Abstract
Background
Central post-stroke pain (CPSP) is a type of neuropathic pain caused by dysfunction in the spinothalamocortical pathway. However, no animal studies have examined comorbid anxiety and depression symptoms. Whether the typical pharmacological treatments for CPSP, which include antidepressants, selective serotonin reuptake inhibitors (SSRIs), and anticonvulsants, can treat comorbid anxiety and depression symptoms in addition to pain remains unclear? The present study ablated the ventrobasal complex of the thalamus (VBC) to cause various CPSP symptoms. The effects of the tricyclic antidepressants amitriptyline and imipramine, the SSRI fluoxetine, and the anticonvulsant carbamazepine on pain, anxiety, and depression were examined.
Results
The results showed that VBC lesions induced sensitivity to thermal pain, measured using a hot water bath; mechanical pain, assessed by von Frey test; anxiety behavior, determined by the open-field test, elevated plus-maze test, and zero-maze test; and depression behavior, assessed by the forced swim test. No effect on motor activity in the open-field test was observed. Amitriptyline reduced thermal and mechanical pain sensitivity and anxiety but not depression. Imipramine suppressed thermal and mechanical pain sensitivity, anxiety, and depression. Fluoxetine blocked mechanical but not thermal pain sensitivity, anxiety, and depression. However, carbamazepine did not affect pain, anxiety, or depression.
Conclusion
In summary, antidepressants and SSRIs but not anticonvulsants can effectively ameliorate pain and comorbid anxiety and depression in CPSP. The present findings, including discrepancies in the effects observed following treatment with anticonvulsants, antidepressants, and SSRIs in this CPSP animal model, can be applied in the clinical setting to guide the pharmacological treatment of CPSP symptoms.
Keywords
Background
Central post-stroke pain (CPSP) is a type of central neuropathic pain caused by lesions or dysfunction in the spinothalamocortical pathway.1,2 The prevalence rate of CPSP is reported at 8%–46% following a hemorrhagic stroke due to the heterogeneity of affected brain areas. 3 After stroke, patients with CPSP experience spontaneous or evoked pain at early 1–2 months. 2 Clinically, CPSP patients are characterized by pain symptoms3,4 and mild motor dysfunctions;2,5 moreover, CPSP is often associated with a variety of psychological symptoms, including sleep disturbance, anxiety, and depression, which place CPSP patients at high risk of suicide.4,6,7 The present study used a previously described animal model of CPSP, in which the administration of collagenase is used to ablate the ventrobasal complex of the thalamus (VBC), causing pain symptoms.8-12 In addition to developing pain symptoms, studies examining this animal model of CPSP have described the existence of comorbidities known to be associated with CPSP, such as anxiety and depression symptoms, facilitating the simultaneous study of all of these comorbidities and better representing the human experience of CPSP.
Previous studies have recommended the tricyclic antidepressant amitriptyline for first-line CPSP treatment.2,13 Among patients with CPSP who respond poorly to amitriptyline administration, the other medications, including antidepressants, anticonvulsants (e.g., carbamazepine [CBZ]), opiates, anesthetics, and N-methyl-D-aspartate (NMDA) antagonists, are often used as second-line drugs to treat the core CPSP symptom of spontaneous or evoked pain.6,13
According to previous clinical studies in patients with CPSP, tricyclic antidepressants, such as amitriptyline and imipramine, inhibit the reuptake of the neurotransmitters serotonin and norepinephrine, resulting in excessive quantities remaining in the synaptic cleft, which may be involved in reducing CPSP-associated symptoms. 14 The anticonvulsant CBZ, which is often used as a second-line medication, suppresses the activity of voltage-dependent sodium ion channels by decreasing electrical activity and glutamate release, which may represent a potential mechanism for reducing pain symptoms in patients with CPSP. 15 Alternatively, selective serotonin reuptake inhibitors (SSRIs, for example, fluvoxamine) are sometimes used in patients with CPSP who experience stroke within 1 year, and its intended effects are independent of its antidepressant functions. 2 In conventional clinical use, fluoxetine is prescribed to treat major depressive disorder. 16 Fluoxetine acts by blocking the serotonin reuptake mechanism in presynaptic neurons, increasing serotonin concentrations in the synaptic cleft, which reduces depression symptoms.16,17 However, the efficacy of fluoxetine against the pain symptoms associated with CPSP has not been thoroughly explored, and fluoxetine is seldom employed for pain symptoms. Currently, the existed animal model allows for the comprehensive exploration of the effects of tricyclic antidepressants, such as amitriptyline and imipramine; SSRIs, such as fluoxetine; and anticonvulsants, such as CBZ, on pain symptoms and comorbid anxiety and depression in CPSP.
The present study aimed to address two issues. First, the present study examined whether the ablation of the VBC in rats could induce pain, anxiety, depression, and motor impairments similar to those experienced by patients with CPSP. Second, the study examined whether amitriptyline and imipramine (tricyclic antidepressants), fluoxetine (an SSRI), or CBZ (an anticonvulsant) could reduce CPSP-related symptoms, including pain, anxiety, depression, and motor impairments.
Results
The experimental procedure was shown in Figure 1. All rats received adaptation, surgery, recovery, and various behavioral tests with antidepressants amitriptyline and imipramine, fluoxetine, and anticonvulsants CBZ (Figure 1). Overview of the experimental procedures for Experiments 1 and 2. All rats experienced adaptation, surgery, recovery, and behavioral tests. (a) In Experiment 1, the rats were tested for thermal pain at baseline, followed by surgery. After recovery from surgery for 7 days, all rats were evaluated for thermal pain test in the hot water bath, motor activity and anxiety behavior in the open-field test, anxiety responses in the elevated plus-maze test, and depression in the forced swim test 1 week for a test on pre-test and Days 7, 14, 21, and 28. (b) In Experiment 2, all rats were subjected to the same procedures as in Experiment 1, but the elevated plus-maze test was replaced with the zero-maze test. The pain tests comprised thermal pain in the hot water bath and mechanical pain in the von Frey test. Amitriptyline and imipramine were intraperitoneally injected on Day 21 and Day 28. Fluoxetine and CBZ were intraperitoneally injected once a day from Day 14 to Day 28. After amitriptyline, imipramine, fluoxetine, and CBZ administrations, the pain, open-field, zero-maze, and forced swim tests were conducted. (c) An example of a collagenase-induced lesion in the ventrobasal complex. CBZ: carbamazepine.
CPSP and comorbidities To identify whether lesioning the right VBC in rats was able to induce the core pain symptoms associated with CPSP, the study tested the right and left withdrawal responses. A 2 × 5 two-way mixed analysis of variance (ANOVA) was performed to analyze withdrawal time in response to pain-inducing stimuli. The results showed significant differences in left paw withdrawal between groups [F (1, 18) = 14.96, p < 0.05], whereas non-significant differences were identified between day [F (4, 72) = 0.30, p > 0.05] and in the group × day interaction [F (4, 72) = 1.64, p > 0.05]. Post-hoc analysis using Tukey’s honestly significant difference (HSD) test showed significant differences during Days 14, 21, and 28 (p < 0.05; Figure 2(a)). For right paw withdrawal, significant differences were observed for group [F (1, 18) = 202.35, p < 0.05], day [F (4, 72) = 4.57, p < 0.05], and the group × day interaction [F (4, 72) = 10.11, p < 0.05]. Post-hoc analysis using Tukey’s HSD test showed significant differences during Days 7, 14, 21, and 28 (p < 0.05; Figure 2(b)). Therefore, the withdrawal time in the CPSP group decreased significantly compared with the withdrawal time of the sham group. Measurement of thermal pain behavior in the hot water bath test in sham and CPSP rats. Mean (±SEM) withdrawal time (sec) of the sham and CPSP groups for pre-test and Days 7, 14, 21, and 28 in (a) the left paw and (b) right paw; *p < 0.05 indicates significant differences compared with the sham group. CPSP: central post-stroke pain induced by the ablation of the ventrobasal complex of the thalamus. The open-field test was used to examine motor activity and anxiety behaviors. A one-way ANOVA revealed no significant differences in total distance traveled [F (1, 18) = 0.79, p > 0.05; Figure 3(a)], maximum speed [F (1, 18) = 0.06, p > 0.05; Figure 3(b)], or time spent time in the center square [F (1, 18) = 3.27, p > 0.05; Figure 3(d)] between the sham and CPSP groups. However, the number of entries into the center square was lower for the CPSP group than for the sham group [F (1, 18) = 19.24, p < 0.05; Figure 3(c)]. Therefore, the VBC lesion did not appear to affect motor activity, but decreased entries into the center square suggested that the VBC lesion increased anxiety behaviors in the CPSP rats. Measurements of motor activity and anxiety behaviors in the open-field test for sham and CPSP rats. (a) Mean (±SEM) total distance traveled (mm), (b) mean (±SEM) maximum speed (mm/sec), (c) mean (±SEM) entries into the center square, and (d) mean (±SEM) time spent in the center square (sec) for the sham and CPSP groups. Measurements of anxiety behaviors in the elevated plus-maze test for the sham and CPSP rats. (e) Mean (±SEM) latency time (sec), (f) mean (±SEM) entries into the open arm, (g) mean (±SEM) time spent in the open arm (sec), (h) mean (±SEM) entries into the closed arm, and (i). mean (±SEM) time spent in the closed arm (sec) for the sham and CPSP groups. All behavioral data were averaged on Day 7, 14, 21, and 28. *p < 0.05 indicates significant differences compared with the sham group. CPSP: central post-stroke pain induced by the ablation of the ventrobasal complex of the thalamus. In the elevated plus-maze test, which was used to assess anxiety behavior, one-way ANOVA showed no significant differences in latency time [F (1, 18) = 0.19, p > 0.05; Figure 3(e)], entries to open arms [F (1, 18) = 0.55, p > 0.05; Figure 3(f)], time spent in open arms [F (1, 18) = 0.53, p > 0.05; Figure 3(g)], entries to closed arms [F (1, 18) = 0.42, p > 0.05; Figure 3(h)], or time spent in closed arms [F (1, 18) = 1.31, p > 0.05; Figure 3(i)]. Therefore, CPSP rats did not display anxiety behaviors during the elevated plus-maze evaluation. The forced swim test was used to examine depression symptoms in the CPSP model. One-way ANOVA demonstrated a significant increase in the floating time for the CPSP model relative to the sham group [F (1, 18) = 10.94, p < 0.05; Figure 4(a)] and a significant decrease in struggling time [F (1, 18) = 25.04, p < 0.05; Figure 4(B)]. However, no significant difference in swimming time [F (1, 18) = 1.99, p > 0.05; Figure 4(c)] was observed between groups. These findings indicated that the CPSP rats displayed depression-like behaviors. Measurements of depression behaviors in the forced swim test for the sham and CPSP rats. (a) Mean (±SEM) floating time (sec), (b) mean (±SEM) struggling time (sec), and (c) mean (±SEM) swimming time (sec) for the sham and CPSP groups. All behavioral data were averaged on Day 7, 14, 21, and 28. *p < 0.05 indicates significant differences compared with the sham group. CPSP: central post-stroke pain induced by the ablation of the ventrobasal complex of the thalamus.


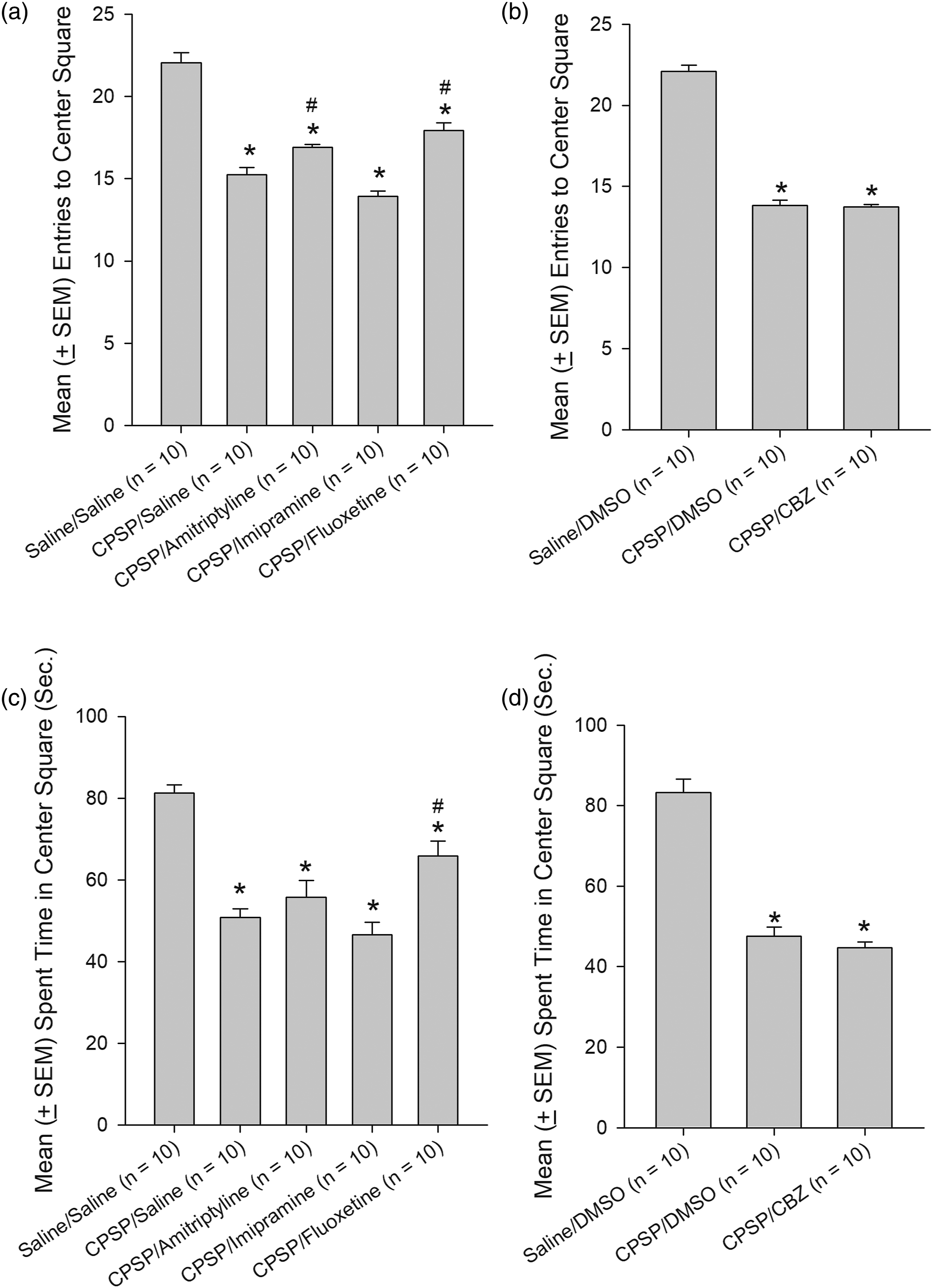
Pharmacological treatments of CPSP symptoms and comorbidities The effects of pharmacological treatments on CPSP symptoms and the comorbidities of anxiety and depression behaviors were tested using multiple behavioral tests. For the thermal pain test in a hot water bath, a 3 × 5 two-way mixed ANOVA was conducted. The results showed that significant differences could be observed in left paw withdrawal following amitriptyline treatment for group [F (2, 27) = 3.96, p < 0.05], day [F (4, 108) = 4.86, p < 0.05], and the group × day interaction [F (8, 108) = 8.66, p < 0.05; Figure 5(a)]; significant differences following imipramine treatment were observed for group [F (2, 27) = 4.42, p < 0.05], day [F (4, 108) = 10.06, p < 0.05], and the group × day interaction [F (8, 108) = 9.94, p < 0.05; Figure 5(b)] for imipramine; significant differences following fluoxetine treatment were observed for group [F (2, 27) = 4.63, p < 0.05], day [F (4, 108) = 20.36, p < 0.05], and the group × day interaction [F (8, 108) = 12.18, p < 0.05; Figure 5(c)]; and significant differences were observed following CBZ treatment for group [F (2, 27) = 53.46, p < 0.05], day [F (4, 108) = 32.17, p < 0.05], and the group × day interaction [F (8, 108) = 8.70, p < 0.05; Figure 5(d)]. In the left paw, the post-hoc with Tukey tests showed that amitriptyline and imipramine significantly increased withdrawal times on Day 28 (p < 0.05). However, fluoxetine and CBZ did not affect withdrawal time on Days 14, 21, and 28 (p > 0.05). Measurement of thermal pain behavior using the hot water bath test following amitriptyline, imipramine, fluoxetine, and CBZ administrations. Mean (±SEM) withdrawal time (sec) of the (a) CPSP/Amitriptyline, (b) CPSP/Imipramine, or (c) CPSP/Fluoxetine groups compared with the Saline/Saline and CPSP/Saline groups on Pre and Days 7, 14, 21, and 28 in the left paw. Mean (±SEM) withdrawal time (sec) of the (e) CPSP/Amitriptyline, (f) CPSP/Imipramine, or (g) CPSP/Fluoxetine groups compared with the Saline/Saline and CPSP/Saline groups on Pre and Days 7, 14, 21, and 28 in the right paw. Mean (±SEM) withdrawal time (sec) of the (d) CPSP/CBZ and (h) CPSP/CBZ groups compared to the Saline/DMSO and CPSP/DMSO groups on Pre and Days 7, 14, 21, and 28 in the right or left paw. Amitriptyline and imipramine were intraperitoneally injected once a day on Day 21 and Day 28, where CBZ and fluoxetine were intraperitoneally injected once a day from Day 14 to Day 28. All drugs were administrated before the pain, open-field, zero-maze, and forced swim tests. No drugs were given on Pre and Day 7. *p < 0.05 indicates significant differences compared with the Saline/Saline or Saline/DMSO group. #p < 0.05 indicates significant differences compared with the CPSP/Saline or CPSP/DMSO group. CPSP: central post-stroke pain induced by the ablation of the ventrobasal complex of the thalamus. Pre: pre-test pain in left or right paw. CBZ: carbamazepine. Arrows represent the injection day. When examining the effects of pharmacological treatments on right paw withdrawal, the results showed significant differences in withdrawal after amitriptyline treatment for group [F (2, 27) = 16.70, p < 0.05], day [F (4, 108) = 20.29, p < 0.05], and the group × day interaction [F (8, 108) = 7.05, p < 0.05; Figure 5(e)]; significant differences were observed following imipramine treatment for group [F (2, 27) = 14.45, p < 0.05], day [F (4, 108) = 35.22, p < 0.05], and the group × day interaction [F (8, 108) = 7.90, p < 0.05; Figure 5(f)]; significant differences were observed following fluoxetine treatment for group [F (2, 27) = 15.52, p < 0.05], day [F (4, 108) = 51.26, p < 0.05], and the group × day interaction [F (8, 108) = 10.98, p < 0.05; Figure 5(g)]; and significant differences were observed following CBZ treatment for group [F (2, 27) = 81.22, p < 0.05], day [F (4, 108) = 36.91, p < 0.05], and the group × day interaction [F (8, 108) = 8.49, p < 0.05; Figure 5(h)]. Amitriptyline and imipramine increase withdrawal time on Day 21 and 28 (p < 0.05). However, fluoxetine and CBZ did not affect withdrawal time on Days 14, 21, and 28 (p > 0.05). The response to mechanical pain was assessed using the von Frey test, with a 3 × 5 two-way mixed ANOVA design, which showed significant differences in left paw response following amitriptyline treatment for group [F (2, 27) = 30.86, p < 0.05], day [F (4, 108) = 23.26, p < 0.05], and the group × day interaction [F (8, 108) = 8.05, p < 0.05; Figure 6(a)]; significant differences were observed following imipramine treatment for group [F (2, 27) = 27.89, p < 0.05], day [F (4, 108) = 29.68, p < 0.05], and the group × day interaction [F (8, 108) = 8.27, p < 0.05; Figure 6(b)]; significant differences were observed following fluoxetine treatment for group [F (2, 27) = 42.99, p < 0.05], day [F (4, 108) = 37.57, p < 0.05], and the group × day interaction [F (8, 108) = 10.18, p < 0.05; Figure 6(c)]; and significant differences were observed following CBZ treatment for group [F (2, 27) = 73.53, p < 0.05], day [F (4, 108) = 83.17, p < 0.05], and the group × day interaction [F (8, 108) = 23.77, p < 0.05; Figure 6(d)]. For left paw response, amitriptyline and imipramine treatment increased weight tolerance on Day 21 and Day 28 (p < 0.05). However, fluoxetine and CBZ did not display any effects in weight tolerance at any days (p > 0.05). Measurement of mechanical pain behavior in the von Frey test following amitriptyline, imipramine, fluoxetine, and CBZ administrations. Mean (±SEM) weight (g) of the (a) CPSP/Amitriptyline, (b) CPSP/Imipramine, or (c) CPSP/Fluoxetine groups compared with the Saline/Saline and CPSP/Saline groups on Pre and Days 7, 14, 21, and 28 in the left paw. Mean (±SEM) weight (g) of the (e) CPSP/Amitriptyline, (f) CPSP/Imipramine, or (g) CPSP/Fluoxetine groups compared with the Saline/Saline and CPSP/Saline groups on Pre and Days 7, 14, 21, and 28 in the right paw. Mean (±SEM) weight (g) of the (d) CPSP/CBZ and (h) CPSP/CBZ groups compared to the Saline/DMSO and CPSP/DMSO groups on Pre and Day 7, 14, 21, and 28 in the right or left paw. Amitriptyline and imipramine were intraperitoneally injected once a day on Day 21 and Day 28, where CBZ and fluoxetine were intraperitoneally injected once a day from Day 14 to Day 28. All drugs were administrated before the pain, open-field, zero-maze, and forced swim tests. No drugs were given on Pre and Day 7. *p < 0.05 indicates significant differences compared with the Saline/Saline or Saline/DMSO group. #p < 0.05 indicates significant differences compared with the CPSP/Saline or CPSP/DMSO group. CPSP: central post-stroke pain induced by the ablation of the ventrobasal complex of the thalamus. Pre: pre-test pain in left or right paw. CBZ: carbamazepine. Arrows represent the injection day. The analysis of the right paw response to the von Frey test indicated significant differences were observed following amitriptyline treatment for group [F (2, 27) = 108.85, p < 0.05], day [F (4, 108) = 39.46, p < 0.05], and the group × day interaction [F (8, 108) = 22.54, p < 0.05; Figure 6(e)]; significant differences were observed following imipramine treatment for group [F (2, 27) = 67.06, p < 0.05], day [F (4, 108) = 39.02, p < 0.05], and the group × day interaction [F (8, 108) = 16.33, p < 0.05; Figure 6(f)]; significant differences were observed following fluoxetine treatment for group [F (2, 27) = 78.36, p < 0.05], day [F (4, 108) = 53.48, p < 0.05], and the group × day interaction [F (8, 108) = 22.24, p < 0.05; Figure 6(g)]; and significant differences were observed following CBZ treatment for group [F (2, 27) = 74.08, p < 0.05], day [F (4, 108) = 80.76, p < 0.05], and the group × day interaction [F (8, 108) = 22.33, p < 0.05; Figure 6(h)]. Amitriptyline, imipramine, and fluoxetine treatment increase weight tolerance on Day 21 and Day 28 (p < 0.05); however, CBZ did not affect weight tolerance over all days. The effects of pharmacological treatments on motor activity and anxiety behavior in the open-field test were assessed using a one-way ANOVA. When comparing among the Saline/Saline, CPSP/Saline, CPSP/Amitriptyline, CPSP/Imipramine, and CPSP/Fluoxetine groups, no significant differences were observed for speed [F (4, 45) = 0.92, p > 0.05; Figure 7(a)], max speed [F (4, 45) = 1.28, p > 0.05; Figure 7(c)], or total distance traveled [F (4, 45) = 0.92, p > 0.05; Figure 7(e)]. Additionally, no significant differences were observed when comparing among the Saline/dimethylsulfoxide (DMSO), CPSP/DMSO, and CPSP/CBZ groups for speed [F (2, 27) = 0.12, p > 0.05; Figure 7(B)], max speed [F (2, 27) = 0.21, p > 0.05; Figure 7(d)], or total distance traveled [F (2, 27) = 0.12, p > 0.05; Figure 7(f)]. Therefore, none of the tested drugs appeared to affect motor activity. Measurements of motor activity in the open-field test following amitriptyline, imipramine, fluoxetine, and CBZ administrations. (a) Mean (±SEM) speed (mm/sec), (c) mean (±SEM) maximum speed (mm/sec), and (e) mean (±SEM) total distance traveled (mm) for the Saline/Saline, CPSP/Saline, CPSP/Amitriptyline, CPSP/Imipramine, and CPSP/Fluoxetine groups. (b) Mean (±SEM) speed (mm/sec), (d) mean (±SEM) maximum speed (mm/sec), and (f) mean (±SEM) total distance traveled (mm) for the Saline/DMSO, CPSP/DMSO, and CPSP/CBZ groups. Amitriptyline and imipramine were intraperitoneally injected once a day on Day 21 and Day 28, where CBZ and fluoxetine were intraperitoneally injected once a day from Day 14 to Day 28. All drugs were administrated before the pain, open-field, zero-maze, and forced swim tests. No drugs were given on Pre and Day 7. All behavioral data were averaged from Days 7, 14 and 21 to Day 28. CPSP: central post-stroke pain induced by the ablation of the ventrobasal complex of the thalamus. CBZ: carbamazepine. When examining the effect of pharmacological treatments on anxiety behavior, as measured by the number of entries and time spent in the center square of the open-field test, one-way ANOVA analysis identified significant differences for the number of entries into the center square [F (4, 45) = 52.50, p < 0.05; Figure 8(a)] and total time spent in the center square [F (4, 45) = 20.24, p < 0.05; Figure 8(c)] when comparing among the Saline/Saline, CPSP/Saline, CPSP/Amitriptyline, CPSP/Imipramine, and CPSP/Fluoxetine groups. Post-hoc Tukey’s HSD tests revealed that amitriptyline and fluoxetine had significant effects on the number of entries into the center square (p < 0.05) and the time spent time in the center square (p < 0.05). One-way ANOVA also indicated significant differences in the number of entries into the center square [F (2, 27) = 244.71, p < 0.05; Figure 8(b)] and the time spent in the center square [F (2, 27) = 77.11, p < 0.05; Figure 8(d)] when comparing among the Saline/DMSO, CPSP/DMSO, and CPSP/CBZ groups. Post-hoc Tukey’s HSD tests showed no significant difference in the number of entries into the center square or the time spent in the center square between the CPSP/CBZ group and the CPSP/DMSO group (p > 0.05). Therefore, amitriptyline and fluoxetine appeared to decrease anxiety behavior, whereas the other tested drugs had no significant effects. Measurements of anxiety behaviors in the open-field test following amitriptyline, imipramine, fluoxetine, and CBZ administrations. (a) Mean (±SEM) entries into the center square and (c) mean (±SEM) time spent in the center square (sec) for the Saline/Saline, CPSP/Saline, CPSP/Amitriptyline, CPSP/Imipramine, and CPSP/Fluoxetine groups. (b) Mean (±SEM) entries into the center square and (d) mean (±SEM) time spent in the center square (sec) for the Saline/DMSO, CPSP/DMSO, and CPSP/CBZ groups. Amitriptyline and imipramine were intraperitoneally injected once a day on Day 21 and Day 28, where CBZ and fluoxetine were intraperitoneally injected once a day from Day 14 to Day 28. All drugs were administrated before the pain, open-field, zero-maze, and forced swim tests. No drugs were given on Pre and Day 7. All behavioral data were averaged from Days 7, 14 and 21 to Day 28. CPSP: central post-stroke pain induced by the ablation of the ventrobasal complex of the thalamus. CBZ: carbamazepine. When testing anxiety behaviors using the zero-maze test, one-way ANOVA analysis showed significant differences in the number of entries into open arm [F (4, 45) = 7.51, p < 0.05; Figure 9(a)] and the time spent in the open arm [F (4, 45) = 13.03 p < 0.05; Figure 9(c)] among the Saline/Saline, CPSP/Saline, CPSP/Amitriptyline, CPSP/Imipramine, and CPSP/Fluoxetine groups. Post-hoc Tukey’s HSD tests revealed that amitriptyline significantly increased entries into the open arm (p < 0.05), and amitriptyline, imipramine, and fluoxetine all significantly increased the time spent in the open arm (p < 0.05) compared with the CPSP/Saline group. One-way ANOVA also identified significant differences in the number of entries into open arm [F (2, 27) = 35.36, p < 0.05; Figure 9(b)] and time in the open arm [F (2, 27) = 13.90, p < 0.05; Figure 9(d)] when comparing among the Saline/DMSO, CPSP/DMSO, and CPSP/CBZ groups. Post-hoc Tukey’s HSD tests showed no significant differences in entries to the open arm or time spent in the open arm between the CPSP/CBZ and CPSP/DMSO groups (p > 0.05). Therefore, amitriptyline, imipramine, and fluoxetine appeared to significantly decrease anxiety behavior, whereas CBZ had no significant effect. Measurements of anxiety behaviors in the zero-maze test following amitriptyline, imipramine, fluoxetine, and CBZ administrations. (a) Mean (±SEM) entries into the open section and (c) mean (±SEM) time spent in the open section (sec) for the Saline/Saline, CPSP/Saline, CPSP/Amitriptyline, CPSP/Imipramine, and CPSP/Fluoxetine groups. (b) Mean (±SEM) entries into the open section and (d) mean (±SEM) time spent in the open section (sec) for the Saline/DMSO, CPSP/DMSO, and CPSP/CBZ groups. Amitriptyline and imipramine were intraperitoneally injected once a day on Day 21 and Day 28, where CBZ and fluoxetine were intraperitoneally injected once a day from Day 14 to Day 28. All drugs were administrated before the pain, open-field, zero-maze, and forced swim tests. No drugs were given on Pre and Day 7. All behavioral data were averaged from Days 7, 14 and 21 to Day 28. *p < 0.05 indicates significant differences compared to the Saline/Saline group. #p < 0.05 indicates significant differences compared to the CPSP/Saline group. CPSP: central post-stroke pain induced by the ablation of the ventrobasal complex of the thalamus. CBZ: carbamazepine. The forced swim test was used to examine the effects of pharmacological treatments on depression symptoms comorbid with CPSP. One-way ANOVA analysis revealed significant differences in floating time [F (4, 45) = 10.72, p < 0.05; Figure 10(a)] and swimming time [F (4, 45) = 7.08, p < 0.05; Figure 10(e)] among the Saline/Saline, CPSP/Saline, CPSP/Amitriptyline, CPSP/Imipramine, and CPSP/Fluoxetine groups. No significant differences in struggling time [F (4, 45) = 2.31, p > 0.05; Figure 10(c)] were observed among the Saline/Saline, CPSP/Saline, CPSP/Amitriptyline, CPSP/Imipramine, and CPSP/Fluoxetine groups. Post-hoc Tukey’s HSD tests indicated that fluoxetine and imipramine but not amitriptyline significantly decreased floating time (p < 0.05) compared with that in the CPSP/Saline group. One-way ANOVA analysis revealed significant differences occurred in floating time [F (2, 27) = 5.91, p < 0.05; Figure 10(b)] and struggling time [F (2, 27) = 13.01, p < 0.05; Figure 10(d)] but not swimming time [F (2, 27) = 1.04, p > 0.05; Figure 10(f)] among the Saline/DMSO, CPSP/DMSO, and CPSP/CBZ groups. Post-hoc Tukey’s HSD tests showed no significant differences in floating time, struggling time, or swimming time between the CPSP/CBZ and CPSP/DMSO groups (p > 0.05). These results suggested that imipramine and fluoxetine significantly decreased depression-like behaviors, but the other pharmacological treatments had no significant effects. Measurements of depression behaviors in the forced swim test following amitriptyline, imipramine, fluoxetine, and CBZ administrations. (a) Mean (±SEM) floating time (sec), (c) mean (±SEM) struggling time (sec), and (e) mean (±SEM) swimming time (sec) for the Saline/Saline, CPSP/Saline, CPSP/Amitriptyline, CPSP/Imipramine, and CPSP/Fluoxetine groups. (b) Mean (±SEM) floating time (sec), (d) mean (±SEM) struggling time (sec), and (f) mean (±SEM) swimming time (sec) for the Saline/DMSO, CPSP/DMSO, and CPSP/CBZ groups. Amitriptyline and imipramine were intraperitoneally injected once a day on Day 21 and Day 28, where CBZ and fluoxetine were intraperitoneally injected once a day from Day 14 to Day 28. All drugs were administrated before the pain, open-field, zero-maze, and forced swim tests. No drugs were given on Pre and Day 7. All behavioral data were averaged from Days 7, 14 and 21 to Day 28. *p < 0.05 indicates significant differences compared with the Saline/Saline group. #p < 0.05 indicates significant differences compared with the CPSP/Saline group. CPSP: central post-stroke pain induced by the ablation of the ventrobasal complex of the thalamus. CBZ: carbamazepine.




Discussion
Summary for pharmacological treatments in CPSP-induced pain and comorbid anxiety and depression.

Note: CPSP compared to the control; amitriptyline, imipramine, fluoxetine, and carbamazepine compared to CPSP; ‐ : non-significant differences; ↓: decreases; ↑: increases.
Central post-stroke pain core and comorbid symptoms in animals and humans
The present data obtained from an animal model of CPSP revealed that the ablation of the VBC induced thermal and mechanical pain, anxiety, and depression symptoms, which are consistent with clinical findings.2, 4,7 Patients with CPSP describe the experience of both spontaneous pain, which can manifest as continuous or intermittent pain (e.g., burning, aching, pricking, squeezing, shooting, lacerating, and throbbing), and evoked pain in response to nociceptive or non-nociceptive stimuli (e.g., movement, touch, and temperature). 2 In addition to the pain symptoms, 18 patients with CPSP often experience motor impairments, 19 cognitive decline,19,20 loss of work, 21 depression,20,21 anxiety, 3 sleep disturbance, 7 poor social interaction, 21 reduced motivation, and interference in thought process, 3 which reduce quality of life, negatively affect rehabilitation processes, 22 and have been associated with an increased suicide rate. 23 However, the present data from an animal model is unable to simulate the full range of comorbid behaviors described in human patients.
In contrast with studies examining primary pain symptoms caused by CPSP, few studies have investigated comorbid symptoms. This study aimed to simulate the comorbid occurrence of anxiety and depression often observed among patients with CPSP. The animal model of CPSP developed in this study can be used to test any interventions for ameliorating CPSP symptoms, in addition to the effects on anxiety and depression symptoms. In addition to specific outcomes associated with CPSP, this novel behavioral model can be used to further explore the neural mechanisms that underly pain, anxiety, and depression symptoms.
The ablation of VBC in this developed animal model of CPSP was found to result in the development of core CPSP symptoms observed in patients, including pain sensitivity and comorbid anxiety and depression. The CPSP animal model can help us to understand the neural mechanisms underlying CPSP and be used to test pharmacological and non-pharmacological interventions, contributing to the clinical understanding of this disease.
Central post-stroke pain’s pharmacological treatments in the animal and human models
In the present study, the results showed that the tricyclic antidepressants amitriptyline and imipramine suppressed thermal pain sensitivity, as assessed by the hot water bath test; mechanical pain sensitivity, as assessed by the von Frey test; anxiety behaviors, as assessed using the open-field test and the zero-maze test; and depression behaviors, as assessed using the forced swim test. The outcomes observed using this CPSP animal model align with the clinical evidence reported in human patients with CPSP, in which amitriptyline was found to ameliorate spontaneous and evoked pain; 13 tricyclic antidepressants have also been reported to have positive outcomes on anxiety, depression, changing mood, and neurotic tendencies in patients with CPSP.2,6
To date, no studies have examined the effects of the SSRI fluoxetine on CPSP symptoms of pain sensitivity, anxiety, or depression, making the present study the first to address these issues. In the present study, fluoxetine decreased mechanical pain, as assessed by the von Frey test; anxiety behaviors, as assessed by the open-field test or zero-maze test; and depression behaviors, as assessed using the forced swim test. These results were consistent with clinical reports that the SSRI fluvoxamine was able to ameliorate symptoms in patients with CPSP. 2 In a pain test measured using the visual analog scale, patients with CPSP patients reported that their average pain value decreased from 7.7 to 6.0 following the administration of fluvoxamine at doses higher than 125 mg daily dose, indicating that fluvoxamine effectively reduced pain symptoms. 2 Therefore, fluoxetine may represent another option for the treatment of CPSP symptoms.
Surprisingly, the present study showed that the anticonvulsant CBZ had no significant effects on thermal pain, as assessed by the hot water bath test; mechanical pain, as assessed by the von Frey test; anxiety behaviors, as assessed by the open-field test or zero-maze test; or depression behaviors, as assessed by the forced swim test. These data are consistent with some clinical data. 24 For example, a randomized controlled trial showed that amitriptyline effectively ameliorated pain and depression symptoms, whereas CBZ had no significant effects compared with placebo. 24 However, a growing body of clinical evidence has supported the use of anticonvulsant drugs as second-line or add-on treatments for the amelioration of pain or depression symptoms in patients with CPSP.6,25 In one study, 14 patients with CPSP were administered CBZ for 4 weeks, with continuously increasing CBZ doses up to 800 mg daily, and five of 14 patients reported pain reduction; however, these changes in pain were not significant compared with the placebo group. 23 Therefore, although CBZ may be considered as an adjuvant therapy to reduce pain, anxiety, and depression symptoms, CBZ should not be considered a first-line drug option for CPSP treatment.
Previous animal models of CPSP have been characterized with motor impairments, which contrasts with the clinical data indicating that patients with CPSPS rarely present with motor impairments or dysfunctions.2,22 Patients with CPSP may present with mild motor dysfunction, including impaired joint positioning or vibrations, and motor impairments are reported less frequently. 2 Therefore, the animal model in the present study, which was able to simulate the pain, anxiety, and depression symptoms associated with CPSP without inducing motor impairment or motor dysfunction, may represent a better CPSP model than prior animal models. However, some clinical studies have indicated that CPSP patients present with minor motor impairments. Thus, whether VBC is involved in the motor deficits associated with CPSP may require further scrutiny in later studies.
Central post-stroke pain pathophysiology and clinical implications
The pathophysiological mechanisms underlying CPSP remain unclear, although dysregulated central disinhibition, central sensitization, or the imbalanced activation of the spinothalamocortical pathway represent likely contributors. 7 Therefore, identifying pharmacological interventions able to act on these pathways represents a potential mechanism for the amelioration of CPSP symptoms. The tricyclic antidepressants amitriptyline and imipramine inhibit serotonin and norepinephrine reuptake mechanisms, increasing serotonin and norepinephrine concentrations in the synaptic cleft, 14 which might block central disinhibition and sensitization, resulting in reduced pain symptoms.
The SSRI fluoxetine is a new and effective pharmacological treatment for patients with major depression disorder, 16 which acts by blocking the serotonin reuptake mechanism to increase the concentration of serotonin in the synaptic cleft, which likely interferes with central disinhibition and sensitization, leading to reduced pain symptoms.
The anticonvulsant CBZ blocks voltage-dependent sodium ion channels, decreasing electrical activity and reducing glutamate release, 15 which might also interfere with central disinhibition and sensitization, reducing pain symptoms.
Tricyclic antidepressants are often considered first-line CPSP treatment options, and many clinical studies in humans have demonstrated that amitriptyline is currently the most effective medication for ameliorating CPSP symptoms. 13 Tricyclic antidepressant drugs activate serotonin and norepinephrine neurons to increase secretion of serotonin and norepinephrine and block reuptake, increasing the neurotransmitter concentration in the synaptic cleft. 14 In clinical studies, tricyclic antidepressants are more effective than SSRI in reducing CPSP symptoms. 2 Therefore, CPSP symptoms likely involve both the serotonin and norepinephrine systems. The limited effects of SSRIs on the serotonin system may not be sufficient to completely combat CPSP symptoms. Anticonvulsants, such as CBZ, which block voltage-dependent sodium channels to decrease neural activity 15 and inhibit central sensitization or central disinhibition, do not appear to be as effective against CPSP symptoms as other types of drugs. CBZ does not target specific neurotransmitter systems, such as the serotonin or norepinephrine systems, which likely explains why CBZ administrations did not demonstrate any significant effects on pain, anxiety, or depression symptoms in the present study.
In conclusion, based on the potential mechanism underlying the pathophysiology of CPSP, pharmacological treatments that target the serotonin and norepinephrine reuptake mechanisms or enhance serotonin and norepinephrine release are likely better approaches for the treatment of CPSP symptoms. The voltage-dependent sodium channel blocker CBZ and the SSRIs fluoxetine might not sufficiently target the critical mechanisms underlying CPSP symptoms. The findings that both SSRIs and tricyclic antidepressants can reduce CPSP symptoms, including pain and depression, the physical pain induced by CPSP may occur through similar mechanisms as the psychological pain and depression induced by adverse emotional events. 26
Regarding the reductions of pharmacological treatments in CPSP symptoms, it is crucial to how and why the pharmacological treatments ameliorate the pathophysiological mechanisms. This emerged issue remains to be investigated in further studies.
Exploring the use of other drugs to treat CPSP and associated symptoms
The present study explored the amelioration effects of antidepressants and SSRIs for CPSP-induced pain and comorbid anxiety and depression symptoms. The underlying mechanisms associated with these medications involve the enhancement of serotonin and norepinephrine release, resulting in reduced CPSP-induced pain and anxiety and depression symptoms. Other drugs may also be able to ameliorate the pain and comorbid anxiety and depression associated with CPSP. A previous review paper reported that the anesthetics lidocaine and mexiletine reduce CPSP-induced pain for a period of time. 2 NMDA receptor antagonists, such as ketamine, also reduce CPSP-induced pain symptoms during the refractory phase; however, ketamine’s analgesic effects are typically short-lived. 2 Morphine has been tested for analgesic effects in CPSP patients but was only able to effectively reduce brush-induced allodynia without ameliorating other CPSP-induced pain symptoms. Moreover, chronic morphine treatments can induce multiple side effects, including drug addiction . 2 In summary, although other drugs have been explored for the treatment of CPSP-induced pain and associated symptoms, they are often associated with drawbacks and limitations. However, these drugs remain to be investigated in the present animal model of CPSP, and exploring the underlying mechanism through which different drugs affect CPSP-induced pain and comorbid anxiety and depression symptoms may lead to the development of novel treatments.
Comparing the present animal model with other existing models
Previously examined animal models of CPSP have required various manipulations to imitate the symptoms observed in CPSP patients. 27 For example, an excitotoxic lesion model involved the administration of kainate into the right thalamus, which resulted in thermal and mechanical pain symptoms but without any other CPSP symptoms.28,29 The ischemic animal model employed cerebral artery occlusion or carotid occlusion to induce CPSP-like responses to mechanical pain and electrical pain tests; however, this CPSP model was not found to be consistent with the surface validity, construct validity, or predictive validity of the hemorrhagic stroke model.30–32 CPSP models have been generated using the GABAa antagonist picrotoxin 33 or electric lesions 34 to ablate the right thalamus to imitate the CPSP symptoms; however, these animal models do not consistently induce pain symptoms and, therefore, not present sufficient surface validity for use as a CPSP model. A powerful and consistent animal model of CPSP was developed through the administration of collagenase to the right thalamus, resulting in thalamic ablation and resulting in thermal and mechanical pain symptoms 12 and consistently recapitulating the motor symptoms observed with CPSP pain. 35 Therefore, animals featuring collagenase lesions in the thalamus provide suitable surface validity, construct validity, and predictive validity for use as CPSP animal models.
In the present study, we used the collagenase-mediated ablation of the VBS to generate an animal model of CPSP, 11 which presented with both CPSP-associated pain symptoms and comorbid anxiety and depression symptoms. The behavioral testing that demonstrated the existence of comorbid anxiety and depression symptoms in this CPSP model are novel, indicating that this model is the first model able to fully imitate all of the known symptoms that present in CPSP patients.
Emerging issues and limitations
The present findings revealed discrepancies among the elevated plus-maze task, open-field task, and zero-maze task for the detection of anxiety behaviors. In Experiment 1, the CPSP group showed a decrease in the number of entries into the central square of the open-field test compared with the Sham group, indicating enhanced anxiety behavior in rats with CPSP. However, the elevated plus-maze task did not reveal significant differences in the numbers of entries or time spent in the open arm between the Sham and CPSP groups. The open-field task is known to be more sensitive than the elevated plus-maze task for testing anxiety behaviors, which may explain these discrepancies.
In Experiment 2, concerning the assessments of antidepressants such as amitriptyline and imipramine, SSRIs fluoxetine, and an anticonvulsant drug CBZ in CPSP-caused anxiety behaviors, the open-field task has the similar results as those of the zero-maze task in anxiolytic effects that amitriptyline, imipramine, and fluoxetine can effectively ameliorate CPSP-induced comorbid anxiety responses. Thus, the results indicated that the open-field task was sensitive to test anxiety behavior as similarly as the zero-maze task.
On the other hand, the elevated plus-maze is known to be less sensitive to the detection of anxiety behaviors than the zero-maze task, and rats have displayed the ability to remain on the middle platform of the elevated plus-maze for long periods of time. By contrast, the design of the zero-maze task does not include a middle platform, forcing the rats to decide between remaining in an open or closed arm. In summary, although the open-field test and the zero-maze tasks are considered to have similar sensitivity for measuring anxiety behaviors, the elevated plus-maze is considered to be less sensitive, which may account for the observed behavioral differences among these tasks in the current study.
The present study found that CBZ administrations did not reduce CPSP-induced thermal and mechanical pain or comorbid anxiety and depression behaviors. However, only one dose of CBZ was used in the present study, limiting our conclusion to the ineffective outcome of CBZ delivered at the teste dose. The crucial issue of whether anticonvulsants can effectively reduce CPSP-induced pain and comorbid anxiety and depression symptoms requires further examination in future studies using various CBZ doses and other anticonvulsants.
Conclusion
The ablation of the VBC in a rat model induced thermal and mechanical pain sensitivity and anxiety and depression symptoms similar to those reported by patients with CPSP. In the animal model, the tricyclic antidepressants amitriptyline and imipramine effectively ameliorated CPSP-induced thermal and mechanical pain sensitivity, anxiety, and depression symptoms. The SSRI fluoxetine also reduced mechanical pain sensitivity, anxiety, and depression symptoms induced by CPSP. However, the anticonvulsant CBZ had no effects on any of the observed CPSP symptoms, which does not align with current clinical findings. This discrepancy should be explored further in future studies. The data derived from this new animal model of CPSP might contribute to the development of approaches to the treatment of CPSP symptoms.
Methods and Materials
Animals
This study consisted of two experiments: Experiment 1 used 20 rats, and Experiment 2 used 80 rats. All 100 male Sprague Dawley rats (220–330 g) were purchased from BioLasco Taiwan Co., Ltd. All rats were allowed to adapt to the environment of the colony room, which was maintained at 22 ± 2°C, for 7 days. The rats were group-housed in pairs in a plastic home cage (47 cm length × 26 cm width × 21 cm height) containing hardwood bedding in the colony room, maintained on a 12-h/12-h light/dark cycle (lights on 6:00 a.m.–6:00 p.m.). Water and food were available ad libitum. All experiments were conducted in accordance with the Animal Scientific Procedures Act of 1986, and the experiments received approval from the Fo Guang University Institutional Animal Care and Use Committee. Experiments were designed in an effort to minimize the number of animals used, and efforts were made to reduce animal suffering.
Experimental surgery
Rats underwent anesthesia and surgery before being subjected to various behavioral tests. 20 minutes before anesthesia application, the rat was intraperitoneally (i.p.) injected with gentamicin (6 mg) and atropine sulfate (0.1 mg). All rats were anesthetized with sodium pentobarbital (50 mg/kg, i.p.). In Experiment 1, the rats were assigned to a sham group (n = 10) and a CPSP group (n = 10). In Experiment 2, the rats were assigned to Saline (n = 40) or CPSP groups (n = 60). The Saline group was separated into the Saline/Saline and Saline/DMSO groups (n = 10 per group). The CPSP groups was divided into the CPSP/Saline, CPSP/DMSO, CPSP/amitriptyline, CPSP/imipramine, CPSP/Fluoxetine, and CPSP/CBZ groups (n = 10, per group). All rats in the CPSP group received a 0.5 μl volume of 0.125 U collagenase type IV (Sigma), which was injected into the right VBC of the thalamus (anterior/posterior: −3.84 mm from bregma, lateral: 2.8 mm from the midline, and ventral: 6.0 mm from the skull surface). 36 The sham or saline group was injected with 0.9% normal saline following the same procedure used for the CPSP group. The injection rate was restricted to 0.25 μl/min. The needle was left in position for an additional 10 min. After surgery, all rats were allowed to recover for 7 days, with free access to food and water.
Apparatus
Hot water bath
The hot water bath apparatus measures thermal analgesia measured by the timing of the paw withdrawal response. This apparatus consists of an empty rectangular tank (30 cm long × 23 cm wide × 15 cm high) that can be filled with water and a temperature control panel. Water was maintained at 48 ± 2°C during the experiment.
von Frey test
Animals are placed on an elevated mesh platform for 30 min. Filaments are applied to the paw with gradually ascending force gradually to determine the minimal force necessary to induce a paw withdrawal response. The minimal force recorded across three trials was averaged to determine the minimum threshold. The von Frey test was performed using consecutive trials, with a 5 min interval between trials.
Open-field test
The open-field test is a square plastic box, sized 86 cm long × 86 cm wide × 50 cm high. The center of the box has a square. In the open-field test, motor activity (e.g., total distance traveled, maximum speed, and average speed) and anxiety behaviors (e.g., entries into the center square and the time spent in the center square) were measured by video tracking software (Video Tracking Record System Version 1.17, SINGA Technology Corporation, Taipei, Taiwan). Increased entries into the center square and the time spent in the center square indicate reduced anxiety behavior.
Zero-maze test
The zero-maze apparatus is comprised of black plastic in a circular track, 10.5 cm wide, 100 cm in diameter, and elevated 49 cm from the ground. This maze is divided into four quadrants of equal lengths. Two opposing quadrants consist of open arms without any walls. The other two opposing quadrants consist of closed arms, comprised of black plastic walls 29.5 cm in height. Animals were measured for 3 min in each trial under constant lighting conditions (i.e., light intensity = 270 lux). The animal is placed in an open quadrant at the start of the trial, and the number of entries into the open arms and the time spent in the open arms were measured to analyze anxiety behavior. More time spent in open arms or more entries into the open arms are indicators of reduced anxiety behavior.
Elevated plus-maze test
The elevated plus-maze apparatus consists of four arms (two open arms without walls and two enclosed arms featuring 30-cm-high walls) that were 50 cm long and 10 cm wide. The open and closed arms are made of dark plastic materials and are perpendicular. The halfway point of the intersection is 10 cm2, and the apparatus is raised 50 cm from the floor using four plastic sticks.37,38 At the beginning of each trial, the animals are placed at one end of an open arm. The latency time to reach the halfway point was recorded. A larger latency time indicates increased avoidance and stronger anxiety. The number of entries into the open arms is measured for 3 min. Fewer entries into the open arms indicate increased anxiety. An entry is defined as placing at least two paws into the open arm. 39
Forced swim test
The forced swim test apparatus is comprised of a plastic cylinder (33 cm diameter × 40 cm high) containing water (25 ± 1°C) at a depth of 25 cm. Time spent floating, swimming, and struggling are measured. Floating was defined as immobility, with the exception of maintaining the head above the water. Swimming was defined as any forward motion through the water with the forepaws maintained on the water surface. Struggling was defined as an upright position in the water with the forepaws breaking the water surface. A longer time spent floating and a shorter time spent swimming or struggling indicate stronger depression. 40
Experimental procedure
All rats were allowed to adapt to the environment of the colony room for 7 days, after which the rats were subjected to baseline pain tests, including the hot water bath and von Frey tests. Surgical procedures were then performed to ablate the VBC and generate CPSP model rats, after which all rats were allowed to recover for 7 days in their home cages. During Experiment 1, all rats were subjected to the pain test, the open-field test for 15 min, the elevated plus-maze for 3 min, and the forced swim test for 5 min; each test was administered once per week pre-test (i.e., prior to surgery) and on Days 7, 14, and 28. During Experiment 2, the behavioral procedures were similar to those used in Experiment 1. However, the elevated plus-maze task was replaced with the zero-maze task to test anxiety behaviors. On Day 7, all rats were subjected to the pain test, open-field test, zero-maze test, and forced swim test without any drug administration. Starting on Day 14, the rats were i.p. injected with saline or specific drugs (e.g., amitriptyline, imipramine, fluoxetine, or CBZ), and behavioral tests were repeated on Days 14, 21, and 28. Amitriptyline and imipramine were administered once per day on Days 21 and 28, whereas CBZ and fluoxetine were injected daily, once per day, from Days 14–28. The procedures used for antidepressant administrations (e.g., amitriptyline and imipramine) were performed according to those described in a previous study; acute amitriptyline and imipramine treatments were administered 60 min before the behavioral tests were performed in each session. 41 Chronic fluoxetine treatments were administered as described by previous studies; fluoxetine was injected once per day for 14 days (from Days 14–28) prior to performing behavioral tests.42,43 Chronic CBZ treatments were performed as described by a previous study; CBZ was injected once per day for 14 days (from Days 14–28) 60 min before performing behavioral tests. 44
Drugs
Amitriptyline hydrochloride and imipramine hydrochloride (tricyclic antidepressants), fluoxetine hydrochloride (SSRI), and CBZ (anticonvulsant) were purchased from Sigma (St Louis, MO, USA). All chemical compounds were administered, i.p. The injection volume of all drugs was 1 mL/kg. Collagenase type IV was purchased from Sigma and prepared with TES buffer. The dose was 0.125 U, and the injection volume was 0.5 μl.11,12 TES buffer was prepared in 0.36 m
Statistical analysis
Two-way (groups vs. sessions) mixed ANOVA was conducted to analyze the mean withdrawal time and the mean force applied during pain tests in the sham and CPSP groups (in Experiment 1) or in the CPSP/Amitriptyline, CPSP/Imipramine, or CPSP/Fluoxetine groups compared to the Saline/Saline and Saline/CPSP groups in the CPSP/CBZ compared to the Saline/DMSO and CPSP/DMSO groups (in Experiment 2). For Experiment 1, mean measurements were obtained for the Sham and CPSP groups on Days 7, 14, 21, and 28 for locomotor activities (i.e., total distance traveled, maximum speed, and average speed) and anxiety behaviors (i.e., entries into the center square and time spent in the center square) in the open-field test, depression (i.e., floating time, struggling time, and swimming time) behaviors in the forced swim test, and anxiety behaviors (i.e., latency time, entries into the open arm, time spent in the open arm, entries into the closed arm, and time spent in the closed arm) in the elevated plus-maze. For Experiment 2, mean measurements were obtained on Days 7, 14, 21, and 28 for the Saline/Saline, CPSP/Saline, CPSP/Amitriptyline, CPSP/Imipramine, or CPSP/Fluoxetine groups for locomotor activities (i.e., total distance traveled, maximum speed, and average speed) and anxiety behaviors (i.e., entries into the center square and time spent in the center square) in the open-field test, depression (i.e., floating time, struggling time, and swimming time) behaviors in the forced swim test, and anxiety behaviors (i.e., entries into the open arm, time spent in the open arm, entries into the closed arm, and time spent in the closed arm) in the zero-maze test. One-way ANOVA was conducted to analyze these behavioral data. When appropriate, Tukey’s HSD post-hoc test was performed. Values of p < 0.05 were considered significant.
Footnotes
Acknowledgments
We also thank the Institute of Biomedical Sciences of Academia Sinica in Taiwan.
Authors’ contributions
BCS designed the study, analyzed data, and prepared the manuscript.ABHH and YHY performed behavioral experiments and analyzed data.ACWH designed the study, analyzed data, and prepared the manuscript.All authors read and approved the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The present study was supported by funding granted from the Ministry of Science and Technology of Taiwan to Dr. Bai-Chuang Shyu (MOST-109-2320-B-001-010) and Dr Andrew Chih Wei Huang (MOST 110-2410-H-431-004).
