Abstract
IB4-positive maxillary trigeminal ganglion (TG) neurons are a subtype of afferent neurons involving nociception in orofacial regions, and excitability of these neurons is associated with orofacial nociceptive sensitivity. TREK-2 channel is a member of two-pore domain potassium (K2P) channel family mediating leak K+ currents. It has been shown previously that TREK-2 channel activity can be enhanced following GABAB receptor activation, leading to a reduction of cortical neuron excitability. In the present study, we have characterized TREK-2 channel expression on maxillary TG neurons and investigated the effect of the GABAB agonist baclofen on electrophysiological properties of small-sized maxillary TG neurons of rats. We show with immunohistochemistry that TREK-2 channels are predominantly expressed in small-sized IB4-positive maxillary TG neurons. Patch-clamp recordings on neurons in ex vivo TG preparations show that baclofen hyperpolarizes resting membrane potentials, increases outward leak currents, and decreases input resistances in IB4-positive maxillary TG neurons. Moreover, baclofen significantly reduces action potential (AP) firing in IB4-positive maxillary TG neurons. In contrast, baclofen shows no significant effect on electrophysiological properties of small-sized nociceptive-like and non-nociceptive-like maxillary trigeminal neurons that are IB4-negatve. Our results suggest that TREK-2 channel activity can be enhanced by baclofen, leading to reduced excitability of IB4-positive maxillary TG neurons. This finding provides new insights into the role of TREK-2 and GABAB receptors in controlling nociceptive sensitivity in orofacial regions, which may have therapeutic implications.
Introduction
Nociceptive afferent neurons (nociceptors) sense stimuli that are potentially harmful under physiological conditions, and these neurons may become hyper-excitable leading to exaggerated pain under pathological conditions. Isolectin B4 (IB4) positive neurons are a subpopulation of nociceptors located in the dorsal root ganglions (DRG) and trigeminal ganglions (TG). IB4-positive neurons are mostly small-sized nonpeptidergic afferent neurons 1 expressing Mas related G protein-coupled receptor D (MrgprD). 2 IB4-positive nociceptors innervate cutaneous regions at the periphery 2 and their central terminals predominantly project to inner lamina II in the dorsal horn. 3 In addition to IB4-positive neurons, the other main subtype of nociceptors is small-sized peptidergic neurons which primarily project to lamina I and outer lamina II regions in the dorsal horn. 4 These two populations of small-sized nociceptors convey large amounts of nociceptive signals to central pathways of nociception.4,5
Excitability of nociceptors is highly regulated during signal transduction at peripheral endings, conduction along their afferent fibers, and transmission at their central terminals. One mechanism of regulating nociceptor excitability is mediated by γ-aminobutyric acid (GABA) and its receptors including both GABAA and GABAB receptors.6,7 GABAA receptors are chloride-permeable channels widely expressed in afferent neurons, and their activation usually suppresses afferent neuron excitability.7,8 On the other hand, GABAB receptors are G-protein coupled receptors, and their activation alters intracellular signal transduction 8 to subsequently affect afferent neuron excitability. 6 GABAB receptors are expressed in nociceptors including their central and peripheral terminals to regulate nociceptor excitability. 6 Baclofen, a GABAB receptor agonist, has been used clinically for treating neuropathic pain and certain types of spasticity (muscle stiffness and tightness) resulting from multiple sclerosis, spinal cord injury, or other spinal cord pathologies. 6 It has been previous reported that baclofen can relieve neuropathic pain in patients who did not respond to opioid treatment. 9 Furthermore, baclofen in combination with carbamazepine have been shown to alleviate trigeminal neuralgia in human patients. 10 More recently, it has been demonstrated that activation of GABAB receptors by endogenous GABA regulates nociceptor excitability at peripheral sites of visceral afferent nerves. 11 However, mechanisms underlying GABAB-mediated suppression of nociceptor excitability remain incompletely understood.
TREK-2 channels belong to the two-pore domain K+ (K2P) channel family which mediate “leak” or “background” K+ currents across cell membranes.12–15 K2P channels play a major role in setting resting membrane potential and membrane input resistance, 12 and are involved in regulating sensory transduction, afferent neuron excitability and nociception. 14 Furthermore, mutations of K2P channels have been found to result in abnormal functions of central and peripheral nervous systems in humans.16,17 nterestingly, TREK-2 channel activity is found to be significantly enhanced in cortical neurons following GABAB activation by baclofen. 18 The activation of TREK-2 channels by GABAB is shown to be mediated by intracellular signaling pathways involving inhibitory G proteins (Gi/o) and cyclic adenosine monophosphate (cAMP), and the ultimate result is the inhibition of neuronal excitability. 18 Immunochemical studies have shown that TREK-2 channels are expressed in a subpopulation of small-sized DRG15,19 and TG neurons. 20 Interestingly, functional K2P channels in small-sized TG neurons have been shown to become downregulated following masseter muscle inflammation in rats, which is thought to be an underlying mechanism of hyperalgesia. 21 Small-sized afferent neurons in maxillary or V2 region of TGs are mainly nociceptors which innervate orofacial regions. If TREK-2 channels are expressed on these nociceptors, GABAB activation may increase TREK-2 activity to subsequently suppress nociceptor excitability. In the present study, we have tested this hypothesis with rat trigeminal ganglions using immunohistochemical as well as patch-clamp recording techniques.
Materials and methods
Animals: Sprague-Dawley rats of both sexes at ages of 10 to 13 weeks were used. Animal care and use conformed to NIH guidelines, and experimental protocols were approved by the Institutional Animal Care and Use Committee (IACUC) of the University of Alabama at Birmingham.
Ex vivo TG preparations: Ex vivo TG preparations were made in the manner described in our previous study. 22 In brief, TGs were rapidly dissected out from animals and placed in ice cold Krebs solution that contained (in mM): 117 NaCl, 3.5 KCl, 2.5 CaCl2, 1.2 MgCl2, 1.2 NaH2PO4, 25 NaHCO3, and 11 glucose. Connective tissues on the surface of the TGs were carefully removed with a pair of fine forceps. TGs were then affixed in a recording chamber by a tissue anchor and submerged in Krebs solution. Krebs solution was saturated with 95% O2 and 5% CO2, had pH of 7.35 and osmolarity of 324 mOsm, and at room temperature of 24 °C. TGs were exposed to 0.025% dispase II and 0.025% collagenase in Krebs solution for 8–10 min, and then TGs were continuously perfused at 2 ml/min with Krebs solution.
FITC-conjugated IB4 labeling: To pre-identify IB4-positive TG neurons for patch-clamp recordings, each TG was incubated with IB4 that was conjugated with fluorescein isothiocyanate (FITC) (IB4-FITC, Sigma). The TG was incubated with IB4-FITC (25 µg/ml in 200 µl Krebs solution) at 24 °C for 30 min to allow IB4-FITC binding to a subpopulation of TG neurons that had membrane binding sites for IB4. The TG was then affixed in a recording chamber and submerged in Krebs solution.
Patch-clamp recordings: Following IB4-FITC labelling, TG preparation was continuously perfused with Krebs solution at 24 °C. Under a fluorescent microscope, TG neurons labeled with IB4-FITC, i.e. IB4-positive neurons, in V2 region of the TG were identified. Then under the DIC-IR objective whole-cell patch-clamp recordings were performed on small-sized IB4-positive maxillary TG neurons. In a different set of experiments, patch-clamp recordings were performed in small-sized maxillary TG neurons that were not labeled by IB4-FITC (i.e., IB4-negative neurons). The recording electrode resistance after filling recording electrode internal solution ranged from 4 to 6 MΩ. Recording electrode internal solution contained (in mM): 105 K-gluconate, 35 KCl, 0.5 CaCl2, 2.4 MgCl2, 5 EGTA, 10 HEPES, 5 Na2ATP and 0.33 GTP-TRIS salt; the pH of the solution was adjusted to 7.35 with KOH. The junction potential between recording electrode internal solution and Krebs solution was 12.3 mV as calculated based on ionic concentrations by using the pCLAMP 11 software (Molecular Devices). After establishing whole-cell access, recordings were performed under voltage-clamp configuration with neurons held at −82.3 mV (voltage command of −70 mV). Unless otherwise indicated, membrane voltages mentioned in the texts for this set of experiments have been corrected for the calculated junction potentials of +12.3 mV. Cells would be discarded if membrane seal currents were negative than −80 pA after establishing whole-cell access. To determine membrane excitability of IB4-postive and IB4-negative maxillary TG neurons, under current-clamp configuration step currents were applied to neurons from −50 pA to 2200 pA with an increment of 25 pA per step and a duration of 500 ms each step. Neuronal excitability was also tested by applying ramp currents from 0 pA to 2000 pA over 1 s under current-clamp configuration. To test the effects of baclofen on electrophysiological properties of IB4-postive and IB4-negative maxillary TG neurons, the aforementioned patch-clamp recording experiments were performed before (control) and following the bath application of 100 µM baclofen for 15 min. Neuronal excitability was characterized by action potential threshold, rheobase, and number of action potentials evoked by a twofold rheobase step current or by a ramp current. Other electrophysiological parameters analyzed included cell capacitance, input resistance, resting membrane potential, and AP amplitude and width. Input resistance was measured with a −10 mV voltage step from membrane holding voltage of −82.3 mV under voltage-clamp configuration or with a −25 pA current step from resting membrane potential under current-clamp configuration. AP amplitude was measured from resting membrane potential to AP peak. AP half width was measured as the duration from 50% AP upstroke to 50% AP repolarization. Signals of patch-clamp experiments were amplified using a Multiclamp 700B amplifier, filtered at 2 kHz, and sampled at 10 kHz (voltage-clamp recordings) or 50 kHz (current-clamp recordings) using pCLAMP 10 software (Molecular Devices).
Immunohistochemistry
TGs were harvested from rats for immunohistochemical staining to characterize TREK-2 channel expression and IB4-FITC staining in TG neurons. In brief, animals were euthanized with overdose isoflurane followed by decapitation, and TGs were rapidly dissected out and fresh frozen on dry ice. The TGs were then embedded in OCT® compound (Baxter Scientific) and cut on a cryostat (Leica Biosystems, Buffalo Grove, IL, USA) into 16 μm sections. TG sections were thaw-mounted onto slides and allowed for air-dry at room temperature of 24 °C for 6 min. The slide-mounted sections were dipped in pre-cooled acetone in a −20°C freezer for 10 min, and then air-dried at room temperature for 30 min. TG sections were then encircled with hydrophobic resin (PAP Pen – The Binding Site), and rinsed 3 times with PBS, 10 min each time, at room temperature. TG sections were incubated with 10% normal goat serum (Cat# 017–000-12, Jackson ImmunoResearch) in PBS for 30 min at room temperature for blocking nonspecific binding of antibodies. TG sections were incubated with a mixture of a polyclonal rabbit anti-TREK-2 antibody 19 (1:500 diluted with 5% normal goat serum in PBS, Cat# APC-055, Alomone Labs, Israel) and IB4-FITC (1:500 diluted from 1 mg/ml stock solution with 5% normal goat serum in PBS, Cat# L2895, Sigma, USA) overnight at 4 °C. Following 3 times of rinses each for 10 min with PBS solution at room temperature, TG sections were incubated with a secondary antibody for 1 hr at room temperature. The secondary antibody was a goat anti-rabbit IgG conjugated with Alexa-594 (1:1000 in 5% normal goat serum in PBS, Cat# A-11012, ThermoFisher Scientific, Waltham, MA USA). TG sections were rinsed 3 times with PBS solution, cover-slipped with the Prolong Diamond Antifade Mountant medium (Cat# P36965, ThermoFisher Scientific). TG sections were viewed under an upright fluorescent microscope (BX-43, Olympus, Tokyo, Japan) and images captured in V2 regions of TGs using a CCD camera (DP80, Olympus, Tokyo, Japan).
Data analysis
In the present study TGs were obtained from both male and female rats, and data from both sexes were pooled together since we did not observe any sex differences in our results. For the data of patch-clamp recording experiments, parameters of electrophysiological properties were analyzed using Clampfit 10 software. For the data of TREK-2 immunostaining, TREK2-ir positive neurons and TREK2-ir negative neurons in V2 TG regions were visualized from the fluorescent images acquired by the Olympus Cell Sens image software (Olympus, Tokyo, Japan). A neuron was considered as TREK-2-ir positive when the fluorescent intensity signal to background noise ratio was ≥ 3:1. For TREK-2-negative cells, they could be clearly differentiated from background and were directly counted with good confidence without using an additional Marker. The total numbers of TG neurons in a field were the sum of TREK-2-ir positive and TREK-2-ir negative neurons. For the data of IB4-FITC staining, IB4-positive neurons could be clearly differentiated from background, and counting IB4 positive neurons was performed in the similar manner as that for TREK-2-ir positive neurons. IB4-negative neurons could not be confidentially differentiated from background and cell counting for IB4-negative neurons could not be made directly. However, since all cells in a field can be visualized in TREK-2 staining, the number of IB4-negative neurons could be counted from the overlay image of TREK-2 staining and IB4 staining. Unless otherwise indicated, all data are reported as mean ± SEM of n independent observations. Statistical comparisons were performed using GraphPad Prism 8 software with *p < 0.05, **p < 0.01, and ***p < 0.001, paired Wilcoxon test or Chi-square test.
Results
Maxillary TG neurons are the afferent neurons whose somas are located in V2 region of the TG. These neurons give rise to afferent fibers primarily innervating orofacial regions. 23 Under an IR-DIC microscope, individual neurons in V2 region could be visualized in our ex vivo TG preparations. Many maxillary TG neurons were small-sized cells with mean diameters smaller than 35 µm (Figure 1(a), left panel). In the present study, we intended to perform recordings from both IB4-positive and small-sized IB4-negative maxillary TG neurons. To achieve this aim, we used IB4-FITC to vital-label IB4-positive neurons in the ex vivo TG preparations (Figure 1(a), right panel). Patch-clamp recordings were then performed on small-sized IB4-positive neurons and small-sized IB4-negative neurons under the visual guidance. Each IB4-positive maxillary TG neuron recorded in the present study exhibited a shoulder in its AP repolarization phase, an electrophysiological signature associated with nociceptors (Figure 1(b), left). IB4-positive maxillary TG neurons sampled for recordings in the present study had diameters of approximately 22 µm (Table 1). We also performed recordings from IB4-negative maxillary TG neurons with cell sizes comparable to those of IB4-positive neurons (Table 1). Many small-sized IB4-negative maxillary TG neurons recorded in the present study also displayed the shoulders in their AP repolarization phase (Figure 1(b), middle). We termed them IB4-negative nociceptive-like maxillary TG neurons in the present study. Some small-sized IB4-negative maxillary TG neurons did not display shoulders in their AP repolarization phases and their AP widths were narrow (Figure 1(b), right, Table 1). These neurons were termed IB4-negative non-nociceptive-like maxillary TG neurons in the present study.

Baclofen hyperpolarizes resting membrane potentials of IB4-positive but not IB4-neagtive small-sized maxillary TG neurons. (a) Images show bright field (left) and fluorescent field (right) of a V2 region in an ex vivo TG preparation. The TG preparation was vital-stained with IB4-FITC. Image was taken under 4X objective. Arrow indicates an IB4-positive (IB4+) small-sized maxillary TG neuron, and asterisk indicates an IB4-negative (IB4-) small-sized maxillary TG neuron. Scale bar: 20 µm. (b) APs recorded from an IB4-positive neuron (left), an IB4-negative nociceptive-like small-sized neuron (middle), and an IB4-negative non-nociceptive-like small-sized neuron (right). (c–e) Summary data of resting membrane potentials before (control, open circles) and following the application of 100 µM baclofen in IB4-postive neurons (c, n = 23), IB4-negative nociceptive-like neurons (d, n = 11), and IB4-negative non-nociceptive-like neurons (e, n = 10). Data represent Mean ± SEM, ns, not significantly different, *P < 0.05, paired Wilcoxon test.
We first investigated whether baclofen may affect passive membrane properties in the three types of small-sized maxillary TG neurons. Interestingly, baclofen hyperpolarized resting membrane potentials in IB4-positive maxillary TG neurons (Figure 1(c)), but not IB4-negative nociceptive-like (Figure 1(d)) and IB4-negative non-nociceptive-like maxillary TG neurons (Figure 1(e)). In IB4-positive maxillary TG neurons, resting membrane potentials were −72.6 ± 1.4 mV (n = 23) in the absence of baclofen and significantly hyperpolarized to −78.5 ± 2.9 mV (n = 23, P < 0.05) following the bath application of 100 µM baclofen (Figure 1(c)). In contrast, in IB4-negative nociceptive-like maxillary TG neurons, resting membrane potentials (-70.63 ± 1.5 mV, n = 11) in the absence of baclofen were not significantly different from those (−68.6 ± 2.3 mV, n = 11) following the bath application of 100 µM baclofen (Figure 1(d)). In IB4-negative non-nociceptive-like maxillary TG neurons, resting membrane potentials (-77.2 ± 0.7 mV, n = 10) in the absence of baclofen were also not significantly different from those (-75 ± 1.4 mV, n = 10) following the bath application of 100 µM baclofen (Figure 1(e)).
We next determined whether baclofen may affect leak current and membrane input resistance in IB4-positive, IB4-negative nociceptive-like, and IB4-negative non-nociceptive-like maxillary TG neurons. Leak current and membrane input resistance are two passive membrane parameters directly associated with the activities of K2P channels. 18 As shown in Figure 2(a) and (b), leak current measured at holding potential of −82.3 mV in IB4-positive maxillary TG neurons was −27.3 ± 6.7 pA (n = 23) in the absence of baclofen and showed significantly less negative (-2.6 ± 15.1 pA, n = 23, P < 0.01, Figure 2(b)) following the bath application of 100 µM baclofen. This change indicated that baclofen enhanced outward leak currents. However, leak currents in IB4-negative nociceptive-like (n = 11, Figure 2(c)) and IB4-negative non-nociceptive-like neurons (n = 10, Figure 2(d)) were not significantly affected by 100 µM baclofen. Consistent with the effects on leak current, membrane input resistance was significantly reduced by baclofen in IB4-positive maxillary TG neurons, but not in IB4-negative nociceptive-like and IB4-negative non-nociceptive maxillary TG neurons. In IB4-positive maxillary TG neurons, membrane input resistance was 382.5 ± 40.41 MΩ (n = 18) in the absence of baclofen and significantly decreased to 300.9 ± 27.2 MΩ (n = 18, P < 0.01) following the bath application of 100 µM baclofen (Figure 2(e)). In IB4-negative nociceptive-like maxillary TG neurons, membrane input resistance was 348.5 ± 43.5 MΩ (n = 11) in the absence of baclofen, and 364.5 ± 60.9 MΩ (n = 11) following the bath application of 100 µM baclofen (Figure 2(f)), and was not significantly different. In IB4-negative non-nociceptive-like maxillary TG neurons, membrane input resistance was 121.6 ± 13.2 MΩ (n = 10) in the absence of baclofen, and 124.1 ± 11.3 MΩ (n = 10) following the bath application of 100 µM baclofen, and was also not significantly different (Figure 2(g)).

Baclofen enhances outward leak currents and decreases membrane input resistance in IB4-positive but not IB4-negative small-sized maxillary TG neurons. (a) Two sets of sample traces show leak currents and membrane responses to testing pulses of ± 10 mV in an IB4-positive maxillary TG neuron. Left panel is before (control) and right panel following of application of 100 µM baclofen. Holding potential was -82.3 mV. (b–d) Summary data of leak currents before (control, open circles) and following the application of baclofen (closed circles) in IB4-positive neurons (b, n = 23), IB4-negative nociceptive-like neurons (c, n = 11), and IB4-negative non-nociceptive-like neurons (d, n = 10). Note that in B leak currents become less negative following baclofen application, which indicates an enhancement of outward leak currents. (e–g) Summary data of membrane input resistance before (control, open circles) and following the application of baclofen (closed circles) in IB4-positive neurons (e, n = 23), IB4-negative nociceptive-like neurons (f, n = 11), and IB4-negative non-nociceptive-like neurons (g, n = 10). Data represent Mean ± SEM, ns, not significantly different, **P < 0.01, paired Wilcoxon test.
We investigated effects of baclofen on AP properties in IB4-positive, IB4-negative nociceptive-like, and IB4-negative non-nociceptive-like maxillary TG neurons. In IB4-positive neurons, AP rheobase and threshold were not significantly different between control in the absence of baclofen and following the bath application of 100 µM baclofen (Table 1). However, AP amplitude was significantly decreased from 101.9 ± 2.3 mV (n = 20) in the absence of baclofen to 92.8 ± 3.4 mV (n = 20, P < 0.01) following the bath application of 100 µM baclofen (Table 1). AP half width was significantly prolonged from 6.4 ± 1.1 ms (n = 20) in the absence of baclofen to 10.0 ± 1.2 ms (n = 20, P < 0.01, Table 1) following the bath application of 100 µM baclofen. In contrast to IB4-positive maxillary TG neurons, none of the aforementioned AP parameters were significantly affected by baclofen in IB4-negative nociceptive-like (n = 11, Table 1) and IB4-negative non-nociceptive-like neurons (n = 10, Table 1).
Effects of baclofen on the aforementioned electrophysiological properties of IB4-positive maxillary TG neurons may be mediated by TREK-2 channels since a previous study 18 showed similar effects of baclofen that were mediated by TREK-2 channels on cortical neurons. This raised the question of whether TREK-2 channels are expressed in IB4-positive maxillary TG neurons. To address this possibility, we performed immunostaining for TREK-2 proteins with a TREK-2 antibody and IB4 staining with IB4-FITC to see if TREK-2 immunoreactivity (TREK-2-ir) was co-localized with IB4 staining in maxillary TG neurons. As shown in Figure 3, both TREK-2-ir and IB4 staining were shown to be present in a subpopulation of small-sized maxillary TG neurons (Figure 3(a) and (g)). In a total of 1445 maxillary TG neurons examined (11 TG sections), 304 neurons (21%) were TREK-2-ir positive (Figure 3(f) and (h)) and 466 neurons (32%) were IB4-positive (Figure 3(g) and (h)). Mean diameters were 17.9 ± 0.3 µm for TREK-2-ir positive neurons and were 17.6 ± 0.3 µm for IB4 positive neurons (Figure 3(g) and (h)). TREK-2-ir largely overlapped with IB4 staining in maxillary TG neurons (Figure 3(a) to (e) and (i)). Of all 304 TREK-2-ir positive neurons in 11 TG sections, 72.8 ± 3.8% (n = 11) of them were co-stained positively with IB4-FITC. For all 466 IB4-positive neurons in 11 TG sections, nearly half of them (45.9 ± 2.6%, n = 11) were co-stained with IB4. Thus, TREK-2 channels are preferentially expressed in IB4-positive maxillary TG neurons.

TREK-2 channels are preferentially expressed on IB4-positive small-sized maxillary TG neurons. (a, b) Fluorescent images show immunostaining for TREK-2 channels (a) and IB4-FITC staining (b) in the same maxillary region of a TG section. Scale bar: 50 µm. (c) Image in the box in A at expanded scale. (d) Image in the box in B at expanded scale. (e) Overlay image of (c) and (d). Scale bar: 20 µm. (f) and (g) Histogram of cell size distribution of TREK-2 immnoreactivity positive (f, n = 304) and IB4 positive (g, n = 466) maxillary TG neurons. (h) Percent of TREK-2-ir positive neurons (red bar, n = 11 TG sections) and IB4 positive neurons (green bar, n = 11 TG sectons) in total maxillary TG neurons. (i) Percent of neurons co-localized with TREK-2-ir and IB4 staining in all TREK-2-ir positive neurons (left bar, n = 11 TG sections) or in all IB4-positive neurons (right bar, n = 11 TG sections). Data represent Mean ± SEM.
Lastly we used patch-clamp recordings and under the current-clamp configuration to investigate whether baclofen may affect action potential (AP) firing in IB4-positive maxillary TG neurons. In this set of experiments, AP firing numbers and firing patterns in the three types of maxillary TG neurons were examined in the absence and following the bath application of 100 µM baclofen. As shown in Figure 4(a) and (b), with 2x rheobase step current stimulation for 500 ms, IB4-positive maxillary TG neurons on average fired 3.4 ± 0.4 (n = 20) APs in the absence of baclofen and AP firing became significantly reduced to 1.9 ± 0.3 (n = 20) following the bath application of 100 µM baclofen (Figure 4(b)). In contrast, AP firing with 2x rheobase step current stimulation were not significantly different between control in the absence of baclofen and following the bath application of 100 µM baclofen in IB4-negative nociceptive-like (n = 11, Figure 4(c)) and IB4-negative non-noniceptive-like maxillary TG neurons (n = 10, Figure 4(d)). The above results were based on averaged AP firing numbers without taking AP firing patterns into consideration. It was noted that some cells fired multiple APs and others could only fire a single AP in response to the step current stimulation. Therefore, we analyzed the effect of baclofen on AP firing patterns (i.e. single AP vs multiple APs) in the three types of small-sized maxillary TG neurons. As shown in Figure 5(a) to (c), while most IB4-positive neurons tested fired multiple APs (n = 15), some only fired a single AP in control in the absence of baclofen (n = 5). Following the bath application of 100 µM baclofen, fewer IB4-positive neurons fired multiple APs (n = 8) and more IB4-positive neurons fired single AP (n = 12), and the changes were significantly different (P < 0.05, Figure 5(c)). In IB4-negative nociceptive-like maxillary TG neurons, the majority fired multiple APs and the minority fired single AP in control, and baclofen did not have any significant effect on AP firing patterns (Figure 5(d)). In IB4-negative non-nociceptive-like maxillary TG neurons, the minority fired multiple APs and the majority fired single AP in control in the absence of baclofen, and baclofen also did not have any significant effect on their AP firing patterns (Figure 5(e)).

Baclofen suppresses AP firing evoked by step current injections in IB4-positive but not IB4-negative small-sized TG neurons. (a) Two sets of sample traces show AP firing at rheobase and 2x rheobase in a IB4-positive neurons before (control, left panel) and following the application 100 µM baclofen (right). (b–d) Summary data of AP numbers at 2x rheobase before (control, open bars) and following the application of baclofen (closed bars) in IB4-positive neurons (b, n = 20), IB4-negative nociceptive-like neurons (c, n = 11), and IB4-negative non-nociceptive-like neurons (d, n = 10). Data represent Mean ± SEM, ns, not significantly different, **P < 0.01, paired Wilcoxon test.
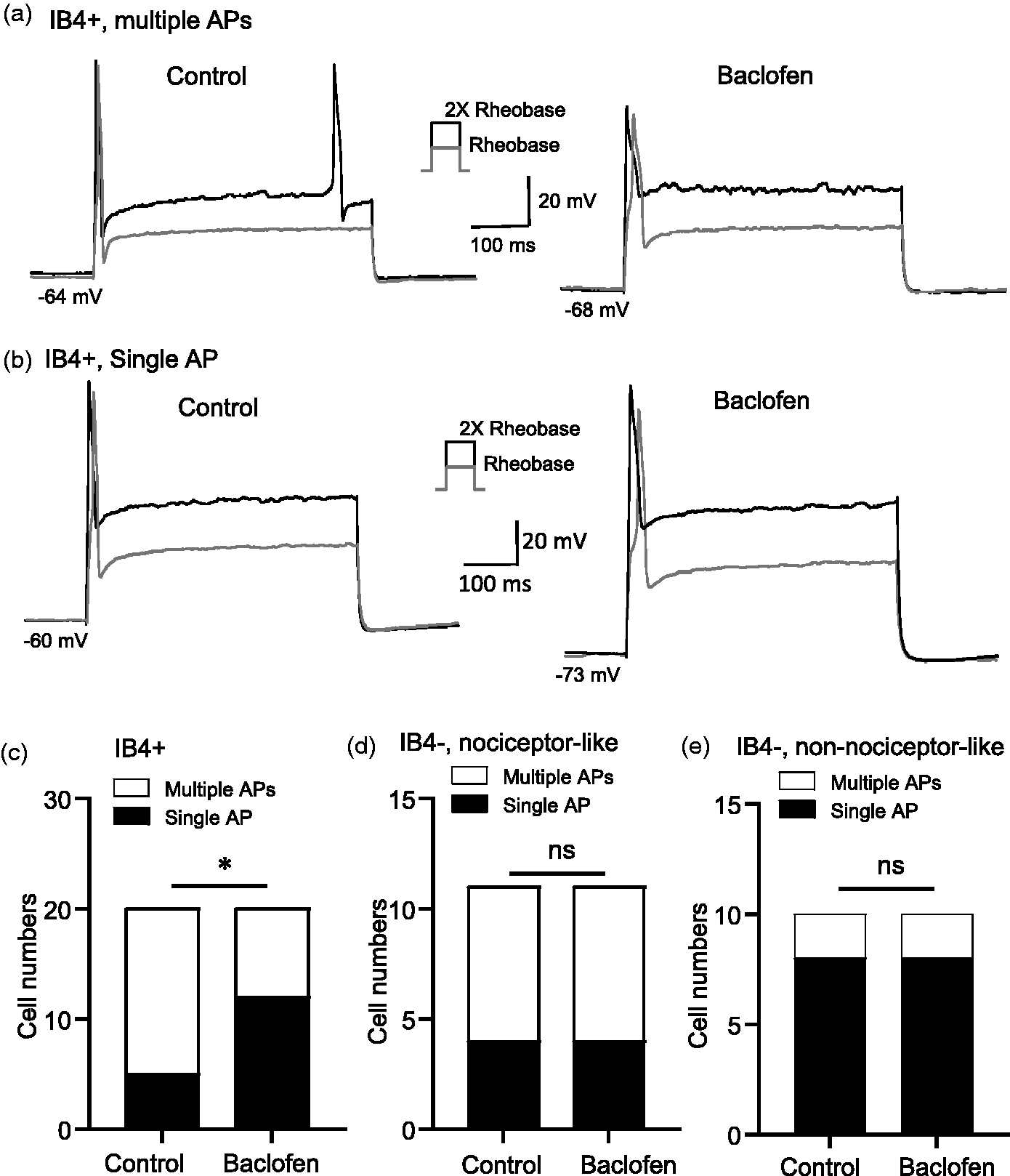
Baclofen alters AP firing patterns evoked by step current injections in IB4-positive but not IB4-negative small-sized TG neurons. (a) Two sets of sample traces show an IB4-positive neuron firing multiple APs before (control, left panel) and firing only a single AP following the application 100 µM baclofen (right). (b) Two sets of sample traces show an IB4-positive neuron firing single AP before (control, left panel) and firing single AP following the application 100 µM baclofen (right). (c–f) Summary data of single AP (open bars) and multiple AP (closed bars) firing patterns at 2x rheobase before (control) and following the application of 100 µM baclofen in IB4-positive neurons (c, n = 20), IB4-negative nociceptive-like neurons (d, n = 11), and IB4-negative non-nociceptive-like neurons (e, n = 10). Data represent cell numbers, ns, not significantly different, *P < 0.05, Chi-square tests.
In the above experiments shown in Figures 4 and 5, AP firings were evoked by step currents. Since physiologically relevant stimuli usually produce relatively slow membrane depolarization, we examined AP firings evoked by ramp currents and tested the effect of baclofen on AP firings. The ramp currents were injected into neurons from 0 pA to 2000 pA in 1 s. For IB4-positive neurons in response to the ramp stimulation, the majority fired APs (n = 12) but some failed to fire any AP (n = 8) in control before baclofen application (Figure 6(a) and (b)). Following the bath application of 100 µM baclofen, most of them failed to fire any AP (n = 14) and only the minority fired APs (n = 6, P = 0.056, Figure 6(b)). For IB4-negative nociceptive-like neurons, the majority fired APs (n = 9) but 2 of them failed to fire any AP in control before baclofen application, and the firing patterns remained unchanged following the application of 100 µM baclofen (Figure 6(c)). For IB4-negative non-nociceptive neurons, there was also no significant change in the numbers of cells firing APs (Figure 6(d)). Pooled results of AP numbers were analyzed and compared between the control in the absence of baclofen and following the application of 100 µM baclofen (Figure 6(e) to (g)). Overall, the averaged AP numbers evoked by the ramp stimulation for all IB4-positive neurons tested were 5.0 ± 1.3 (n = 20) in control and significantly reduced to 1.4 ± 0.4 (n = 20) following the bath application of 100 µM baclofen. On the other hand, for both IB4-negative nociceptive-like (Figure 6(f), n = 11) and IB4-negative non-nociceptive-like neurons (Figure 6(g), n = 10), there was no significant difference in AP numbers between control in the absence of baclofen and following the bath application of 100 µM baclofen.

Baclofen alters AP firing in response to ramp current injections in IB4-positive but not IB4-negative small-sized TG neurons. (a) Two sample traces show an IB4-positive neuron firing APs in response to a ramp current stimulation before (control, left panel) and following the application 100 µM baclofen (right). (b–d) Summary data of AP firing numbers before (control, open circles) and following the application of 100 µM baclofen (closed circles) in IB4-positive neurons (b, n = 20), IB4-negative nociceptive-like neurons (c, n = 11), and IB4-negative non-nociceptive-like neurons (d, n = 10). (e–g) Summary data of numbers of neurons firing APs (open bars) or without firing APs (closed bars) before (control) and following the application of 100 µM baclofen in IB4-positive neurons (e, n = 20), IB4-negative nociceptive-like neurons (f, n = 11), and IB4-negative non-nociceptive-like neurons (g, n = 10). Data represent cell numbers, ns, not significantly different, *P < 0.05, Chi-square tests.
Discussion
In the present study we have provided immunohistochemical evidence showing that TREK-2 channels are predominantly expressed in IB4-positive maxillary TG neurons. We have further provided electrophysiological evidence indicating that the GABAB receptor agonist baclofen selectively alters electrophysiological properties reducing excitability of IB4-positive maxillary TG neurons. More importantly, we have found that the selective effect by baclofen on the excitability of IB4-positive maxillary TG neurons is closely linked with the changes of passive membrane properties controlled by K2P channel activity.
The predominant expression of TREK-2 channels on IB4-positive maxillary TG neurons is generally consistent with a previous study showing that TREK-2-ir and IB4 staining are co-localized in a subpopulation of rat IB4-positive DRG neurons. 19 However, the co-localization of TREK-2-ir and IB4 staining in DRG neurons was more than 95% in the previous study 19 but substantially less in TG neurons in the present study. This may be due to the differences in defining positive staining between our study and the previous study. 19 In our study, positive staining was considered only when signal intensity was ≥ 3 fold of background so that only strong positive staining was considered as TREK-2 expression and/or IB4 positive neurons. TREK-2-ir positive neurons were approximately 21% in total maxillary TG neurons in the present study, consistent with our previous study performed in rat DRG neurons. 15 IB4-positive neurons are approximately 30% in total maxillary TG Neurons in the present study, consistent with a previous study in rat TG neurons. 24
In our patch-clamp recording experiments, we used IB4-FITC vital staining in ex vivo TG preparations to label IB4-positive maxillary TG neurons for visual guidance. IB4-FITC vital staining has been used previously for patch-clamp recordings from dissociated afferent neurons, and is not reported to affect electrophysiological properties of afferent neurons. 25 Therefore, the differential effects of baclofen on electrophysiological properties between IB4-positive and IB4-negative neurons shown in the present study is unlikely due to IB4-FITC staining. We have performed patch-clamp recordings from maxillary TG neurons at 24 °C since at this temperature cell membranes can be maintained heathy for long time recordings. Healthy membranes are important in our studies on leak K+ currents mediated by K2P channels. It should be noted that TREK-2 channels are temperature-sensitive leak K+ channels thereby leak currents measured at 24 °C would be smaller than those measured at higher temperatures.
Three groups of neurons, IB4-positive, IB4-negative nociceptive-like, and IB4-negative non-nociceptive like maxillary TG neurons, are tested with baclofen in the present study. Previous studies have indicated that IB4-positive afferent neurons are mainly non-peptidergic C-fiber nociceptors, and peptidergic C-fiber nociceptors are mostly IB4-negative afferent neurons. 2 Therefore, in the present study IB4-positive maxillary TG neurons are most likely non-peptidergic C-fiber nociceptors, and IB4-negative nociceptive-like maxillary TG neurons are most likely peptidergic C-fiber nociceptors. However, we cannot exclude the possibility that some IB4-negative nociceptive-like maxillary TG neurons are actually C-fiber low threshold mechanoreceptors (C-LTMRs) since C-LTMRs are IB4-negative with shoulders in their repolarization phases. 26 In our study, IB4-negative non-nociceptive-like maxillary TG neurons have relatively larger membrane capacitance and shorter AP widths, raising the possibility that they may be Aδ-LTMR. 26 In the three types of maxillary TG neurons tested in the present study, we show that the GABAB agonist baclofen selectively acts on IB4-positive maxillary TG neurons to alter their electrophysiological properties reducing excitability. This result is consistent with the expression of GABAB receptors in afferent neurons, 27 particularly in P2X3 receptor-expressing and IB4-positive TG neurons. 28 Suppression of AP firing in IB4-postive maxillary TG neurons by baclofen is at least partially due to baclofen-induced hyperpolarization of resting membrane potentials and reduction of input resistance. GABAB-receptors are coupled with Gi/o-proteins in DRG neurons,29,30 and it has been shown in cortical neurons that GABAB-receptor activation by baclofen stimulates Gi/o-proteins and cAMP signaling pathways to enhance TREK-2 channel activity, which in turn suppress neuronal excitability. 18 This may also be the mechanism underlying the effect of baclofen on IB4-positive maxillary TG neurons in the present study, but this possibility remains to be validated in future studies. In a recent study, it has been shown that GABAB activation affects TRPV1 activity in a manner independent of canonical G protein signaling, but relies on direct interactions between GABAB1 receptor subunits and TRPV1 receptors. 31 It would be interesting to investigate whether GABAB-receptor subunits may have direct interaction with TREK2-channels to affect their activity.
Membrane and action potential parameters before and following bath application of baclofen.

**P < 0.01.The membrane and action potential parameters were measured before (control) and 15 min following the continuous bath application of 100 µM baclofen. AP: action potential; Cm: membrane capacitance; IB4+: IB4-positive neurons; IB4–: IB4-negative TG neurons. Comparisons are made between the control and following baclofen application using the Wilcoxon test.
Baclofen has been clinically used for treating patients with neuropathic pain including trigeminal neuralgia. 10 Baclofen mediated suppression of IB4-positive maxillary TG neuron excitability, possibly through TREK-2 channel activation, provides a new mechanism underlying the therapeutic uses of Baclofen for pain management. Preferential expression of TREK-2 channels in IB4-positive afferent neurons offers a potentially selective therapeutic target for managing pain with restricted side-effects. In addition to baclofen, compounds which can directly activate TREK-2 channels 12 may also be therapeutically useful for managing pathological pain.
Footnotes
Acknowledgments
We thank Dr Ryan Vaden for reading and commenting on the manuscript.
Author Contributions
JGG conceived and designed the experiments and wrote the paper. YC performed electrophysiology experiments, and JL performed immunostaining experiments.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by NIH grants DE018661 and DE023090 to JGG.
