Abstract
Mirogabalin is a novel α2δ ligand approved in Japan for the treatment of peripheral neuropathic pain. However, the sites of action of α2δ ligands to produce analgesic effects on inflammatory pain remain unclear. In this study, we investigated the analgesic effect and site of action of mirogabalin using the rat formalin test, an acute inflammatory pain model. Mirogabalin was administered orally, intrathecally, and intracerebroventricularly. Open field tests were performed to evaluate the effect of oral-, intrathecally, and intracerebroventricularly administered mirogabalin on locomotor activity and orientation ability. Oral mirogabalin produced an analgesic effect when the formalin test was performed 4 h, but not 1 or 2 h, after oral administration. Intrathecal, but not intracerebroventricular, administration of mirogabalin produced analgesic effects when mirogabalin was administered 10 min before formalin injection. These analgesic effects were not antagonized by idazoxan, an α2 adrenergic antagonist; WAY100135, a 5-HT1A antagonist; or naloxone, an opioid receptor antagonist. Mirogabalin attenuated moving distances 1 and 2 h after oral administration and 10 min after intracerebroventricular administration, but not 10 min after intrathecal administration. In the oral administration group, the time course of the analgesic effect was different from that of moving distance. In the intracerebroventricular group, mirogabalin attenuated moving distances but did not produce an analgesic effect. In the intrathecal group, mirogabalin produced an analgesic effect but did not affect moving distances. These findings suggest that the analgesic effect of mirogabalin on the rat formalin test is mediated by spinal action and not by the activation of α2, 5-HT1A, or opioid receptors, and that the inhibitory effect of mirogabalin on moving distances is mediated by the supraspinal brain.
Although guidelines for the pharmacological treatment of neuropathic pain lists α2δ ligands, which act on the α2δ subunit of voltage-dependent calcium channels, as first-line drugs, 1 the number needed to treat (NNT) for pregabalin, an α2δ ligand, was reported to be 7.7 (95% CI: 6.5–9.4). 2 Moreover, α2δ ligands have several adverse effects such as drowsiness, dizziness, peripheral edema, and visual blurring. 3 The number needed to harm (NNH) for pregabalin was reported to be 13.9 (95% CI: 11.6–17.4), 2 a difference of only 6.2 from the NNT. These findings clearly indicate that more effective and safer α2δ ligand alternatives to pregabalin for the treatment of neuropathic pain are needed.
Recently, a new α2δ ligand, mirogabalin, has been developed in Japan. 4 The calcium channel α2δ subunit has four subtypes: α2δ-1, -2, -3, and -4. 5 The α2δ-1 subunit has been shown to be a main target of α2δ ligands in the production of analgesic effects, and the α2δ-2 subunit contributes to central nervous system (CNS) side effects.6,7 Neither pregabalin nor mirogabalin shows subtype selectivity for α2δ-1 or α2δ-2, and gabapentin shows no binding affinity for α2δ-3 or α2δ-4.8,9,10 Mirogabalin has a slower dissociation rate for α2δ-1 than α2δ-2, while pregabalin shows the same dissociation rate for both. 4 This suggests that mirogabalin may have the same analgesic effect as pregabalin with less harmful side effects. In clinical trials, mirogabalin has been reported to be effective in patients with diabetic peripheral neuropathic pain and postherpetic neuralgia, but not fibromyalgia.11,12,13 These findings suggest that mirogabalin is an effective drug for the treatment of neuropathic pain conditions.
The sites of action for α2δ ligands to produce their analgesic effects are still unclear.14,15 One of the putative sites of action is the spinal cord since α2δ ligands decrease neurotransmitter release and inhibit the development of spinal sensitization. 15 In the supraspinal region, the locus coeruleus (LC) has been presumed to be a site of action since α2δ ligands increase activity of coerulo-spinal neurons and induce spinal noradrenalin release.16,17
Although the analgesic effects of α2δ ligands have been mainly investigated for neuropathic pain conditions, α2δ ligands have also been reported to be effective for inflammatory pain. 15 In this study, we investigated the primary site of action of α2δ ligand analgesia on inflammatory pain using mirogabalin in the rat formalin test, an acute inflammatory pain model. We also compared the time course of analgesic effects with the effect on locomotor activity and orientation ability to determine the site of action of mirogabalin.
Methods
Animal Care
This study was performed according to a protocol approved by the Institutional Animal Care Committee of Kumamoto University, Kumamoto, Japan. Male Sprague-Dawley rats weighing 250–300 g (Japan SLC, Shizuoka, Japan) were examined for the effect of mirogabalin on their responses to the formalin and open field tests. Animals were housed in groups of two, maintained on a 12-h dark-light cycle, and permitted food and water ad libitum. Animals were handled on arrival, housed for at least 3 days before testing, and euthanized immediately after the behavioral study.
Drugs and Administration
Mirogabalin besylate (mirogabalin) was obtained from Daiichi Sankyo Co., Ltd. (Tokyo, Japan). WAY100135, a 5-HT1A receptor antagonist; idazoxan, an α2 adrenergic receptor antagonist; and naloxone, an opioid receptor antagonist were purchased from Sigma-Aldrich (St Louis, MO). The dose levels of the test compounds are expressed as free form.
For peroral (PO) administration, a stainless steel tube was inserted through the esophagus to the stomach of restrained individuals. Mirogabalin was dissolved in 2.5 mL distilled water for PO administration. For intrathecal (IT) administration, the drugs were dissolved in 10 μL saline and injected into the L5-6 intervertebral space under brief isoflurane anesthetic using a 30-gauge needle. Successful entry into the intrathecal space was evidenced by a tail flick response upon the insertion of the needle. For intracerebroventricular (ICV) injection, cannulae were implanted into the right lateral ventricle under isoflurane anesthesia. Briefly, thin-wall stainless steel guide cannulae (24-gauge, 0.64 mm outer diameter, 15 mm long) were stereotaxically placed through a burr hole (0.5 mm caudal to coronal suture and 1 mm lateral to sagittal suture; 3 mm deep to the dura) and affixed to the skull with stainless steel screws and cranioplastic cement. The ICV-administered drugs were delivered in a total volume of 5 μL saline. For mirogabalin, 150 μg was the highest dose administrable by ICV injection because of the limitation of solubility. For the intraperitoneal (IP) injection of naloxone, 1 mg/kg was dissolved in 1 mL saline.
Formalin Test
In the formalin test, 50 μL of 5% formalin was injected subcutaneously into the dorsal surface of the hind paw with a 26-gauge needle under brief isoflurane anesthesia. Within 1 min of formalin injection, spontaneous flinching of the injected paw was observed. Flinching was readily discriminated as a rapid and brief withdrawal or flexion of the injected paw. This pain-related behavior was quantified by counting the number of flinches for 1-min periods at 5-min intervals from 0 to 60 min after injection. Two phases of spontaneous flinching behavior were observed: an initial acute phase (phase 1: during the first 6 min after formalin injection) and a prolonged tonic phase (phase 2: beginning about 10 min after formalin injection). The phase 1 response is mediated by direct stimulation of nociceptors by formalin, and the phase 2 response is mediated by the inflammatory response induced by formalin. 18
Experimental Protocol
Formalin test
To determine the most effective timing of mirogabalin PO administration, mirogabalin was administered 1, 2, or 4 h before formalin injection. To obtain control data, the vehicle (distilled water) was administered PO. After determination of the appropriate timing, the dose-response study was conducted.
In the IT and ICV injection studies, mirogabalin was administered 10 min before formalin injection. To obtain control data, the vehicle (saline) was injected IT or ICV.
To examine whether the analgesic effects of PO- (4-h study) or IT-administered mirogabalin were mediated by the activation of spinal 5-HT1A, spinal α2 adrenergic, or μ-opioid receptors, the inhibitors WAY100135 (46 μg/10 μL, IT), idazoxan (30 μg/10 μL, IT), or naloxone (1 mg/kg, IP) were administered 10 min before formalin injection in the PO study and mirogabalin IT injection in the IT study. The doses of the antagonists were determined according to our previous report. 19
Open field test
To evaluate the effect of mirogabalin on locomotor activity and orientation ability, a computerized open field video tracking system (ANY-maze behavioral tracking software; Stoelting Co., IL) was used to record moving distance (m) and rest time (sec). The rats were placed in a round field 750 mm in diameter, which was enclosed by a wall 400 mm in height. Open field tests were started 1, 2, or 4 h after PO mirogabalin or vehicle administration and 10 min after ICV/IT mirogabalin or vehicle injection. Once the rats were placed in the open field, data were obtained for 1 h.
Statistical Analysis
Formalin test
The time-response data are presented as the mean numbers of flinches ± SEM per min for the periods 1–2 min and 5–6 min after formalin treatment and then for 1-min periods at 5-min intervals for 60 min. The data from phase 1 (0–6 min) and phase 2 (10–60 min) observations were analyzed separately. In each case, the cumulative instances of formalin-evoked flinches during phase 1 and phase 2 were calculated for each rat. For the mirogabalin dose-response study, one-way ANOVA was used with Tukey’s post hoc test, while the Student’s t-test was used for the antagonist study. At each time point of the time-response graph, the Student’s t-test was used to compare the instances of flinches in the mirogabalin group with that in the vehicle group during mirogabalin-effective phases.
Open field test
Moving distance and rest time data were analyzed using two-way ANOVA with Tukey’s post hoc test. At each time point of the time-response graph, the Student’s t-test was used to compare the moving distances and resting times between the mirogabalin-effective groups with vehicle-treated groups.Where appropriate, results are expressed as means ± SEM. Critical values that reached p < 0.05 were considered significant. Statistical analysis was performed using SigmaPlot 14.0 (Systat Software, Inc, CA).
Results
The subcutaneous injection of formalin resulted in a highly reliable biphasic display of flinching of the injected paw, and this behavior was comparable to a previous report. 18
Formalin Test
PO administration study
When 10 mg/kg mirogabalin was administered orally 1 h before paw formalin injection, mirogabalin produced no analgesic effect (phase 1: p = 0.926; phase 2: p = 0.196). However, when administered 2 h before formalin injection, an analgesic effect was seen on phase 1 (p = 0.0132), but not phase 2, responses (p = 0.622), and a significant analgesic effect was observed when administered 4 h before formalin injection (phase 1: p = 0.00,253; phase 2: p = 0.000,327; Figure 1), which was dose-dependent between 1 and 10 mg/kg (phase 1: p = 0.009; phase 2: p < 0.001; Figure 2). In the 4 h study, 10 mg/kg mirogabalin significantly attenuated the number of flinching behaviors at 1 (p = 0.0103), 10 (p = 0.0481), 20 (p = 0.0115), 30 (p = 0.00,187), and 40 min (p = 0.00,735) after the formalin injection as compared with vehicle-treated rats. In the 2-h study, 10 mg/kg mirogabalin significantly attenuated the number of flinching behaviors at 1 min after the formalin injection (p = 0.0277). Effects of 10 mg/kg oral mirogabalin or vehicle on the rat formalin test. Mirogabalin or vehicle was administered 1, 2, or 4 h before formalin injection. Each group contained five rats. Each point represents the mean ± SEM. Abscissa: time after formalin injection; Ordinate: number of flinches per min. *: p < 0.05 as compared with vehicle-treated rats; **: p < 0.01 as compared with vehicle-treated rats. Dose-response effect of oral mirogabalin administered 4 h before formalin injection on phase 1 and phase 2 responses. Each point represents the mean ± SEM of five rats. Abscissa: mirogabalin dose (mg/kg); Ordinate: sum of flinches per min. *: p < 0.05 compared with vehicle-treated rats. **: p < 0.001 compared with vehicle-treated rats.

In the antagonist study, neither IT WAY100135 (phase 1: p = 0.587; phase 2: p = 0.327), IT idazoxan (phase 1: p = 0.769; phase 2: p = 0.517), nor IP naloxone (phase 1: p = 0.0672; phase 2: p = 0.319) affected the analgesic effect of PO mirogabalin (Figure 3). Effects of IT WAY100165, IT idazoxan, and IP naloxone on the analgesic effect of 10 mg/kg oral mirogabalin in the rat formalin test. As a comparison, water was administered PO. WAY100165, idazoxan, and naloxone were administered 10 min before formalin injection. Mirogabalin or water was administered 4 h before formalin injection. Each group contained five rats. Each point represents the mean ± SEM. Abscissa: time after formalin injection; Ordinate: number of flinches per min.
ICV study
ICV injection of mirogabalin produced no analgesic effect compared with saline-treated rats at doses between 5 and 150 μg (phase 1: p = 0.250; phase 2: p = 0.746; Figure 4). Effect of ICV mirogabalin on the formalin test. Mirogabalin was administered 10 min before formalin injection. ICV mirogabalin had no effect on the formalin test. A: Time course of 150 μg ICV mirogabalin in the rat formalin test. Abscissa: time after formalin injection; Ordinate: number of flinches per min. Each group contained five rats. Each point represents the mean ± SEM. B: Dose-response effect of ICV mirogabalin on phase 1 and phase 2 responses. Each point represents the mean ± SEM of five rats. Abscissa: mirogabalin dose (μg); Ordinate: sum of flinches per min.
IT study
Mirogabalin injected IT 10 min before paw formalin injection produced a significant dose-dependent analgesic effect on phase 2 (p = 0.009), but not phase 1 (p = 0.129), responses at doses between 0.5 and 5 μg (Figure 5). In the 5 μg mirogabalin study, mirogabalin significantly attenuated the number of flinching behaviors at 20 (p = 0.0233), 35 (p = 0.00,559), and 40 min (p = 0.0344) after the formalin injection as compared with vehicle-treated rats. This indicated that the onset of the analgesic effect occurred 30 min after mirogabalin injection. Effect of IT mirogabalin on the formalin test. Mirogabalin was administered 10 min before formalin injection. IT mirogabalin produced a significant analgesic effect in the formalin test. A: Time course of 5 μg IT mirogabalin in the rat formalin test. Abscissa: time after formalin injection; Ordinate: number of flinches per min. Each group contained five rats. Each point represents the mean ± SEM. B: Dose-response effect of IT mirogabalin on phase 1 and phase 2 responses. Each point represents the mean ± SEM of five rats. Abscissa: mirogabalin dose (μg); Ordinate: sum of flinches per min. *: p < 0.05 compared with vehicle-treated rats.
In the antagonist study, neither IT WAY100135 (p = 0.110), IT idazoxan (p = 0.435), nor IP naloxone (p = 0.272) had an antagonistic effect on the phase 2 analgesic effect of IT mirogabalin (Figure 6). Effects of IT WAY100165, IT idazoxan, IP naloxone on the analgesic effect of 5 μg IT mirogabalin in the rat formalin test. As a comparison, saline was administered IT. WAY100165, idazoxan, and naloxone were administered 10 min before the mirogabalin IT injection. Mirogabalin or saline was administered 10 min before formalin injection. Each group contained five rats. Each point represents the mean ± SEM. Abscissa: time after formalin injection; Ordinate: number of flinches per min.
Open Field Test
PO study
When 10 mg/kg mirogabalin was administered PO 1 or 2 h, but not 4 h, before the open field test, moving distances in the mirogabalin-treated rats were significantly reduced compared with vehicle-treated rats (1 h: p = 0.020; 2 h: p = 0.0020; 4 h: p = 0.127; Figure 7). Mirogabalin at 10 mg/kg administered PO 1, 2, or 4 h before the open field test significantly increased resting times compared with vehicle-treated rats (1 h: p = 0.011; 2 h: p = 0.002; 4 h: p = 0.048; Figure 7). A significant effect on moving distances was observed at 0–10 min for the 2 h group (p = 0.0263), and a significant effect on resting times was observed at 0–10 min for the 2 (p = 0.0256) and 4 h (p = 0.0347) groups (Figure 7). Effects of 10 mg/kg oral mirogabalin or vehicle administration on the open field test. The open field test was started 1, 2, or 4 h after oral mirogabalin or vehicle administration. Left and right panels represent the time course of the effect of mirogabalin on moving distances (m) and resting times (sec), respectively. Each group contained five rats. Each point represents the mean ± SEM. Abscissa: time (min) after the start of open field test; Ordinate: left panel, moving distance (m); right panel, resting time (sec). *: p < 0.05 compared with vehicle-treated rats.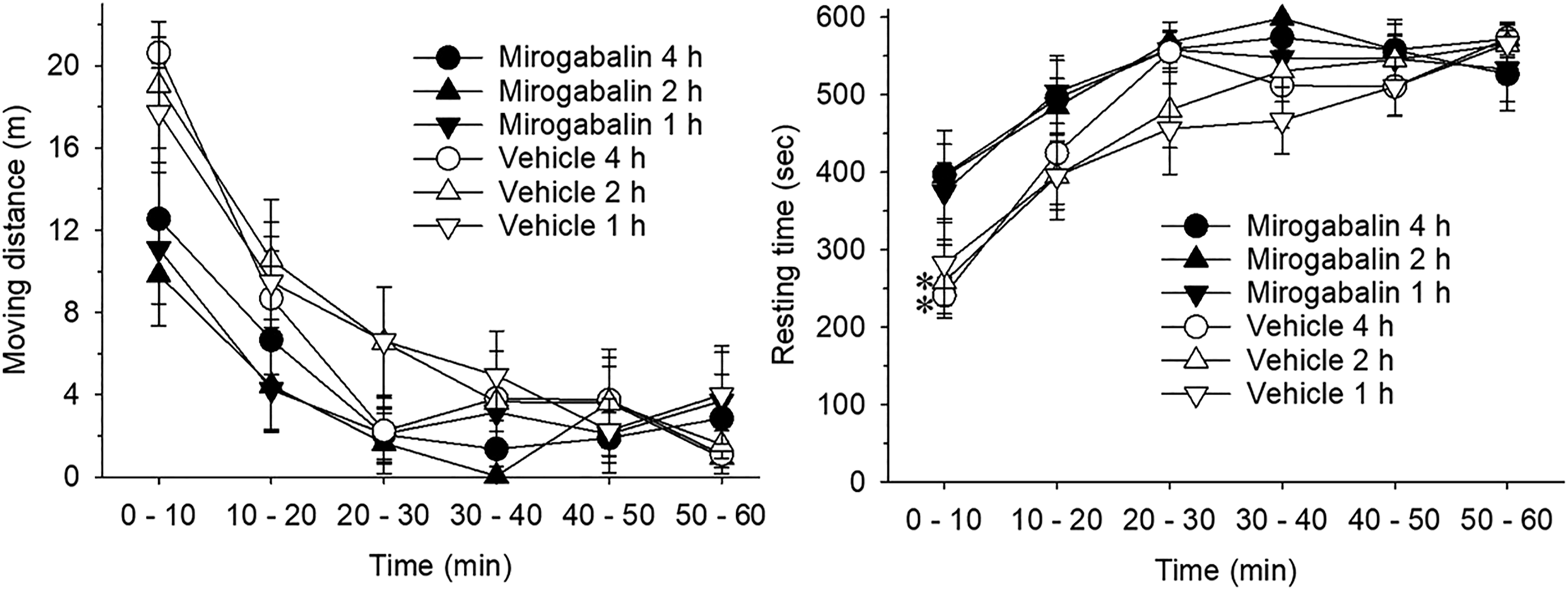
ICV study
When 150 μg mirogabalin was administered ICV 10 min before the open field test, mirogabalin significantly decreased moving distances (p = 0.035; Figure 8) but had no effect on resting times (p = 0.078; Figure 8) compared with vehicle-treated rats. A significant effect on moving distances was observed at 50–60 min (p = 0.0248). Effects of 150 μg ICV (upper panel) or 5 μg IT (lower panel) mirogabalin on the open field test. The open field test was started 10 min after ICV or IT mirogabalin. Vehicle was injected as a control. Left and right panels represent the time course of the effect of mirogabalin on moving distance (m) and resting time (sec), respectively. Each group contained 10 rats. Each point represents the mean ± SEM. Abscissa: time (min) after the start of open field test; Ordinate: left panel, moving distance (m); right panel, resting time (sec). *: p < 0.05 compared with vehicle-treated rats.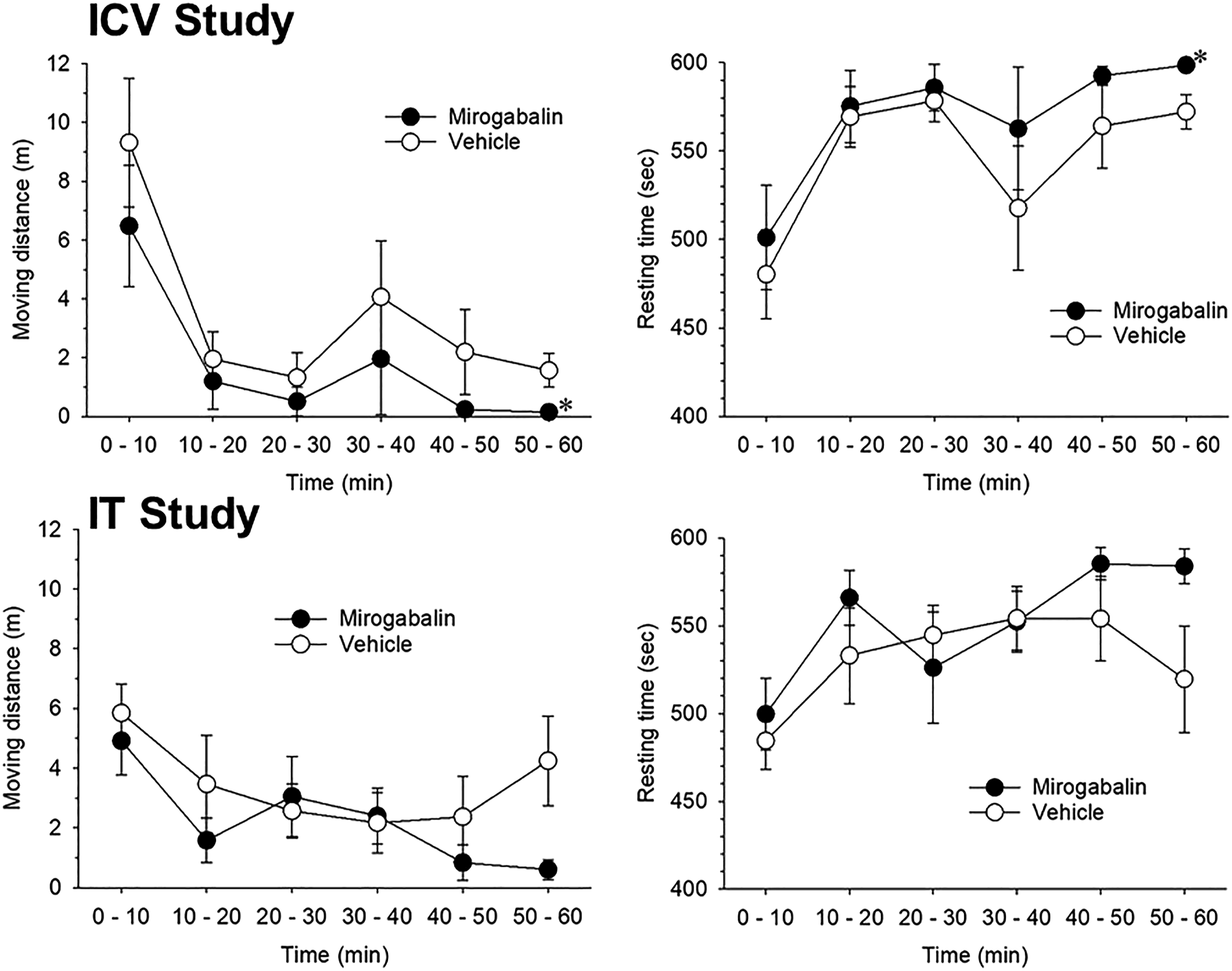
IT study
The IT administration of 5 μg mirogabalin 10 min before the open field test did not affect moving distances (p = 0.059) or resting times (p = 0.092; Figure 8) compared with vehicle-treated rats.
DISCUSSION
This study demonstrated that mirogabalin administered PO or IT, but not ICV, produced a significant analgesic effect in the rat formalin test. This suggests that the site of analgesic action of PO mirogabalin during inflammation is the spinal cord and not the supraspinal brain. It has been suggested that the dorsal root ganglion is a possible candidate for an alternate peripheral site of action. Biggs et al. 20 reported that exposure of cultured dorsal root ganglion neurons to gabapentin or pregabalin for 5–6 days attenuated high-voltage-activated calcium channel currents. Kitano et al. 21 also reported that little or no effect on the calcium current density in dorsal root ganglion culture neuron was observed following acute exposure to mirogabalin and pregabalin. In this study, an analgesic effect was observed 30 min after IT administration of mirogabalin, suggesting that the site of action may not be the dorsal root ganglion but the spinal cord.
The analgesic effect of mirogabalin on the phase 1 response was observed 2 and 4 h after PO administration and on the phase 2 response 4 h after PO administration. These data suggest that PO mirogabalin attenuates the phase 1 response more effectively than the phase 2 response. Hummig et al. 22 used the orofacial formalin test to show that systemic pregabalin produced an analgesic effect on phase 2, but not phase 1, responses. It is possible that, even in the same inflammatory model, the sensitivity of the α2δ ligand to orofacial pain is different from that to hind paw pain. Another possibility is that the analgesic characteristics of pregabalin differ from that of mirogabalin, with pregabalin preferentially attenuating the phase 2 response, while mirogabalin attenuates both phase 1 and phase 2 responses.
In the IT study, mirogabalin produced an analgesic effect on phase 2, but not phase 1, responses, while no analgesic effect was seen in the ICV study. These data suggest that the site of analgesic action of PO mirogabalin is not only the spinal cord but also sites other than the supraspinal brain, although these sites require further clarification.
The effect of mirogabalin on moving distances was observed 1 and 2 h, but not 4 h, after PO administration and on resting times at 1, 2, and 4 h after PO administration. The time-course discrepancy between the analgesic effects and effects on moving distances and resting times may be attributed to the different sites of action. Moreover, ICV, but not IT, injection of mirogabalin decreased moving distances. This also indicates that the analgesic effect of mirogabalin during inflammation is mediated by spinal cord action, while its effect on moving distance is mediated by supraspinal action.
Domon et al. 4 reported that peak plasma concentrations of mirogabalin were observed within 1 h after PO administration at doses between 2.5 and 10 mg/kg and that this indicated that the time course of the effect of PO mirogabalin on moving distance and resting time but not analgesic effect was similar to that of the plasma concentration of mirogabalin. In humans, it has been reported that peak plasma concentrations of pregabalin after PO administration are observed within 1 h 23 and that tmax of mirogabalin is 1 h. 24 Although mirogabalin data were not reported, peak cerebrospinal fluid concentrations of pregabalin were observed 8 h after PO administration in humans. 25 This study demonstrated that the target site for the analgesic effect of mirogabalin is the spinal cord, with an analgesic effect observed 4 h after PO administration. Thus, in rats, it may take 4 h for mirogabalin to reach the spinal cord after PO administration.
As shown in Figures 7 and 8, in the open field test, moving distance values for the vehicle injection group 0–10 min after injection showed considerable differences between PO, ICV, and IT injections. It has been reported that the open field test is sensitive to acute stress and that handling procedures may lead to changes in several parameters over varying test sessions. 26 ICV and IT injections were performed 10 min before the open field test, and oral administration was performed 1, 2, and 4 h prior. Therefore, differences in the drug administration routes and intervals of drug administration may have influenced the moving distances in the open field test.
In this study, mirogabalin was administered by IT or ICV 10 min before formalin injection. It is possible that the lack of an observed analgesic effect in the ICV study was due to this 10 min interval between ICV and formalin injections. In the IT study, we observed an analgesic effect when mirogabalin was administered 10 min before formalin injection. This strongly suggests that the 10 min interval between injection of IT or ICV and formalin was adequate to examine the central effect of mirogabalin. Moreover, we found that mirogabalin injected ICV 10 min before the open field test decreased moving distances, which also suggests that 10 min was long enough for a sufficient amount of mirogabalin to penetrate to the CNS.
The mechanisms that produce analgesic effects on inflammatory pain are thought to differ from that on neuropathic pain. The suppressive effects of α2δ ligands on the forward trafficking of α2δ subunit and calcium channels to neurotransmitter release sites is proposed to be one of the mechanisms for their analgesic effect on neuropathic pain. 27 It takes a long duration before this effect is seen. In this study, the analgesic effect of mirogabalin was observed 10 min after IT injection. α2δ subunit increases the density of high-voltage-activated calcium channels at release sites and promotes increased exocytosis. 28 α2δ ligands interrupt a facilitation of high-voltage-activated calcium channels at neurotransmitter release sites and decrease neurotransmitter release. 15 This mechanism may produce analgesic effects in both neuropathic and inflammatory pain. It has been reported that, in the neuropathic pain model, one of the action sites of α2δ ligands is the LC.16,17,29 It has also been reported that systemic administration of gabapentin induces noradrenalin release in the spinal cord in spinal nerve ligation model rats, a neuropathic pain model, and that the analgesic effect of LC gabapentin is antagonized by IT idazoxan. 17 In this study, we found that the analgesic effect of PO mirogabalin was not antagonized by IT idazoxan in the rat formalin test and that ICV mirogabalin did not produce an analgesic effect in the rat formalin test. This indicates that the analgesic effects of systemic α2δ ligands may not be mediated by the activation of spinal α2 receptors in the inflammatory pain model.
Spinal-supraspinal 5-HT circuits have been reported to regulate the analgesic effect of gabapentin in the neuropathic pain model and that this effect is mediated by 5-HT3 receptor activation. 30 Furthermore, 5-HT7, but not 5-HT1A, receptors have been reported to play pronociceptive roles at spinal sites in the rat formalin test. 31 In this study, the analgesic effects of neither PO nor IT mirogabalin were antagonized by IT WAY100135, indicating that 5-HT1A receptors are not involved in the analgesic effects of PO or IT mirogabalin in the rat formalin test.
IP naloxone was not shown to antagonize the analgesic effects of either oral or IT mirogabalin. To our knowledge, there have been no reports on the μ-opioid receptor activation-mediated analgesic effects of α2δ ligands. Our mirogabalin data are consistent with previous reports on pregabalin and gabapentin.
In conclusion, oral and IT, but not ICV, mirogabalin produced analgesic effects, strongly suggesting that the site of analgesic action of mirogabalin during inflammation is the spinal cord and not the supraspinal brain. Furthermore, the site of the depressant effect of mirogabalin on locomotor activity and orientation ability is the supraspinal brain. Last, the analgesic effect of mirogabalin is not mediated by μ-opioid, spinal α2 adrenergic, or spinal 5-HT1A receptor activation.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was partly supported by Grant-in-Aid for Scientific Research (C) 17K09036 and Kumamoto University. Mirogabalin was provided from Daiichi Sankyo Co., Ltd. (Tokyo, Japan).
