Abstract
Background
Spinal GABAergic neurons act as a critical modulator in sensory transmission like pain or itch. The monosynaptic or polysynaptic primary afferent inputs onto GABAergic neurons, along with other interneurons or projection neurons make up the direct and feed-forward inhibitory neural circuits. Previous research indicates that spinal GABAergic neurons mainly receive excitatory inputs from Aδ and C fibers. However, whether they are controlled by other inhibitory sending signals is not well understood.
Methods
We applied a transgenic mouse line in which neurons co-expressed the GABA-synthesizing enzyme Gad65 and the enhanced red fluorescence (td-Tomato) to characterize the features of morphology and electrophysiology of GABAergic neurons. Patch-clamp whole cell recordings were used to record the evoked postsynaptic potentials of fluorescent neurons in spinal slices in response to dorsal root stimulation.
Results
We demonstrated that GABAergic neurons not only received excitatory drive from peripheral Aβ, Aδ and C fibers, but also received inhibitory inputs driven by Aδ and C fibers. The evoked inhibitory postsynaptic potentials (eIPSPs) mediated by C fibers were mainly Glycinergic (66.7%) as well as GABAergic mixed with Glycinergic (33.3%), whereas the inhibition mediated by Aδ fibers was predominately both GABA and Glycine-dominant (57.1%), and the rest of which was purely Glycine-dominant (42.9%).
Conclusion
These results indicated that spinal GABAergic inhibitory neurons are under feedforward inhibitory control driven by primary C and Aδ fibers, suggesting that this feed-forward inhibitory pathway may play an important role in balancing the excitability of GABAergic neurons in spinal dorsal horn.
Introduction
The spinal dorsal horn contains both excitatory (glutamatergic) and inhibitory (GABAergic and Glycinergic) interneurons receiving primary inputs, therefore making it an imperial center for integrating different sensory information. 1 GABAergic interneurons utilizing Gama-aminobutyric acid as their neural transmitter account for about 30% of neurons located in lamina I to III. 2 GABAergic neurons are principal in mammalian spinal cord which involve in mediating postsynaptic inhibition.3,4 Numerous physiological and behavioral studies in rodents confirmed the imperial role of GABAergic neurons in the control of nociceptive and non-nociceptive information.5,6 For instance, GABAergic disinhibition by blocking GABA receptors induces hyperalgesia and tactile allodynia.7–9 Loss of GABAergic inhibitory interneurons also induces sustained itch, 10 which can be reversed by transplanting GABAergic neurons. 5
Modulation of spinal GABAergic neurons on sensory modalities is mainly mediated by receiving inputs from different type afferents innervating peripheral skin.11–13 Myelinated Aδ fibers and unmyelinated C fibers transmitting noxious information terminate in spinal lamina I and II, while myelinated Aβ fibers mainly responding to non-noxious touch information innervate deeper layers from lamine IIi to IV.1,14–16 In gate-control theory of pain proposed 55 years ago, nociceptive and innocuous information from peripheral area can be integrated by spinal inhibitory interneurons acting as gate units. 17 We have reported a gate like spinal feedforward inhibitory pathway from Glycinergic interneurons to PKCγ positive excitatory interneurons which both receive Aβ fiber inputs. 18
The characteristics and inputs of spinal interneurons immunoreacting with GABA antibody or containing GABA-synthesizing enzyme GAD in the dorsal horn have been previously described.19–21 GABAergic neurons labeled by enhanced green fluorescent protein in the spinal cord receive prominent high-threshold C fiber input and small amount of low-threshold Aβ fiber input and contribute to gating innocuous as well as noxious information.22,23 Also, spinal GABAergic islet neurons, one of neural types classified by the dendritic morphology, invariably have inputs from low threshold C-fibers.16,24 Even though the primary fiber’s drive on GABAergic spinal neurons have been elucidated in details,16,25,26 evidence about spinal GABAergic neurons receiving inhibitory innervation from other inhibitory interneurons driven by primary afferents is still delusive.
Here by establishing the Gad2 td-Tomato mice, we examined the synaptic inputs (both excitatory and inhibitory) to GABAergic neurons in spinal lamina II evoked by electric stimulation to the dorsal root. We found that GABAergic neurons not only receive primary inputs from C fibers and Aδ fibers, but also receive inhibitory inputs from other inhibitory interneurons driven by C fibers and Aδ fibers. We also testified that the feedforward inhibition was mediated by merely Glycinergic interneurons or both Glycinergic and GABAergic interneurons. The present study indicated that spinal GABAergic inhibitory neurons are under feedforward inhibitory control driven by C and Aδ fibers.
Materials and methods
Animals
Gad2::Cre mice obtained from The Jackson Laboratory were crossed with Rosa26LSL-tdTomato Ai14 reporter mice line in our lab to label the Gad2::Cre derived (referred to as Gad2+) neurons. They were fed in relatively sterile environment in 12/12 day-night cycle with appropriate temperature at 22°C which were in accordance with the rules and schedule of the Ethics Committee for Animal Experimentation of the Fourth Military Medical University.
Immunohistochemistry
Gad2tdTomato mice (3-4 weeks old), anesthetized deeply with pentobarbital (50 mg/kg), were perfused by saline and subsequently with 4% paraformaldehyde. Then the spinal cord containing lumber segment (L1-L5) was separated and post-fixed in the same fixative for 12 hours followed by immersing the tissue in 30% sucrose for 48 hours. The spinal cord slices (30 µm) were obtained by a cryostat and pre-blocked in 5% donkey serum and 0.3% Triton X-100 for 2 hours at RT (room temperature). Afterwards, incubating the sections with primary antibodies: goat anti-CGRP (1:500, Abcam, ab36001), rabbit anti-PKCγ (1:400, GeneTex, GTX107639), rabbit anti-Gad65 antibody (1:1500, Proteintech, 20746-1-AP) for 12 hours at 4°C. Then tissue slices were incubated with secondary donkey anti rabbit 488 (1:500, Abcam, ab150073), donkey anti goat 488 (1:500, Abcam, ab150129) and Lectin IB4 (1:200, Vectorlabs) for 2 hours at RT. The slices were photographed and viewed using confocal microscopy with fluorescence to identify td-Tomato expressing neurons. Three nonadjacent spinal sections were selected randomly from lumber segments (L4-L5) from 3 young (3-4 weeks) mice to observe the co-expression of tdTomato and Gad65. For the specificity of GAD65 antibody, control slices were incubated in the donkey serum without primary antibody. No specific labeling was observed in the control slices compared with those immersed in primary antibodies (Supplementary Figure 1).
Spinal cord slices preparation
The lumber spinal cord of deeply anesthetized male Gad2tdTomato mice (3–4 weeks old) was acquired by heart perfusion in a cold room and subsequent detachment in the cutting solution preoxygenated with 95% O2 and 5% CO2 for 30 min containing (in mM) sucrose 75, NaCl 80, KCl 2.5, NaH2PO4 1.25, CaCl2 2.5, MgCl2 1.2, NaHCO3 25, sodium ascorbate 1.3, and sodium pyruvate 3.0 (pH at 7.4 and osmolality at 310–320 mOsm). All ventral and dorsal roots except one dorsal root were torn off after the spinal pia mater was stripped out. The remaining tissue was parasagittally cut by a vibrating microtome. Spinal slice (450–500 µm) attached with one dorsal root (0.8–1.0 cm) was then quickly immersed in the perfusate made of (in mM) NaCl 125, KCl 2.5, NaH2PO4 1.25, NaHCO3 26, D-glucose 25, CaCl2 2, MgCl2 1, sodium ascorbate 1.3, and sodium pyruvate 3.0 (pH at 7.2 and osmolality at 310–320 mOsm). The perfusate was continuously filling with 95% O2 and 5% CO2 for 1 hour at room temperature.
Patch clamp recordings
The procedures for electrophysiological recording have been reported previously.15,16,18 Spinal slices fixed in a groove with an anchor were immersed in artificial cerebrospinal fluid. They were viewed using a microscope with a 40x objective installed with fluorescence. The Gad2 td-Tomato neurons in translucent band of spinal dorsal horn (lamina II) were recorded by tight seal whole cell patch recordings using an Axopatch 200B amplifier with glass pipettes (5–10 MΩ) containing internal solution. The pipette solution included (in mM) K-gluconate 130, KCl 5, Mg-ATP 4, phosphocreatine 10, Li-GTP 0.3, and HEPES 10 (pH at 7.3 and osmolality at 300 mOsm). The acquisition and analysis of data were performed through pCLAMP10 software. The resting membrane potential (RMP) was identified after the establishment of whole-cell configuration in I = 0 model. Recordings were considered to be from “healthy” neurons if stable membrane potentials were more negative than −45 mV, and overshooting action potentials were generated by depolarizing current introduced through the recording electrode. Series resistances typically were 10 MΩ and were monitored throughout the recording period. Recording from a cell was discarded if the series resistance changed significantly (25%). 16 In current clamp mode, the firing pattern of each neuron was determined by 1-s depolarizing pulses with 25 pA step from the holding potential of −60 mV. Tonic firing neurons featured persistent firing of action potentials. Delayed firing neurons were characterized with a delay between the onset of current injection and the discharge of action potentials. Initial bursting neurons were characterized with only a few action potentials at the beginning. Neurons with firing patterns occurring at irregular intervals were classified as irregular firing type.15,16
The procedures for dorsal root stimulation have been reported previously.15,16,18 The dorsal root was connected with a suction electrode (Figure 3(a)). Graded 0.1 to 0.5 ms pulses delivered through a suction electrode were used to initiate afferent volleys in the dorsal roots. The conduction velocities (CVs) of primary afferent fibers evoking monosynaptic EPSPs were estimated from the latency of the evoked response and the conduction distance. The dorsal root evoked postsynaptic potentials were elicited by graded intensity to recruit Aβ, Aδ and C fibers. Classification of whether synaptic responses were from Aβ, Aδ and C fibers was based on a combination of response threshold and CVs. A or C fiber evoked EPSPs were judged to be monosynaptic if they had a constant latency and absence of failures in 20 Hz or 1 Hz repetitive trials.15,27,28 The response thresholds and CVs for Aβ, Aδ, and C fibers were determined under our recording conditions. 18 The dorsal root was stimulated orthodromically (0.1 ms for A fiber, 0.5 ms for C fiber) and compound APs were recorded extracellularly at its central end. The stimulus intensities for activation of Aβ, Aδ, and C fibers were determined at 0.1 to 0.5 V, 0.6 to 1.3 V, and 2 to 10 V in our recording conditions. 18
Strychnine (glycine receptor antagonist, 2 µM), together with bicuculline (GABA receptor antagonist, 10 µM) were used to differentiate the component of eIPSPs. The amplitude of 10 eIPSPs was averaged and compared before and after drug application.
To label the cells during whole cell recording, biocytin (0.5%) in the internal solution was essential. After recording, the whole slice was fixed in 4% paraformaldehyde overnight and blocked with Tris-Triton buffer containing 4% donkey serum. Then, the slice was incubated with Fluorescein green conjugated Streptavidin Avidin (1:500 Vector) for 12 hours followed by washing in Tris buffer. All the fluorescent signals were captured by confocal microscope (Olympus FV1200). The determination of morphological features was based on the combination of dendrite length and orientation.15,16 Briefly, islet cells were characterized by elongating their dendritic trees rostrocaudally. Neurons similar with islet types but whose dendrites extended moderately in the rostro-caudal planes were identified as central cells. Vertical cells had extensive dendrites distributed dorsoventrally.16,29
Data analysis
The data were reported as mean ± SEM. The number of GABAergic neurons was quantified by the software Image J. One-way ANOVA was applied to confirm the significance of value changes. P < 0.05 was regarded as significant.
Results
GABAergic neurons were labeled successfully in Gad2tdTomato mice
To selectively visualize GABAergic neurons in the spinal cord, we established the Gad2tdTomato mice to mark the neurons expressing Gad2 gene coding for one isoform of the enzyme Gad 65 which synthesizes GABA. The distribution of td-Tomato expressing neurons in lumbar spinal slices of Gad2tdTomato mice was observed. Fluorescence intensity of td-Tomato expressing spinal neurons was distinguished considerably which ranged from weak to strong as is shown in Figure 1(a), (d), and (g). The td-Tomato positive neurons were highly expressed in lamine I-III of the dorsal horn. To accurately determine the laminar expression of these fluorescent neurons, immunohistochemistry staining of calcitonin gene-related peptide (CGRP) and isolectin B4 (IB4) (Figure 1(b) and (e)) were used to distinguish lamina I, lamina IIo and lamina IIi. Protein kinase C γ (PKC γ) (Figure 1(h)) was also used as a marker to direct the boundary of lamina II–III. Figure 1 illustrated that Gad2 td-Tomato neurons were mainly located in lamina II (53.27 ± 3.88%) and III (32.15 ± 3.79%), while a few were in lamina I (14.58 ± 1.44%). In addition, a small part of red fluorescence positive neurons was also observed in deeper layers.

The expression pattern of td-Tomato neurons in lumber section of Gad2-td mice. (a, d, g) Images of Gad2-tdTomato positive neurons (red) in the dorsal spinal cord of lumber section. IHC images labeling with antibodies of CGRP (b), IB4 (e) and PKCγ (h) (green). Double fluorescence images depicting Gad2-tdTomato distribution with CGRP (c), IB4 (f) and PKCγ (i). n = 3 mice and 3 sections of each mice. IHC: immunohistochemistry; CGRP: calcitonin gene-related peptide; IB4: isolectin B4; PKCγ: Protein kinase Cγ.
To confirm that the td-Tomato labeled neurons of Gad2tdTomato mice were mainly GABAergic neurons, we performed immunohistochemistry of GABAergic neurons on transverse spinal slices from Gad2tdTomato mice by applying anti-Gad 65 antibody (Figure 2(a) to (f)). The results showed that Gad2 td-Tomato neurons constitute 64.5 ± 3.0% neurons marked by anti-Gad 65 antibody in lamine I–III (Figure 2(g)). Simultaneously, as is shown in Figure 2(g), 75.9 ± 2.3% Gad2 td-Tomato cells were also labeled with Gad65 antibody. The above results indicated that td-Tomato convincingly marked most GABAergic interneurons.

Gad2-td Tomato successfully recapitulates expression of spinal GABAergic neurons. (a–c) IHC images of Gad65 (green) with Gad2-tdTomato fluorescence(red). (d–f) High power confocal images of selected area in (a) to (c). Arrows indicate Gad2 td-Tomato neurons co-expressed with Gad 65. (g) Percentage of td-Tomato fluorescence co-expressing Gad65 and Gad65 neurons co-expressing td-Tomato. n = 3 mice and 3–4 sections of each mice. Data was shown as mean ± SEM.
Spinal GABAergic interneurons possess diverse morphological and electrophysiological properties
Previous research demonstrated four main types of neural morphology based on the patch clamp recordings combined with morphological analysis from mice, guinea pigs and rats which are islet, central, vertical and radial.15,16,30 GABAergic neurons labeled by different methods exhibited distinct cell morphology.23,29,31 To test this, we explored the morphology of GABAergic neurons in the parasagittal spinal cord slices from Gad2tdTomato mice. We patched the fluorescence positive neurons and subsequently applied biocytin intracellularly (Figure 3(a)). Figure 3(b) demonstrated a neuron having both fluorescence (red) and biocytin (green) located on the border between lamina II and III and had morphology accordant with islet cells which accounted for 42.9% (6/14) of the neurons labeled. Central cells (35.7%, 5/14) were also noticed in superficial layer of spinal cord as is depicted in Figure 3(c). Neuron accordant with vertical type (21.4%, 3/14) was shown in Figure 3(d). Table 1 revealed concrete measurement of different cell types. These results are in accordance with previous research that most GABAergic neurons are islet or central type.29,31

Morphologic features of Gad2-tdTomato labeled neurons in parasagittal spinal slice. (a) Schematic image of spinal slices with a dorsal root of Gad2-td mice in electrophysiological recordings. Examples of Gad2-td neurons (red) filled with biocytin (green) match with islet (b), central (c) and vertical (d) properties. (e) Proportions of cells with 3 morphological features. 14 neurons of 6 mice were recorded.
Morphological properties of Gad2-tdTomato labeled neurons.

Data shown as means ± SEM.
The firing pattern of neurons was used as an important index in identifying the subclass of inhibitory interneurons in the spinal cord. 16 We recorded the firing pattern of 66 fluorescent cells in current clamp mode. The criteria for classifying firing pattern were based on previous research. 31 The most common type of GABAergic neurons exhibited was tonic firing pattern which included 69.7% of the total recorded neurons (Figure 4(a)). Irregular firing pattern was also observed which account for 15.2% of the recorded neurons (Figure 4(b)). We also found delayed firing and initial burst firing which made up 6.1% of the recorded neurons respectively (Figure 4(c) and (d)). Simultaneously, only a small part of neurons (2.9%) showed regular and single spike firing patterns (Figure 4(e)). In addition, the membrane properties (RMP, Rh) and characteristics of action potential (Firing threshold, Amplitude, Duration, Half-width) were shown in Table 2.
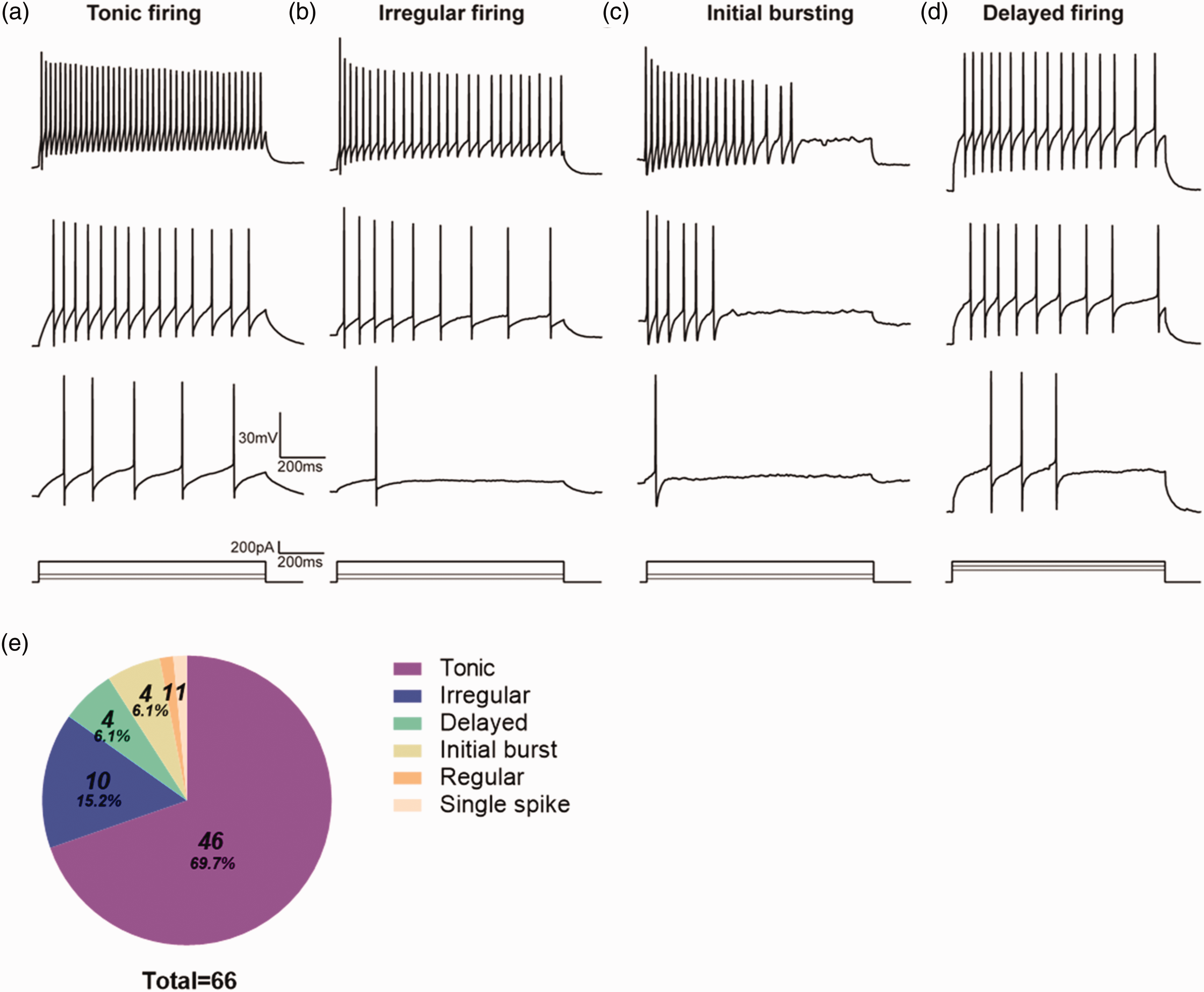
Representative AP firing patterns of Gad2-tdTomato neurons in lamina II. Examples of tonic (a), irregular (b), initial bursting (c), delayed (d) firing patterns recorded from GABAergic neurons by injecting different intensities of depolarizing current. (e) Proportion of different AP firing patterns acquired from GABAergic neurons. n = 7 mice and 9–12 neurons per mice were recorded.
Electrophysiological features of Gad2-tdTomato labeled neurons.

Data shown as means ± SEM. RMP, resting membrane potential, Rh, rheobase.
GABAergic interneurons receive both excitatory and inhibitory inputs
The clarification of different afferent types from peripheral which activate specific GABAergic neurons can give a better understanding on the role of neurons in sensory processing. To address the primary afferents to spinal GABAergic neurons, we recorded the evoked post synaptic potentials of GABAergic neurons by stimulating the dorsal roots at different intensities. We found that GABAergic neurons were mainly activated by monosynaptic C fiber which accounted for 53.0% of total evoked postsynaptic potentials (Figure 5(a)). Activation mediated by monosynaptic Aδ and Aβ fiber contributed with lower percentage of 16.5% and 7.8% respectively (Figure 5(b) and (c)). The eEPSPs mediated by Aδ and C (2.6%), or Aβ and C fibers (0.9%) (Figure 5(d) and (e)) were also observed. After blocking the excitatory components by holding membrane potential at 0 mV, eIPSPs were detected evidently (Figure 5(f) and (g)) in 9 neurons. We also found that 13 of total GABAergic neurons responded merely with eIPSPs at RMP which was mediated by C or Aδ fibers (Figure 5(h) and (i)). Therefore, significant number of GABAergic neurons received inhibitory inputs from other inhibitory interneurons driven by Aδ and C fibers (Figure 5(f) to (j)). Strikingly, none of the inhibitory inputs mediated by Aβ fibers were detected.

Examples of primary afferent drive onto Gad2-tdTomato neurons. (a) EPSPs were evoked at the strength of C fiber (3.7 V, 0.5 ms, 0.1 Hz) through stimulating the dorsal root. (b) eEPSPs mediated by Aδ fiber (0.6V, 0.1 ms, 0.1 Hz). (c) eEPSPs mediated by Aβ fiber (0.3 V, 0.1 ms, 0.1 Hz). (d) Mixed eEPSPs of Aδ (0.7 V, 0.1 ms, 0.1 Hz) and C fiber (2.2 V, 0.5 ms, 0.1 Hz). (e) eEPSPs mediated by Aβ (0.3 V, 0.1 ms, 0.1 Hz) and C fiber (5.5 V, 0.5 ms, 0.1 Hz). Examples of mixed eIPSP and eEPSP of C fiber (3.8 V, 0.5 ms, 0.1Hz) (f) and Aδ fiber (1.3 V, 0.1 ms, 0.1 Hz) (g). Purely inhibitory inputs by C fiber (4.5 V, 0.5 ms, 0.1 Hz) (h) and Aδ fiber (0.7V, 0.1 ms, 0.1 Hz) (i). (j) Percentage of eEPSPs and eIPSPs of different primary afferents recorded from GABAergic neurons. n=14 mice and 7–9 neurons per mice were recorded. eEPSPs: evoked excitatory postsynaptic potentials; eIPSPs: evoked inhibitory postsynaptic potentials.
To further distinguish if the inhibition to GABAergic neurons were mediated by other GABAergic or Glycinergic interneurons, we applied bicuculine and strychnine when encountering eIPSPs. A large part (57.1%) of eIPSPs executed by Aδ fibers was partly blocked after applying bicuculine (−8.00 ± 0.57 mV vs −4.40 ± 0.38 mV, n = 4, P < 0.05) and also suppressed almost completely by subsequent strychnine application (−4.40 ± 0.38 mV vs −1.05 ± 0.30 mV, n = 4, P < 0.05) as is shown in Figure 6(b) and (e). Purely Glycinergic inhibition (−10.02 ± 0.08 mV vs −1.50 ± 0.31 mV, n = 3, P < 0.05) was observed in the remaining Aδ fiber driven eIPSPs (42.9%) (Figure 6(a) and (e)). Conversely, a large part (66.7%) of C-fiber mediated eIPSPs was unaffected after applying bicuculine (−7.84 ± 0.3 0 mV vs −5.41 ± 1.3 6 mV, n = 10, P > 0.05) and they were almost totally abolished (−5.41 ± 1.36 mV vs −0.49 ± 0.17 mV, n = 10, P < 0.05) by subsequently adding strychnine (Figure 6(c) and (e)). The remaining C fiber driven eIPSPs were due to the mixed contribution of both GABA (−16.11 ± 1.54 mV vs −8.63 ± 1.80 mV, n = 5, P < 0.05) and Glycine (−8.63 ± 1.80 mV vs −0.55 ± 0.29 mV, n = 5, P < 0.05) as they were almost completely blocked after perfusion of both antagonists (Figure 6(d) and (e)). Notably, we didn’t acquire purely bicuculine sensitive components of eIPSPs driven by both types of fibers. The above results suggested that C fiber driven feed-forward inhibition onto GABAergic interneurons was mainly glycinergic, while Aδ driven feed-forward inhibition onto GABAergic interneurons was rather both glycinergic and GABAergic (Figure 6(f)).
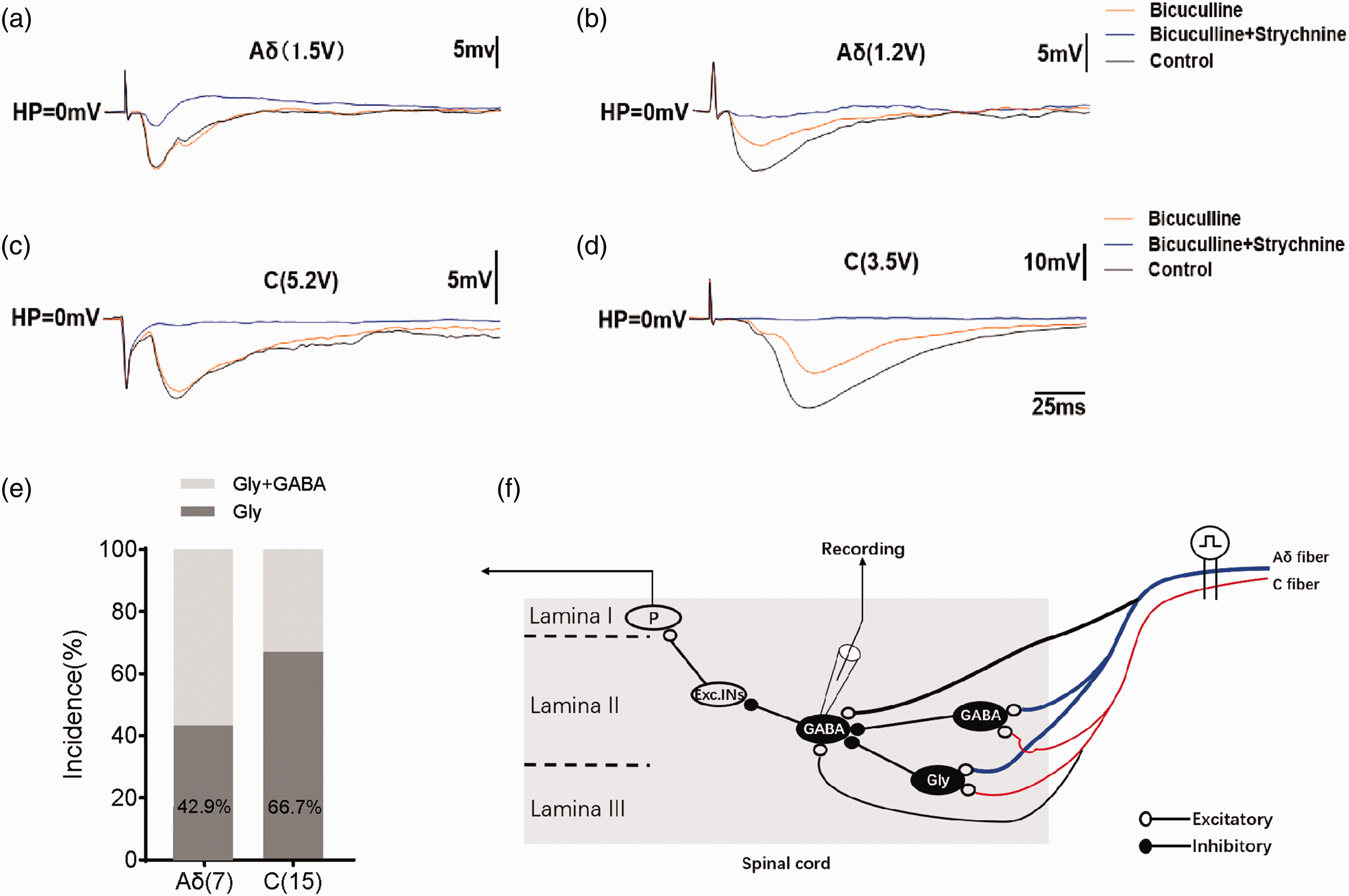
Glycine and/or GABA mediated eIPSPs driven by Aδ and C fibers. (a) Example of GABAergic neuron receiving purely Glycinergic input driven by Aδ fiber. IPSPs were acquired in the presence of bicuculine (10 µM), bicuculine (10 µM) and strychnine (2 µM) and under control conditions. (b) Mixed inhibition by Glycine and GABA driven by Aδ fiber. Examples of only Glycine dominant IPSPs (c) and GABAergic with glycinergic dominant IPSPs (d) driven by C fibers. (e) Bar chart demonstrating the relative contribution of Glycine and GABA to IPSPs. (f) Schematic summarizing the feed forward inhibition to spinal GABAergic neurons driven by primary C and Aδ fibers. 22 neurons of 14 mice were recorded. eIPSPs: evoked inhibitory postsynaptic potentials.
We have analyzed the correlations between the inhibitory inputs to spinal GABAergic neurons and their electrophysiological properties. Of the total 66 neurons recorded in the current experiments, 4/46 of cells displaying tonic firing received inhibitory inputs, while nearly half of irregular firing neurons (3/10) received inhibitory inputs. No inhibitory components were observed in the rest firing types. We also analyzed inputs received by GABAergic neurons with different morphological features. 2 of 6 islet cells received inhibitory inputs compared with 3 of 5 central cells and 2 of 3 vertical cells. From the above results, we can conclude that tonic and irregular cells are more likely to be controlled by feedforward inhibitory inputs. All morphological types of GABAergic neurons might be under feedforward inhibitory control.
Discussion
Previous research has demonstrated excitatory primary afferents to GABAergic neurons in the spinal cord, but whether if there are inhibitory inputs onto GABAergic neurons is not mentioned. 22 By using the Gad2-tdTomato mice, the present study characterized both excitatory and inhibitory synaptic inputs to spinal GABAergic neurons. The results indicated that spinal GABAergic inhibitory interneurons are under feed-forward inhibitory control from other inhibitory interneurons driven by primary C and Aδ fibers. Although we did not encounter any inhibitory input to spinal GABAergic neurons driven by Aβ fibers, we cannot exclude the possibility that some GABAergic neurons receive inhibition driven by Aβ fibers.
Neurons marked by td-Tomato fluorescence are mainly located in lamine II-III especially in lamina II which is paralleled with preceding research about the location of spinal GABAergic neurons. 31 Meanwhile, we confirmed GABAergic neurons by applying antibody combining Gad65 (the GABA synthesizing enzyme) which stained 75.9% of td-Tomato neurons for the reason that Gad65 is linked with Gad2 gene.32,33 The morphologic results indicated that GABAergic neurons in lamina II primarily met the features of islet type, which is consistent with previous work. 16 In addition, some td-Tomato neurons were also characterized as central type neurons which contains both excitatory and inhibitory phenotypes. 1 Taking in account the fact that the firing patterns in response to current steps were diverse, it cannot be used to classify spinal GABAergic neurons. However, studies about the relationship between the function and firing properties of interneurons in the superficial layer indicate that neurons displaying tonic firing are inhibitory.34,35 Our results described that the most common firing patterns of td-Tomato neurons was tonic which had also been observed in EGFP labeled Gad 67 and Gad 65 mice from previous reports.22,25 Also, islet neurons mainly displayed tonic firing mode which is accordant with previous study. 16 While the central neurons displayed tonic, initial bursting and transient firing mode, the vertical neurons mainly displayed delayed firing mode. In this study, we also found that tonic and irregular cells are more likely to be controlled by feedforward inhibitory inputs. All morphological types of GABAergic neurons might be under feedforward inhibitory control. Previous studies have usually classified neurons according to their basic electrophysiological characteristics such as discharge patterns and morphological characteristics, but rarely involved the distinction of primary afferent fibers.18,30,31,36–38 Our recent research documented that it is not reliable to judge the type of spinal neurons only by discharge patterns and morphological characteristics. 18
Moreover, apart from excitatory drive to GABAergic neurons mediated by different fibers, 39 we provided direct evidence proving inhibitory inputs to Gad2 td-Tomato neurons driven by Aδ and C fibers. The inhibitory inputs to GABAergic neurons may be on account of normal physiological conditions. For example, the connections between brain inhibitory interneurons mediate diverse memory expression or emotional behaviors related to sensory inputs.40,41 The disinhibition of this pathway could permit enormous activation of excitatory interneurons and contribute to the synaptic plasticity which involved in the integration of complicated sensory inputs.42,43 As for the spinal cord where peripheral pain or itch information were processed primarily, this consecutive inhibitory pathway may gate sensory discrimination when pain and itch signals simultaneously exist. 44 Previous evidence suggests that GlyT2 immunoreactivity was highly involved in many Gad-immunoreactive boutons. 20 With the exception of the possibility that some spinal neurons use glycine and GABA as co-transmitters, it's natural to assume that the GABAergic neurons receive inhibitory inputs from spinal glycinergic and/or GABAergic neurons. Although the inhibitory inputs recorded from GABAergic neurons only occupied a small part of whole primary inputs, it is reasonable to assume that activation of this consecutive inhibitory pathway will induce the decreased excitability of downstream GABAergic neurons which may generate hypersensitivity to spinal pain or itch pathway. Furthermore, this astonishing phenomenon prompted us to hypothesize whether if there is a potential balance between inhibitory and excitatory components on the GABAergic neurons in the spinal cord, which can be altered in pathological conditions such as neuropathic pain or itch.
The sources of inhibitory inputs to GABAergic neurons in the spinal dorsal horn deserve comments. Previous research has categorized inhibitory interneurons in the spinal cord into four non-overlapped types according to the neurochemical identity, that is, galanin+, NPY+, NOS+ and dynorphin+ neurons. 45 These 4 types of inhibitory neurons are GABAergic or Glycinergic.46–49 However, for the reason of the technological limits of single cell patch clamp recording, we could only elucidate the neurochemical features of recorded neurons combining with intracellular biocytin labeling. The neurochemical phenotypes of presynaptic inhibitory interneurons in the spinal cord couldn’t be verified through electrophysiological recordings applied in our experiment. Combining with our results, it seems that all phenotypes of inhibitory neurons (galanin+, NPY+, NOS+ and dynorphin+) can be potential candidates for the inhibitory inputs to GABAergic neurons. Future research through double patch clamp whole cell recordings may give a definite description about this issue.
It has been reported that GABAergic neurons predominantly displayed delayed firing or initial burst firing patterns.31,50 Our results depicted that the tonic firing pattern was highly prevalent. This could be due to the diverse location of randomly selected neurons with different resting membrane potentials (RMPs). For instance, GABAergic neurons with its RMP around −48 mV tend to discharge tonic firing in lamina II. 30 When neurons are at a hyperpolarized RMP, they are more liable to display gap or initial burst firing with the potential mechanism associated with A-type potassium currents. 51 In addition, the electrophysiological properties of GABAergic neurons in the spinal cord could be changed during development.22,52–55 However, the primary inputs to GABAergic neurons in lamina II didn’t display significant changes between young (P21) mice and adult (P49) mice. 56
GABAergic neurons of Gad67-EGFP mice has been comprehensively observed in other body regions which has been found highly expressed in the neocortical and hippocampal parts. 57 We have not addressed fluorescence expression patterns among other part of body in Gad2-td mice. Further study on td-Tomato expression in the brain is indispensable. Furthermore, Glycinergic neurons in the spinal cord have also been extensively researched by using the GlyT2-EGFP mouse line and it showed that Glycine and/or GABA mediated inhibition also existed in EGFP positive neurons. 58 We have not addressed if the fluorescent neurons in our experiment actually indicated the purely GABAergic or GABAergic mixed with Glycinergic neurons because GABAergic neurons exhibited a large overlay with Glycinergic neurons in the lamine I-III. 20 Moreover, Duan et al. showed that the same type of spinal interneurons in different layers could receive diverse afferent drive. 59 Additional research is needed to examine whether this potential pathway of GABAergic neurons also exist in more superficial or deeper layers. Lastly, further studies are essential to determine if this pathway could balance itch and pain transmission and if GABA and/or Glycine dominant inhibition will be altered under pathological states.
Equally imperial, neurons marked with red fluorescence in our experiment couldn’t label the entire GABAergic neurons located in the spinal cord, since gad2 only encode one phenotype of GABA synthetase gad65 in central nervous system. Additional research is needed to clarify whether GABAergic neurons marked with gad1 will also exhibited such a feedforward inhibition with similar proportion.
The present study demonstrated a complicated circuit in the spinal cord containing GABAergic and Glycinergic inhibition. Peripheral sensory information can be transmitted to the spinal inhibitory interneurons by Aδ and C fibers which may further release Glycine or GABA, thereby acting on GABAergic neurons and finally achieving the disinhibition effect of GABAergic neurons (Figure 6(f)). This study may contribute to understanding the function of spinal inhibitory interneurons in different sensory modalities and provide novel targets to treat pathologic conditions.
Supplemental Material
sj-pdf-1-mpx-10.1177_1744806921992620 - Supplemental material for Spinal GABAergic neurons are under feed-forward inhibitory control driven by Aδ and C fibers in Gad2 td-Tomato mice
Supplemental material, sj-pdf-1-mpx-10.1177_1744806921992620 for Spinal GABAergic neurons are under feed-forward inhibitory control driven by A
Footnotes
Acknowledgements
We thank Christopher G. Myers (PhD, Johns Hopkins University) for language review of the manuscript.
Author Contributions
PL and YL designed the study. PL, XZ and ZJ performed these experiments. QW and XH analyzed the data and prepared the figures. PL and XZ wrote the manuscript. YL revised the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China to Dr. Yan Lu (Grant code: 31530090, 81971058).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
