Abstract
Background
Rotator cuff tears (RCTs) are often associated with severe shoulder pain. Non-steroidal anti-inflammatory drugs, not recommended for long-term use, do not effectively manage RCT-induced pain, resulting in reduced quality of life. To improve management, a better understanding of the fundamental properties of RCT pain is needed. Here, we aimed to compare the expression levels of nerve growth factor (NGF) and cyclooxygenase-2 (COX-2) mRNA in the synovial tissues of patients with RCT-induced pain and patients with non-painful recurrent shoulder dislocation (RSD).
Methods
The study included 32 patients with RCT who underwent arthroscopic rotator cuff repair and 28 patients with non-painful RSD who underwent arthroscopic Bankart repair. Synovial tissue samples were harvested from subacromial bursa and rotator interval of RCT patients and from the rotator interval of RSD patients. Samples were analyzed quantitatively expression levels for NGF and COX2 mRNA and NGF protein.
Results
NGF mRNA and protein levels were significantly higher in the rotator interval of RCT patients than in the rotator interval of RSD patients (p = 0.0017, p = 0.012, respectively), while COX2 mRNA levels did not differ significantly between the two patient groups. In RCT patients, COX2 mRNA was more highly expressed in the rotator interval than in the subacromial bursa (p = 0.038), whereas the mRNA and protein levels of NGF did not differ between the two tissues. The expression of NGF mRNA in the synovium of the rotator interval was significantly correlated with the numeric rating scale of pain (ρ = 0.38, p = 0.004).
Conclusion
NGF mRNA and protein levels were elevated in patients with painful RCT compared with those in patients with non-painful RSD, whereas COX-2 levels were comparable in the two patient groups. These findings provide insights into novel potential strategies for clinical management of RCT.
Introduction
Rotator cuff tears (RCTs) increase with age1,2 and are often associated with severe shoulder pain that leads to a reduced quality of life. However, the size of a RCT is not associated with pain severity, 3 similar to many other painful musculoskeletal pain conditions. 4 Although the population of patients with asymptomatic RCT is notably larger than that with symptomatic RCT,1,2 the mechanisms underlying why some RCT patients experience pain while others are pain-free, remain unclear.
Anti-inflammatory medications, such as non-steroidal anti-inflammatory drugs (NSAIDs) and corticosteroids, are used for pain control in RCT, 5 although long-term use of traditional NSAIDs is not recommended6–9 due to the risk of adverse effects.10,11 Steroid injection is another treatment modality for the management of pain in RCT.8,9,12 However, local steroid injections also include the risk of tendon injury progression and other adverse effects.13–16 Therefore, new therapeutic targets for painful RCT are needed.
Nerve growth factor (NGF), a member of the neurotrophin family of proteins, 17 is a modulator of pain that is regulated during injury, inflammation, and chronic pain conditions; and can facilitate pain.18–22 NGF localization is associated with joint and tissue injuries, such as osteoarthritic knees23,24 and degenerated intervertebral discs,25,26 both of which are often associated with pain. We recently reported that NGF levels are elevated in a rat RCT model, 27 and are observed in the synovial tissue of patients with RCT 28 ; however, a control experiment is needed to draw definitive conclusions. Moreover, considering that NGF monoclonal antibodies are currently being tested in various musculoskeletal conditions, 29 it is important to elucidate which conditions may represent suitable candidates for anti-NGF treatment, from a mechanistic standpoint.
The aim of this study was, therefore, to investigate NGF and cyclooxygenase-2 (COX-2) expression levels (using mRNA and protein expression) in synovial membrane tissues from patients with RCT and recurrent non-painful shoulder dislocation (RSD). To the best of our knowledge, this is the first study to examine and compare NGF expression levels in patients with RCT and RSD.
Materials and methods
Ethical approval and consent to participate
This study was approved by our institutional Ethics Review Board (reference number: KMEO B13-113). All participants (or parents, in case of kids) provided written informed consent prior to surgery. This study was registered in the clinical trial registry of our country (ID, UMIN000041077).
Patients
Samples were collected from 32 patients with painful RCT and 28 patients with non-painful RSD between October 2017 and November 2019. The clinical characteristics of patients are summarized in Tables 1 and 2. For evaluation of shoulder pain, the numeric rating scale (NRS) was used (none, 0; mild, 1–3; moderate, 4–7; and severe, >8). 30 If patients experienced negligible pain, such as feeling pain only with specific motions or during moments of heavy exertion, their pain was assessed as 1. Additionally, the Constant score 31 was used to evaluate clinical scores for RCT patients, while RSD patients were assessed using the Rowe score. 32 The Constant score consists of four variables used to assess shoulder function. The subjective variables are pain and activities of daily living (sleep, work, recreation/sport) which account for a total of 35 points. Meanwhile, the objective variables comprise range of motion and strength accounting for a total of 65 points. Alternatively, the Rowe score consists of 100 points divided into three variables, stability (0–50 points), mobility determined by range of shoulder motion (0–20 points) and function determined by patient activity (0–30 points).
Clinical characteristics of patients whose samples were used for qRT-PCR analysis.
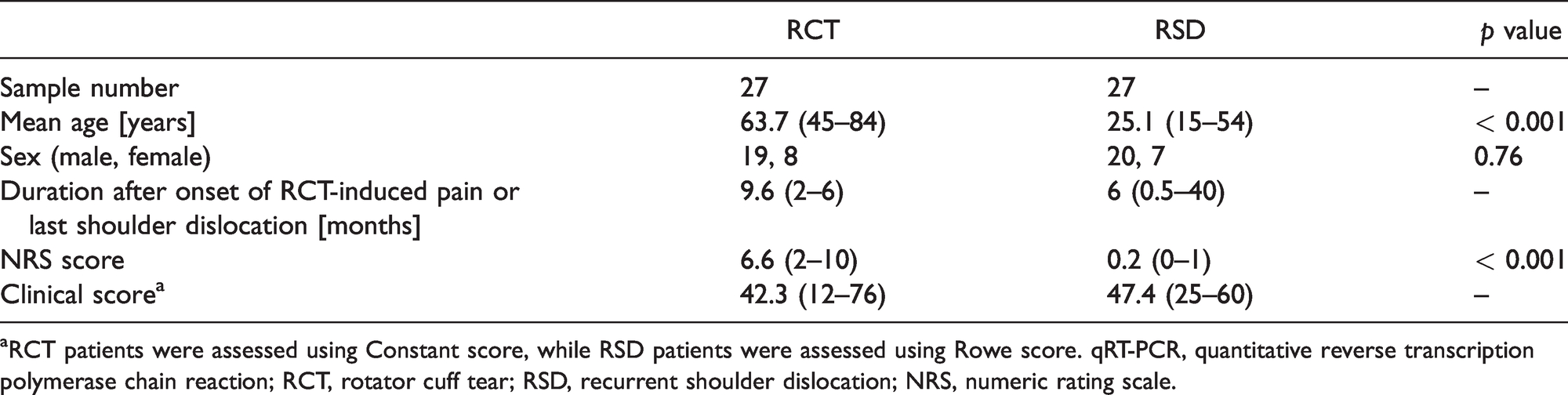
aRCT patients were assessed using Constant score, while RSD patients were assessed using Rowe score. qRT-PCR, quantitative reverse transcription polymerase chain reaction; RCT, rotator cuff tear; RSD, recurrent shoulder dislocation; NRS, numeric rating scale.
Clinical characteristics of patients who provided samples for western blotting.

aRCT patients were assessed using Constant score, while RSD patients were assessed using Rowe score. RCT, rotator cuff tear; RSD, recurrent shoulder dislocation; NRS, numeric rating scale.
Exclusion criteria for this study included: 1) diagnosed rheumatoid arthritis or other collagen diseases, 2) patients undergoing dialysis, 3) history of corticosteroid use including injection and/or oral formulations within one month of operation.
Synovial tissue harvesting and preparation
RCT patients
All patients underwent arthroscopic rotator cuff repair surgery performed by a single experienced shoulder surgeon (K.T.). Two synovial membrane samples were harvested from each RCT patient. One sample was obtained from the site of the most marked synovitis in the rotator interval (Figure 1(a) and (b)) and the other was obtained from a site in the subacromial bursa (Figure 1(a) and (c)) around a coracoacromial ligament where the greater tuberosity of the humeral head was usually impinged.

Scheme of the shoulder joint anatomy (a), where tissue was obtained from each group of patients. Representative arthroscopic view of intra articular (b) and subacromial space (c). *Synovial tissue of rotator interval; **Synovial tissue of subacromial bursa; †rotator cuff tear; HH, humeral head; Ac, acromion; SSc, subscapularis; SSp, supraspinatus; LHB, long head of biceps tendon.
RSD patients
All patients underwent arthroscopic Bankart repair by a single experienced shoulder surgeon (K.T.). Samples of the synovial membrane of the rotator interval were harvested from each RSD patient. Samples were not harvested from the subacromial space around a coracoacromial ligament in these patients as there is a possibility of iatrogenic pain in patients with RSD, and the institutional Ethics Review Board did not permit sampling from healthy or unaffected areas in these patients.
All samples were immediately frozen in liquid nitrogen and stored at −80°C until RNA extraction for quantitative reverse transcription polymerase chain reaction (qRT-PCR) and protein extraction for western blotting.
qRT-PCR analysis
To investigate differences in mRNA expression of NGF and COX2 between RCT and RSD patients, qRT-PCR analysis was performed on synovial membrane tissue obtained from each group (n = 27 per group). TRIzol (Invitrogen, Carlsbad, CA, USA) was used to extract total RNA from the synovial membrane, according to the manufacturer’s protocol. Total RNA was used as a template for first-strand cDNA synthesis using a SuperScript III RT (Invitrogen). A 25-µL reaction comprising 2 µL cDNA, a specific primer set (0.2 µM final concentration; Table 3), and 12.5 µL SYBR Premix Ex Taq (Takara, Shiga, Japan) were used for qRT-PCR. Primers for NGF and COX2 were designed using the Primer-Blast tool from NCBI, and synthesized by Hokkaido System Science Co., Ltd. (Sapporo, Japan). qRT-PCR was conducted on a CFX-96 Real-Time PCR Detection System (Bio-Rad, Hercules, CA, USA) using the following protocol: initial denaturation at 95°C for 1 min, 40 cycles at 95°C for 5 s, and 60°C for 30 s. The specificity of the primer-amplified products was confirmed using melting curve analysis, and NGF and COX2 mRNA expression was determined by normalizing to that of glyceraldehyde-3-phosphate dehydrogenase (GAPDH) using the delta-delta Ct method. 33 We then evaluated the correlation between gene expression and NRS.
Sequences of the primers used in this study.

F, forward; R, reverse; GAPDH, glyceraldehyde 3-phosphate dehydrogenase; NGF, nerve growth factor; COX2, cyclooxygenase-2.
Western blotting
To investigate differences in NGF abundance between RCT and RSD patients, western blotting was performed on synovial membrane obtained from both study groups (n = 5 per group). Synovial membranes were lysed in radioimmune precipitation buffer (Wako Pure Chemical Co., Inc., Osaka, Japan) containing a protease inhibitor cocktail (Roche, Madison WI, USA), and the protein concentration in each lysate was quantified using the bicinchoninic acid (BCA) assay (Pierce, Rockford, Illinois, USA). Proteins (10 µg/lane) were separated by sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) and were electro-transferred onto polyvinylidene fluoride (PVDF) membranes (Pierce), which were then blocked with PVDF blocking reagent (DS Pharma Biomedical, Suita, Japan) for 1 h at room temperature (22‒25 °C). The blocked membranes were incubated overnight at 4°C with rabbit polyclonal primary antibodies against NGF (cat. no: ab52918; Abcam) diluted 1:1000 in blocking reagent (ImmunoBlock, DS Pharma). The membranes were washed with phosphate-buffered saline (PBS, pH 7.4) containing 0.05% Tween and incubated with secondary antibodies (GE Healthcare, Piscataway NJ, USA) diluted 1:1000 in blocking reagent. Immunoreactive proteins were visualized based on chemiluminescence using an ECL detection system (GE Healthcare) and exposing the membranes to x-ray film. Each band was quantified by densitometric scanning using the NIH software ImageJ version 1.8.0_112. The densitometric readings of the bands were normalized to those of β-actin.
Immunohistochemistry
Synovial tissue samples were embedded in paraffin and sliced into 3-µm-thick sections, which were deparaffinized in xylene for 1 h, hydrated in serial dilutions of ethanol, and rinsed in distilled water. For antigen retrieval, deparaffinized sections were heated in Tris/EDTA (pH 9) at 98°C for 40 min. Endogenous peroxidases were blocked by incubating the sections in 3% hydrogen peroxide prepared in methanol for 15 min. The samples were washed with PBS and incubated with 10% goat serum (Nichirei, Tokyo, Japan) for 10 min at room temperature (22–25°C). Next, the sections were incubated with anti-NGF monoclonal rabbit IgG (cat.no: ab6199, Abcam) for 3 h and proteins were visualized using the streptavidin-biotin-peroxidase method (Histofine SAB-PO Kit; Nichirei). Sections were counterstained with Meyer's hematoxylin. Negative controls were also included in which incubation with the primary antibody was omitted.
Statistical analysis
Data are presented as mean ± standard error. The results of qRT-PCR and western blotting did not have equal variance. Hence, differences between 1) the subacromial bursa and rotator interval samples from RCT patients, and 2) rotator interval samples from RCT and RSD patients were determined using the Mann-Whitney-U test. Correlations between the mRNA levels of NGF, COX2 and NRS, and age were determined using Spearman’s rank correlation coefficient. All statistical analyses and power analyses were performed using the SPSS software (version 19.0; SPSS, Inc., Chicago, IL, USA), and p < 0.05 was considered statistically significant. The classification scheme for the correlation was defined as follows: 0 < |ρ| ≤ 0.2, negligible; 0.2 < |ρ| ≤ 0.4, low; 0.4 < |ρ| ≤ 0.7, moderate; 0.7 ≤ |ρ|, high.
Results
Expression levels of NGF and COX2 mRNA in RCT and RSD patients
The samples obtained from sites with the highest degree of synovitis in the rotator intervals showed a significant difference in NGF mRNA expression between the RCT and RSD groups (p = 0.0017, ρ = 0.92; Figure 2(a), Table 4). However, no significant difference was observed in COX2 mRNA expression between these two groups (p = 0.59, ρ = 0.07; Figure 2(b), Table 4).

Quantitative polymerase chain reaction for analyzing the mRNA expression of NGF (a) and COX2 (b) in the synovial tissue of the RI of patients with RSD, the SAB of patients with RCT, and synovial tissue of the RI of patients with RCT and. Gene expression was normalized to the expression of glyceraldehyde 3-phosphate dehydrogenase (GAPDH). *p = 0.0017, between the RI of RCT and RSD patients. †p = 0.039, between the SAB of RCT patients and the RI of RCT patients. NGF, neural growth factor; COX2, cyclooxygenase-2; SAB, subacromial bursa; RI, rotator interval; RSD, recurrent shoulder dislocation; RCT, rotator cuff tear.
NGF and COX2 expression levels in RCT and RSD patients.

Boldface to enhance the values that shows significant difference between two groups.COX2, cyclooxygenase-2, NGF, nerve growth factor; RCT, rotator cuff tear; RSD, recurrent shoulder dislocation; RI, rotator interval; SAB, subacromial bursa.
In the RCT group, no significant difference was observed in the expression levels of NGF mRNA between the subacromial bursa and synovial membrane in the rotator interval (p = 0.28, power = 0.06; Figure 2(a), Table 4). Meanwhile, the expression of COX2 mRNA in the subacromial bursa was significantly lower than that in the rotator interval synovial membrane (p = 0.039, ρ = 0.24; Figure 2(b), Table 4).
Correlation between mRNA expression of NGF and COX2 and age
Samples obtained from sites with the most marked synovitis in the rotator intervals showed no significant correlation between NGF and COX2 mRNA expression and age (p = 0.12, 0.59, respectively; ρ = 0.17, −0.061, respectively; Figure 3).

Correlation between the mRNA levels of NGF (a), COX2 (b) and age in the synovial tissue of rotator intervals. NGF, nerve growth factor; COX2, cyclooxygenase-2; white circle, synovium in rotator interval of recurrent shoulder dislocation; black circle, synovium in rotator interval of rotator cuff tears; black triangle, synovium in subacromial bursa of rotator cuff tears; dotted line, regression line.
Correlation between the mRNA levels of NGF, COX2 and NRS
NGF mRNA expression in the rotator interval correlated with NRS (ρ = 0.38, p = 0.004; Figure 4(a)). However, no correlation was observed between COX2 mRNA expression in the rotator interval and NRS (ρ = -0.006, p = 0.97; Figure 4(b)). Additionally, no correlation was observed between NGF and COX2 mRNA expression in the subacromial bursa of RCTs and NRS (ρ = 0.08, p = 0.69; ρ = −0.24, p = 0.22, respectively; Figure 5).

Correlation between the mRNA levels of NGF (a), COX2 (b) and numeric rating scale of pain in the synovial tissue of rotator intervals. NGF, nerve growth factor; COX2, cyclooxygenase-2; white circle, synovium in rotator interval of recurrent shoulder dislocation; black circle, synovium in rotator interval of rotator cuff tears; dotted line, regression line.

Correlation between the mRNA levels of NGF (a) and COX2 (b) and the numeric rating scale of pain in the synovial tissue of the subacromial bursa. NGF, nerve growth factor; COX2, cyclooxygenase-2; black circle, synovium in the subacromial bursa of rotator cuff tears; dotted line, regression line.
NGF protein expression and localization in synovial tissue
The abundance of NGF protein in the synovial membrane is shown in Figure 6. Rotator interval samples from the RCT patients showed significantly higher NGF protein abundance than those from RSD patients (RCT vs RSD, 1.08 ± 0.34 vs 0.12 ± 0.05, p = 0.012, power = 0.68; Figure 6(b)). However, no significant differences were observed between the subacromial bursa and synovial membrane in the rotator interval of RCT patients (subacromial bursa of RCT vs rotator interval of RCT, 1.08 ± 0.34 vs 0.46 ± 0.20, p = 0.095, power = 0.28; Figure 6(b)). Furthermore, immunohistochemistry revealed the presence of NGF-positive cells in the lining layer of the synovial tissue of RCT patients (Figure 7(a) and (b)), while few NGF-positive cells were observed in the synovial tissue of RSD patients (Figure 7(c)). NGF staining was not seen in negative control sections (Figure 7(d) to (f)).

NGF expression measured in synovial membrane from the SAB and RI of patients with RCT and RI of patients with RSD patients by (a) western blotting and (b) normalized to the expression of β-actin. (n = 5 per group). *p = 0.012, between the RI of RCT and RSD patients. NGF, nerve growth factor; SAB, subacromial bursa; RI, rotator interval, RCT, rotator cuff tear; RSD, recurrent shoulder dislocation; white circle, synovium in rotator interval of recurrent shoulder dislocation; black circle, synovium in rotator interval of rotator cuff tears.

Immunolocalization of NGF (black arrows) in the synovial tissue of the SAB (a) and RI (b) of RCT patients, and RI of RSD patients (c. (d to f) Negative control sections in each patient are shown. NGF, nerve growth factor; SAB, subacromial bursa; RI, rotator interval; RCT, rotator cuff tear; RSD, recurrent shoulder dislocation; Scale bar = 100 μm.
Discussion
Results show that NGF expression was significantly higher in the synovial tissue of patients with RCT than in patients with RSD, while no significant difference was observed in COX2 expression between the two groups. NGF protein expression was higher in the synovium of the rotator interval in patients with RCT than in patients with RSD. Further, the mRNA expression of NGF in the rotator interval synovium significantly correlated with NRS. Moreover, NGF-positive cells localized in the lining layer of the synovial tissue in patients with RCT, but this was not observed in the synovial tissue of RSD patients.
NGF mRNA has been detected in a human mast cell line 34 and NGF is also reportedly present in, and released from, human CD14+ T cell clones and human monocytes that differentiate rapidly during tissue damage.35,36 The data of the relationship between NGF and peripheral sensitization supports the pronociceptive functions of NGF that may include driving local neuronal sprouting at the site of tissue injury. 37 In addition, the administration of anti-NGF antibody has been found to inhibit ectopic sprouting of sensory and sympathetic nerve fibers in an experimental mouse model of arthritis. 38 Sensory nerves, free nerve endings, and expression levels of nociceptive receptors are all increased in the subacromial bursa and around the rotator interval with RCTs. 39 Meanwhile, NGF has recently become the focus of research for the treatment of musculoskeletal chronic pain,4,23–26 with NGF monoclonal antibodies currently being tested in clinical settings. 40 NGF is associated with sensitizing nociceptors through the transient receptor potential cation channel subfamily V member 1 receptor 41 ; which is believed to be one of the links to pain and, hence, peripheral sensitization induced by NGF may be one of the causes of pain associated with RCT. In the present study, NGF mRNA and protein expression levels were found to be higher in the synovial membrane of patients with RCT than in that of patients with RSD. Considering that NGF expression in the rotator interval samples showed a significant positive correlation with the NRS of pain and immunohistochemistry revealed the presence of NGF-positive cells in the lining layer of the synovial tissue of RCT patients, rotator cuff injury may trigger increased NGF expression in the synovial tissue. Our findings, together with existing literature, suggest that elevated NGF expression may be a promising target (TRKA-inhibition or NGF monoclonal antibodies) for pain management in patients with RCT.
NGF levels in synovial fluid are increased in patients with osteoarthritis (OA), rheumatoid arthritis, and spondyloarthritis.42–44 Additionally, NGF levels are increased in synovial specimens from patients with advanced OA compared with those in non-OA controls, as well as in specimens from patients exhibiting symptomatic chondropathy compared with those from patients with asymptomatic chondropathy. 45 Here, we confirmed that NGF mRNA and NGF protein expression levels in the synovial membrane were significantly higher in patients with RCT than in patients with RSD. In addition, no significant difference was observed in mRNA and protein levels of NGF between the subacromial bursa and rotator interval synovium. These results are consistent with those from animal experiments that showed that NGF levels are elevated for up to 56 d after RCT. 27 Taken together, these studies suggest that NGF could be associated with some of the underlying mechanisms of rotator cuff-related pain.
NGF levels are frequently elevated in inflamed states and have been linked to OA pain and lower back pain that could both be reduced with an NGF-neutralizing monoclonal antibody.46–48 Moreover, the anti-NGF antibody improved pain behavior in a COX-2 inhibitor-resistant rat OA model. 49 In the present study, COX2 expression in the rotator interval samples from the RCT group showed no significant difference compared to that in the rotator interval samples from the RSD group. Previous reports have shown that COX-2 levels in the subacromial bursa of patients with RCT are increased compared with those in the subacromial bursa of patients with RSD based on an immunohistochemical grading scale; however, these levels have not been quantitatively assessed. 50 We previously reported that COX2 upregulation was reduced 14 d after RCT in a rat model. 27 Moreover, a COX-2 inhibitor can induce NGF expression in the human synovium. 28 Our findings suggest that degenerative RCT-associated pain depends on factors other than COX-2, and, therefore, some patients with RCT may not benefit from short-term NSAID treatment.
In this investigation, the mean age of patients with RCT was significantly higher than that of patients with RSD; therefore, age may affect NGF and COX-2 expression. However, in our study, no correlation existed between the mRNA expression of NGF and COX2 and age (Figure 3). Further, RCT are potentially degenerative disorders,1–3 while the rotator cuff is commonly damaged during shoulder dislocation in elderly patients. 51 Therefore, it is difficult to obtain a large enough number of RSD patients without RCT in middle or elderly age. In addition, COX-2 expression and immunoreactivity are significantly decreased with aging in mouse hippocampus. 52 The mean expression level of COX2 may be associated with the difference in age between the RCT and RSD groups. In addition, our findings indicate that COX-2 may not be a main contributor to pain in RCT.
Limitations
This study had some limitations. First, the power of the western blotting results is relatively low, due to the small sample size. PCR and western blotting could not be performed on the same samples due to the limited sample volume collected from each patient. Therefore, the scatter plot of mRNA and protein levels for each patient could not be created. A healthy control group was not included in this study. Second, we could not—for ethical reasons—harvest synovial membrane samples from the subacromial bursa of RSD patients, and every patient with RSD in this study did not show subacromial impingement symptoms. A previous report showed that COX2 mRNA levels in the subacromial bursa of RSD patients were significantly lower than those in the subacromial bursa of RCT patients, 50 while in this study, there was no significant difference in either NGF or COX2 expression between the subacromial bursa and rotator interval synovial membrane in RCT patients. Therefore, our findings have not been influenced by the expression of these genes in the subacromial bursa of patients with RSD.
In conclusion, the synovial membrane of patients with RCT exhibits a significantly higher expression of NGF mRNA and protein than that of patients with RSD, but no significant difference in COX2 expression.
Conclusion
This study supports further investigations of NGF as a potential therapeutic target for pain in patients with RCT, whereas the interaction with the COX-2 pathways may not be a promising target.
Footnotes
Acknowledgment
We acknowledge Ms. Yuko Onuki for her help with qRT-PCR analysis.
Author Contributions
RT wrote the manuscript. TK enrolled the patients and organized this study. KU performed PCR and WB analysis and contributed to manuscript writing. LAN provided logistic support and interpreted PCR and WB data and revised the manuscript. NN and MN performed PCR analysis. TW performed western blotting analysis and contributed to manuscript writing. GI is responsible for the integrity of this study, especially method and ethical issues. MT is responsible for the integrity of this study, and all authors approved the final version of the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This investigation was supported in part by the Japan Society for the Promotion of Science (JSPS) Grant-in-Aid for Scientific Research (KAKENHI) [Grant No. 18K16633]. The Center for Neuroplasticity and Pain (CNAP) is supported by the Danish National Research Foundation (DNRF121).
