Abstract
Interferons (IFNs) are cytokines secreted by infected cells that can interfere with viral replication. Besides activating antiviral defenses, type I IFNs also exhibit diverse biological functions. IFN-β has been shown to have a protective effect against neurotoxic and inflammatory insults on neurons. Therefore, we aimed to investigate the possible role of IFN-β in reducing mechanical allodynia caused by Complete Freund’s Adjuvant (CFA) injection in rats. We assessed the antinociceptive effect of intrathecal IFN-β in naïve rats and the rats with CFA–induced inflammatory pain. After the behavioral test, the spinal cords of the rats were harvested for western blot and immunohistochemical double staining. We found that intrathecal administration of IFN-β in naïve rats can significantly increase the paw withdrawal threshold and paw withdrawal latency. Further, the intrathecal injection of a neutralizing IFN-β antibody can reduce the paw withdrawal threshold and paw withdrawal latency, suggesting that IFN-β is produced in the spinal cord in normal conditions and serves as a tonic inhibitor of pain. In addition, intrathecal injection of IFN-β at dosages from 1000 U to 10000 U demonstrates a significant transient dose-dependent inhibition of CFA-induced inflammatory pain. This analgesic effect is reversed by intrathecal naloxone, suggesting that IFN-β produces an analgesic effect through central opioid receptor-mediated signaling. Increased expression of phospho-µ-opioid receptors after IFN-β injection was observed on western blot, and immunohistochemical staining showed that µ-opioids co-localized with IFN-α/βR in the dorsal horn of the spinal cord. The findings of this study demonstrate that the analgesic effect of IFN-β is through µ-opioid receptors activation in spial cord.
Introduction
Interferons (IFNs) are antiviral molecules that are known for their ability to interfere with viral replication in mammalian cells. 1 They are secreted from infected cells and activate innate immunity by enhancing natural killer (NK) cell function and antigen presentation. 2 The type I IFN family includes numerous IFN-α subtypes, a single IFN-β and the less understood IFN-ε, IFN-κ, and IFN-ω.3,4 Type I IFNs, especially INF-α and IFN-β, share significant amino acid homology, 5 bind to a common receptor (IFN-α/βR),6,7 and display both separate and overlapping roles in the interaction between host and infecting microbe. 8 Beyond their role in the activation of antiviral and antimicrobial defenses, type I IFNs also exhibit diverse biological functions, including modulation of immune cells, differentiation and survival of cells. 9 Therefore, type I IFNs are currently the major treatment of hematologic malignancies and several non-malignant diseases such as hepatitis B and C and multiple sclerosis.10–12
IFNs may be constitutively expressed even in the absence of an acute infection. 9 It has been suggested that IFN-α may be produced at a low “physiologic” level within the central nervous system (CNS). 9 In this sense, several studies indicate that IFNs can participate in modulating the function of neurons, microglia, astrocytes and oligodendrocytes in CNS.13–15 Furthermore, IFN-α can also directly modulate central nociceptive signal transmission.16,17 Spinal administration of INF-α in rats and mice has been shown to reduce Complete Freund’s Adjuvant (CFA)-induced inflammatory pain.16,17 The analgesic effect of IFN-α may be due to its mediation of nociceptive signal transmission by dorsal horn neurons 17 and its interaction with the opioid receptor.16,18 Another report supported the correlation of analgesia of IFN-α with opiates by showing that serum IFN-α in mice induced by endotoxin was reduced after a single injection of morphine. 19 Moreover, IFN-α was reported to produce potent endophine-like effect. 20 IFN-β is also a type I IFN in addition to IFN-α and has a homologous sequence to IFN-α. In addition, IFN-β has been found to induce direct protective effect on neurons against neurotoxic and neuroinflammatory effect.21,22 Whether IFN-β could produce antinociceptive effect like IFN-α is still unclear. In this study, the antinociceptive effect of IFN-β administered intrathecally and the mechanism of antinociceptive effect of IFN-β were investigated.
Materials and methods
Animals
Adult male Sprague-Dawley rats (weight 200–240 g; National Laboratory Animal Center, Taiwan) were housed in a room in individual cage and provided with free access to standard food and water. The room was kept at 22°C and with light for 12 h from 7AM to main light/dark cycle. All experimental procedures were carried out during the light phase and animals were randomly included in different treatment groups. The experimental protocols were approved by the Animal Care Committee of E-Da hospital in accordance with E-Da hospital Animal institutional guidelines. Minimum rats were recommended to be used in study by Animal Care Committee, adhering to the principles of the 3Rs (Replacement, Reduction and Refinement). 23 The sample size was determined on the basis of our previous studies experience16,17 and recent literature 24 with similar the experimental models and anticipated biological variables.
Pharmacological treatments
IFN-β and IFN-β neutralizing antibody were purchased from Sigma-Aldrich (St. Louis, MO, USA) and used without further purification except when mentioned specifically. It was diluted in sterile saline and administered intrathecally (i.t.) in a volume of 10 µL to give a final concentration of 10000 U IFN-β. Saline control rats received an equivalent volume of saline vehicle. Intrathecal injection was made with 25 G needle between L5 and L6 intervertebral spaces to deliver the drugs to the subarachnoid space. 25
CFA-induced inflammatory pain and behavioral testing
To induce persistent inflammatory pain, subcutaneous injection of twenty microliters of Complete Freund’s Adjuvant (CFA) (Sigma-Aldrich, St. Louis, MO, USA) into the plantar surface of the left hind paw was performed. Local inflammation, paw swelling and pain that persists for at least 2 weeks after CFA injection. Mechanical allodynia was determined by presenting von Frey filament (Stoelting Co., Wood Dale, IL,USA) perpendicularly to the plantar surface of each hind paw. 26 The rats were placed on a metal mesh table for at least three times before test to adapt to the metal mesh. The bending force of the von Frey filaments was exerted to the plantar surface of the hind paw by a serially increased manner from 0.6 to 26 g. Following application of each filament four times with sufficient forces causing bending of filament, the 50% paw withdrawal threshold was determined using Dixon’s up–down method. 27 Thermal hyperalgesia was examined by delivering an infrared heat source under the plantar surface of the hind paw. 28 Rats were placed on glass plates and allowed to adapt to the new environment. The thermal stimulus elicited hind paw withdrawal at a latency of approximately 12 to 15 sec. The mean paw withdrawal latencies were determined from the 3 separate trials taken at 5 min intervals to prevent thermal sensitization and behavioral disturbances.
Western blot analysis
Protein samples were separated on sodium dodecyl sulphate–polyacrylamide gel electrophoresis gel (4–15%, Bio-Rad) and transferred to polyvinylidene difluoride membrane (Immobilon-P, Millipore, Bedford, MA, USA). The blots were blocked with phosphate-buffered saline (PBS) containing 5% non-fat milk for 1 h and incubated overnight at 4°C with polyclonal antibody against Phospho-µ-Opioid Receptor (1:1000, Bioss, bs-3724R, USA) and mouse monoclonal anti-actin (1:5000, Santa Cruz Biochemicals, Inc., Santa Cruz, CA, USA). Then, the membranes were incubated with 2nd Antibodies of horseradish peroxidase-conjugated donkey anti-rabbit 1:5000 immunoglobulin G (Jackson ImmunoResearch, West Grove, PA, USA) and 1:5000 horseradish peroxidase-linked sheep anti-mouse IgG (Amersham Biosciences, Arlington Heights, IL, USA) for 1 h. The blots were shown by incubation with chemiluminescent solution (Immobilon Western Chemiluminescent HRP Substrate; Millipore, Billerica, MA, USA) and were visualized on the UVP BioSpectrum 500 Imaging System (UVP, Upland, CA, USA). β-actin was used as loading control for Phospho-µ-Opioid receptor proteins. The final density of each band was determined after being normalized to its corresponding loading control.
Immunohistochemistry
Animals were deeply anesthetized with isoflurane followed by perfusion with 0.9% saline followed by 4% paraformaldehyde through their ascending aorta. After perfusion the L4–L5 spinal cord segments were harvested and post fixed in paraformaldehyde overnight. Spinal cord sections were cut at a thickness of 30 µm free-floating sections in a cryostat and processed for immunofluorescence. In addition to blocked with 2% goat serum, spinal cord sections were then incubated over night at 4°C with the following primary antibodies: IFN-α/βR (mouse, 1:50, Santa Cruz, sc-30015, M-300) and Phospho-µ-Opioid Receptor (1:400, Bioss, USA). After incubating with primary antibodies, spinal cord sections were then incubated with Cy3- or FITC- conjugated secondary antibodies (1:200, Alexa Fluor488, 588, abcam immunolab) for 1 h at room temperature. Spinal cord sections were incubated in mixture of IFN-α/βR and Phospho-µ-Opioid Receptor antibodies followed by a mixture of FITC- and CY3-congugated secondary antibodies for double immunofluorescence. The specificity of immunostaining and antibodies was tested by omitting the primary antibody.
Experimental designs
Assessing the effects of the spinal administration of IFN-β and naloxone on CFA-induced inflammatory pain
Two days after CFA injection rats were divided into seven groups (n = 5 each group). Three groups received intrathecal IFN-β (1000, 2500 and 10000 U), one group received intrathecal saline at an equivalent volume. The paw withdrawal tests for the nociceptive response in these four groups were performed 0.5, 1, 2, 4 and 24 h later after injection. The two groups received intrathecal IFN-β 10000 U, and naloxone 20 nmol was injected intrathecally in one of the two groups one hour later. The remaining one group is CFA alone group. The paw withdrawal test was again performed at 0.5, 1, 2 and 4 h after naloxone injection.
Assessing the effect of the intrathecal IFN-β and IFN-β antibody on naïve rat
Rats were divided into four groups (n = 8 each group). Two groups had intrathecal injections of IFN-β 10000 U, while two control groups received intrathecal saline at an equivalent volume. The IFN-β groups were tested for a nociceptive response by the von Frey filaments test or the thermal hyperalgesia test after treatment. The control group was also tested by the same behavioral tests after injection. After the behavioral tests one hour after injection, the spinal cords of the rats of one IFN-β group and one control group were harvested for western blot of the phospho-µ-opioid receptor (n = 5 each group) and immunohistochemical double staining for the IFN-α/β and phospho-µ-opioid receptors (n = 3 for each group). The other two groups of IFN-β and control groups were tested on 1, 12, 24, 28 and 32 hours for a nociceptive response by the von Frey filaments test or the thermal hyperalgesia test after treatment. Based on the dose used in our previous study, 17 a neutralizing IFN-β antibody at a dose of 30 ng or the serum at an equivalent volume were administrated intrathecally to another two groups of naïve rats (n = 5 each group). The behavioral tests were performed to test mechanical allodynia or thermal hyperalgesia in rats of IFN-b antibody group and serum group 0.5, 1 and 3 h later after injection.
Statistics
The data were statistically analyzed with Prism 8 (GraphPad Software, La Jolla CA, USA). All data were expressed as mean ± standard deviation (SD). The sample sizes used in each experiment were based on our previous studies16,17 and recent literature. 24 A power analysis was performed to detect significant differences with minimal animal numbers based on our experimental experience with 80% power. Therefore, n = 5–10 was chosen. All data were included in the analyses. The data from the behavioral test were analyzed with two-way repeated-measures analysis of variance (RM ANOVA) followed by Tukey’s or Bonferroni’s post hoc test. Western blot data was compared using two-tailed t tests. A value P < 0.05 was considered significant. Image-Pro Plus analysis software (Media Cybernetics, Silver Spring, MD, USA) was used to measure the density of specific bands from western blotting. The results were shown by the ratio of phospho-µ-opioid receptor to β-actin immunoreactivity.
Results
IFN-β treatment inhibits CFA-induced inflammatory pain
To determine whether an intrathecal injection of IFN-β would regulate pain sensitivity, different doses of IFN-β (1000, 2500 and 10000 U) were injected intrathecally 2 days after plantar CFA injection. Significant dose-dependent attenuation of CFA-induced mechanical allodynia by intrathecal administration of IFN-β was noted (two-way RM ANOVA; F(6, 112) = 134.6 for factor time, P < 0.001; F(3, 112) = 124.9 for factor group, P < 0.001; F(18, 112) = 14.45 for time x group interaction, P < 0.001; n = 5 each group, Figure 1), and the highest dosage (10000 U) exerted a significant effect 24 hours after injection (IFN-β 10000 U vs. Saline, 0.5 h: mean difference [95% CI]: 3.260 [0.9528 to 5.567], 1 h: mean difference [95% CI]: 7.600 [5.293 to 9.907], 2 h: mean difference [95% CI]: 9.550 [7.243 to 11.86], 4 h: mean difference [95% CI]: 9.350 [7.040 to 11.66], 24 h: mean difference [95% CI]: 11.91 [9.603 to 14.22]; n = 5 each group, Figure 1).

Effect of the intrathecal (i.t.) administration of IFN-β on CFA-induced mechanical allodynia. Two days after CFA injection, four groups of rats received an i.t. injection of either IFN-β 1000 U, 2500 U, 10000 U or an equivalent volume of saline. After treatment, the paw withdrawal test was used to test mechanical allodynia at 0.5, 1, 2, 4 and 24 h. Rats that received i.t. injection of 10000 U IFN-β showed significant inhibition of CFA-induced mechanical allodynia from 0.5 h to 24 h. * P = 0.002, **P < 0.001, IFN-β 10000 U vs. Saline; #P < 0.001, IFN-β 10000 U vs. IFN-β 1000 U, 2500 U; +P = 0.03, IFN-β 2500 U vs. Saline; &P = 0.001, IFN-β 1000 U vs. Saline; followed by two-way RM ANOVA Tukey’s post-hoc test; n = 5 rats/group. All data are written as mean ± S.D.
Spinal administration of IFN-β increased the paw withdrawal threshold and paw withdrawal latency
Intrathecal IFN-β 10000 U significantly increased the paw withdrawal threshold (two-way RM ANOVA; F(5, 84) = 6.676 for factor time, P < 0.001; F(1, 84) = 58.29 for factor group, P < 0.001; F(5, 84) = 5.268 for time x group interaction, P < 0.001; n = 8 each group, Figure 2(a)) and paw withdrawal latency (two-way RM ANOVA; F(5, 84) = 6.824 for factor time, P < 0.001; F(1, 84) = 45.68 for factor group, P < 0.001; F(5, 84) = 6.919 for time × group interaction, P < 0.001; n = 8 each group, Figure 2(b)) in naïve rats for at least 28 h (paw withdrawal threshold, 1 h: mean difference [95% CI]: 3.200 [1.452 to 4.948], 12 h: mean difference [95% CI]: 3.500 [1.752 to 5.248], 24 h: mean difference [95% CI]: 2.800 [1.052 to 4.548], 28 h: mean difference [95% CI]: 2.200 [0.4516 to 3.948]; paw withdrawal latency, 1 h: mean difference [95% CI]: 2.600 [1.164 to 4.036], 12 h: mean difference [95% CI]: 2.200 [0.7637 to 3.636], 24 h: mean difference [95% CI]: 2.700 [1.264 to 4.136], 28 h: mean difference [95% CI]: 1.900 [0.4637 to 3.336]) . Further, a neutralizing IFN-β antibody decreased the paw withdrawal threshold (two-way RM ANOVA, F(3, 32) = 19.02 for factor time, P < 0.001; F(1, 32) = 60.11 for factor group, P < 0.001; F(3, 32) = 12.58 for time x group interaction, P < 0.001; n = 5 each group, Figure 3(a)) and paw withdrawal latency (two-way RM ANOVA; F(3, 32) = 16.60 for factor time, P < 0.001; F(1, 32) = 74.58 for factor group, P < 0.001; F(3, 32) = 19.05 for time x group interaction, P < 0.001; n = 5 each group, Figure 3(b)) in naïve rats 0.5 and 1 hour after intrathecal injection (paw withdrawal threshold, 0.5 h: mean difference [95% CI]: −5.410 [−7.622 to −3.198], 1 h: mean difference [95% CI]: −6.160 [−8.372 to −3.948], 3 h: mean difference [95% CI]: paw withdrawal latency, 0.5 h: mean difference [95% CI]: −4.400 [−5.934 to −2.866], 1 h: mean difference [95% CI]: −4.890 [−6.424 to −3.356]) . Therefore, these results suggested that the antinociceptive effect of IFN-β could have persisted for 28 hours and the duration of the pronociceptive effect of IFN-β antibody was not more than 3 hours on naïve rats.
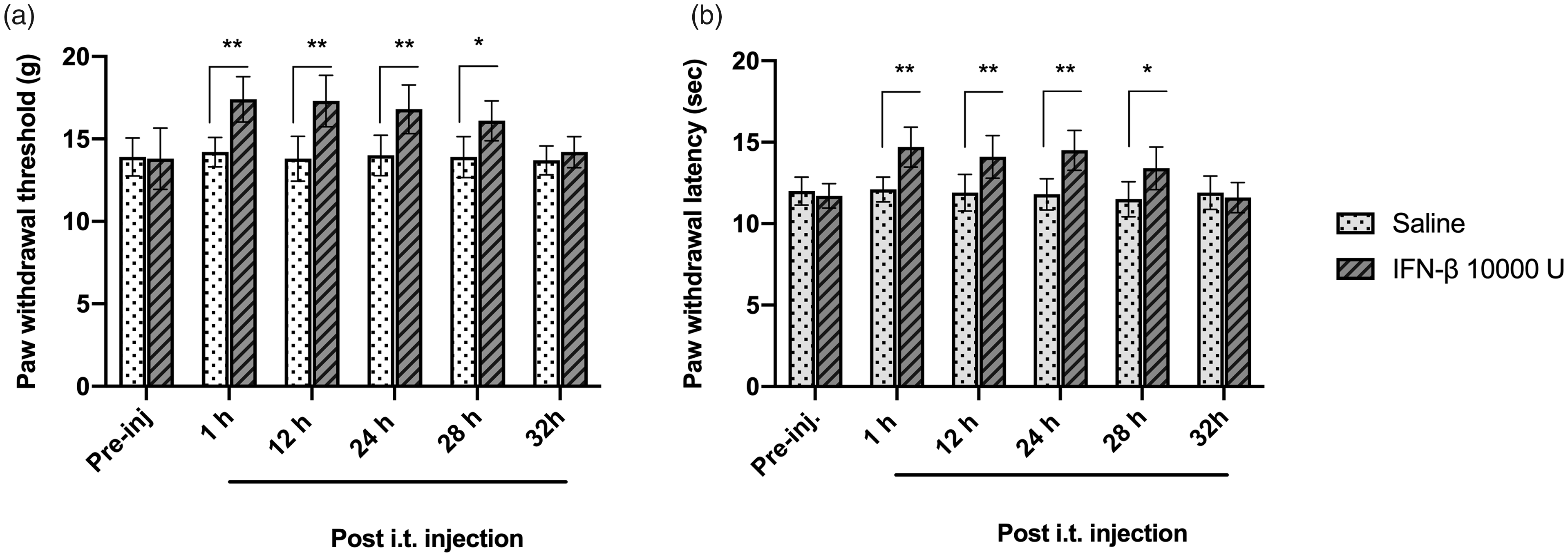
Analgesic effect of intrathecal (i.t.) administration of IFN-β on naïve rats. Paw withdrawal threshold and latency were examined after i.t. injection of 10000 U IFN-β. Intrathecal injection of 10000 U IFN-β significantly increased the paw withdrawal threshold (a) and paw withdrawal latency (b) in naïve rats at 1, 12, 24, 28 h. *P = 0.006 (a), 0.004 (b), **P < 0.001, IFN-β vs. Saline; followed by two-way RM ANOVA Bonferroni’s post-hoc test; n = 8 rats/group. All data are written as mean ± S.D.

Effects of intrathecal (i.t.) injection of IFN-β neutralizing antibody on the nociceptive response of naïve rats. After i.t. Injection of IFN-β neutralizing Ab 30 ng, the paw withdrawal threshold (a) and paw withdrawal latency (b) were significant decreased in naïve rats at 0.5 h and 1 h. *P < 0.001, IFN-β Ab vs. Serum; followed by two-way RM ANOVA Bonferroni’s post-hoc test; n = 5 rats/group. All the data are written as mean ± S.D.
Spinal administration of naloxone reverses the analgesic effect of IFN-β
In rats with CFA-induced mechanical allodynia, the antinociceptive effect of intrathecal injection of IFN-β was reversed after the spinal administration of 20 nmol naloxone one hour later (two-way RM ANOVA, F(6, 84) = 128.6 for factor time, P < 0.001; F(2, 84) = 222.8 for factor group, P < 0.001; F(12, 84) = 25.56 for time × group interaction, P < 0.001; n = 5 each group, Figure 4). Intrathecal naloxone decreased the paw withdrawal threshold 0.5, 1, 2 h and 4 h after injection (0.5 h, mean difference [95% CI]: −7.05 [−8.563 to −5.537], 1 h, mean difference [95% CI]: −6.31 [−7.823 to −4.797], 2 h: mean difference [95% CI]: −5.60 [−7.113 to −4.087], 4 h, mean difference [95% CI]: −1.73 [−3.243 to −0.2169], n = 5 each group, Figure 4).

Intrathecal (i.t.) injection of naloxone reverses the analgesic effect of IFN-β. All rats received plantar injection of CFA for 2 days. Behavior tests were examined in three groups: 1 hour after i.t. injection of IFN-β 10000 U (IFN-β 10000 U 1 h); injection of 20 nmole naloxone after receiving IFN-β for 1 hour (IFN-β 10000 U 1 h+ Naloxone 20 nmole); and only injection of CFA (CFA alone). Nociceptive responses were accessed at 0.5, 1, 2, 4 h after naloxone injection. I.t. IFN-β significantly produced anti-allodynic effect that was reversed by administration of naloxone after 0.5, 1, 2, 4 h. *P = 0.02, **P < 0.001, IFN-β vs. IFN-β + Naloxone; #P < 0.001, IFN-β vs. CFA alone; + P < 0.001, IFN-β + Naloxone vs. CFA alone; followed by two-way RM ANOVA Tukey’s post-hoc test; n = 5 rats/group. All data are written as mean ± S.D.
IFN-β treatment on the expression of µ-opioid receptors in spinal cord dorsal horn
To identify the mechanism of spinal IFN-β in pain modulation we further investigated the expression of the phospho-µ-opioid receptor and IFN-α/βR after the intrathecal administration of IFN-β. The expression of the phospho-µ-opioid receptor was significantly increased after the administration of IFN-β, indicating that IFN-β could induce the phosphorylation of the µ-opioid receptor (t = 14.41, df = 4, P < 0.001, Figure 5). Remarkably, the IFN-α/βR expression was highly expressed in the superficial dorsal horn (laminae I-II) of the spinal cord, where the nociceptive primary afferents (C/Aδ) terminate (Figure 6(a) and (c)). Colocalization of IFN-α/βR and the phospho-µ-opioid receptor (Figure 6(e) and (f)) was further found in lamina I-II by double staining, suggesting that part of µ-opioid receptor may also be expressed with IFN-α/βR.
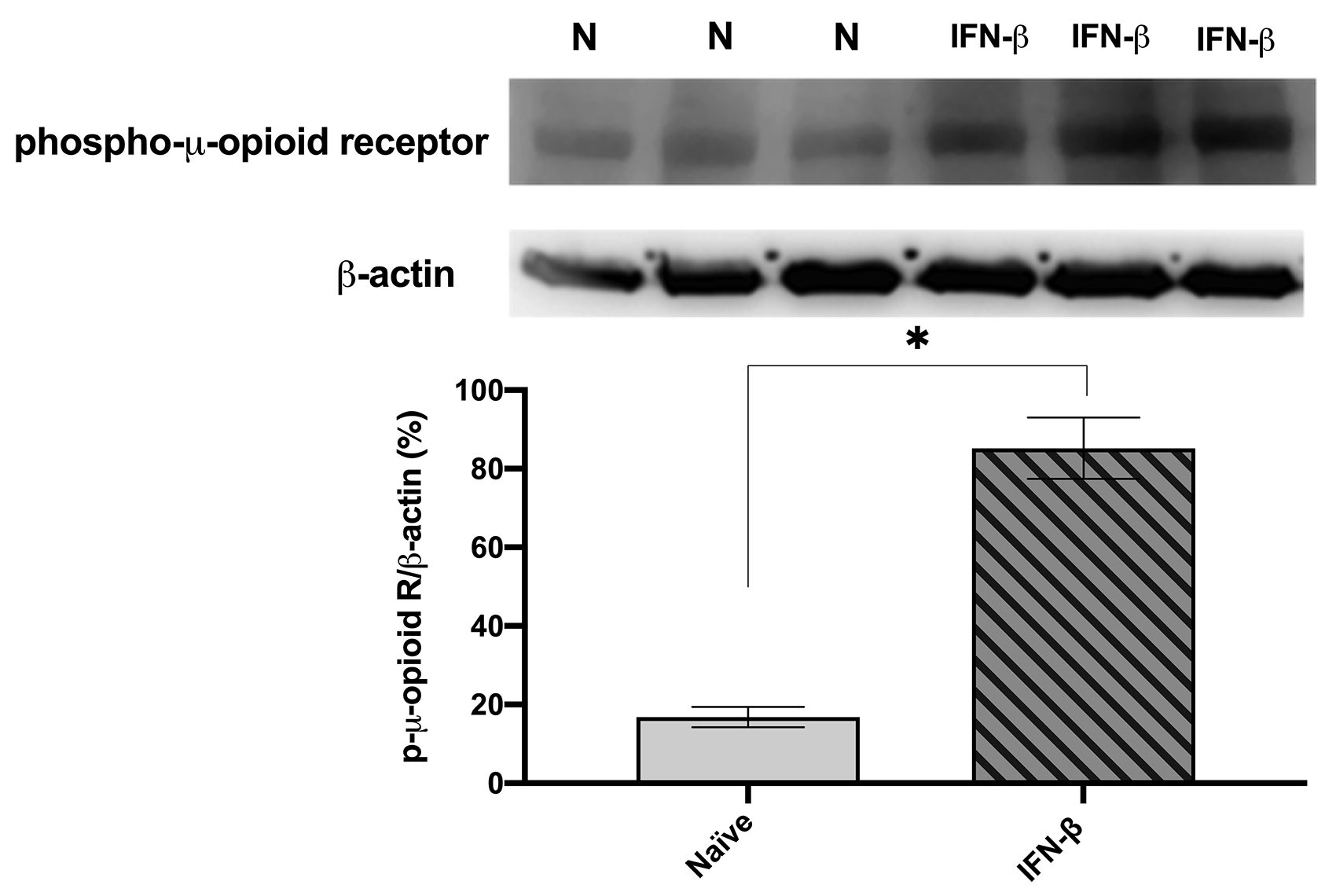
Expression of phospho-µ-opioid receptors after intrathecal (i.t.) injection of IFN-β. One group of naïve rats received an i.t. injection of IFN-β 10000 U, and the other group were given intrathecally an equivalent volume of saline. Tissues were collected one hour after IFN-β treatment. N is indicated as naïve. *P < 0.001, IFN-β vs. naïve; unpaired t test, two-tailed; n = 5 rats/group. All data were mean ± S.D.

Expression of IFN-β receptors and phosphor-µ-opioid receptors in the spinal cord dorsal horn. (a, c) Immunostaining of the Type I-IFN (IFN-α/βR) and µ-opioid receptors in the superficial dorsal horn. Scale, 100 µm. (e) Colocalization of IFN-α/βR and the µ-opioid receptor in the superficial dorsal horn (laminae I-II). (b, d) Staining of the Type I-IFN receptor and the μ-opioid receptor with omission of the 1st Ab. (f) High magnification image showed colocalization of IFN-α/βR and the µ-opioid receptor. Scale, 100 µm in (e) and 10 µm in (f). Arrows in (e) and (f) indicate double-labeled receptors.
Discussion
The present study characterized the antinociceptive properties of IFN-β and its mechanism of action at the spinal cord level. Our results show that the spinal administration of IFN-β significantly increased baseline paw withdrawal threshold and paw withdrawal latency in naïve rats. By contrast, spinal administration of IFN-β antibody could reduce baseline paw withdrawal threshold and paw withdrawal latency in naïve rats, thereby demonstrating the ability of IFN-β to produce a spontaneous antinociceptive effect. Furthermore, intrathecal administration of IFN-β could suppress mechanical allodynia in rats with CFA-induced inflammatory pain. These data indicate that IFN-β has an antinociceptive effect on CFA-induced inflammatory pain and serves as a tonic inhibitor of pain in the spinal cord in normal conditions. Another family member of type I IFNs, IFN-α, is also reported to maintain spinal cord pain circuit homeostasis. 17 Spinal injection of IFN-α has been shown to inhibit CFA-induced inflammatory pain. 17 Several studies have suggested that the analgesic effect of IFN-α in the CNS may be the result of underlying opioid signaling. First, similarities in the structure and function of IFN-α and endorphin have been demonstrated. 29 Second, IFN-α can bind to opioid receptors in the brain, inducing an analgesic effect. 30 Third, the analgesic effect induced by intracerebroventricular injection of IFN-α could be reversed both by a non-selective opioid receptor antagonist (naloxone) and a µ selective opioid antagonist β-FNA. 18 Dafny and Reyes-Vazquez 31 reported that the administration of IFN-α could alter the neuronal activity of brain regions that participate in the opioid response either in the presence of opioids or when given alone. IFN-β and IFN-α are type I IFNs that act on the same receptor and share similar amino acid sequences. Thus, we attempted to investigate whether IFN-β suppresses nociceptive transmission in the spinal cord through opioid signaling, much like IFN-α.
In support of our behavioral studies, we further provided data to prove that IFN-β inhibition of nociception occurs at the spinal cord level. First, intrathecal administration of IFN-β increased the expression of the phospho-µ-opioid receptor, indicating that IFN-β could activate the µ-opioid receptor (Figure 5). Second, our immunohistochemical data showed that the type I IFN receptor (IFN-α/βR) was co-localized with the phospho-µ-opioid receptor and both were expressed on laminae I and II in the superficial dorsal horn of the spinal cord (Figure 6). Together with our finding that the antinociceptive effect of IFN-β on CFA-induced mechanical allodynia could be reversed by naloxone, we conclude that the antinociception of IFN-β is associated with µ-opioid receptor binding and activation in the spinal cord.
The ratio of IFN-α and IFN-β produced by cells is different and depends on various tissue and species. 4 Generally, IFN-α is produced by leukocytes while IFN-β is produced by fibroblasts, and both are anti-inflammatory cytokines. Type I IFNs share the same receptor composed of two distinct subunits (IFNAR1 and IFNAR2) that interact with a member of the Janus activated kinase (JAK) family. 7 Upon binding, the initial step of type I IFNs mediated signaling is structural rearrangement and dimerization of the two receptor subunits, followed by cross-phosphorylation of tyrosine kinase 2 (Tyk2) and Jak1 and activation of the classical JAK–STAT (signal transducer and activator of transcription) pathway. 7 JAK-STAT activation can regulate, either indirectly or directly, the downstream pathways and alter the expression of a variety of genes that mediate various biological reactions. 32 The many variable activities of type I IFNs are based on the induction or repression of thousands of genes. 33 Although type I IFNs activate their receptors on the cell surface, their effects are often cell type-specific and related to the subtype used, the duration of activation, and concentration. It is because of these factors that many of the findings of prior studies on type I IFNs are difficult to replicate within different milieus, making the assessment of the generalizability of those findings a serious challenge. 34 Our data provides evidence that IFN-β-induced antinociceptive effects are the result of µ-opioid receptor activation that is co-localized with the type I IFN receptor (IFN-α/βR). It implies that there may be interactions between these two types of receptors that need to be explored in the future.
Within type I IFNs, it has been reported that only IFN-α could bind to opioid receptors and possess analgesic properties.18,20,29 Blalock et al. 29 indicated that human leukocyte interferon (HuIFN-α) could be neutralized by antiendorphin antiserum. These data show that only HuIFN-α shares antigenic and structural similarities with endorphins. Moreover, the same authors 20 demonstrated that only HuIFN-α is capable of reducing the specific binding of dihydromorphine to a membrane preparation from the mouse brain. In addition, HuIFNs were injected intracerebrally in mice, and only HuIFN-α showed evidence of analgesic activity. These analgesic effects could be reversed by naloxone when given after HuIFN-α. However, the administration of IFN-α could induce obvious adverse effects in animals or humans. IFN-α treated mice would have lower activity and decreased muscle strength and endurance. 35 It has also been reported that patients receiving long term IFN- α treatment display significant side effects such as anorexia, cognitive dysfunction and psychiatric symptoms.36,37
In our study, the spinal administration of IFN-β could inhibit CFA-induced inflammatory pain, and its antinociceptive effects could be reversed by intrathecal naloxone. Moreover, intrathecal injection of IFN-β could induce phosphorylation of the µ-opioid receptor that was co-localized with IFN-α/βR to lamina I-II of the dorsal horn of the spinal cord. Gironi et al. 38 reported that treatment with IFN-β induces an increase in β-endorphin level in the peripheral blood mononuclear cells of patients with multiple sclerosis. Stokes et al. 39 demonstrated that intrathecal IFN-β attenuated allodynia induced by Toll-like receptor (TLR) 2, 4 ligands. Ifnar1−/− mice developed prolonged allodynia following intrathecal TLR 3, 4 ligands compared with wild mice. Type I IFN signalling is required for the rapid resolution of allodynia induced by TLR 3, 4 ligands. Intrathecal IFN-α and IFN-β have also been shown to inhibit CFA-hypersensitivity 16 and upregulate proinflammatory cytokines. 40 These reports provide evidence of the antinociceptive effects of IFN-β and its activation of the type I IFN receptor. Our data provides evidence of the potential interaction between IFN-α/βR and the µ-opioid receptor in the generation of the antinociceptive effect of IFN-β. There are conflicting reports claiming that intraplantar administration of either IFN-α (300 U/25ul) or IFN-β (300 U/25ul) produced a rapid mechanical hypersensitivity for 3 days. DRG neurons were exposed to IFN-α (300 U/ml) and measured neuronal excitability using patch-clamp electrophysiology. Hyperexcitability in small diameter DRG neurons was found to be promoted by type I IFN. 41 Notably, Donnelly et al. 42 reported on the stimulation of interferon genes (STING)-mediated antinociception in mice and nonhuman primates, which is governed by type-I IFNs. The antinociception by STING agonists is associated with increased expressions of IFN-α and IFN-β in DRG and spinal cord by stimulating STING resides in the endoplasmic reticulum. Intrathecal administration of IFN-α could block mechanical allodynia induced by intraplantar IFN-α in this study. 42 Furthermore, peripheral IFN-β cannot directly penetrate blood brain barrier (BBB) to the CNS. 13 Taken together, the analgesic effect resulted from intraplantar injection of IFN-β was arguably due to a local effect. The difference effect of intraplantar and intrathecal injection of IFN-α and IFN-β is possibly resulted from different acting sites on peripheral nervous system and central nervous system. In another study, formalin-evoked inflammatory nociceptive behavior was potentiated by subcutaneous IFN-α administration for 8 days at doses of 8000 IU/gram body weight/day, but not altered by single injection of IFN-α. 43 This administered dose of IFN-α administered is much higher than dose administered in previous reports.12,39,42 Therefore, the pronociceptive effects induced by intraplantar administration of type-I IFN is possibly a dose-dependent effect.
Conclusion
We have demonstrated that spinal administration of IFN-β could effectively reduce mechanical allodynia in naïve rats or those with CFA-induced inflammatory pain. The analgesic effect of IFN-β is mediated through µ-opioid receptors that are co-expressed with IFN-α/βR in the dorsal horn of the spinal cord. Although the present work is limited to rats and may not completely recapitulate the human clinical pain condition, it provides evidence of a new analgesic role of IFN-β in the modulation of nociceptive transmission at the spinal cord level.
Footnotes
Acknowledgments
We thank Dr. Jiin Tsuey Cheng for her theoretical and statistical support.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was supported by grants from the Taiwan Ministry of Science and Technology (MOST 106–2314-B-214 –003-MY3), Chi Mei Hospital Grants (CMFHR10809, 10902), CMNDMC11012 and E-Da Hospital Grants (EDAHP 107030, 108010).
