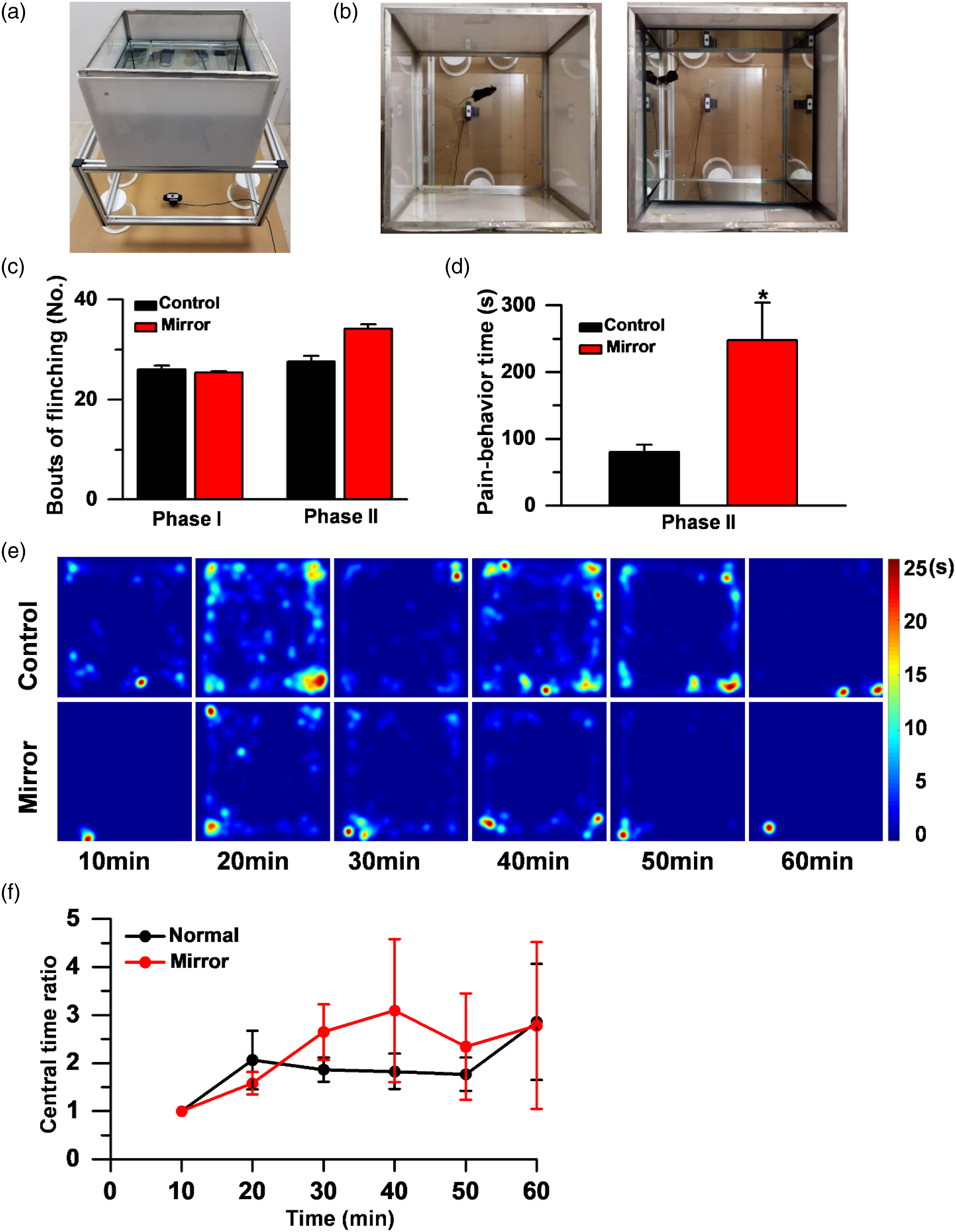
Discussion
Many studies have reported that different animals are able to pass the mirror test and have good self-recognition ability, whereas there were very few studies on mice. In our present study, we want to understand whether the mice can recognize the images in the mirror, as well as their behavioral changes. Different kinds of mice behaviors were successfully observed when they were in the normal or mirror surrounding open field by using the newly developed video system to record and analyze the mice trajectory visualized as a heatmap. We found the mice saw the objects in the mirror and felt less anxiety in the mirror surrounding open field, as if they saw their companions. We considered that the mouse saw the images in the mirror. Because the mice trajectory and dwell time in mirror surround open field and companion-around open field are highly similar, we speculate that mice cannot recognize the images are themselves. After injecting histamine or formalin, the anxiolytic effect by viewing mirror was reduced or even disappeared. However, nociceptive behaviors are enhanced, while itch responses are not affected.
The mirror test is a test of self recognitive ability by distinguishing its own face or body in the mirror. It is not clear whether the mice could further recognize the mirror image on their own. Some animals could recognize the self-image in the mirror through certain learning process, 24 while many other animals would fail the mirror test.25,26 There is debate on how to interpretate the results of the mirror test. 7 Some studies suggested that the application of this test to animals that mainly rely on perception other than vision is not appropriate. 1 It’s well known that mice have poor eyesight. 27 While in our present study, we found that mice did see the objects in the mirror by showing less anxiety. It is revealed in previous reports regarding the social behavior of mice that, mice would surround their peers, even same gender peers, and interact with each other. 28 In our study, since the mice could not smell each other in open field surrounded by either mirror or companions, it is clearly observed that although the anxiety of mice was reduced, they did not present more interaction with their peers. Hence, whether the social behavior of mice needs to be assisted by olfaction in addition to vision needs further study.
In the present studies, we found that the effects of the mirror were not significant under itch or pain conditions. In the itch and short-duration pain, Phase I of formalin-induced pain, the presence of the mirror resulted in almost no difference in mice behavior. Interestingly, in Phase Ⅱ, the effect of mirror on the behavior of mice, by analyzing the trajectory of mice action, show no difference. On the other hand, while the numbers of flinching were about the same, the mice spent more time on licking or biting their hindpaw. These results clearly indicate that behavioral nociceptive responses are clearly enhanced. This finding is consistent with recent results reported by our group and others in regard of observed or spread of pain.11,29 Recently, our group reported sibling but not stranger observer mice exhibit observational pain. 11 Smith et al. reported that a brief social interaction with a mouse experiencing pain or morphine analgesia resulted in the transfer of these experiences to its social partner. 29 These results consistently indicate that rodents can communicate with each other of their social emotions, although how they may communicate such information remain to be determined. However, we did not observe the contagious itch behavior under our mirror condition. And in our group previous study, after watching itch-like videos, normal mice did not show itch empathy. 9 Dr. M. Catherine Bushnell group also reported that they failed to be reproduced contagious itch behaviors for normal mice. 8 Observing each other’s images and possible face expression may be one of several communication methods. In the present study, we found that mice cannot distinguish their own images from others in the mirror. It is possible that they may employ other communication methods such as ultrasonic vocalization 30 or odors to convey the information. Future studies are clearly needed to determine the exact mediators for social emotion.
To better analyzes the track of mice behavior, in our present study, we adopted a new Moustat system to visualize the trajectory from the videos. Comparing Moustat to traditional behavior recording system, this system can extract information from the video, no matter recorded from the top or the bottom, correct the distortion caused by the lens, reset the ROI and generate the heat map, which is conducive to our intuitive analysis of the trajectory, dwell position, and time of the mouse in the open field. This new system allowed us to collect data which demonstrates that mice did recognize objects in the mirror, and we found that mice showed less anxiety when they were in the mirrored open field or under the companion-around condition. We believe that this system will be useful for future investigation of mouse behaviors in the open field.
In conclusion, our results indicate that mice saw the objects in the mirror, hence the mouse can be a useful experimental model for elucidating the neural mechanisms of mirror image cognition. Meanwhile, since mice could not further distinguish between images of themselves from others, the mechanism of mirror effects requires further investigation. Anyway, our results clearly indicates that the mice may help us to reveal the relationship between the pathway of pain and anxiety.
Footnotes
Acknowledgement
The authors would like to thank Yong-Min Liu for his constructions of the experiment.
Authors’ contributions
S.B.Z, and M.Z. designed the experiments. S.B. Z., M.X., W. S., K. F., Y.X.C. performed behavioral experiments and analyzed data. J.J.W. built the Moustat system, S.B.Z, M.X. J.S.L. Q.Y. C. and X.H.L, M.Z. drafted the manuscript and finished the final version of the manuscript. All authors read and approved the final manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: M.Z. is in part supported by grants from the Canadian Institute for Health Research (CIHR) project grants (PJT-148648 and 419286). X.H.L. is supported by grants from the National Science Foundation of China (32100810).
