Abstract
Low threshold mechanoreceptors (LTMRs) are important for environmental exploration, social interaction, and tactile discrimination. Whisker hair follicles are mechanical sensory organs in non-primate mammals that are functionally equivalent to human fingertips. Several functional types of LTMRs have been identified in rodent whisker hair follicles, including rapidly adapting (RA), slow adapting type 1 (SA1), and slowly adapting type 2 (SA2) LTMRs. Properties of these LTMRs have not been fully characterized. In the present study, we have used pressure-clamped single-fiber recording technique to record impulses of RA, SA1, and SA2 LTMRs in mouse whisker hair follicles, and tested effects of 5-HT, Cd2+, tetraethylammonium (TEA), 4-aminopyridine (4-AP), and Ba2+ on the LTMR impulses. We show that 5-HT at 2 mM suppresses SA1 impulses but has no effects on RA and SA2 impulses. Cd2+ at 100 μM suppresses both SA1 and SA2 impulses but has no effects on RA impulses. TEA at 10 mM has no effects on RA and SA1 impulses but increased SA2 impulses. However, TEA at 1 mM and 200 μM decreases SA2 impulses. 4-AP at 1 mM suppresses both SA1 and SA2 impulses but has no effects on RA impulses. Ba2+ at 5 mM increases both RA and SA1 impulses but suppresses SA2 impulses. Collectively, RA, SA1, and SA2 LTMRs show distinct pharmacological properties, suggesting that these LTMRs may use different mechanisms to tune their mechanical signaling.
Introduction
Low threshold mechanoreceptors (LTMRs) transduce tactile stimuli into afferent impulses, which is essential for tactile tasks such as environmental exploration, social interaction, and tactile discrimination.1,2 LTMRs are highly enriched in fingertips of humans as well as in whisker hair follicles of non-primate mammals.1,3,4 Whisker hair follicles in many non-primate animals are functionally equivalent to human fingertips, and are key sensory organs for animals to use their whisker hairs to perform tactile tasks. Therefore, rodent whisker follicles often serve as a model tactile organ to investigate cellular and molecular mechanisms underlying mechanical transduction by LTMRs. 4 Different types of LTMRs have been identified in tactile sensitive spots in the body, and based on their anatomic features, they are classified as Merkel discs, Meissner’s corpuscles, Ruffini endings, Pacinian corpuscles, lanceolate endings, and free nerve endings.3,5 Functionally, at least three general types of LTMRs have been defined in whisker hair follicles based on patterns of afferent impulses elicited following mechanical stimulation. 6 These three functional subtypes of LTMRs are rapidly adapting (RA) LTMRs, slowly adapting type 1 (SA1) LTMRs, and slowly adapting type 2 (SA2) LTMRs. 6 RA, SA1, and SA2 LTMRs in whisker hair follicles are shown to be largely due to the activation of lanceolate endings, Merkel discs, and Ruffini-like endings, respectively. 2
Previously, we have shown RA LTMRs in whisker hair follicles are activated by rapid mechanical indentation. 6 RA impulses are elicited during dynamic phase but not during static phase of mechanical indentation. 6 In contrast to RA, SA1 LTMRs and SA2 LTMRs in whisker hair follicles respond to mechanical indentation by firing impulses during both dynamic phase and static phase of the mechanical stimulation, and total impulse numbers increase with increased indentation distance. 6 Previous studies have shown that impulses firing of LTMRs can be affected by compounds that affected different ligand-gated receptors. For example, ATP suppressed activity in slowly adapting but not rapidly adapting LTMRs in toad skin, 7 metabotropic glutamate receptor antagonists selectively enhanced responses of SA1 LTMRs, 8 NMDA receptor blockers ifenprodil and MK801 inhibited SA1 LTMR responses in rat whisker hair follicles, 9 a broad spectrum ionotropic glutamate receptor antagonist kynurenate caused dose-dependent reductions in SA1 LTMR responses. 10 5-HT2 and 3 receptor antagonists suppressed the response of rat SA1 LTMRs. 11 Previously, we have shown that 5-HT at low concentrations potentiated but at high concentrations suppressed mechanically evoked impulses that were recorded from whole whisker afferent nerve bundles, 12 but it was not determined in the study which type(s) of LTMRs were sensitive to 5-HT. In addition to compounds for ligand-gated receptors, previous studies also have shown that a number of other compounds including metal ions and inhibitors of voltage-gated ion channels modulated LTMR activities. For example, manganese and magnesium showed differential effects on two types of slowly adapting LTMRs in the skin of frogs, 13 Gd3+ and Cd2+ suppressed the responses of SA1 LTMRs in frog dorsal skin, 14 KCNQ inhibitor linopirdine potentiated activity of RA1 and Skin Down-hair (D-hair) LTMRs. 15
The previous pharmacological studies have suggested that LTMRs are tuned by different receptors and ion channels which may be expressed at the LTMRs. To help further understand the tuning of LTMRs, in the present study, we have used pressure-clamped single-fiber recording technique to record impulses of RA, SA1, and SA2 LTMRs in mouse whisker hair follicles, and examined effects on the activities of these LTMRs by 5-HT, Cd2+, tetraethylammonium (TEA), 4-Aminopyridine (4-AP), and Ba2+.
A reason for investigating effects of Cd2+ in the present study was to confirm our previous finding about the key role of voltage-gated Ca2+ channels in SA1 responses. 4 For testing effects of TEA, 4-AP and Ba2+ in the present study, we intend to investigate whether tactile responses in different types of LTMRs may be differentially tuned by K+ channels.
Materials and methods
Whisker hair follicle preparations
C57BL/6 male mice aged 8–10 weeks were used for making whisker hair follicle preparations and whisker afferent single-fiber recordings. Animal care and use conformed to NIH guidelines for care and use of experimental animals. Experimental protocols were approved by the Institutional Animal Care and Use Committee (IACUC) at the University of Alabama at Birmingham. In brief, animals were anesthetized with 5% isoflurane and then sacrificed by decapitation. Whisker pads were dissected out from orofacial areas of mice by using a pair of dissecting scissors and placed in a 35-mm petri dish that contained 5 mL ice cold L-15 medium. Under a dissection microscope, fat tissues that covered whisker hair follicles were removed by using a pair of fine tweezers to expose individual whisker hair follicles. Each whisker hair follicles together with its whisker afferent bundle and hair shaft was then gently pulled out. The capsule of each whisker hair follicle was cut open to two small holes at the enlargement and end parts of the capsule to facilitate solution exchange and drug diffusion into each whisker hair follicle. The whisker hair follicle preparations with attached whisker afferent nerve bundles were affixed into a recording chamber by a tissue anchor and submerged in a Krebs bath solution. The Krebs solution contained (in mM): 117 NaCl, 3.5 KCl, 2.5 CaCl2, 1.2 MgCl2, 1.2 NaH2PO4, 25 NaHCO3, and 11 glucose. The pH of the Krebs solution was adjusted to 7.3 with HCl or NaOH and osmolarity adjusted to 325 mOsm with sucrose. The Krebs solution was saturated with 95% O2 and 5% CO2 during experiments.
Pressure-clamped single-fiber recordings from whisker afferent fibers
The pressure-clamped single-fiber recording was performed in the same manner described in our previous studies.6,16 In brief, the recording chamber was mounted on the stage of an Olympus BX50WI microscope. The whisker hair follicle preparations were then briefly exposed for 3–5 min to a mixture of 0.05% disipase II plus 0.05% collagenase in the Krebs solution, and the enzymes were quickly washed off with the normal Krebs bath solution. This very gentle enzyme treatment was to help separating individual afferent fibers so that a single fiber could be aspirated into a recording electrode and pressure-clamped for single fiber recordings. Recording electrodes for pressure-clamped single-fiber recordings were made by thin walled borosilicate glass tubing without filament (inner diameter 1.12 mm, outer diameter 1.5 mm, World Precision Instruments, Sarasota, FL, USA). They were fabricated by using P-97 Flaming/Brown Micropipette Puller (Sutter Instrument Co., Novato, CA, USA) and fire polished using a microforge (World Precision Instruments, Sarasota, FL, USA) to have a final tip diameter of 5–10 μm. The recording electrode was filled with Krebs bath solution, mounted onto an electrode holder which was connected to a high-speed pressure-clamp (HSPC) device (ALA Scientific Instruments, Farmingdale, NY, USA) for fine controls of intra-electrode pressures. Under a 40x objective, the ending of a single whisker afferent nerve was first separated from whisker afferent nerve bundle by applying a low positive pressure (∼10 mmHg) from the recording electrode. The nerve end was then aspirated into the recording electrode by a negative pressure at approximately −10 mmHg. Once the nerve end within the electrode reached ∼10 μm, the electrode pressure was readjusted to −5 to −1 mmHg and maintained at the same pressure throughout the experiment. Nerve impulses conducted along a single whisker afferent fiber were recorded using a Multiclamp 700A amplifier (Molecular Devices, Sunnyvale, CA, USA) and signals were sampled at 20 KHz with low pass filter set at 1 KHz. Unless otherwise indicated, all recording experiments were performed at the room temperature of 23°C–24°C.
Mechanical stimulation
Mechanical stimulation was applied to the body of a whisker hair follicle using a 20 gauge needle as a mechanical stimulation probe. The needle was mounted on a pipette holder and attached to a piezo device (E-625 PZT, Physik Instrumente, Auburn, MA, USA). The tip of the probe was positioned at an angle of 45° to the surface of the hair follicle. The piezo device with the mechanical probe was mounted on a micromanipulator (Sutter Instrument Co., Novato, CA, USA) to allow to manually search receptive fields. The piezo device was also computer-programmable with the pCLAMP10 software (Molecular Devices, Sunnyvale, CA, USA) to generate forward movement for the mechanical probe to deliver stepwise mechanical stimulation. In each experiment, a receptive field was first probed by multiple forward movements of the mechanical probe using the micromanipulator. Once a receptive field was identified, the vertical position of the probe tip was adjusted such that no nerve impulses were evoked at this position but a 1-μm forward movement of the probe would evoke nerve impulses. To quantitatively applying mechanical stimuli, stepwise forward movements were applied via the piezo device. Unless otherwise indicated, the stepwise forward movement of the probe consisted of a 100-ms ramp to 38-μm step (dynamic phase) followed by a 2500-ms holding position at the 38-μm step (static phase) and then a 100-ms ramp back to baseline. The probe movement and nerve impulses were simultaneously recorded using the pCLAMP 10 software.
Pharmacology
Mechanical responses were elicited by the 38-μm displacement before (control) and following the bath application of testing compounds. Testing compounds included 5-HT (2 mM), Cd2+ (100 μM), TEA (0.2, 1, 5, and 10 mM), 4-AP (0.02, 0.1, and 1 mM), and Ba2+ (5 mM). 5-HT was bath applied for 10 min, Cd2+ bath applied for 20 min, TEA, 4-AP and Ba2+ were bath-applied for 5 min.
Statistical analysis
Impulses recorded from single whisker afferent fibers were analyzed using the CLAMPFIT 10 software. In most cases for the mechanically evoked responses, impulses during the dynamic phase of 100 ms ramp and impulses during the static phase of 2500-ms holding position were counted separately. Unless otherwise specified, impulses refer to the total impulse numbers during both dynamic and static phases. Data of the mechanically evoked impulses were presented as impulse numbers.
Spontaneous impulses were counted for 1 min during the single-fiber recordings without mechanical stimulation. Data of the spontaneous impulses were reported as impulse frequencies. Data are presented as mean ± SEM. Statistical significance was evaluated using paired Student’s t test, ns; no significant difference, *p < 0.05, **p < 0.01, and ***p < 0.001.
Results
We used the pressure-clamped single-fiber recording technique recently developed by us to record whisker afferent impulses evoked by mechanical probing of whisker hair follicles (Figure 1(a)). Using this technique, we have previously identified in mouse whisker hair follicles three functional types of mechanoreceptors, RA, SA1, and SA2, following mechanical probing whisker hair follicles.
6
In the present study, mechanical impulses of all three types of mechanoreceptors were evoked by a 38-μm displacement with a mechanical probe. In addition to the mechanically evoked responses, we also recorded spontaneous impulses which are defined in the present study as impulses not due to mechanical stimulation, and the impulses induced by testing drugs were considered to be spontaneous impulses in the present study. Effects of a high concentration of 5-HT on mechanical responses of RA, SA1, and SA2 mechanoreceptors. (a) Graph illustrates the pressure-clamped single-fiber recordings from the whisker afferent nerve of a mouse whisker hair follicle. To elicit mechanical responses, a mechanical probe controlled by a piezo device is used to deliver displacement stimulations to whicker hair follicles. (b) Left panel, sample traces of RA responses before (control, top) and following the application of 2 mM 5-HT for 10 min (bottom). Insets, impulses at an expanded time scale. Right panel, summary data of RA impulse numbers in control (n = 5) and following the application of 2 mM 5-HT (n = 5). (c) Left panel, sample traces of SA1 responses before (control, top) and following the application of 2 mM 5-HT for 10 min (Bottom). Right panel, two sets of bar graphs show summary data of SA1 impulse numbers in dynamic phase (Left) and static phase (Right) before (control, n = 6) and following the application of 2 mM for 10 min 5-HT (n = 6). (d) Left panel, sample traces of SA2 responses before (control, top) and following the application of 2 mM 5-HT for 10 min (bottom). Right panel, two sets of bar graphs show summary data of SA2 impulse numbers in dynamic phase (Left) and static phase (Right) before control (n = 7) and following the application of 2 mM for 10 min 5-HT (n = 7). Data represent Mean ± SEM, ns; no significant difference, **p < 0.01, ***p < 0.001, paired t-test.
We tested effects of 5-HT on mechanical responses of RA, SA1, and SA2 mechanoreceptors of whisker hair follicles. In a previous study, we have shown that 5-HT at 2 mM significantly reduced mechanically evoked impulses on whole-bundle whisker nerves using a suction recording electrode. 12 In the present study, we determined which types of mechanoreceptors in whisker hair follicles were affected by the high concentration of 5-HT. For RA responses, impulse numbers were not significantly different before (control, n = 5) and following the application of 2 mM 5-HT (n = 5, Figure 1(b)). For SA1 responses, impulses in both dynamic phase and static phase were suppressed following the bath application of 2 mM 5-HT (Figure 1(c)). The impulse numbers in the dynamic phase were 14.17 ± 2.55 before (control, n = 6) and reduced to 8.0 ± 2.54 (n = 6, p < 0.001) following the bath application of 2 mM 5-HT (Figure 1(c)). The impulse numbers in the static phase were 88.5 ± 14.18 before (control, n = 6) and reduced to 7.0 ± 5.05 (n = 6, p < 0.01) following the bath application of 2 mM 5-HT (Figure 1(c)). Thus, 5-HT at the high concentration of 2 mM moderately suppressed SA1 impulses in the dynamic phase and almost completely abolished the SA1 impulses in the static phase. For SA2 responses, impulse numbers in both static phase and dynamic phase were not significantly different before (control, n = 7) and following the bath application of 2 mM 5-HT (Figure 1d). For all the three types of mechanoreceptors, bath application of 2 mM 5-HT did not induce spontaneous impulses (not illustrated).
Effects of Cd2+, a voltage-gated Ca2+ channel blocker, on RA, SA1, and SA2 mechanoreceptors in whisker hair follicles were tested. RA impulses were not significantly affected by 100 μM Cd2+ (Figure 2(a), n = 7). In contrast to RA impulses, SA1 impulse numbers in both dynamic phase and static phase were significantly reduced following the bath application of 100 μM Cd2+ (Figure 2b). The SA1 impulse numbers in the dynamic phase were 11.2 ± 1.9 before (control, n = 6) and reduced to 7.5 ± 1.9 (n = 6, p < 0.05, Figure 2(b)) following the bath application of 100 μM Cd2+. The SA1 impulse numbers in the static phase were 82.8 ± 24.2 before (control, n = 6) and reduced to 17.5 ± 6.6 (n = 6, p < 0.05, Figure 2(b)) following the bath application of 100 μM Cd2+. Thus, the effects of Cd2+ were more prominent on the static phase than on the dynamic phase of SA1 impulses (Figure 2(b)). Similar to SA1 responses, the SA2 impulse numbers in both dynamic phase and static phase were significantly reduced following the bath applications of 100 μM Cd2+ (Figure 2(c)). The SA2 impulse numbers in the dynamic phase were 15.1 ± 2.0 before (control, n = 8) and reduced to 11.4 ± 2.0 (n = 8, p < 0.01, Figure 2(c)) following the bath application of 100 μM Cd2+. The SA2 impulse numbers in the static phase were 276.5 ± 44.6 before (control, n = 8) and reduced to 166.4 ± 53.7 (n = 8, p < 0.01, Figure 2(c)) following the bath application of 100 μM Cd2+. For all the three types of mechanoreceptors, bath application of 100 μM Cd2+ did not induce spontaneous impulses (not illustrated). Effects of Cd2+ on mechanical responses of RA, SA1, and SA2 mechanoreceptors. (a) Left panel, sample traces of RA responses before (control, top) and following the application of 100 μM Cd2+ (Bottom). Insets, impulses at an expanded time scale. Right panel, summary data (n = 7) of RA impulse numbers in control (n = 7) and following the application of 100 μM Cd2+ (n = 7). (b) Left panel, sample traces of SA1 responses before (control, top) and following the application of 100 μM Cd2+ (Bottom). Right panel, two sets of bar graphs show summary data (n = 6) of SA1 impulse numbers in dynamic (Left) and static phases (Right) in control and following the application of 100 μM Cd2+. (c) Left panel, sample traces of SA2 responses before (control, top) and following the application of 100 μM Cd2+ (bottom). Right panel, two sets of bar graphs show summary data (n = 8) of SA2 impulse numbers in the control and following the application of 100 μM Cd2+ (n = 8). In each set of experiments 100 μM Cd2+ was applied for 20 min. Data represent Mean ± SEM, ns; no significant difference, *p < 0.05, **p < 0.01, paired t-test.
We examined effects of TEA (tetraethylammonium), a commonly used voltage-gated K+ channel blocker,
17
on mechanically evoked impulse firing of the three types of mechanoreceptors in whisker follicles (Figures 3(a)-(f)). Neither RA (n = 6) nor SA1 (n = 5) impulses were significantly affected following the bath application of 10 mM TEA (Figure 1a, b). No spontaneous impulses were detected before (control) and following the bath application of 10 mM TEA in RA mechanoreceptors (n = 6, Figure 3(a)). In SA1 mechanoreceptors, very low frequency spontaneous impulses were observed before (control) bath application of TEA, and no significant changes in spontaneous impulses were observed following the bath application of 10 mM TEA (n = 5, Figure 3(b)). In contrast, 10 mM TEA significantly increased impulse numbers in both dynamic and static phases of SA2 responses (Figure 3c). For the dynamic phase, SA2 impulse numbers were increased from 12.2 ± 1.8 in control before TEA application to 15.0 ± 2.0 following the bath application of 10 mM TEA (n = 5, p < 0.01, Figure 3(c)). For the static phase, SA2 impulse numbers were increased from 253.2 ± 37.3 in control before TEA application to 472.6 ± 52.8 following the bath application of 10 mM TEA (n = 5, p < 0.01, Figure 3(c)). In addition to enhancing mechanically evoked SA2 responses, TEA induced spontaneous impulses (Figure 3(c)). As shown in Figure 3(c), there was no any spontaneous impulse in SA2 mechanoreceptors in the absence of TEA (control) but spontaneous impulses occurred at the frequency of 18.4 ± 4.8 Hz (n = 5, p < 0.05) following the bath application of 10 mM TEA. Similar to evoked SA2 impulses, spontaneous impulses also had regular firing pattern (Figure 3(c)). Interestingly, spontaneous impulses usually were briefly stopped immediately after the evoked SA2 impulses and resumed shortly (Figure 3(c)). Similarly, TEA at 5 mM increased SA2 impulses and induced spontaneous impulses (Figure 3(d)). In contrast, SA2 impulses were significantly reduced rather than increased by TEA at lower concentrations of 1 mM (Figure 3(e), n = 6, p < 0.01) and 200 μM (Figure 3(f), n = 6, p < 0.001), but spontaneous impulses were not induced at these lower concentrations of TEA (Figure 3e, f). Effects of tetraethylammonium on mechanical responses of RA, SA1, and SA2 mechanoreceptors. (a) Left panel, two sets of sample traces show RA responses before (control, top) and following the bath application of 10 mM tetraethylammonium (TEA, bottom). Insets, impulses at an expanded time scale. Right panel, two sets of bar graphs show summary data (n = 6) of RA impulses (left) and spontaneous impulses (right) before (control) and following the application of 10 mM TEA. (b) Left panel, sample traces of SA1 responses before (control, top) and following the bath application of 10 mM TEA. Right panel, three sets of bar graphs are summary data (n = 5) of SA1 impulse numbers in the dynamic phase (Left), in the static phase (Middle), and spontaneous impulses (Right). (c) Left, sample traces of SA2 responses before (control, top) and following the application of 10 mM TEA (bottom). Right panel, three bar graphs are summary data of SA2 impulse numbers in the dynamic phase (Left), in the static phase (Middle), spontaneous impulses (Right). (d–f) Similar to C except TEA was applied at the concentrations of 5 mM (D, n = 6), 1 mM (E, n = 6), and 200 μM (F, n = 6). Impulse numbers are impulses in both dynamic and static phases. Data represent Mean ± SEM, ns; no significant difference, *p < 0.05, **p < 0.01, ***p < 0.001, paired t-test.
We determined 4-AP (4-aminopyridine), another commonly used voltage-gated K+ channel blocker,
17
on the three types of mechanoreceptors. Mechanically evoked RA impulses were not significantly affected following the bath application of 1 mM 4-AP (n = 6), and there was no spontaneous impulses before and following the application of 1 mM 4-AP. (Figure 4(a), n = 6). However, mechanically evoked SA1 impulses in both dynamic and static phases were significantly suppressed by 1 mM 4-AP (Figure 4(b)). SA1 impulse numbers in the dynamic phase were 14.2 ± 1.6 (n = 6) in the control before the 4-AP application and reduced to 7.3 ± 1.3 (n = 6, p < 0.001, Figure 4(b)) following the bath application of 1 mM 4-AP. SA1 impulse numbers in the static phase were 75.8 ± 8.4 (n = 6) in the control before the 4-AP application and reduced to 42.2 ± 11.2 (n =6, p < 0.01, Figure 4(b)) following the bath application of 1 mM 4-AP. Very low frequency spontaneous impulses of SA1 mechanoreceptors were observed before the 4-AP application and were not significantly affected following the bath application of 1 mM 4-AP (Figure 4(b)). At a lower concentration of 100 μM 4-AP, neither mechanically evoked SA1 impulses nor spontaneous impulses of SA1 mechanoreceptors were significantly affected (n = 6, Figure 4(c)). For SA2 mechanoreceptors, impulse numbers in the dynamic phase were 11.6 ±1.5 (n = 6) in the control before the 4-AP application and significantly reduced to 5.4 ± 1.2 (n = 6, p < 0.001, Figure 4(d)) following the bath application of 1 mM 4-AP. Impulse numbers in the static phase were 237.0 ± 29.8 (n = 6) before the 4-AP application (control) and significantly reduced to 86.8 ± 18.8 (n = 6, p < 0.001, Figure 4(d)) following the bath application of 1 mM 4-AP. Spontaneous impulses in SA2 mechanoreceptors were absent before the application of 1 mM 4-AP (control) and became 12.0 ± 1.0 Hz (n = 5, p < 0.001, Figure 4(d)) following the bath application of 1 mM 4-AP. Similarly, 4-AP at a lower concentration of 100 μM significantly decreased SA2 impulses and increased spontaneous impulses (Figure 4(e), n = 5). At 20 μM, 4-AP still significantly decreased SA2 impulses (n = 4, p < 0.01) but did not induce any spontaneous impulses (Figure 4(f), n = 4). Effects of 4-aminopyridine on mechanical responses of RA, SA1, and SA2 mechanoreceptors. (a) Left panel, sample traces of RA responses before (control, top) and following the application of 1 mM 4-aminopyridine (4-AP, bottom). Insets, impulses at an expanded time scale. Right panel, summary data (n = 6) show RA impulse numbers (right) and spontaneous impulse numbers (left) in control and following the application of 1 mM 4-AP. (b) Left panel, sample traces of SA1 responses before (control, top) and following the application of 1 mM 4-AP. Right panel, three sets of bar graphs are summary data of SA1 impulse numbers in dynamic phase (Left, n = 7), in static phase (Middle, n = 7), and spontaneous impulses (Right, n = 6) before (control) and following the application of 1 mM 4-AP. (c) Similar to B except 4-AP effects on SA1 responses were tested with a lower concentration at 100 μM (n = 6). (d) Left panel, sample traces of SA2 responses before (control, top) and following the application 1 mM 4-AP (bottom). Right panel, three sets of bar graphs are summary data (n = 6) of SA2 impulse numbers in dynamic phase (Left), in static phase (Middle), and spontaneous impulses (Right) before (control) and following the application of 1 mM 4-AP. (e) Summary data (n = 5) of total SA2 impulse numbers (Left) and spontaneous impulses (Right) before (control) and following the application of 100 μM 4-AP. (f) Similar to E except 20 μM 4-AP was tested (n = 4). Data represent Mean ± SEM, ns; no significant difference, *p < 0.05, **p < 0.01, ***p < 0.001, paired t-test.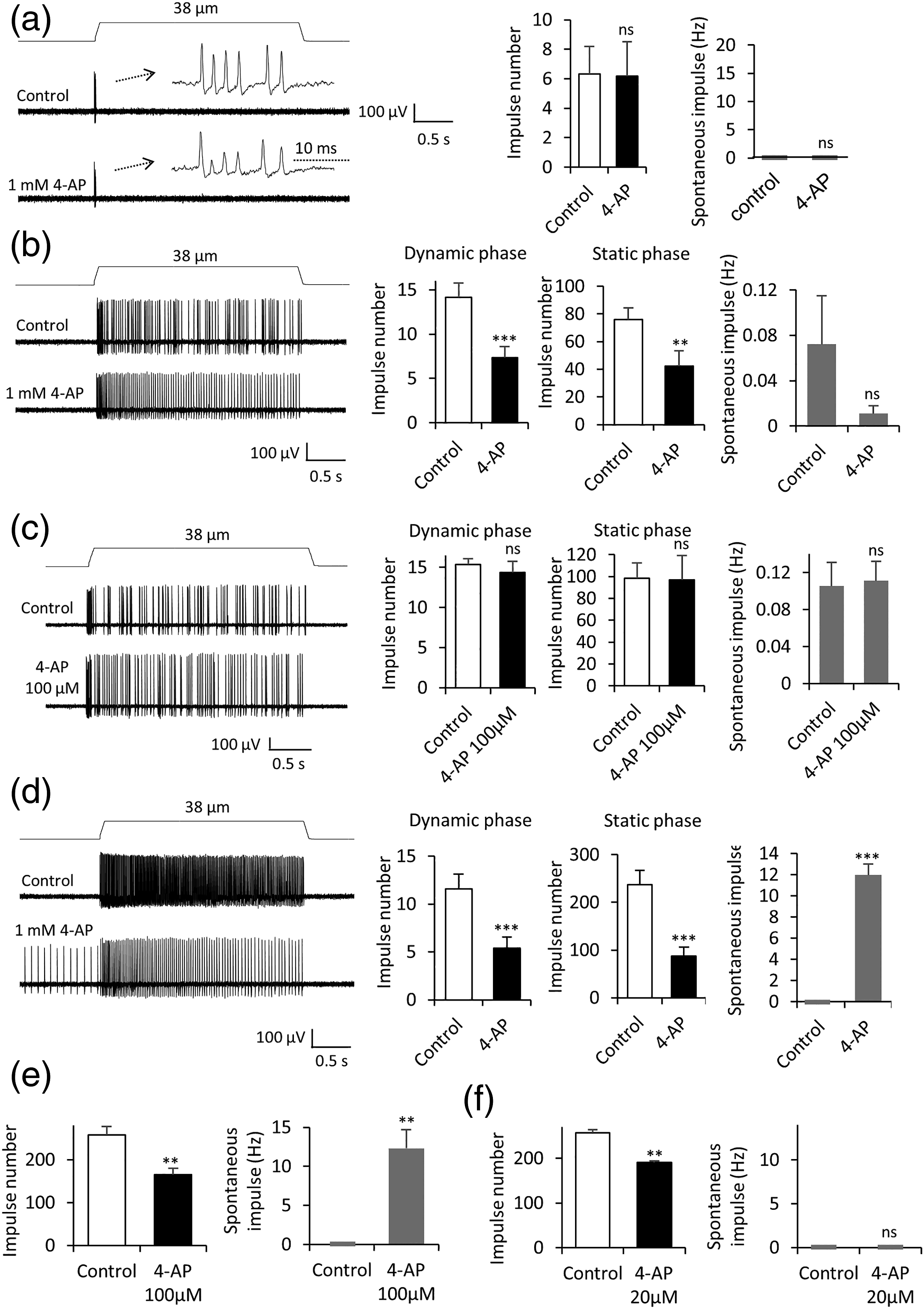
Effects of Ba2+ on mechanoreceptors in whisker hair follicles were examined. RA impulse numbers were significantly increased from 5.3 ± 0.5 before the Ba2+ application (n = 10, control) to 10.7 ± 2.4 (n = 10, p < 0.05, Figure 5(a)) following the bath application of 5 mM Ba2+. RA impulses occurred only in the dynamic phase in all 10 RA mechanoreceptors recorded before Ba2+ application, but 3 of them had impulses extended into the static phase following bath application of Ba2+. For SA1 mechanoreceptors, Ba2+ at 5 mM significantly increased mechanically evoked SA1 impulses in the static phase but not in the dynamic phase (Figure 5(b)). The impulse numbers in the static phase were 60.2 ± 11.4 before the Ba2+ application (control, n = 5) and increased to 141.2 ± 17.2 (n = 5, p < 0.01, Figure 5(b)) following the bath application of 5 mM Ba2+. Ba2+ also increased numbers of spontaneous impulses of SA1 mechanoreceptors from 0.03 ± 0.02 Hz (n = 5) in control before the Ba2+ application to 2.6 ± 0.6 Hz (n = 5, p < 0.01, Figure 5(b)) following the bath application of 5 mM Ba2+. For SA2 mechanoreceptors, Ba2+ at 5 mM reduced mechanically evoked SA2 impulses in both dynamic and static phases (Figure 5(c)). The impulse numbers in the dynamic phase were 12.0 ± 2.0 (n = 6) in the control and decreased to 7.6 ± 1.21 (n = 6, p < 0.05) following the bath application of 5 mM Ba2+. The impulse numbers in the static phase were 222.4 ± 23.2 (n = 6) in the control and decreased to 163.8 ± 27.1 (n = 6, p < 0.05) following the bath application of 5 mM Ba2+ (Figure 5(c)). The numbers of spontaneous impulses of SA2 mechanoreceptors were absent in the control and increased to 20.8 ± 4.6 Hz (Figure 5(c), n = 6, p < 0.001) following the bath application of 5 mM Ba2+. Effects of Ba2+ on mechanical responses of RA, SA1, and SA2 mechanoreceptors. (a) Left panel, sample traces of RA responses before (control, top) and following the bath application of 5 mM Ba2+ (bottom). Insets, impulses at an expanded time scale. Right panel, two sets of bar graphs are summary data (n = 10) of RA impulse numbers and spontaneous impulses in control and following the bath application of 5 mM Ba2+(closed bars). (b) Left panel, sample traces of SA1 responses before (control, top) and following the application of 5 mM Ba2+. Right panel, three sets of bar graphs are summary data (n = 5) of SA1 impulse numbers in dynamic phase (Left), in static phase (Middle), and spontaneous impulses (Right). (c) Left panel, sample traces of SA2 responses before (control, top) and following the application 5 mM Ba2+ (bottom). Right panel, three sets of bar graphs are summary data of SA2 impulse numbers in dynamic phase (Left, n = 6), in static phase (Middle, n = 6), and spontaneous impulses (Right, n = 6). Data represent Mean ± SEM, ns; no significant difference, *p < 0.05, **p < 0.01, ***p < 0.001, paired t-test.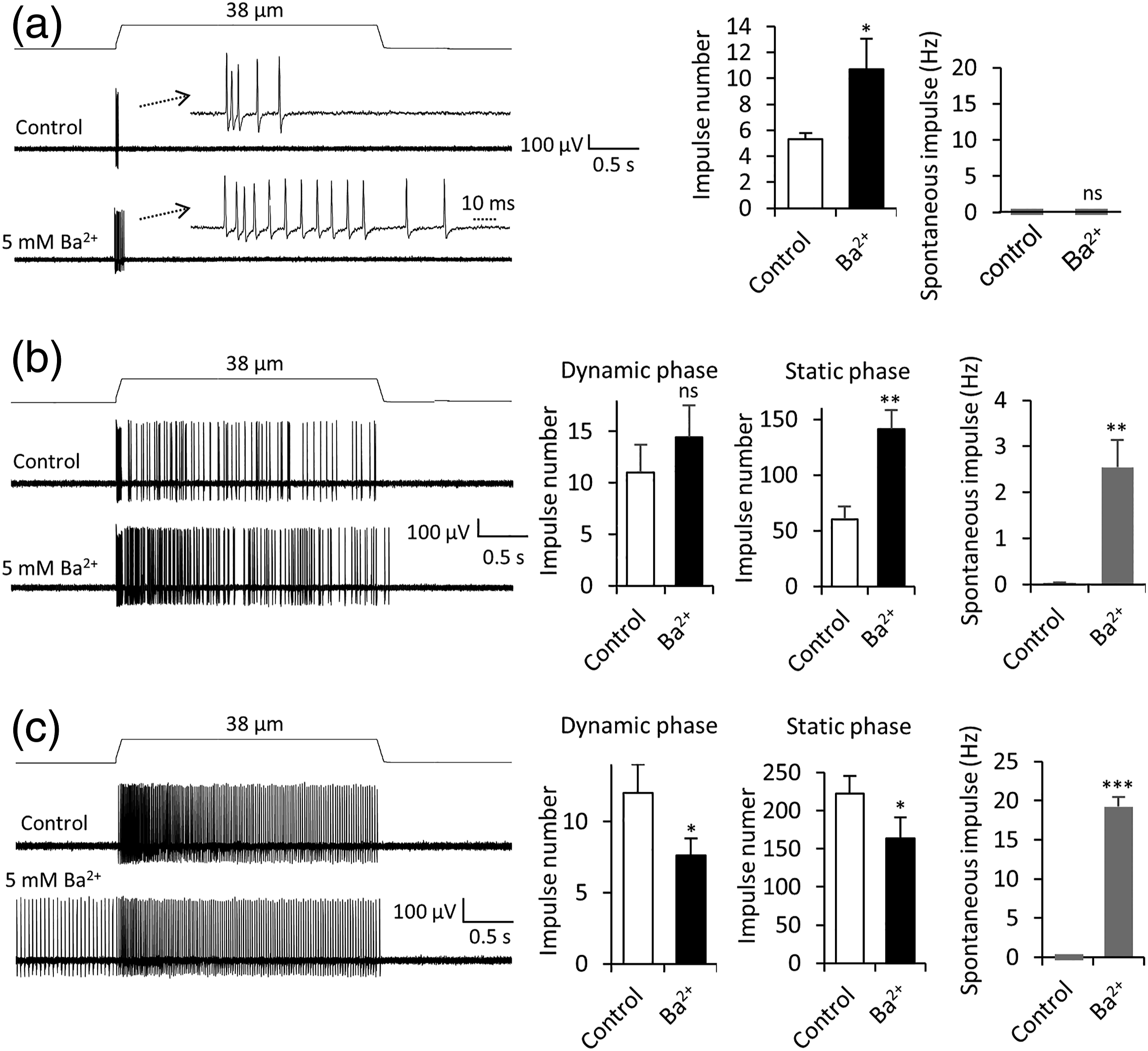
Discussion
Applying our newly developed pressure-clamped single-fiber recording technique to mouse whisker hair follicles, the present study has characterized effects on RA, SA1, and SA2 LTMRs by several compounds including 5-HT, Cd2+, TEA, 4-AP, and Ba2+. We show these compounds have differential effects on the three functional types of LTMRs, suggesting these three types of LTMRs may use different molecules at their terminals to tune tactile signaling.
We show 5-HT at 2 mM largely suppresses SA1 impulses but has no significant effect on RA and SA2 impulses. Although SA1 impulses in both dynamic and static phases are suppressed by 2 mM 5-HT, impulses in the static phase are more severely suppressed, and almost completely abolished by 2 mM 5-HT. The present result with our single fiber recordings is consist with our previous studies performed using suction electrode recordings form whole nerve bundle of whisker hair follicles. 12 We show 2 mM 5-HT specifically suppress SA1 impulses, and has no effect on RA and SA2 impulses. This result may suggest a specific role of 5-HT in tactile signaling at Merkel discs. Interestingly, a recent study has suggested that norepinephrine may be a transmitter at Merkel discs in the skin touch domes, 18 but this idea has been challenged in a more recent study. 19 Further studies are needed to elucidate the nature of the transmitter(s) at Merkel dicks.
We show that Cd2+, a voltage-gated Ca2+ channel blocker, has no effect on RA impulses but significantly suppress both SA1 and SA2 impulse. These results indicate that Cd2+-sensitive voltage-gated Ca2+ channels do not play a significant role in RA impulse generation but are important in the generation of SA1 and SA2 impulses. Voltage-gated Ca2+ channels on Merkel cells 20 would be inhibited by Cd2+ to abolish Merkel cell Ca2+ action potentials, 4 which would subsequently block tactile transmission from Merkel cells to the afferent terminals of SA1 LTMRs. We show that SA2 impulses are suppressed by Cd2+, which may suggest voltage-gated Ca2+ channels are present at afferent terminals of SA2 LTMRs and play an essential role in SA2 impulse generation.
We show that TEA, a widely used voltage-gated K+ channel blocker, has differential effects on the three types of LTMRs in whisker hair follicles. SA1 impulses are not significantly affected by TEA. RA impulses are shown to be slightly increased in the present of TEA although the change is not statistically significant. On the other hand, SA2 impulses are significantly increased by high concentrations of TEA but reduced by lower concentrations of TEA. TEA at high concentrations also induced spontaneous impulses in SA2 LTMRs. These results may suggested that TEA-sensitive voltage-gated K+ channels are not expressed or expressed at low levels in SA1 ad RA LTMRs. On the other hand, at least two types of TEA-sensitive voltage-gated K+ channels may be present in SA2 LTMRs involving setting their SA2 impulse firing. TEA-sensitive voltage-gated K+ channels include Kv1.1 and all Kv3 channels. 17 On the other hand, TEA cannot effectively block other Kv1 channels and Kv4 channels. 17
We show that 4-AP at high concentrations had no effect on RA mechanoreceptors but significantly inhibited both SA1 and SA2 impulses. 4-AP preferentially blocks A-type voltage-gated K+ channels. 17 Therefore, the lack of effects by 4-AP on RA mechanoreceptors suggest that A-type voltage-gated K+ channels do not play a significant role in controlling impulse generation in RA mechanoreceptors. On the other hand, A-type voltage-gated K+ channels may be essential for impulse generation in afferents of both SA1 and SA2 mechanoreceptors. For SA1 mechanoreceptors, Merkel cell action potentials may also require IA currents 20 and block of A-type voltage-gated K+ channels may impair action potential firing at Merkel cells and thereby suppress SA1 impulses. Interestingly, while 4-AP suppressed both SA1 and SA2 impulses, it induced spontaneous impulses in SA2 but not SA1 mechanoreceptors. This differential effects of 4-AP suggest that afferents of these two types mechanoreceptors have different electrophysiological properties. Although both TEA and 4-AP had no effect on RA, we show that Ba2+ significantly increased RA impulses, and in some cases the impulses fired in static phase in the presence of Ba2+. This result may suggest the presence of Ba2+-sensitive K+ channels which are not TEA- and 4-AP-senstive on the afferents of RA mechanoreceptors. One such type of K+ channels may be mechanosensitive K2P channels. 21 Activation of these channels by mechanical stimulation may limit impulses of RA mechanoreceptors, and therefore the inhibition of these channels by Ba2+ may produce disinhibition to increase RA impulses. While TEA had no effect and 4-AP inhibit SA1 impulses, Ba2+ increased SA1 impulses and spontaneous impulse of SA1 mechanoreceptors. These results may also suggest the involvement of mechanosensitive K2P channels in the afferents of SA1 mechanoreceptors. In contrast to RA and SA1, Ba2+ inhibited SA2 impulses but induced spontaneous impulses. This may suggest the presence of a different type of Ba2+-sensitive K+ channels in the afferents to SA2 mechanoreceptors.
The present study explored differences in tactile signaling among RA, SA1, and SA2 LTMRs with respect to the effects of 5-HT, Cd2+, TEA, 4-AP, and Ba2+ on the impulses of these LTMRs. Future studies are needed to use subtype selective blockers to pinpoint specific subtypes of 5-HT receptors, voltage-gated Ca2+ channels and K+ channels that are involved in regulating tactile signaling in these different types of LTMRs. Other approaches including immunohistochemical and genetic methods in combination with electrophysiological recordings will be more helpful in elucidating molecular mechanisms underlying tactile signaling in different types of LTMRs.
Footnotes
Authors’ Contributions
MS performed experiments
MS, HY and NN analyzed data
JGG wrote the manuscript
All authors read and approved the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by NIH grants DE018661, DE023090, and NS109059 to JGG.
