Abstract
Management of chronic pain is still hard, and new analgesic drugs are needed. Evodiamine (Evo) and rutaecarpine (Rut) are two major active components of Evodia rutaecarpa, a Chinese traditional medicine that has been used as an analgesic for a long time. However, their effects on peripheral hypersensitivity remain unknown. Similar to capsaicin, the Evo and Rut were docked to the transient receptor potential cation channel subfamily V member 1 (TRPV1) in molecular simulation experiments. Moreover, Evo (10 µM) and Rut (50 µM) activated TRPV1 on human embryonic kidney 293 (HEK293) cells in electrophysiological recording experiments. Behaviorally, the application of Evo and Rut reduced peripheral hypersensitivity in a dose-dependent manner, which was blocked by capsazepine (a selective inhibitor of TRPV1). Furthermore, both Evo and Rut increased time in the open arms of the elevated plus maze on mice with nerve injury. These observations suggested that Evo and Rut reduced peripheral hypersensitivity and anxiety in mice with nerve injury or inflammation via TRPV1.
Keywords
Introduction
Chronic pain refers to the pain that lasts for over three months, which is characterized as allodynia, hyperalgesia, and spontaneous pain. The prevalence of chronic pain reaches 20%, with an 8% high impact on the quality of life, 1 and it is hard to manage chronic pain. 2 New analgesic drugs are needed for chronic pain treatments. 3 Transient receptor potential cation channel subfamily V member 1 (TRPV1) is an outwardly rectifying nonselective cation channel, which is mainly activated by noxious heat or its agonist, capsaicin (Cap). 4 TRPV1 distributes in both periphery and central nervous system,5,6 which is involved in pain generation and information processing.7,8 Recently, Cap has been used to treat chronic pain, 9 and some agonists of TRPV1 are also in clinical trials. 10 Therefore, new chemicals targeting TRPV1 would have significant potentials for chronic pain management in the future.
Evodiamine (Evo) and rutaecarpine (Rut) are the main medicinal components of traditional Chinese medicine WU-ZHU-YU (Fructus evodiae), the dried fruit of Evodia rutaecarpa (Juss.Benth). 11 Previous studies focused on the anti-inflammatory,12,13 as well as anti-cardiovascular disease 14 and anti-Alzheimer’s disease15,16 effects of these two components. Evo and Rut activate TRPV1,17,18 and this indicates that they may have effects on chronic pain hypersensitivity. However, few studies were performed to evaluate this point.
In the current study, we evaluated the possible role of Evo and Rut on peripheral hypersensitivity. In behavioral aspects, the application of Evo or Rut increased PWTs on mice with peripheral nerve injury or inflammation in a dose-dependent manner, and the preapplication of capsazepine (CPZ) blocked this effect. Both chemicals increased entries and time in open arms of the elevated plus maze (EPM) but did not change performance in the novel objects’ recognition task. Our data suggested that Evo or Rut may be a useful analgesic candidate for pain treatments in the future.
Materials and methods
Experiment animals
C57BL/6 mice from six to eight weeks were used. Four or five animals were kept in a cage and average room temperature (25 ± 5°C) with a humidity of 60 ± 5% under a 12-h light/dark schedule (from 7:00 to 19:00). Food and water were provided ad libitum. The animal care and use committee of Zhejiang University approved all mouse protocols.
Experiment reagents
The reagents as follows were used: Evo (Purity: ≥98.0%, Sinopharm, China), Rut (Purity: ≥98.0%, Sinopharm, China), Cap (Purity: ≥98.0%, Sinopharm, China), and CPZ (Purity: >99.0%, Macklin, China). In vivo, Evo, Rut, Cap, and CPZ were dissolved in 10% DMSO, 10% Tween-80, and 80% physiological saline, respectively. In vitro, they were dissolved in DMSO. Anti-TRPV1 (Abcam, 1:1000, ab203103; Cambridge, MA, USA) and anti-Actin (Solarbio, 1:3000, RG000120; Beijing, China) were used for Western blot.
Animal models
Common peroneal nerve (CPN) model, we performed CPN ligation following the previous reports.19,20 Briefly, after the anesthetization by isoflurane (1%–3%, as needed), the left CPN was ligated slowly with chromic gut suture 5-0 (Ethicon, Blue Ash) until the appearing of twitching of related digits. The skin was sutured using a 5-0 silk suture and cleaned with povidone-iodine. Sham surgery was conducted in the same manner, but the nerve was not ligated. All animals were kept in a healthy living chamber postsurgery. The mice were given the behavioral test on postsurgical days 3 to 14.
Complete Freund’s adjuvant (CFA) model, 10 µl of 50% CFA (Sigma, St. Louis, MO, USA) was injected into the left hind paw of the mouse using a 50 µl microinjector. The mice were given the behavioral test on postsurgical days 1 to 14.
Mechanical allodynia test
The mechanical response threshold was quantified by measuring left hind paw withdrawal response to von Frey filament stimulation according to a method described by Chaplan et al. 21 To let the mouse calm down, we put it on wire mesh covered with a clear plastic box for half an hour. Then, left hind paw was stimulated by von Frey using up-down methods. The positive response was marked as “X,” and no withdrawal was recorded as “O.” Fifty percent response threshold was calculated using a formula: 50% g threshold = 10(xf+kδ)/10000, where, xf = value (in log units) of final von Frey hair used; k = tabular value for pattern of positive/negative responses; and d = mean difference (in log units) between stimuli (here 0.296).
Spontaneous pain behavior test
Paw spontaneous flinching: The mice were placed in a plexiglass box (15 cm × 15 cm × 12 cm) and adapted to the environment for 30 min. A mirror was placed obliquely at 45° under the test box to observe the mouse’s flinching behavior artificially. Injecting 20 µl of the reagents into the left hind paw caused nociceptive behaviors such as flinching and licking the paws. After injecting the reagents, the number of times of flinching was recorded in units of 5 min for a total of 20 min.
Conditioned place preference (CPP) test: CPP test was utilized from the behavioral paradigm built by King et al. for adult rats.22–24 The experimental mice were preadapted for three days and explored freely for 15 min each time. On the last day of preadaptation, mice spend more than 80% (time spent >720 s) or less than 20% (time spent <120 s) of total time in a chamber would be excluded for further tests. On the morning of the fourth day, we randomly placed the mouse into a chamber for 30 min after saline injection. Four hours later, the mouse was placed in another chamber after the intraperitoneal administration of chemicals (Evo: 0.30 mg/kg; Rut: 0.29 mg/kg). After 20 h, on the fifth day, we retook videos and analyzed place preference.
Intrathecal administration
After deeply anesthetizing with isoflurane, we penetrated through the fifth and sixth lumbar vertebrae with a microinjector (1 ml insulin syringe, Kangfulai, Changzhou, China). The operator felt a clear feeling of falling and saw the mouse’s tail moved, indicating that the needle has penetrated the meningeal cavity, and then slowly pushed the reagents (50 µl/each mouse). After the injection, the operator held the needle for 10 s and disinfected the skin with alcohol after the needle was pulled. After leaving the isoflurane environment, the mice can wake up within 2 to 3 min, and it did not affect the subsequent experiments. We performed mechanical allodynia tests at 10 min and 30 min. Moreover, finally, we took out the spinal cord at 1 h.
EPM test
EPM was performed as described previously. 25 We performed the EPM test after seven days on CPN ligation. Mice were placed in the experimental area for 1 h to adapt to the environment two days before tests. The experimental area was soundproofed, and the lighting and ambient temperature were suitable. Behavioral test was performed after injection of reagents (Evo: 0.30 mg/kg; Rut: 0.29 mg/kg) for 30 min. The mice were gently moved into the cross-center area with the head facing open arms. Video recording was performed for 5 min. After testing, we cleaned stool, urine, and odor with 75% alcohol, and the next experiment was performed after equipment was arid. The time stayed in the open arms, and the total distance of movement was counted by ANY-maze software.
Novel object recognition test
The novel object recognition test was adapted from the paradigm reported by Leger et al. 26 First, we placed a mouse in a rectangular box-shaped plastic box and adapted to a new environment for 5 min. The next day, we placed two identical objects in the box 10 cm from the inner wall. Behavioral test was performed after injection of reagents (Evo: 0.30 mg/kg; Rut: 0.29 mg/kg) for 30 min. The mouse could explore two identical objects freely; one of the objects was replaced with a new one 24 h later; the mouse explored two objects freely. The discrimination index was calculated as the percentage of the difference between times spent on new and old objects.
Measuring body temperature
First, we removed hair in the middle of the abdomen and left groin of a mouse. This area was about 0.5 cm2 and used as a scanning position for an electronic thermometer (GM400, Guangdong, China) to measure body temperature. Second, we allowed the mouse to adapt to the experimental environment for two days (20 min/each time). On the third day, we placed the mouse in a plastic box to adapt for 20 min and measured the body temperature three times (10 min/each time) before injecting chemicals (Evo: 0.30 mg/kg; Rut: 0.29 mg/kg). The average value was used as a measurement reference. After chemicals injection, we measured the body temperature six times at an interval of 10 min.
Reverse transcription quantitative polymerase chain reaction and Western blot
RNA was extracted from the spinal cord using an RNA-quick purification kit (ES Science, Shanghai, China). cDNA was synthesized using 1 µg of isolated RNA, and 2 µl of synthesized cDNA library was used for quantitative polymerase chain reaction (qPCR) with specific primers and Hieff™ qPCR SYBR Green Master Mix (Yeasen, Shanghai, China). Primers were as follows: TRPV1 forward (5′-
Mice were lightly anesthetized with isoflurane and then decapitated. Spinal cord was dissected and then homogenized in a radioimmunoprecipitation assay buffer (50 mM Tris-Cl, pH 7.6, 150 mM NaCl, 1 mM ethylenediaminetetraacetic acid (EDTA), 1% Nonidet P-40, 0.1% sodium dodecyl sulfate (SDS), 1 mM dithiothreitol, and 0.5% sodium deoxycholate) containing a protease inhibitor cocktail. After centrifugation, supernatants were used for protein quantification by Bradford assay. Then, the supernatants were boiled in SDS sample buffer, and SDS-polyacrylamide gel electrophoresis and immunoblotting were performed. The density of immunoblots was measured with ImageJ software (NIH).
Electrophysiology
The whole-cell patch-clamp was used to recording current from TRPV1-expressing cells by a HEKA EPC 10 amplifier controlled with PatchMaster software (HEKA). Patch pipette was prepared from borosilicate glass and fire polished to the resistance of ∼6MΩ. Both pipette solution and bath solution contained 130 mM NaCl, 3 mM 4-(2-hydroxyethyl)-1-piperazineethanesulfonic acid (HEPES), and 0.2 mM EDTA (pH 7.3). After transfecting cells (HEK293T) for 12 to 14 h with lipo2000, yellow fluorescent protein fluorescence could be seen with exciting light. Membrane potential was held at 0 mV at the beginning and elicited by +100 mV for 200 ms and –80 mV for 200 ms, then 0 mV for 50 ms as a cycle. The current was sampled at 10 kHz and filtered at 2.9 kHz. All recordings were performed at room temperature (22°C) with a maximum variation of 1°C. Ligands such as Cap, Evo, and Rut and inhibitor (CPZ) were perfused to membrane patch by a gravity-driven system (RSC-200, Bio-Logic, France). They were dissolved in DMSO as stocks and diluted with bath solution before the experiment. Bath, ligands, and inhibitor solutions were delivered through separate tubes to minimize the mixing of solutions. Patch pipette was placed in front of the perfusion tube outlet.
Molecular docking
All simulations were performed on DiscoveryStudio2016 software (Neotrident, Beijing, China). The ligand molecules used were downloaded from PubChem Project, and they needed to be subjected to energy minimization before docking in order to facilitate docking into the nest of the receptor molecule later. TRPV1 receptor (PDB ID: 5IS0) also needed to be treated before docking: removing the ligand and removing the water molecules, adding polar hydrogen atoms, imparting charges, and adding magnetic fields. We used the CDOCKER module to perform molecular docking and then analyzed the data after molecular docking.
Data analysis
Graph-pad Prism 8 was used for data processing and analysis, and all data were expressed as means ± SEM. Unpaired t-test, paired t-test, one-way analysis of variance (ANOVA), or two-way ANOVA was used to conduct data analysis. All data were presented as mean ± SEM. In all cases, P values less than 0.05 was treated as statistical significance.
Results
The activating effects of Evo and Rut on TRPV1
Given that Evo and Rut may activate TRPV1,17,18 here, we confirm this point by performing molecular docking analysis. We employed the CDOCKER module of DiscoveryStudio2016 software and examined the hydrophobic interactions and hydrogen bonds between chemicals and TRPV1. In this analysis, hydrogen bonds increase the stability of the complex, and hydrophobic forces make ligand and receptor bonds better. The score was an assessment of the binding energy of the chemical-receptor complex, and a higher score indicated better binding of molecular to the receptor. We examined the docking scores of Cap, Evo, and Rut to TRPV1, respectively. As shown in Figure 1(a) and (b), Thr550 of TRPV1 formed hydrogen bonds to Cap, and hydrophobic residues were Leu515, Leu553, ALA546, MET547, and ALA665, and the optimal combination structure score was 47.73 Kcal/mol. We also performed electrophysiological recordings on TRPV1-transfected HEK293T cells. As shown in Figure 1(c), the puff application of Cap (3 µM) induced TRPV1-mediating currents. Thr550 and Asn551 of TRPV1 formed hydrogen bonds with Evo, while residues, including Tyr511, Leu515, Leu553, Ile573, and Leu669, formed a hydrophobic pocket that interacted with Evo (Figure 1(d) and (e)). The docking score of the best combination structure is 36.89 Kcal/mol, which was smaller than that of Cap. In electrophysiological recording experiments, the application of Evo (10 µM) activated TRPV1, and the currents were blocked by CPZ (10 µM) (Figure 1(f)). In simulation experiments of Rut, Tyr511, Thr550, and Asn551 formed hydrogen bond interactions with Rut. Moreover, hydrophobic residues were Tyr511, Leu515, Met547, Leu553, and Leu669 (Figure 1(g) and (h)). The best combination structure score is 35.93 Kcal/mol, and this was smaller than that from Cap and Evo. Consistently, Rut at 50 µM could activate TRPV1, which was also blocked by CPZ (10 µM) (Figure 1(i)). According to the above data, the common residue that formed a hydrogen bond with these three compounds was Thr550, and the common hydrophobic amino acid residues were Leu515 and Leu553. These results indicated that Evo and Rut could bind with TRPV1-like Cap and produce biological effects.

Molecules docking and electrophysiological results of Cap, Evo, and Rut with TRPV1 receptor, respectively. (a) Cap was surrounded by hydrophobic pockets composed of hydrophobic residues of TRPV1 (Leu515, Leu553, ALA546, MET547, and ALA665). (b) Interaction between Cap and TRPV1 residues with 2D interaction diagram. The major residue of TRPV1 that formed hydrogen bonds to capsaicin was Thr550. (c) Representative trace showing that TRPV1 was activated by capsaicin (3 µM). (d and e) Thr550 and Asn551 of TRPV1 formed hydrogen bonds with Evo, and hydrophobic residues (Tyr511, Leu515, Leu553, Ile573, and Leu669) formed a hydrophobic pocket to enhance the interaction with it. (f). Evo (10 µM) activated TRPV1-like capsaicin (3 µM), and Evo-induced currents could be perfectly blocked by CPZ (10 µM). (g and h) The key residues that formed hydrogen bond interactions with Rut were Tyr511, Thr550, and Asn551, and hydrophobic residues were Tyr511, Leu515, Met547, Leu553, and Leu669. (i). Rut (50 µM) activated TRPV1-like capsaicin (3 µM) and was completely suppressed by CPZ (10 µM). (Hydrogen bonds contained conventional hydrogen bond and carbon hydrogen bond, and hydrophobic bonds include Pi–Pi, Alkyl, and Pi–Alkyl.)
The involvements of TRPV1 to peripheral hypersensitivity
Was TRPV1 involved in the development of peripheral hypersensitivity? We examined the expression of TRPV1 in the spinal cord at seven days after CPN ligation. As shown in Figure 2(a) and (b), the mRNA and protein level of TRPV1 in spinal cord reached two times of those in the sham group, respectively (protein: P < 0.01, n = 4 for sham, n = 5 for injury; mRNA: P < 0.05, n = 6 for each group). We applied Cap (0.15 mg/kg, i.p.) and observed an increase of paw withdrawal thresholds (PWTs) on mice with CPN ligation 30 min later (Figure 2(c)); this result suggests that activating TRPV1 could reduce peripheral hypersensitivity induced by nerve injury. What was the effect of antagonizing TRPV1? We applied CPZ (2 mg/kg, i.p.), a selective antagonist of TRPV1, to mice with CPN ligation; we did not observe any effects on PWTs (Figure 2(d)). However, the preapplication of CPZ 30 min in advance blocked the effects of Cap on PWTs (Figure 2(e)). These data suggested that activating TRPV1 could reduce peripheral hypersensitivity induced by nerve injury.

The involvements of TRPV1 to peripheral hypersensitivity. (a and b) Expression levels of TRPV1 in the spinal cord were increased at seven days after nerve injury. ((a) Western blot: P < 0.01, n = 4 for sham, n = 5 for CPN and (b) RT-qPCR: P < 0.05, n = 6 for each group). (c) Cap at 0.15 mg/kg raised the PWT of CPN group (two-way RM ANOVA, sham vs. CPN, F(1, 18) = 34.52, P< 0.01; time points, F(1, 18) = 31.63, P< 0.01; interaction, F(1, 18) = 17.78, P< 0.01, n=10 per group, ** P< 0.01 under Sidak’s test). (d) When CPZ was applied alone, no effect was observed on PWTs (two-way RM ANOVA, sham vs. CPN, F(1, 14) = 83.69, P < 0.01; time points, F(1, 14) = 0.23, P = 0.64; interaction, F(1, 14) = 0.64, P = 0.44, n = 8 each group). (E) Preapplication of CPZ half an hour earlier blocked the effects of Cap on PWTs on mice with CPN ligation (two-way RM ANOVA, sham vs. CPN, F(1, 8) = 54.12, P< 0.01; time points, F(1, 8) = 0.01, P = 0.94; interaction, F(1, 8) = 0.12, P = 0.74, n= 5 per group, **P< 0.01 under Sidak’s test). (ANOVA indicates analysis of variance; RM, repeated measures; Sidak’s test, Sidak’s multiple comparisons test. Data are represented as mean ± SEM.)
The application of Evo reduced peripheral hypersensitivity after CPN ligation or CFA injection
We next evaluated the effects of Evo on peripheral hypersensitivity. As shown in Figure 3(a), CPN ligation significantly decreased PWTs compared to the sham group (two-way ANOVA, sham vs. CPN, n = 8 for sham and n = 10 for CPN, Sidak’s test, P < 0.01). We then applied Evo (i.p.) and examined PWTs at 0.5 h after injection. A low dose of Evo (0.03 mg/kg, i.p., Figure 3(a)) has no effect, but higher doses of Evo at 0.3 mg/kg (Figure 3(b)) and 3.0 mg/kg, i.p. (Figure 3(c)) significantly increased PWTs of mice with nerve injury. There were no effects on the sham group. Were the effects of Evo related to TRPV1? We applied CPZ 30 min before Evo (0.3 mg/kg, i.p.) application on mice with or without CPN ligation. We found that CPZ blocked the effects of Evo (Figure 3(d)) on PWTs. We injected CFA subcutaneously into the plantar of the left hind paw, as shown in Figure 3(e), CFA injection decreased PWTs significantly, which could be elevated by Evo (0.3 mg/kg, i.p.), and this effect disappeared within 2 h. To further confirmed the effects of Evo on peripheral hypersensitivity, we applied Evo intrathecally (50 µl/mouse, Evo: 10 µM/l). We observed the higher PWTs on the mice with CPN ligation at 10 min after Evo injection (Figure 3(f)). These data suggested that Evo could alleviate peripheral hypersensitivity induced by both nerve injury and inflammation via TRPV1.

The application of Evo reduced peripheral hypersensitivity after CPN ligation or CFA injection. (a) Nerve injury decreased the PWTs of mice, and Evo at 0.03 mg/kg had no effect on the threshold in the sham and CPN groups (two-way RM ANOVA, sham vs. CPN, F(1, 16) = 86.93, P < 0.01; time points, F(3, 48) = 11.62, P < 0.01; interaction, F(3, 48) = 8.42, P < 0.01, n = 8 for sham, n = 10 for CPN,** P < 0.01 under Sidak’s test). (b) Evo at 0.30 mg/kg increased the PWTs in CPN but not in sham (two-way RM ANOVA, sham vs. CPN, F(1, 16) = 30.19, P< 0.01; time points, F(2, 32) = 25.61, P< 0.01; interaction, F(2, 32) = 22.15, P< 0.01, n= 8 for sham, n= 10 for CPN, ** P< 0.01 under Sidak’s test). (c). Evo at 3.0 mg/kg increased the threshold and last for over 2 h (two-way RM ANOVA, sham vs. CPN, F(1, 15) = 19.28, P< 0.01; time points, F(2, 30) = 11.37, P< 0.01; interaction, F(2, 30) = 12.41, P< 0.01, n = 8 for sham, n= 9 for CPN, ** P<0.01 under Sidak’s test). (d) Preapplication of CPZ half an hour earlier inhibited the effects of Evo on elevation of PWTs of mice with CPN (two-way RM ANOVA, sham vs. CPN, F(1, 14) =170.0, P < 0.01; time points, F(1, 14) = 0.23, P = 0.60; interaction, F(1, 14) = 0.64, P = 0.44, n = 8 each group, ** P < 0.01 under Sidak’s test). (e) The application of Evo (0.3 mg/kg, i.p.) increased the PWTs of mice with CFA injection (two-way RM ANOVA, interaction: F(3, 54) = 12.50, P <0.01, saline vs. CFA: F(1, 18) = 21.26, P < 0.01, time: F(2.07, 37.29) = 14.43, P < 0.01, n=10 per group, ** P < 0.01 under Sidak’s test). (f) The application of Evo (10 µM/50 µl/mice, i.t.) at seventh day after CPN ligation elevated the PWTs of mice with CPN ligation (one-way RM ANOVA, F(3, 23) = 11.85, P <0.01, n = 6, *P < 0.05 under Tukey’s test). (ANOVA indicates analysis of variance; RM, repeated measures; Sidak’s test, Sidak’s multiple comparisons test. Data are represented as mean ± SEM.)
The application of Rut reduced peripheral hypersensitivity after CPN ligation or CFA injection
In the same way, we found that Rut at 0.029 mg/kg had no effect on PWTs (two-way ANOVA, sham vs. CPN, n = 10, Sidak’s test, P < 0.01, Figure 4(a)), but higher doses at 0.29 mg/kg (Figure 4(b)) or 2.9 mg/kg (Figure 4(c)) increased PWTs at 0.5 h after injection. Similarly, the preapplication of CPZ blocked the effects of Rut (0.29 mg/kg, i.p.) on PWTs (Figure 4(d)). Furthermore, the application of Rut (0.29 mg/kg, i.p.) also increased PWTs of mice with CFA injection (two-way ANOVA, saline vs. CFA, n = 8 for saline and n = 10 for CFA, Sidak’s test, P = 0.74, Figure 4(e)) but not that from control group. To further confirm the analgesic effect of Rut, we also employed more administration route and applied the Rut intrathecally. Similarly, the application of Rut in the spinal cord increased the PWTs of mice with CPN ligation (Figure 4(f)). In short, our data suggested that Rut reduced peripheral hypersensitivity induced by peripheral nerve injury or inflammatory via TRPV1.

The application of Rut also reduced peripheral hypersensitivity after CPN ligation or CFA injection. (a) Rut at 0.029 mg/kg had no effect on the threshold in the sham and CPN groups (two-way RM ANOVA, sham vs. CPN, F(1, 18) = 125.30, P< 0.01; time points, F(3, 54) = 11.33, P< 0.01; interaction, F(3, 54) = 8.09, P<0.01, n = 10 per group, ** P < 0.01 under Sidak’s test). (b) Rut at 0.29 mg/kg increased the PWTs in CPN but not in sham (two-way RM ANOVA, sham vs. CPN, F(1, 14) = 26,49, P < 0.01; time points, F(2, 28) = 14.58, P< 0.01; interaction, F(2, 28) = 10.40, P< 0.01, n= 7 for sham, n= 9 for CPN, ** P< 0.01 under Sidak’s test). (c) Rut at 2.9 mg/kg increased the threshold and last for over 2 h (two-way RM ANOVA, sham vs. CPN, F(1, 18) = 10.18, P< 0.01; time points, F(2, 36) = 20.17, P< 0.01; interaction, F(2, 36) = 8.05, P< 0.01, n=10 per group, ** P<0.01 under Sidak’s test). (d) Preapplication of CPZ half an hour earlier blocked the effects of Rut on elevation of PWTs of mice with CPN (two-way RM ANOVA, sham vs. CPN, F(1, 14) = 140.90, P < 0.01; time points, F(1, 14) = F(1, 14) = 2.505e-005, P = 0.99; interaction, F(1, 14) = 0.11, P = 0.74, n=8 per group, ** P < 0.01 under Sidak’s test). (e) The application of Rut (0.29 mg/kg, i.p.) increased the PWTs of mice with CFA injection (two-way RM ANOVA, interaction: F(2, 32) = 26.08, P <0.01, saline vs. CFA: F(1, 16) = 68.35, P < 0.01, time: F(2, 32) = 25.02, P < 0.01, n=8 for saline, n=10 for CFA, **P <0.01 under Sidak’s test). (f) The application of Rut (10 µM/50 µl/mice, i.t.) at seventh day after CPN ligation elevated the PWTs of mice with CPN ligation (one-way RM ANOVA, F(3, 23) = 9.85, P <0.01, n = 6, *P < 0.05 under Tukey’s test). (g) The administration of Evo intrathecally decreased the expression of TRPV1 in spinal cord of mice with CPN ligation (one-way RM ANOVA, F(2, 12) = 9.24, P < 0.01, n = 5 per group, *P < 0.05 under Tukey’s test). (h) CFA injection increased the expression of TRPV1 in the spinal cord, and the application of Evo or Rut partially normalized the changes (one-way RM ANOVA, F(3, 13) = 11.48, P < 0.01, n = 4 for Saline + Veh, CFA + Veh, and CFA + Rut, n = 5 for CFA + Evo, *P < 0.05 under Tukey’s test). (i) The intraplantar injection of Evo induced increased the lifting numbers of lift hind paw of normal mice (two-way RM ANOVA, interaction: F(6, 45) = 0.46, P > 0.05, time: F(2.54, 38.09) = 48.71, P < 0.01, treatments: F(2, 15) = 1.55, P > 0.05, n = 6 per group, *P <0.05 under Tukey’s test). (ANOVA indicates analysis of variance; RM, repeated measures; Sidak’s test, Sidak’s multiple comparisons test. Data are represented as mean ± SEM.)
Would the application of Evo or Rut change the expression of TRPV1? To answer this question, we examined the expression of spinal TRPV1 1 h after the intrathecal application of Evo or Rut, respectively. We observed a significant decrease of TRPV1 in the spinal cord of mice with CPN ligation (Figure 4(g)). Moreover, we checked the effects of two chemicals on the TRPV1 of mice with CFA injection. Similarly, we observed a higher expression of TRPV1 in the spinal cord at third day after CFA injection. Furthermore, the application of Evo or Rut partially normalized this change examined 1 h after the chemical application (Figure 4(h)). These data suggest that the application of Evo or Rut could change the expression of spinal TRPV1.
Given that the activities of TRPV1 regulate the spontaneous nociception, 27 whether the Evo or Rut have the same effect? We also injected the Evo or Rut into the plantar of normal mice, and we observed a higher number of lifting hind paw on mice with Evo injection, but not that with Rut, and this effect only lasted about 5 min (Figure 4(i)). Therefore, similar to Cap, the Evo could also induce spontaneous nociception.
The application of Evo and Rut did not induce place preference
Spontaneous pain is one of the significant pathological phenomena of chronic pain; would Evo and Rut modulate spontaneous pain induced by CPN ligation 24 ? We investigated this point by employing the CPP behavioral paradigm. As shown in Figure 5(a), mice did not show a place preference during the preconditioning phase. Unlike our previous studies that showed clonidine induced place preference,23 the mice did not spend a long time in the Evo-paired chamber (0.3 mg/kg, i.p.) at the test period (Figure 5(b)). Similarly, Rut did not induce place preference (Figure 5(c) and (d)). These data suggest that Evo and Rut may not regulate spontaneous pain induced by nerve injury.
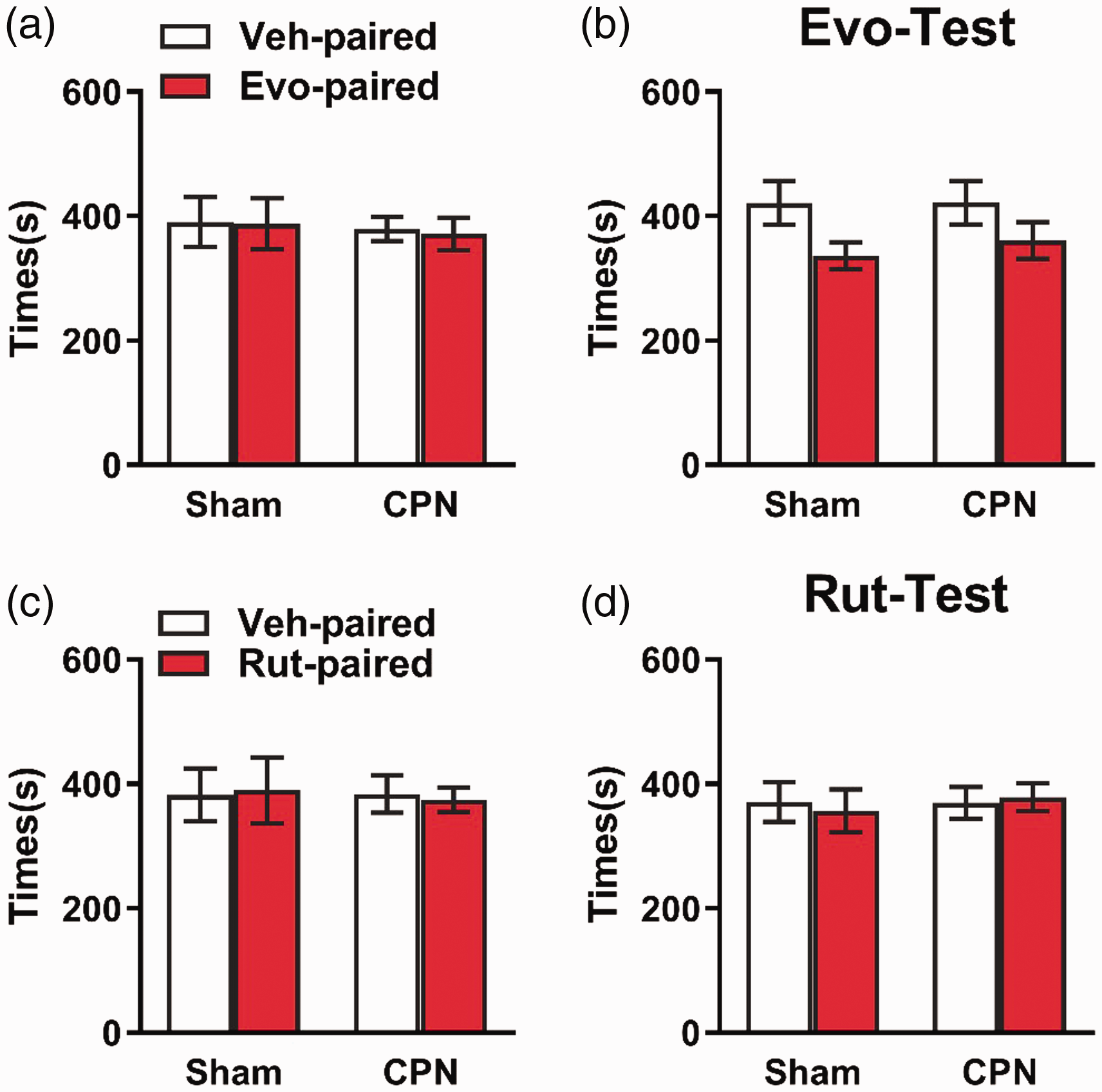
The application of Evo or Rut did not induce place preference. (a) No differences were detected on the time spent in the tested chamber in precondition test (two-way ANOVA, Veh vs. Evo, F(1, 18) = 0.03, P = 0.86; sham vs. CPN, F(1, 18) = 0.19, P = 0.67; interaction, F(1, 18) = 0.01, P = 0.93, n = 5 for sham, n = 6 for CPN). (b) Evo did not induce preference for the Evo-paired chamber in CPN group (two-way ANOVA, Veh vs. Evo, F(1, 18) = 5.41, P = 0.03; sham vs. CPN, F(1, 18) = 0.16, P = 0.69; interaction, F(1, 18) = 0.15, P = 0.70, n = 5 for sham, n = 6 for CPN). (c) No differences were detected on the time spent in the tested chamber in precondition test for Rut (two-way ANOVA, Veh vs. Evo, F(1, 16) = 0.01, P = 0.98; sham vs. CPN, F(1, 16) = 0.03, P = 0.86; interaction, F(1, 16) = 0.05, P = 0.83, n = 5 for both groups). (d) Evo did not induce preference for the Evo-paired chamber in CPN group (two-way ANOVA, Veh vs. Evo, F(1, 16) = 0.01, P = 0.94; sham vs. CPN, F(1, 16) = 0.13, P = 0.73; interaction, F(1, 16) = 0.16, P = 0.69, n = 5 for both groups). (ANOVA indicates analysis of variance; RM, repeated measures; Sidak’s test, Sidak’s multiple comparisons test. Data are represented as mean ± SEM.)
Anti-anxiety effects of Evo and Rut on mice with CPN ligation
Previous articles have reported that aversion and curiosity to a new environment would come to a balance in EPM experiments. When mice were in an anxious state, the aversion to open arms would be stronger than curiosity, and mice spent less time in open arms.24,28,29 As shown in Figure 6(a) and (b), the mice with nerve injury had a smaller number of open arms entries. However, the application of Evo or Rut increased the number of open arms entries in the mice with nerve injury. Also, mice with nerve injury spent less time in the open arm than the sham group (Figure 6(c)). Moreover, the application of Evo or Rut significantly increased the time in open arms (Figure 6(c)). There was no difference in the total distance among groups (Figure 6(d)). Therefore, Evo and Rut can alleviate anxiety after nerve injury.

The application of Evo and Rut changed the behaviors in the elevated plus maze test. (a) Representative trace of sham and CPN mice treated with Evo or Rut in EPM (horizontal rectangles indicate open arms and vertical rectangles represent closed arms). (b) Summarized data showed the entries to open arms in the EPM (one-way ANOVA, treatments, F(4, 37) = 5.21, P < 0.01, n = 14 for Veh-Sham, n = 5 for Evo-Sham, n = 8 for Veh-CPN, n = 9 for Evo-CPN, n = 6 for Rut-CPN, ** P < 0.01 under Sidak’s test). (c) Veh-CPN group spent less time in open arms compared with other three groups in the EPM (one-way ANOVA, treatments, F(3, 37) = 11.44, P < 0.01, n = 14 for Veh-Sham, n = 5 for Evo-Sham, n = 8 for Veh-CPN, n = 9 for Evo-CPN, n = 6 for Rut-CPN, ** P < 0.01 under Sidak’s test). (d) A difference in total distance was detected among groups (one-way ANOVA, treatments, F(3, 37) = 3.14, P < 0.05, n = 14 for Veh-Sham, n = 5 for Evo-Sham, n = 8 for Veh-CPN, n = 9 for Evo-CPN, n = 6 for Rut-CPN, * P < 0.01 under Sidak’s test). (ANOVA indicates analysis of variance; RM, repeated measures; Sidak’s test, Sidak’s multiple comparisons test. Data are represented as mean ± SEM.)
The application of Evo and Rut did not affect the learning process
The failure of induction of place preference might come from impacts of Evo and Rut on learning and memory. To exclude this possibility, we employed novel objects recognition (NOR) behavioral task and applied Evo 30 min before the first phase of training (Figure 7(a)). As shown in Figure 7(b), mice in both groups spent equal time on identical objects. Twenty-four hours later, we replaced one of the objects with a new one. Mice injected with Evo (0.30 mg/kg) performed similarly to the control group; they all spent a significantly longer time to explore the new object (Figure 7(c)). Furthermore, the application of Rut did not change the behaviors of mice in the NOR task (Figure 7(d) and (e)). These data suggest that the application of Evo or Rut did not impair the learning ability of mice.

Evo and Rut had no effect on learning tested in novel objects recognition test. (a) (a) Experimental paradigm for the effects of Evo and Rut on learning tested in NOR. Representative trajectories of mouse to explore two identical objects (b) and two different objects (c). (b) During the training period, there was no difference between Evo group and vehicle group (two-way RM ANOVA, objects, F(1, 8) = 0.02, P = 0.90; treatments, F(1, 8) = 0.05, P = 0.83; interaction, F(1, 8) = 1.25, P = 0.30, n = 5 per group). (c) Mice of Evo and Veh groups preferred new objects (two-way RM ANOVA, objects, F(1, 8) = 24.63, P < 0.01; treatments, F(1, 8) = 3.78, P = 0.09; interaction, F(1, 8) = 3.50, P = 0.10, n=5 each group, *P < 0.05 under Sidak’s test). (d) Exploration times for the two identical objects had no difference in each group (two-way RM ANOVA, objects, F(1, 8) = 0.01, P = 0.94; treatments, F(1, 8) = 3.11, P = 0.12; interaction, F(1, 8) = 0.33, P = 0.58, n = 5 per group). (e) The Veh and Rut groups spent more time exploring the novel object (two-way RM ANOVA, objects, F(1, 8) = 35.53, P < 0.05; treatments, F(1, 8) = 3.55, P = 0.13; interaction, F(1, 8) = 0.69, P = 0.45, n = 5 each group, *P < 0.05 under Sidak’s test). (f) (a) Experimental paradigm for the effects of Evo and Rut on memory retrieval tested in NOR. Representative trajectories of mouse to explore two identical objects (b) and two different objects (c). (g) Mice from both Evo and Veh groups preferred new objects (two-way RM ANOVA, objects, F(1, 15) = 22.84, P < 0.01; treatments, F(1, 15) = 0.16, P = 0.70; interaction, F(1, 15) = 0.01, P = 0.96, n = 7 for Veh, n = 10 for Evo group, *P < 0.05 under Sidak’s test). (h) The Veh and Rut groups spent more time exploring the novel object (two-way RM ANOVA, objects, F(1, 15) = 26.81, P < 0.05; treatments, F(1, 15) = 3.67, P = 0.07; interaction, F(1, 15) = 0.02, P = 0.89, n = 7 for Veh, n = 10 for Evo group, **P < 0.01 under Sidak’s test). (ANOVA indicates analysis of variance; RM, repeated measures; Sidak’s test, Sidak’s multiple comparisons test. Data are represented as mean± SEM.)
The application of Evo and Rut did not affect retrieval in NOR task
Did the applications of Evo or Rut affected retrieval of memory? We further tested this point by using the NOR behavioral paradigm and applied the chemicals at 30 min before the second phase (Figure 7(f)). Similarly, the mice from both groups spent equal time on identical objects. The application of Evo (Figure 7(g)) or Rut (Figure 7(h)) did not change the behaviors in the second phase; they all spent a long time to explore the new object. Moreover, there was no difference between the Evo or Rut application group and the control group. These data suggested that the application of Evo or Rut could not affect memory retrieval in the NOR task.
The application of Rut did not change the body temperature of mice
Given that TRPV1 is involved in temperature detection, and application of Cap can increase the body temperature of subjects. We next wondered whether Evo or Rut would have some effects on body temperature. We measured the body temperature on the left groin and abdomen before and after Evo or Rut application. Interestingly, we found that application of Evo increased the body temperature on left groin and abdomen (two-way repeated measures (RM) ANOVA, Sidak’s test, 30 min, P = 0.01, 40 min, P = 0.02, 50 min, P < 0.01, Figure 8(a)) at 30 min and 50 min after injection (two-way RM ANOVA, Sidak’s test, 30 min, P < 0.01, 40 min, P = 0.02, 50 min, P < 0.01, Figure 8(b)), while Rut did not change the body temperature at all (Figure 8(a) and (b)).
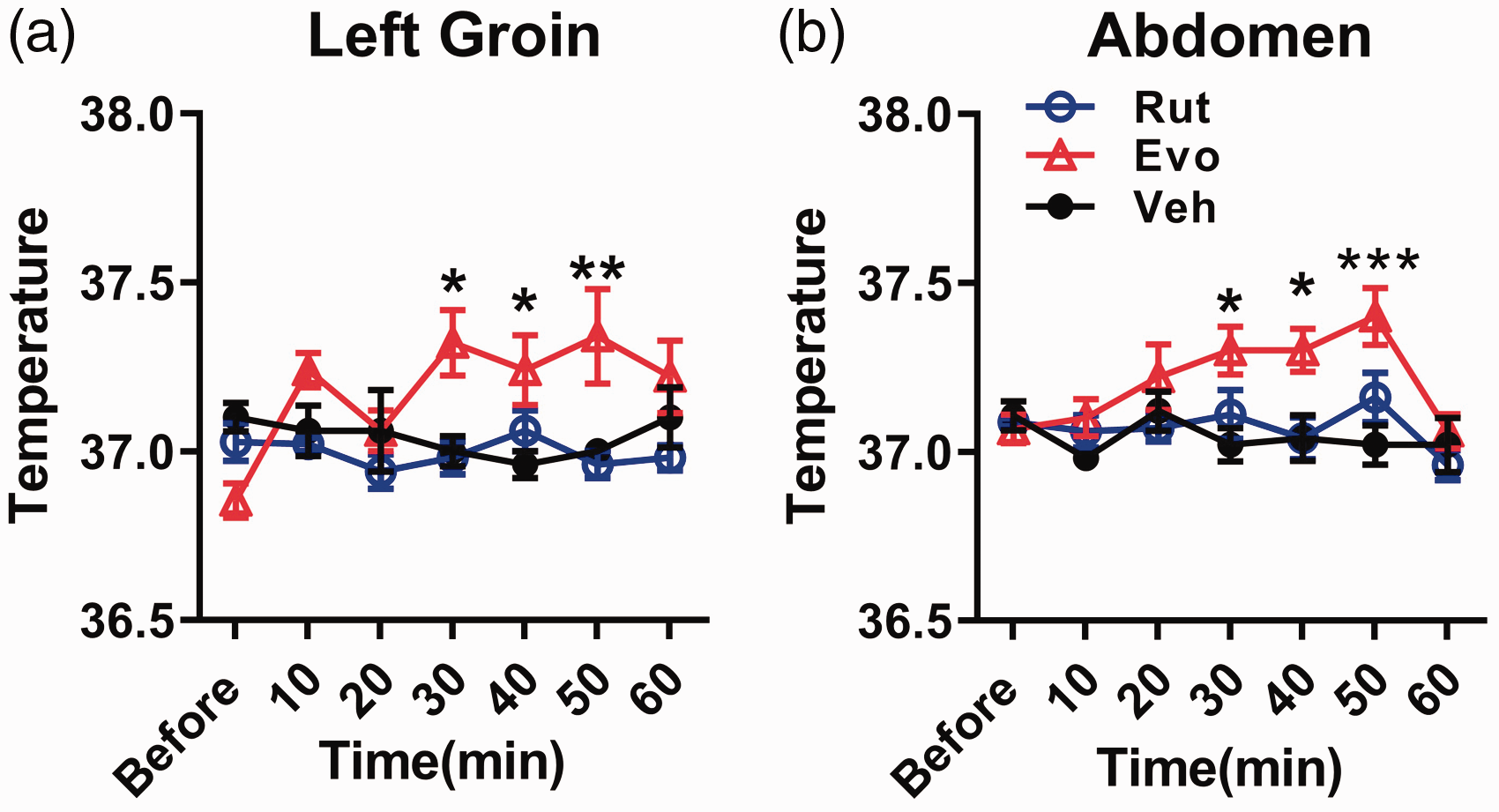
Evo, but not Rut, increased the body temperature of mouse. (a) In the left groin region, the body temperature was elevated relative to Veh 30 min after the injection of Evo, but not the Rut, (two-way RM ANOVA, Veh vs. Evo vs. Rut, F(2, 12) = 4.39, P < 0.05; time points, F(6, 72) = 1.78, P = 0.12; interaction, F(12, 72) = 3.87, P <0.01, n = 5 per group,* P < 0.05 under Sidak’s test). (b) In the abdomen part, the body temperature can be raised after applying Evo (two-way RM ANOVA, Veh vs. Evo vs. Rut, F(2, 17) = 4.82, P = 0.02; time points, F(6, 102) = 3.62, P < 0.05; interaction, F(12, 102) = 1.83, P > 0.05, n = 5 for Veh and Evo, n = 10 for Rut, * P < 0.05 under Sidak’s test). (ANOVA indicates analysis of variance; RM, repeated measures; Sidak’s test, Sidak’s multiple comparisons test. Data are represented as mean ± SEM.)
Discussion
In the current study, we evaluated analgesic effects of Evo and Rut on chronic pain mouse models, we found that both chemicals could alleviate mechanical allodynia but failed to induce a place preference in CPP behavioral test. They increased the time stayed in open arms of EPM at the same doses, but Rut did not change the body temperature at all. Our data suggested that Evo and Rut had analgesic and anti-anxiety effects, and Rut had potential advantages to be developed as an analgesic drug in the future.
The previous data showed that TRPV1 was upregulated in both inflammatory pain and neuropathic pain conditions, 30 and TRPV1 has been treated as an analgesic target for pain treatments. Previous studies showed that these two compounds could bind to TRPV1, 31 chemical method, 32 and computational simulation methods.18,33 Here, our electrophysiological and pharmacological experiments showed that Evo and Rut directly activated TRPV1. The previous study showed that Evo decreased acetic acid-induced writhing, 34 which indicated that Evo could alleviate acute visceral pain. The administration of Evo or Rut intraperitoneally or intrathecally increased PWTs of mice with CPN ligation or CFA injection, respectively. Our data provided the first evidence that Evo and Rut have analgesic effects on chronic pain.
Spontaneous pain is one of the pathological phenomena of neuropathic pain.2,3 Our previous studies have shown that CPN ligation could induce spontaneous pain, 19 examined by condition place preference behavioral paradigm. 35 In the current study, we did not observe the place preference induced by Evo or Rut. This may come from the aversion effects of TRPV1 because we observed spontaneous nociception after the intraplantar injection of chemicals. Evo or Rut may affect the learning ability. However, we did not observe any effects on learning and memory retrieval. Therefore, it would not be possible that the Evo or Rut affect the learning ability and further affect the CPP.
Chronic pain was often accompanied by negative emotions, such as anxiety and depression.36,37 Here, we showed that the application of Evo and Rut increased the entries and time in open arms of EPM, which suggested that they have anti-anxiety effects. The analgesic and anxiolytic effects of Evo and Rut may share the same pathways and neurotransmitter systems. Anterior cingulate cortex was involved in the processing of information about pain and mood (especially anxiety and depression). They might also act directly or indirectly on other brain regions, causing anxiety levels to drop, such as hippocampus, 38 medial prefrontal cortex (mPFC), 39 amygdale, 40 etc. Additionally, the perceived anti-anxiety effects could primarily derive by analgesic effects of these compounds rather than direct anti-anxiety effects. Further studies are needed to investigate detail mechanisms mediating this effect.
Different molecular mechanisms may mediate the effects of Evo and Rut on body temperature. After the application of Cap, an initial burning sensation would be produced, which was an apparent deficiency in the analgesic application of Cap. 41 In the current study, it turned out that Evo raised the body’s temperature; this may come from the acceleration of generated heat and may further lead to weight loss. 42 Rut reduced obesity by inhibiting the expression of two neuropeptides that promoted appetite in the hypothalamus. 43 The application of Rut did not change body temperature. This may also come from the different permeability to blood–brain barrier and thus have different effects on body temperature. Further studies are needed to investigate the exact molecular mechanisms.
Highlights
Application of evodiamine and rutaecarpine reduced peripheral hypersensitivity induced by nerve injury or CFA injection.
Application of capsazepine (i.p.), an inhibitor of TRPV1, blocked the effects of evodiamine and rutaecarpine on peripheral hypersensitivity.
Applications of evodiamine and rutaecarpine increased time in the open arms of the elevated plus maze.
Applications of rutaecarpine did not change the body temperature of the mouse.
Footnotes
Acknowledgments
We thank Dr San-Hua Fang and Dr Li Liu at Core Facilities of the School of Medicine, Zhejiang University. The authors apologize to colleagues whose work could not be cited due to space and reference restrictions.
Author Contributions
W-DZ conducted behavioral experiments, molecular docking, and biochemical experiments; analyzed the data; and prepared the manuscript. X-YC conducted electrophysiology experiments and analyzed the data. CW helped conduct behavioral experiments and biochemical experiments, analyzed the data, and prepared the manuscript. Y-NL, Y-JW, and J-HW helped to analyze the data and prepare the manuscript. FY helped to design the electrophysiology study and analyze the data. C-HL designed the study, analyzed the data, and prepared the manuscript. X-YL designed the study, analyzed the data, and prepared the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the National Program on Key Basic Research Project of China (973 Program; 2014CB548200), the National Natural Science Foundation of China (81571068, 31871062, and 81801102), and the China Postdoctoral Science Foundation (2018M642413).
