Abstract
Itch and pain are both unpleasant, but they are discrete sensations. Both of these sensations are transmitted by C-fibers and processed in laminae I-II of the dorsal horn. To examine whether pruriception modulates pain, we first confirmed the activation of cells in the itch-related circuits that were positive for gastrin-releasing peptide (GRP) and GRP receptor (GRPR) using a paw formalin injection model. This pain model with typical biphasic pain behavior increased c-Fos but did not affect the expressions of GRP and GRPR mRNAs in the dorsal horn. Using c-Fos expression as a marker for activated cells, we confirmed that formalin injection increased the number of cells double-labeled for c-Fos and GRP or GRPR in the dorsal horn. The emergence of these neurons indicates the activation of itch-related circuits by acute pain signals. The effect of an antagonist for a GRPR was examined in the paw formalin injection model. Intrathecal chronic antagonization of spinal GRPR enhanced the onset of phase II of paw formalin injection-induced pain behavior. Exogenous intrathecal GRP infusion to the paw-formalin injection model not only showed significant reduction of pain behavior but also increased c-Fos in the inhibitory neurons in the dorsal horn. The anti-nociceptive effect of spinal GRP infusion was observed in the peripheral inflammation model (complete Freund’s adjuvant injection model). In this study we suggest that painful stimuli activated itch-related neuronal circuits and uncovered the spinal activation of the itch-induced analgesic effect on acute and established inflammatory pain.
Keywords
Introduction
Pain and itch are two distinct yet related sensations. Both of these sensations are detected mainly by C-fiber dorsal-root ganglia (DRG) neurons and transmitted to the central nervous system. Indeed, chemical compounds that induce pain and itch can evoke characteristic behavioral responses, such as withdrawal (to avoid tissue injury) and scratching (to remove irritants), respectively, in animal models. Recent studies have elucidated the key elements of itch sensor neurons in the primary afferent neurons.1–4 Unmyelinated primary afferents are the major neuronal types that mediate pain and itch transmission to the spinal cord.5,6 In the last decade, several key molecules (peptides and receptors) of the peripheral and spinal itch pathways have been identified. In the periphery, Mas-related G-protein-coupled receptor member A3 (MrgprA3)-positive pruriceptors are sensitive to variety of itch stimuli. 4 Additionally, other subsets of peripheral pruriceptors are identified by the expression of brain-type natriuretic peptide (Nppb) or somatostatin.7,8 At the spinal cord dorsal horn level, the critical roles of the local neural circuits expressing neuropeptide gastrin-releasing peptide (GRP) and GRP receptor (GRPR) in itch processing have also been elucidated.9–11 These reports uncovered the specific neuronal components of primary afferent and spinal circuits for itch, which can be experimentally differentiated by molecular characteristics. Therefore, pathways carrying itch information centrally are considered to form “labeled lines,” meaning that they each convey a particular stimulus. Accompanying this “labeled line theory,” it is well known that the different types of stimuli modify each other at the dorsal horn circuit level. This spinal modification plays an essential role in the processing and recognition of sensory information in the physiological and pathological conditions. For example, peripheral nerve stimulation activates feed-forward inhibitory pathways in the dorsal horn and decreases pain sensitivity, leading to an analgesic effects in an animal model.12,13 Dysfunction of the feed-forward inhibitory circuit induces mechanical allodynia. 14 It has been reported that scratch-induced mechanical signals can reduce itch, and analgesic opioids induce itching, which indicates an antagonistic interaction. 15 These modifications provide important aspects of the spinal cord circuitry of itch that shows attenuation by counter stimuli. Conversely, it remains unclear whether itch signals in the spinal cord can act as counter stimuli to modify pain signals or how much of the itch circuit is activated by acute painful inflammatory information in the dorsal horn. In addition, the effects of activation or inhibition of spinal itch circuits on pain have not yet been reported. In this study, we demonstrated the effect of acute inflammatory pain on the itch circuits and examined the effect of the activation or inhibition of itch circuits on pain behaviors.
Material and methods
Animals
Adult male Sprague–Dawley rats weighing 250–300 g were purchased from Japan SLC, Inc and used in all experiments in this study. The animals were maintained in a 12-h light/dark cycle with light on at 8:00 a.m. in a temperature-controlled (25.0°C±1.0°C) room, were group-housed in cages (two animals per cage), and had free access to food and water. Rats were anesthetized with sevoflurane (3.0%–5.0% inhalation administration, in air) during all operations.
All animal experimental procedures were approved by the Hyogo College of Medicine Committee on Animal Research (approval number, 20–024) and were conducted in accordance with the National Institutes of Health guidelines on animal care. All efforts were made to minimize suffering.
Animal model
We used formalin and Complete Freund’s adjuvant (CFA) for the acute and inflammatory pain models, respectively. Rats were anesthetized with sevoflurane (3.0%–5.0% inhalation administration, in air), and formalin (50 μL of a 1.5% solution of formaldehyde with saline) or CFA (100 μL of a 1:1 mixture of saline/CFA) was injected into the plantar surface of the left hind paw using a 1-mL tuberculin syringe equipped with a 30-gauge needle.
Immunohistochemistry (IHC)
Immunohistochemistry (IHC) was performed as previously described. 16 Briefly, 2 h after formalin administration, rats were deeply anesthetized with sodium pentobarbital and perfused transcardially with 300 mL of 1% paraformaldehyde (PFA) in 0.1 M phosphate buffer (PB, pH 7.4), followed by 500 mL of 4% PFA in 0.1 M PB.
The spinal cords were dissected out and post-fixed in the same fixative at 4°C overnight, followed by immersion in 20% sucrose in 0.1 M PB at 4°C for 3 days. The bilateral L4–L5 spinal cord was dissected out, rapidly frozen in powdered dry ice, and cut on a cryostat to a thickness of 14 μm. Sections were mounted onto MAS-coated glass slides (Matsunami, Osaka, Japan).
After washing in Tris-buffered saline (TBS) (0.1 M Tris-HCl, pH 7.4, and 0.15 M NaCl), the sections were immersed in 50% ethanol for 10 min, 70% ethanol for 10 min, and 50% ethanol for 10 min to enhance antibody penetration.
For single immunohistochemistry (IHC) staining of c-Fos, the sections were preincubated in TBS containing 5% normal horse serum (NHS) for blocking for 30 min, followed by incubation with polyclonal rabbit anti-c-Fos antibody (1:500, Synaptic Systems, Germany, Cat# 226 003, RRID: AB_2231974) in 5% NHS for 2 h at room temperature. The sections were washed in TBS and then incubated in ImmPRESS horse anti-rabbit IgG Polymer reagent (Vector Laboratories, Cat# MP-7401, RRID: AB_2336529) for 30 min at room temperature. The sections were washed in TBS and the peroxidase reaction was developed with 0.05% diaminobenzidine (DAB) (Wako) and 0.01% hydrogen peroxide in TBS. The sections were then washed in TBS and dehydrated in a graded ethanol series, cleared in xylene, and coverslipped.
For double-labeling IHC of c-Fos with paired box 2 (Pax2), the sections were processed as previously described. 17 In brief, TBS containing 5% NHS was used for the incubation of sections with guinea pig anti c-Fos polyclonal antibody (1:200, Synaptic Systems, Germany, Cat# 226 308, RRID: AB_2905595) and rabbit anti-Pax2 monoclonal antibody (1:10,000, abcam, UK, Cat# ab79389, RRID: AB_1603338) for 24 h. Fluorescence-conjugated secondary antibodies, donkey anti-rabbit IgG Alexa Fluor 488 (1:1,000, Thermo Fisher Scientific, USA, Cat# A-21206, RRID: AB_2535792) and donkey anti-guinea pig IgG CF555 (1:500, Biotium, USA, Cat# 20276–1, RRID: AB_10853597) were used for the visualization of immunoreactivity. For quantification of c-Fos immunoreactive cells, the immunoreactive intensity was measured by a computerized image analysis system (NIH Image, version 1.61). In 256 Gy scale signal gradients, we considered signal intensities above 192 as positive signals. For the fluorescent image of c-Fos and Pax2, signals on the DAPI were counted as the positive signal.
In situ hybridization histochemistry (ISHH)
Using enzyme-digested clones, 35S UTP-labeled rat GRP and GRPR cRNA probes (GRP: accession number M31176.1, 418–974; in pCRII TOPO vector, GRPR: accession number NM_012706.2, 566–1166; in pCRII TOPO vector) were synthesized. For autoradiography, the sections were coated with NTB emulsion (Eastman Kodak, Rochester, NY, USA), diluted 1:1 with distilled water at 42°C, and exposed for 4 weeks (GRP) or 6 weeks (GRPR) in light-tight boxes at 4°C. After development in D-19 and fixation in 24% sodium thiosulfate, the sections were rinsed in distilled water, stained with hematoxylin, dehydrated in a graded ethanol series, cleared in xylene and coverslipped.
In order to examine the distribution of c-Fos, and GRP or GRPR mRNA, we combined IHC with ISHH. The treatment of sections and the method of double-labeling with IHC and ISHH were described previously. 18 The sections were washed in TBS and immersed in 50% ethanol for 10 min, 70% ethanol for 10 min, and 50% ethanol for 10 min to enhance antibody penetration, and 0.3% H2O2 in methanol for 5 min to block endogenous peroxidase activity. After washing in TBS, the sections were preincubated in 2.5% NHS for 30 min, followed by incubation in rabbit anti-c-Fos antibody (1:500) in 5% NHS with 100 U/mL RNase inhibitor overnight at 4°C. The sections were washed in TBS and then incubated in ImmPRESS horse anti-rabbit IgG Polymer reagent for 30 min at room temperature. The sections were washed in TBS and the horseradish peroxidase reaction was developed in TBS containing 0.05% DAB and 0.01% hydrogen peroxide. Sections were then washed in TBS. After immunohistochemistry, sections were immediately processed for ISHH.
To measure the number of cells positive for GRP or GRPR mRNAs, neuronal profiles that contained nuclei or c-Fos positivity were used for quantification. Using the ISHH sections with sense probe, we determined the background level of grains. In a diameter 10 μm circle of the ISHH section of sense probe, the average number of grain was 0.35 for GRP ISHH and 0.52 for GRPR ISHH. We used a signal-to-noise (S/N) ratio for each cell in each tissue. Cell nuclei or c-Fos-immunoreactivity (ir) with a density of grains ten-fold the background level or higher (approximately more than four or six grains on the nuclei of c-Fos-ir cells labeled for GRP and GRPR, respectively) were considered positively labeled for GRP or GRPR mRNA.
Drug administration
Gastrin-releasing peptide peptide (R&D Systems, MN) and RC-3095 (Sigma-Aldrich, MO) were dissolved in sterile water. Under adequate inhalation anesthesia with sevoflurane (3%–5%, in air), the vertebral arch of the L5 vertebra was resected and a 5 mL soft tube (SILASTIC laboratory tubing, O.D.: 0.64 mm; Dow Corning Corporation, MI, USA) filled with saline solution was inserted. The tube was inserted 10 mm into the subarachnoid space (toward the head). For a single intrathecal injection of GRP, the end of the soft tube was ligated and fixed subcutaneously. The next day, under sevoflurane inhalation anesthesia, the ligated tube was cut and GRP was administered using a Hamilton syringe. Furthermore, 0.03 nmol and 0.3 nmol doses of GRP were administered (diluted in saline at 8.5 μg/mL and 85 μg/mL, respectively).19,20 For intrathecal administration of RC-3095 for 1 day, a pump (MINI-OSMOTIC PUMP MODEL 2001D; DURECT Corporation, CA) was connected to a soft tube and the pump was held subcutaneously with sutures. The dosages of RC-3095 were 0.3 nmol and 3 nmol per day (diluted in saline at 14.2 ng/mL and 142 ng/mL, respectively), and the solutions were injected using a mini-pump.
Behavioral experiments
After injection of formalin into the plantar of the left paw, the rats were immediately returned to the cage. The number of flinching bouts occurring at 5 min intervals for 60 min after injection was recorded. The GRP-treated group received formalin 3 min after GRP administration and the RC-3095-treated group received formalin 24 h after continuous administration using a mini pump as described above, and pain behavior was evaluated.
For the analysis of the CFA model, heat hypersensitivity was tested using the plantar test (7370, Ugo Basile, Italy). Briefly, a radiant heat source beneath a glass floor was aimed at the plantar surface of the hind paw. Three measurements of latency were taken for each hind paw in each test session. The hind paws were tested alternately with greater than 5 min intervals between consecutive tests. The three measurements of latency per side were averaged. All the behavioral experiments were assessed by researchers blinded to the treatment type.
Statistical analysis
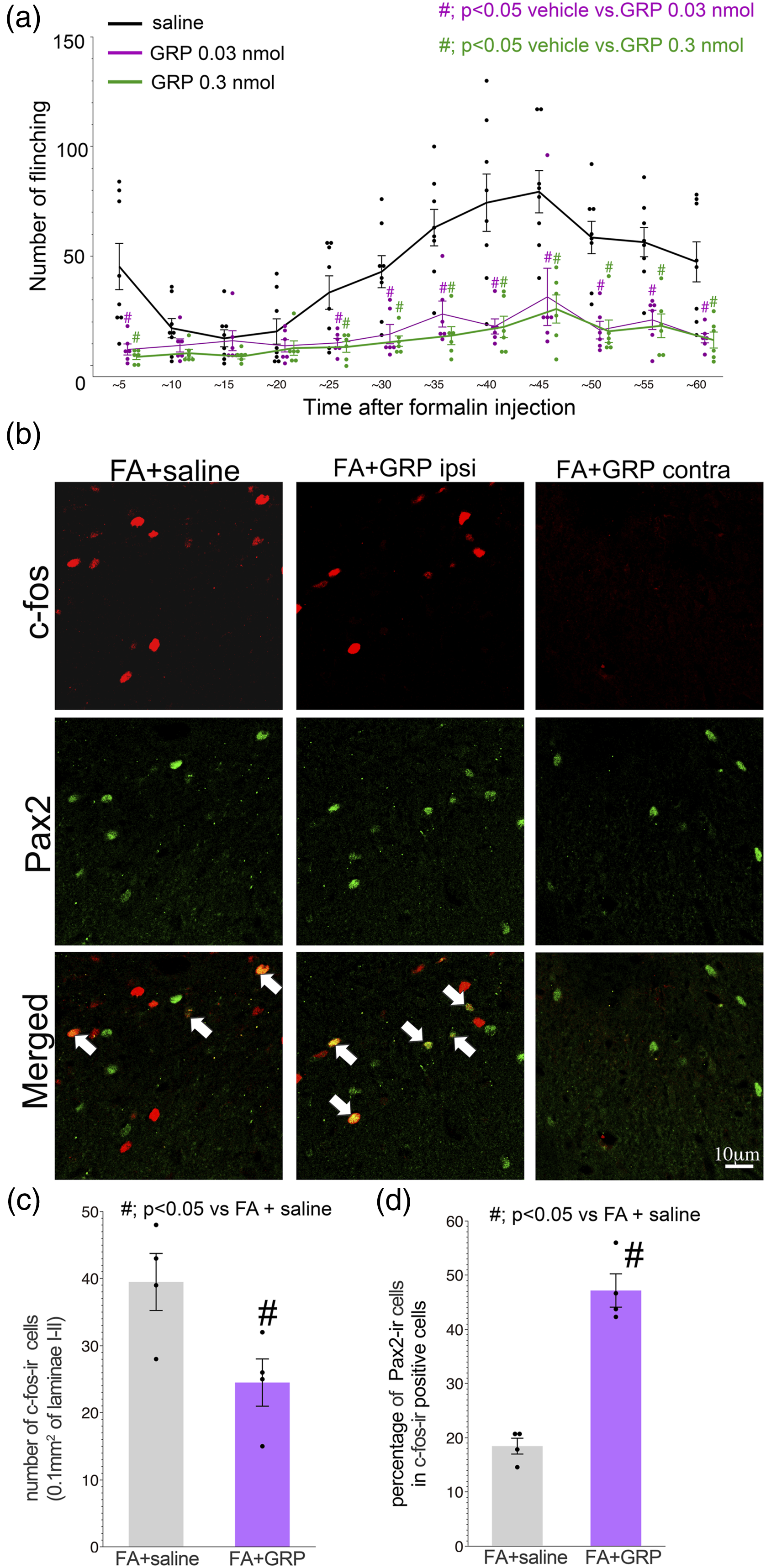
All data are presented as the mean ± standard error of mean (SEM). Statistical analyses were performed using Statview (Statview, SAS Institute Inc., RRID: SCR_017411) and/or JMP Pro 15 (JMP, SAS Institute Inc. RRID:SCR_014242). Parametric statistical tests were used to compare the different experimental groups. The paired Student’s t-test was used to assess statistical significance between two groups (Figure 1(b), (d), (g), (j), and (l), and Figure 3(c) and (d)). When more than three groups were compared, one-way non-repeated measures analysis of variance (ANOVA) was used, followed by a Fisher’s Protected Least Significant Difference (PLSD) post-test (Figures 2, 3(a), 4(a) and (b)). A difference was accepted as significant if the probability was less than 5% (p < 0.05). Intradermal formalin injection activates dorsal horn neurons expressing mRNAs for gastrin-releasing peptide receptor (GRP) and GRP receptor (GRPR). (a), Representative images of DAB staining photomicrographs of c-Fos-ir in the dorsal horn 2 h after the formalin injection in the contralateral (left) and ipsilateral (right) sides. Bar; 100 µm. (b), Quantification of the c-Fos-ir in the dorsal horn of formalin-injected rats (n = 4, five slides for each rat). Y-axis represents total number of c-Fos-ir per animal (mean ± SEM). # indicates significance compared with the contralateral side (p < 0.05; t-test). (c)-(h), Double labeling analysis of c-Fos-ir with mRNA for GRP or GRPR in the dorsal horn of the formalin injection model. (c) and (f), Representative image of double labeling of c-Fos with GRP mRNA (c) or GRPR mRNA (f) at 2 h after formalin injection. Arrows indicate double-labeled cells with mRNA and c-Fos. Bars; 25 μm. (d) and (g), Quantification of the percentages of double labeling profiles (c-Fos-ir with mRNAs) in total GRP (d) or GRPR (g) positive cells in the dorsal horn of formalin injection model. Y-axis represents the percentage of double labeling profiles of c-Fos-ir with mRNAs for GRP (d) or GRPR (g) positive signals. Data are expressed as mean ± SEM. For GRP/c-Fos analysis, n = 4, four sections from each animal, total 1584 GRP positive cells from contralateral and 1581 cells from ipsilateral side. For GRPR/c-Fos analysis, n = 5, four sections from each animal, total 714 GRPR positive cells from contralateral and 680 cells from ipsilateral side. # indicates significance compared with the contralateral side (p < 0.05; t-test). (e) and (h), Percentage of double labeled cells of c-Fos-ir with mRNAs (GRP or GRPR) in total c-Fos-ir profiles (for GRP/c-Fos-ir, mean ± SEM, n = 4, four sections from each animal, total 1071 c-Fos-ir, and for GRPR/c-Fos, n = 5, four sections from each animal, total 1183 c-Fos-ir cells). (i) and (k), Dark field images of the single labeling ISHH of mRNA for GRP (i) and GRPR (k) in the dorsal horn at 2 h after formalin injection. Insets of (i) and (k) show bright field images of positive cells for GRP and GRPR in the contra (left) and ipsilateral side (right) of formalin injection. Bar; 100 μm (dark field images).; 10 μm (insets). (j) and (l), Quantification of GRP and GRPR positive cells in the dorsal horn showing that the formalin injection did not affect the expression of GRP and GRPR (for GRP mRNA, n = 4, four sections per animal and for GRPR mRNA, n = 5, four sections per animal). Y-axis represents the mean number of positive cells per animal (mean ± SEM, n = 4–5, four sections from each animal). The effects of intrathecal administration of RC-3095, an antagonist for GRPR, on formalin-induced spontaneous paw flinching. Formalin was intradermally injected into the hindpaw followed by the administration of RC-3095 at 5 min after formalin injection (0.3 and 3 nmol). The low dose of RC-3095 (0.3 nmol) partially enhanced the formalin-induced pain behavior from 25 min to 35 min after formalin injection. In all graphs, values are represented as mean ± SEM (n = 8 for saline and 3 nmol RC-3095 group, n = 5 for 0.3 nmol RC-3095 group). # indicates p < 0.05 vs. saline group, one-way ANOVA followed by individual post hoc comparisons (Fisher’s PLSD). (a), The effects of intrathecal administration of GRP on formalin-induced spontaneous paw flinching. Formalin was intradermally injected to the hindpaw followed by GRP administration at 5 min after formalin injection (0.3 and 0.03 nmol). Both doses of GRP administration partially reversed the formalin-induced pain behavior from 5 min to 60 min after formalin injection. In all graphs, values are represented as mean ± SEM (n = 8 for saline group, n = 6 for each of the GRP groups). # indicates p < 0.05 vs. saline group, one-way ANOVA followed by individual post-hoc comparisons (Fisher’s PLSD). (b)-(d), Double labeling analysis of c-Fos-ir with Pax2 in the dorsal of paw formalin injection model treated with intrathecal saline or GRP. (b), Representative images of c-Fos-ir (red) with Pax2-ir (green). Arrows indicate double-labeled cells with c-Fos and Pax2. (c), Quantification of the c-Fos-ir in the dorsal horn of formalin injected rats treated with intrathecal saline or GRP (n = 4, 0.1 mm
2
from each rats). Y-axis represents total number of c-Fos-ir per animal (mean ± SEM). # indicates significant compared with the saline treatment group (p < 0.05; t-test). (d), Graph shows the percentage of Pax2-ir profiles in the total c-Fos-ir in the dorsal horn of the formalin injection model treated with intrathecal saline or GRP. # indicates significance compared with the saline treatment group (p < 0.05; t-test). Effect of intrathecal administration of GRP on the thermal sensitivity of CFA model rats. GRP (0.03 nmol) was infused into the intrathecal space at 1 day after CFA injection. (a), Paw withdrawal threshold of the side contralateral to CFA injection. GRP injection did not change the thermal sensitivity of the contralateral side. (b), Intrathecal infusion of GRP significantly inhibited the established thermal hyperalgesia. Note that the withdrawal thresholds for thermal stimuli at 0 and 5 min after GRP infusion were not changed. GRP almost completely reversed thermal allodynia at 60 min after infusion. # indicates p < 0.05, by t-test compared with corresponding vehicle controls (saline) (n = 5).


Results
Activation of gastrin-releasing peptide (GRP) and GRP receptor-positive neurons in the dorsal horn by painful stimulation
To test whether the paw formalin injection activated GRP- and GRPR-expressing neurons, we first assessed c-Fos expression in the dorsal horn neurons. Two hours after formalin injection, the induction of c-Fos-ir was evident and showed a significant increase compared to contralateral side (Figure 1(a)), and we found that we could use c-Fos-ir as a marker of neuronal activation in specific neurons in the dorsal horn (Figure 1(b)).
Using c-Fos-ir with double labeling of mRNAs for GRP or GRPR, we examined whether the painful stimuli induced by paw formalin injection could activate neurons positive for GRP or GRPR in the L4/5 dorsal horn (Figure 1(c)-(h)). In the double-labeling images of the spinal cord taken 2 h after formalin injection, we observed the induction of c-Fos-ir in cells expressing GRP or GRPR mRNAs (Figure 1(c) and (f)). Quantitative analysis showed that the formalin injection significantly increased the number of c-Fos-positive neurons double-labeled with GRP mRNA or with GRPR mRNA in the ipsilateral dorsal horn compared with contralateral side. About 25% of GRP mRNA expressing neurons were labeled for c-Fos-ir in the ipsilateral dorsal horn (Figure 1(d)). Conversely, about 40% of c-Fos-ir cells co-expressed GRP mRNA (Figure 1(e)). Quantification of cells double-labeled for c-Fos and GRPR mRNA revealed that the intradermal formalin injection significantly increased the number of double-labeled cells, and about 34% of GRPR positive cells expressed formalin-induced c-Fos-ir (Figure 1(g)). Conversely, about 20% of formalin-induced c-Fos-ir cells were co-labeled with GRPR mRNA (Figure 1(h)). Therefore, we considered that the formalin injection-derived painful signals could activate a population of neurons positive for GRP or GRPR in the dorsal horn.
We confirmed the effect of paw formalin injection on the expression of mRNAs for GRP and GRPR in the L4/5 dorsal horn neurons by ISHH at 2 h after formalin injection (Figure 1(i) - (l)). The expression of mRNAs for GRP and GRPR showed specific patterns in laminae I-II of the dorsal horn, respectively (Figure 1(i) and (k)). Quantitative analysis revealed that the paw formalin injection did not change the number of cells positive for GRP or GRPR mRNA expression (Figure 1(j) and (l)). In addition, we did not observe a difference in signal intensity of the mRNAs for GRP or GRPR between the ipsi- and contralateral dorsal horn (Insets of Figure 1 (i) and (k)).
Effect of intrathecal injection of GRPR antagonist on the formalin induced pain behavior
Induction of c-Fos-ir in GRP- and GRPR positive neurons suggested a possible involvement of GRP in the pain processing in the dorsal horn in the formalin model. In order to elucidate the roles of the endogenous GRP-GRPR system in pain processing, we administered two doses of the GRPR antagonist RC-3095 intrathecally and examined pain behavior using the formalin injection model (Figure 2). As previously reported, the formalin-induced flinching occurred in two phases, which were observed at 0–10 min and at 25–60 min after formalin injection (Figure 2, vehicle treated group). The series of RC-3095 treatments did not affect the first phase of pain behavior (0–10 min after injection). However, in the second phase of the pain behavior from 25 to 35 min after formalin injection, both doses of RC-3095 increased the number of flinches (Figure 2). From 35 to 60 min formalin injection, intrathecal injection of RC-3095 showed a similar pain behavior pattern compared to the control group (Figure 2).
Effects of intrathecal GRP injection on behavior in the formalin model
Enhancement of pain behavior by the intrathecal GRPR antagonist RC-3095 (Figure 2) indicates the anti-nociceptive role of GRP in the acute pain state. In order to examine the anti-nociceptive effect of GRP in the dorsal horn, we examined the impact of exogenous administration GRP on pain behavior in the rat formalin model. To this end we intrathecally administered GRP and subsequently injected formalin into the left hind paw, and examined the change in the number of flinches compared to vehicle control (Figure 3(a)). After formalin injection we could observe a number of spontaneous flinches in the pre-injected saline group. For GRP pre-treatment we tested two doses (0.03 and 0.3 nmol), and both doses of the intrathecal GRP treatment apparently suppressed the formalin-induced flinching behavior not only in the first but also in the second phase (Figure 3(a), GRP treated group). This data suggests that itch related to GRP may have an anti-nociceptive effect on pain behavior after formalin injection into the hind paw.
Using the formalin model treated by intrathecal GRP, we performed double labeling analysis of
Effect of GRP on inflammation-induced thermal hyperalgesia
So far, we found that the acute pain signal activated a part of the itch-related neuronal circuit, and that the intrathecal GRP injection reduced pain behavior associated with the activation of inhibitory neurons in the dorsal horn in this model. Next, we examined the effect of exogenous GRP on the intraplantar CFA injection-induced inflammatory pain model. CFA injection resulted in increased sensitivity to noxious heat, which is mediated by dorsal horn sensitization. 21 On the contralateral side of this model, CFA injection did not change the thermal sensitivity at 1 day after the CFA injection. Gastrin-releasing peptide (0.03 nmol) injection did not affect the contralateral paw withdrawal latency (Figure 4(a)). CFA injection induced thermal hyperalgesia that was characterized by the reduction of the withdrawal threshold to thermal stimuli on the ipsilateral side 1 day after injection (Figure 4(b)). Intrathecal infusion of GRP showed a delayed effect on thermal hyperalgesia. Spinal GRP administration did not affect thermal sensitivity at the early time point but significantly reversed thermal hyperalgesia at 60 min after administration (Figure 4(b) purple bars).
Discussion
In this study, we demonstrated that peripheral acute inflammation induced c-Fos expression in a subset (∼28% for GRP, ∼34% for GRPR) of itch-related neurons in the dorsal horn without affecting the expression level of GRP and GRPR themselves. In this model animal, about 40% and 20% of c-Fos-ir cells were labeled with GRP and GRPR, respectively. Based on the increased number of cells double-labeled for c-Fos-ir and GRP or GRPR, we considered that some parts of acute painful signals were transmitted to the itch processing circuits of the dorsal horn. Using the paw formalin injection model, we examined the effect of intrathecal administration of the GRPR antagonist and of GRP on spontaneous paw-flinching behavior. The inhibition of spinal GRPR partially enhanced the pain response, and the intrathecal infusion of GRP attenuated spontaneous paw-flinching. The inhibitory effect of exogenous GRP on pain behavior was associated with the activation of Pax2-positive inhibitory neurons in the dorsal horn. These data indicate that nociceptive inputs activate some parts of the GRP-GRPR system and that the pain circuit may utilize itch-related signals that are tuned to spinal inhibitory neurons.
The “labeled line theory” has been experimentally proven, suggesting a specific role of GRP and GRPR neurons in itch.3,9 However, studies showed the possible cross-talk between itch and pain information both at the primary afferent and dorsal horn levels. Indeed, nociceptors are known to be equipped with receptors for pruritic mediators. 22 Relevant receptors on sensory nerve fibers for pruritus have nociceptive potency, e.g. histamine receptors, proteinase activated receptor-2 and endothelin A-receptors.23–25 In addition, it has been reported that the dorsal root ganglion pruriceptors anatomically represent a subpopulation of nociceptors positive for transient receptor potential cation channel V1 (TRPV1), indicating their contribution to nociception. 4 Furthermore, topically applied capsaicin elicited itch, 26 but intradermal injections of capsaicin always evoked only pain in humans.27,28 Indeed, recent optogenetic and designer Receptor Exclusively Activated by Designer Drugs (DREADD) based study have revealed the multimodality of c-fiber through which pain and itch signals were transmitted to the dorsal horn. 29
In this study we observed the emergence of c-Fos-ir in the itch-related neurons (GRP or GRPR) in the dorsal horn of formalin injection model. According to the previous reports that characterized GRPR positive neurons, these neurons were essential to itch sensation and co-expressed the neurokinin-1 receptor (NK1R), a well known receptor for Substance P.9,30,31 NK1R positive neurons were often highlighted as a subset of the spinal projection neurons.32–34 It has been reported that the nociceptive and pruritic signals were transmitted to the GRP or GRPR positive neurons in mono-synaptic manner from primary afferent terminals.15,35 Therefore coding and processing of itch and pain information in the primary afferent and dorsal horn circuit are still controversial. However taken together of these lines of literatures and our results, we supposed that pruriceptors and their circuit could receive nociceptive stimuli, which was processed and transmitted to the brain.4,29
We found that intrathecal pre-injection of RC-3095, an antagonist for GRPR, partially enhanced pain and that intrathecal GRP infusion attenuated spontaneous pain behavior in the formalin injection model. In addition, spinal infusion of GRP decreased the total number of c-Fos-ir but increased the c-Fos-positive inhibitory neurons in the dorsal horn ipsilateral to formalin injection. These data suggest that painful stimuli reinforce the analgesic effect of GRP, such that GRPR positive neurons activate the analgesic signal in the dorsal horn. We believe that RC-3095 turned off the activity of inhibitory circuits that are presumably activated by GRPR-positive neurons, resulting in the enhancement of pain behavior after formalin injection. It has been reported that a subpopulation of neurons in the dorsal horn of the spinal cord receives itch signals as well as mechanical, thermal, or chemical noxious stimuli.36–40 Ultimately, the convergence of pain and itch in the spinal cord circuits suggest physiological roles of cross-sensory modulation, which is involved in the reciprocal enhancement or inhibition of these sensations. Modulation of spinal inhibitory neurons on sensory modalities is mainly mediated by receiving inputs from different types of afferents.41–43 Indeed C and A-delta fibers drive spinal inhibitory interneurons in a feed-forward manner. Therefore, inhibition of pain behavior and the induction of c-Fos in the subsets of inhibitory neurons by the administration of intrathecal GRP suggests the possible involvement of GRPR positive neurons in the feed-forward activation of inhibitory circuits.
Sun et al. proposed an interesting neuronal gate theory by showing that GRP positive neurons produced both pain and itch sensations in an intensity-dependent coding manner. They also found evidence that the GRP positive neurons transmit both itch and weak pain signals; however, for strong painful stimuli, the recruitment of endogenous opioids serves to reduce excessive pain. 15 We considered that Their findings were consistent with our data showing the modification of pain behavior by the intrathecal treatment with GRPR antagonist and of intrathecal GRP on the formalin-induced pain model.
In the formalin injection model, pre-treatment with GRP immediately reduced pain behavior, and the GRP-induced analgesia continued throughout the experimental period (∼60 min after formalin injection). In contrast to the continuous inhibitory effect of GRP on pain behavior, intrathecal pre-injection of RC-3095 partially enhanced pain behavior in the formalin injection model. This enhancement was observed only 25–35 min after formalin injection (Figure 2). This may be due to the saturation of the number of pain behavior at the peak timepoints (35–50 min after formalin injection) or to the limited duration of RC-3095 activity when administered into the subarachnoid space.
We observed the analgesic effect of GRP on CFA-induced thermal hyperalgesia not at the early stage (∼5 min) but at 1 h after GRP injection. It is well known that central sensitization, a hypersensitivity of the dorsal horn neurons, produces thermal hyperalgesia in the CFA treated model. For the formation and establishment of sensitization, several types of molecular modifications are required, such as changes of receptor channel properties, activation of intracellular signaling cascades, and enhancement of transcription. 44 Therefore, due to such enzymatic activities in the sensitized neurons, the analgesic time window of post-GRP infusion on the CFA model may be different from that of the acute inflammatory pain model.
In the present study we demonstrated that the neuronal circuit containing a GRP- and GRPR positive neuronal population possibly acts on inhibitory signals and ultimately reduces pain signals in the dorsal horn. For the understanding of modulatory mechanisms of itch signal on pain processing, further works are required to confirm the roles of GRP and GRPR in nociception as well as itch. This study may contribute to understanding the itch and pain modalities in the dorsal horn and provide novel insight to treat unpleasant sensations.
Footnotes
Acknowledgements
Author contributions
AS: animal surgery, behavioral studies, histological studies and pictures; HY: research concept, and interpretation, drafting of the manuscript; KK: data analysis, histological studies and pictures, drafting of the manuscript; MO: histological studies; KN: research concept, supervision of studies, critical revision of the manuscript. All authors read and approved the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Japan Society for the Promotion of Science Grants (JSPS KAKENHI) (Grant Number: 17H04113), “Hyogo Medical University Diversity Grant for Research Promotion” under MEXT Funds for the Development of Human Resources in Science and Technology, “Initiative for Realizing Diversity in the Research Environment (Characteristic-Compatible Type)”, and a Grant-in-Aid for Researchers, Hyogo Medical University.
