Abstract
Background
Management of bone cancer pain is difficult because of its complex mechanisms, which has a major impact on the quality of patients’ daily life. Recent studies have indicated that endoplasmic reticulum stress is involved in many neurological and inflammatory pathways associated with pain. However, the factors that contribute to endoplasmic reticulum stress and its causes in bone cancer pain are still unknown. In this study, we examined whether the endoplasmic reticulum stress response is involved in caspase signaling pathway-dependent apoptosis in neurons in the spinal dorsal horn of tumor-bearing rats and whether it thereby induces bone cancer pain.
Methods
Bone cancer pain was measured behaviorally by the paw withdrawal threshold. The expression levels of endoplasmic reticulum stress markers, namely, immunoglobulin-binding protein/glucose-regulated protein 78 (BIP/GRP78), activating transcription factor-6 (ATF6), phosphorylated inositol-requiring enzyme-1 (p-IRE1), phosphorylated protein kinase RNA-like endoplasmic reticulum kinase (p-PERK) and cleaved caspase-3, were quantitatively assessed by western blot. The distribution of p-eIF2α (an endoplasmic reticulum marker) and cleaved caspase-3 in the lumbar enlargement of the spinal cord was determined by immunohistochemistry in spinal dorsal horn slices.
Results
Bone cancer pain suffered bone damage and persistent mechanical allodynia. Bone cancer pain increased the expression of GRP78, ATF6, p-PERK, p-IRE1, and cleaved caspase-3 in a rat model of bone cancer pain. In addition, p-eIF2α and cleaved caspase-3 were localized to neurons. The intrathecal injection of tauroursodeoxycholic acid as a specific inhibitor of endoplasmic reticulum stress attenuated bone cancer pain and reduced the expression of GRP78, ATF6, p-PERK, p-IRE1, and cleaved caspase-3 in the spinal cord. Moreover, Z-DEVD-FMK (FMK) was also shown to attenuate bone cancer pain.
Conclusion
Endoplasmic reticulum stress causes the activation of caspase signaling pathway-dependent apoptosis in neuronal cells and induces bone cancer pain-related hyperalgesia.
Introduction
Bone cancer pain (BCP) is regarded as a specific type of pain with overlapping but distinct features of both inflammatory and neuropathic pain processes, including background pain, spontaneous pain, and excitation pain.1,2 Despite the availability of many advanced therapeutic methods, BCP is notoriously underreported and undertreated. 3 The treatment of BCP remains a clinical challenge 4 due to relatively frequent adverse side effects.5,6 Therefore, elucidating the basic mechanisms that contribute to BCP in preclinical animal models is key for providing practicable and efficient therapies to patient populations.
The endoplasmic reticulum (ER) is the cellular organelle in which protein folding (the maturation of transmembrane, secretory, and ER-resident proteins), lipid biosynthesis, and Ca2+ storage principally occur. The stability of the internal environment of the ER is important for the function of the ER. Therefore, the ER has a strong internal steady-state system. However, many factors can cause an imbalance in the homeostasis of ER function, resulting in ER stress. Such ER stress initiates an evolutionarily conserved signaling cascade called the unfolded protein response (UPR), which is a self-protective signaling pathway. 7 In other words, multiple perturbations can cause the accumulation of unfolded proteins in the ER and activate the UPR. 8 The role of the UPR is to restore ER function by activating protein kinase R (PKR)-like endoplasmic reticulum kinase (PERK), inositol-requiring enzyme 1 (IRE1), activating transcriptional factor 6 (ATF6), and immunoglobulin-binding protein/glucose-regulated protein 78 (BIP/GRP78). Increased ER stress may be a self-protective signal transcription pathway after mild injury. 9 In contrast, failure to relieve prolonged or excessive ER stress may cause the cell to undergo apoptosis.10,11 Apoptosis refers to the orderly death of cells, and it is controlled by genes to maintain homeostasis. Apoptosis involves the activation, expression, and regulation of a number of genes, and caspase-3 plays an irreplaceable role in this process.
Currently, it is widely accepted that ER stress plays an important role in neuropathic pain.12–16 Inceoglu et al. 14 reported that ER stress in the peripheral nervous system is a significant inducer of pain. Moreover, Zhang et al. 16 reported that ER stress impairment plays a critical role in the spinal dorsal horn (SDH) in a rat model of spinal nerve ligation. In addition, studies have shown that ER stress induces apoptosis. 17 In addition, there is accumulating experimental evidence on the critical role played by apoptosis in hyperalgesia. 18 However, this mechanism has not been tested in a rat model of BCP.
Hence, we formulated a hypothesis that the emergence of ER stress and the subsequent activation of its signaling pathways in cells participates in the facilitation of BCP-like behavior in rats. In the present study, we aimed to investigate the relationship between ER stress and BCP. Initially, we detected changes in the expression and cellular localization of spinal ER stress marker proteins in a rat model of BCP. Furthermore, we investigated whether the ER stress signaling pathway regulates BCP. To further test our hypothesis, we examined whether ER stress signaling influences the production of cleaved caspase-3, the activation form of caspase-3 (caspase-3a), after the inoculation of Walker 256 cells. In addition, changes in the expression and cellular localization of caspase-3a in the SDH were also examined.
Materials and methods
Animals
Female adult Sprague-Dawley rats (180–200 g) were obtained from the Experimental Animal Center of Zhejiang Province Academy of Medical Sciences and housed in plastic cages. Water and food were provided ad libitum, and a 12-h light/dark cycle was maintained constantly. All animal experiments and protocols were approved by the Jiaxing University, Institutional Animal Care and Use Committee (IACUC) and complied with the ethical guidelines of investigations of experimental pain in conscious animals. 19
Sprague Dawley (SD) rats were randomly assigned to several groups: the Sham group, the BCP group, the BCP + Vehicle group (BCP +Veh veh) group (5% DMSO; 30 µl; Sigma-Aldrich), the BCP + Z-DEVD-FMK (BCP + FMK) groups (12.5 µg, 25 µg, and 50 µg; MedChemExpress (MCE)), the BCP + tauroursodeoxycholic acid (BCP + TUD) groups (10 µg, 20 µg, and 40 µg; Sigma), and the BCP + thapsigargin (BCP + Tg) groups (20 µM, 40 µM and 60 µM; Sigma). Z-DEVD-FMK was used as a specific inhibitor of caspase-3.20,21 TUD acid was used as an ER stress inhibitor.22,23 Thapsigargin (Tg) served as an ER stress inducer. 15 These compounds were prepared as 30-µl solutions of different concentrations and injected intrathecally into 9-day-old BCP rats.
Preparation of tumor cells
The protocol for tumor cell preparation was the same as previously described.24,25 Walker 256 rat mammary gland carcinoma cells were obtained from the Cell Bank of Type Culture Collection of Chinese Academy of Sciences (Shanghai, China). The cells were administered to female rats by intraperitoneal injection. Carcinoma cells were extracted and immediately washed with phosphate-buffered saline (PBS) solution. These cells were diluted to a concentration of 107 cells/ml. The Sham groups were injected with the same concentration of carcinoma cells that were heat-killed for 30 min. It is widely accepted that using heat-killed cancer cells as a control is better than simply using normal saline (NS).6,26
Animal model of BCP
To establish an animal model of the clinical pathological process of BCP, we used SD rats and injected Walker 256 tumor cells into the intramedullary space of the left leg as previously reported.24,25,27,28
In summary, each animal was anesthetized with pentobarbital sodium (60 mg/kg, i.p.). After a superficial incision was made in the left leg, the lower one third of the left tibia of the rat was exposed, and a hole was cautiously drilled. Then, Walker 256 cells (107 cells/ml) or heat-killed cells (107 cells/ml for the sham group) were carefully injected into the intramedullary space of the bone. The syringe was then kept in place for 1 min to prevent the cells from leaking out. After removing the syringe, the injection site was immediately blocked with bone wax. The wound was closed and covered with chlortetracycline eye ointment. The animals were placed on a heated blanket for natural recovery.
Intrathecal catheterization
To reduce systemic effects on bone tumors, drug administration was performed through an intrathecal catheter. As previously described, 29 we inserted a PE-10 microtube into the intervertebral space between L4 and L5 for drug delivery. The back side of each rat was shaved after the animal was subjected to pentobarbital sodium anesthesia (60 mg/kg, i.p.). When the intrathecal catheter (PE-10) was inserted into the intervertebral disc and extended to the subarachnoid space of the lumbar enlargement, the microtube was fixed to the adjacent ligament with a 3-0 suture so that 2 cm of the free end of the microtube was exposed and plugged. Then, the surgical incision was sutured. The day after surgery, the paralysis of the two hind limbs within 15 s of after intrathecal delivery of 10 µl of lidocaine through the tube indicated that the catheter was functional. All intrathecal catheterizations were performed immediately after BCP model preparation, and drug administration was reliable nine days after operation. 30
Von Frey test for behavioral assessment
Mechanical allodynia in the left hind paw was measured with von Frey monofilaments (BME-404; Institute of Biological Medicine, Academy of Medical Science, Beijing, China) as previously described. 24 Before each test, the rats were individually arranged in transparent Plexiglas compartments (25 × 20 × 20 cm) on a platform made of metal wires for 30 min to adapt to the new environment. The test was repeated three times with at least a 5-min interval between stimulations. The paw withdrawal threshold (PWT) was expressed as the maximal tolerance in grams, and the mean PWT of each hind paw was calculated as the average value of the three tests. Experimenters blinded to the animal groups performed all of the behavioral testing procedures to ensure the accuracy and reliability of the results.
Western blot analysis
Rats were sacrificed at 5, 9, and 14 days after intramedullary inoculation or at the peak of drug onset by an overdose injection of pentobarbital sodium (80 mg/kg; i.p.). The L3–L5 enlargement tissues was immediately collected and stored in liquid nitrogen until further use. The L3–L5 enlargement was homogenized with buffer containing 150 mM NaCl, 20 mM Tris-HCl (pH 7.6), 1 mM EDTA, 1% Nonidet P-40 (NP-40), 1 mM phenylmethanesulfonyl fluoride (PMSF), protease inhibitor cocktail (Sigma), and phosphatase inhibitor cocktail (Sigma, St. Louis, MO, USA) on ice. After centrifugation at 15,000 r/min for 15 min at 4 °C, the supernatant was collected, and then the protein concentration of each sample was measured using a bicinchoninic acid (BCA) protein concentration assay kit (Pierce, Rockford, IL, USA). A 10% SDS-PAGE gel was used as the separation gel. Equal amounts of protein (30 µg) were loaded in each lane, constant pressure electrophoresis was performed until the proteins were effectively separated, and then electrophoresis was stopped. Next, the separated proteins were transferred to nitrocellulose blots. After the transfer, the blots were immersed in skim milk powder as a blocking solution and shaken gently for 2 h on a shaker at room temperature. Next anti-GRP78 (1:1000, rabbit, Abcam), anti-p-PERK (1:1000, rabbit, Cell Signaling Technology), anti-p-IRE1 (1:1500, rabbit, Affinity), anti-ATF6 (1:1500, rabbit, Affinity), and anti-cleaved caspase-3 (1:1000, rabbit, Affinity) primary antibodies were diluted in blocking buffer and gently shaken overnight at 4 °C on a shaker. Nonspecifically bound primary antibodies were washed by shaking in PBS+0.1%Tween-20 (PBST), and the membrane was placed in a skim milk powder containing an horseradish peroxidase (HRP)-labeled secondary antibody (1:10,000, Jackson Immunolab) at room temperature for 1.5 h. Immunoreactive bands were detected by using enhanced chemiluminescence (Thermo Scientific) and exposed to X-ray films. β-Tubulin was used as an internal control.
Immunofluorescence
Immunofluorescence staining and double immunostaining were performed as previously described. 28 Briefly, four rats from each group were selected, deeply anesthetized, and exsanguinated. Then, the lumbar enlargements were removed, soaked in 4% phosphate-buffered paraformaldehyde for 4 to 6 h at 4 °C, and subsequently dehydrated in a 10% to 30% gradient of sucrose in sterile water for five to seven days at 4 °C. Next, the lumbar enlargement of the spinal cord tissues was frozen in optimal cutting temperature (OCT) compound (Sakura, America) in a cryostat at −25°C and then sliced at a thickness of 20 µm. The sections were first blocked with 4% normal donkey serum, 0.03% Triton X-100, and PBS for 1 h at room temperature. The sections were then incubated overnight at 4 °C with the following primary antibodies: p-eIF2α (1:200, rabbit; Affinity), cleaved caspase-3 (1:150, rabbit, Affinity), glial fibrillary acidic protein (1:1000, mouse; Sigma), IBA-1 (1:250, mouse; Abcam), and NeuN (Mouse,1:1000, Abcam). The sections were washed with PBST and incubated for 2 h with fluorescein isothiocyanate- or Cy3-conjugated secondary antibodies (1:500, Abcam) at room temperature. Finally, the stained sections were surveyed with an Olympus fluorescence microscope, and images were acquired with a CCD Spot camera. Finally, the images were analyzed using Image Pro-Plus 6.0 (Image Pro-Plus Kodak, USA).
Histological analysis of bone
Rats were injected with an overdose of pentobarbital (80 mg/kg; i.p.) and sacrificed nine days after tumor inoculation. Tissues from the left tibia around the inoculation site (a total of 1 cm) were collected. The tibia bones were decalcified in 10% EDTA solution for 24 h. Then, the bone tissues were dehydrated, embedded in paraffin, and sliced into 8-µm-thick sections by a rotary microtome (Reichert-LEICA RM2235, Germany). The sections were stained with hematoxylin and eosin (H&E) to verify the extent of tumor cell infiltration. All images were captured using a 10× or 20× objective with a microscope (OLYMPUS BX 51, Japan).
Statistical analysis
Statistical significance was examined by SPSS version 20.0. The results are expressed as the mean ± standard error of the mean. Differences between the groups were analyzed with one-way analysis of variance (ANOVA) followed by Bonferroni multiple comparison posttest or two-way repeated measures or three-way repeated measures ANOVA followed by Newman–Keuls post hoc test. The Shapiro–Wilk test was used to evaluate the distribution of the data. For homogeneity analysis, we used the F test to test equality of variances for T tests and Levene’s test of equality of error variances for ANOVA. P < 0.05 was considered to be statistically significant.
Results
Intramedullary inoculation of Walker 256 breast carcinoma cells induces bone destruction
The BCP model that was used exhibited an obvious decrease in the PWT at 5 to 21 days after intramedullary inoculation. Specifically, the PWT of tumor-bearing rats was obviously decreased on day 9 (***P < 0.001; BCP group vs. Sham group; one-way ANOVA; n = 10) after inoculation and progressively decreased throughout the following 12-day post-operation observation period. Rats that were inoculated with heat-killed cells did not show pain-related behavior (Figure 1(a)). As previously described, nine days after operation is optimal for observing cancer-induced pain,24,31,32 and molecular investigations were performed on days around this time point, namely, 0, 5, 9, and 14 days after intramedullary inoculation. Histopathological analysis was performed to confirm the development of bone cancer in these rats. Histopathological images showed that a large number of morphologically altered cells infiltrated the medullary cavity. These abnormal cells exhibited a large nucleus, a high proportion of nucleoplasm, and typical pathological changes, such as nuclear pyknosis, which indicates infiltration in tumor cells. Collectively, the histopathological images indicated obvious tumor infiltration nine days after intramedullary inoculation. In contrast, no destruction was observed in rats inoculated with heat-killed cells on day 9 after operation (Figure 1(b)).
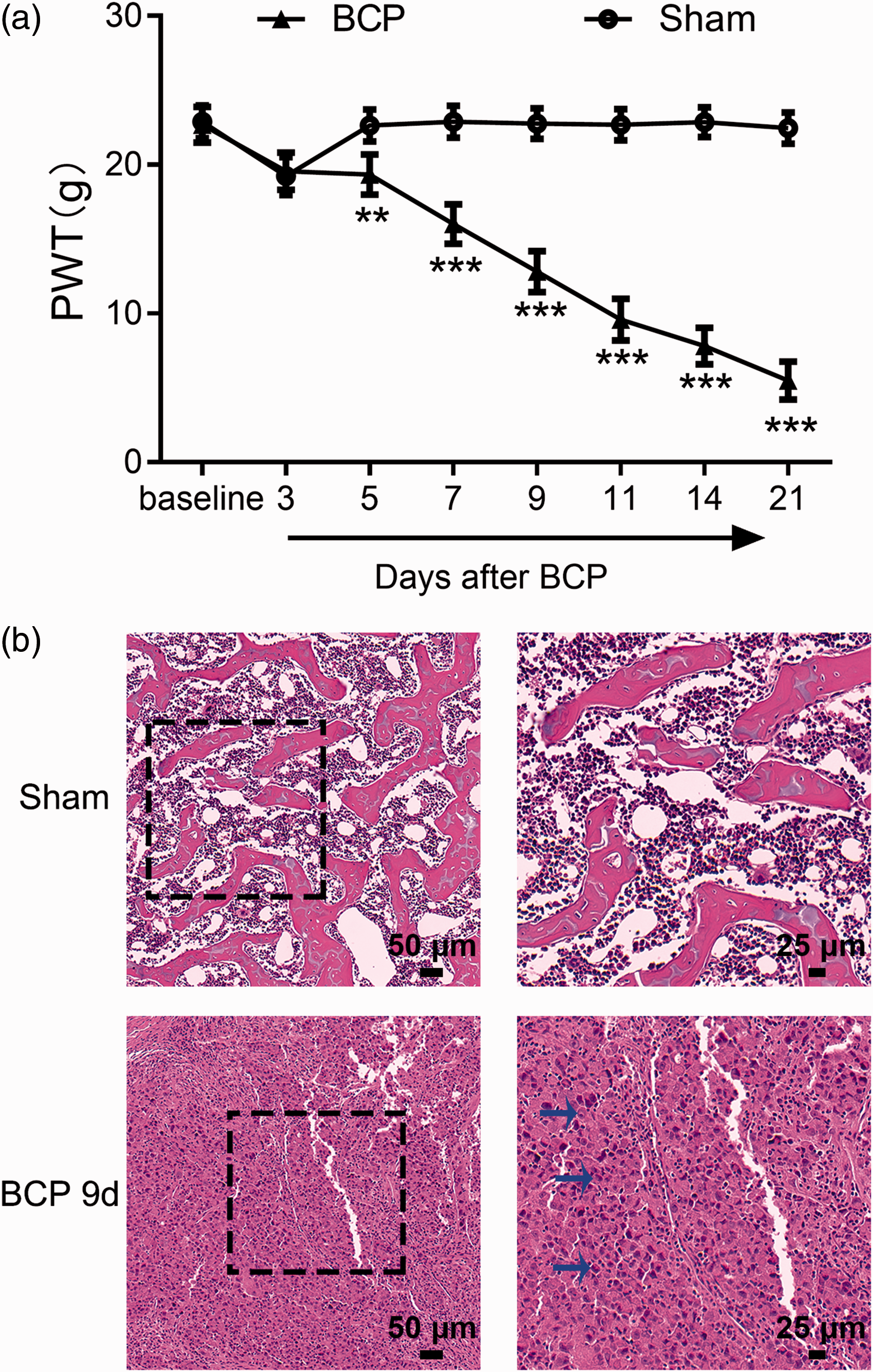
Behavioral and histochemical analysis of tumor development in the left tibia of BCP rats. (a) The PWT of the left hind paw after tumor inoculation. Compared with that in the Sham group, the PWT of the left hind paw in the BCP group began to decrease on the fifth day. Sham group rats showed no significant change in pain sensitivity (**P < 0.01, ***P < 0.001; vs. Sham group; one-way ANOVA followed by the Bonferroni test; n = 10). (b) Histopathological images of hematoxylin and eosin staining of tibia samples on the ninth day after tumor cell inoculation showed a large number of cells infiltrating the medullary cavity (blue arrow). The data are presented as the mean ± standard error of the mean in all figures and throughout the text. BCP: bone cancer pain; PWT: paw withdrawal threshold.
Tumor inoculation induced the overexpression of ER stress markers and cleaved caspase-3 in SDH
We established an animal model of BCP in rats and then observed the participation of ER stress in the pain process. The expression of ER stress markers was detected in spinal tissues of rats by western blotting. The significant upregulation of GRP78 (*P < 0.05, **P < 0.01; BCP group vs. Sham group; One-way ANOVA; n = 4), ATF6 (*P < 0.05, **P < 0.01; BCP group vs. Sham group; One-way ANOVA; n = 4), p-PERK (*P < 0.05, **P < 0.01; BCP group vs. Sham group; One-way ANOVA; n = 4), and p-IRE1α (*P < 0.05, **P < 0.01; BCP group vs. Sham group; One-way ANOVA; n = 4) was observed in BCP rats (Figure 2(a)), which suggests that UPR pathways were activated. We also observed a significant increase in the expression of cleaved caspase-3 (Figure 2(b)) (*P < 0.05, **P < 0.01; BCP group vs. Sham group; One-way ANOVA; n = 4), which is only present in cells that are undergoing apoptosis in the pain process. Spinal tissues from sham rats (nine days after sham operation) and vehicle-treated rats (nine days post catheterization) served as controls for all western blot experiments.

Western blot analysis of changes in the expression of ER stress marker proteins (GRP78, ATF6, p-IRE1, and p-PERK) and the levels of caspase-3a over time in the spinal horn. The spinal expression of ER stress markers and caspase-3a was enhanced in BCP rats. Compared with that in the Sham group, the expression of GRP78 (c) and ATF6 (d) in the spinal dorsal horn in the BCP group showed significant changes on five days after the establishment of the model. The expression levels of ER stress markers and cleaved caspase-3 (caspase-3a) in the dorsal horn of the spinal cord were increased 9 days and 14 days after the establishment of the model, and the differences were statistically significant (*P < 0.05, **P < 0.01, ***p < 0.001; vs. Sham group, two-way or one-way ANOVA followed by the Bonferroni test; n = 4). BCP: bone cancer pain; GRP78: glucose-regulated protein 78; ATF6: activating transcription factor-6; p-IRE1: phosphorylated inositol-requiring enzyme-1; p-PERK: phosphorylated protein kinase RNA-like endoplasmic reticulum kinase.
Effects of intrathecal injections of TUD acid and thapsigargin on hyperalgesia behavior in BCP rats
To examine whether ER stress is sufficient to induce BCP, we conducted the following experiment. Nine days after the establishment of the BCP model, agonists and inhibitors of ER stress were administered.
We found that, compared to the administration of vehicle, the intrathecal administration of TUD acid significantly increased the PWT. In other words, nociceptive behavior was attenuated by TUD (Figure 3(a)). The analgesic effect of a 20-µg dose was observed 4 h after injection, peaked at 7 h, and lasted for 8 h (P < 0.001; BCP + TUD group vs. BCP + Veh group; One-way ANOVA; n = 10). Subsequently, we selected the optimal dose and time points for western blot verification, and the successive experimental parameters were in accordance with this standard. The injection of TUD reduced the expression of a series of markers of ER stress in the SDH tissue of BCP rats (Figure 4(a)).
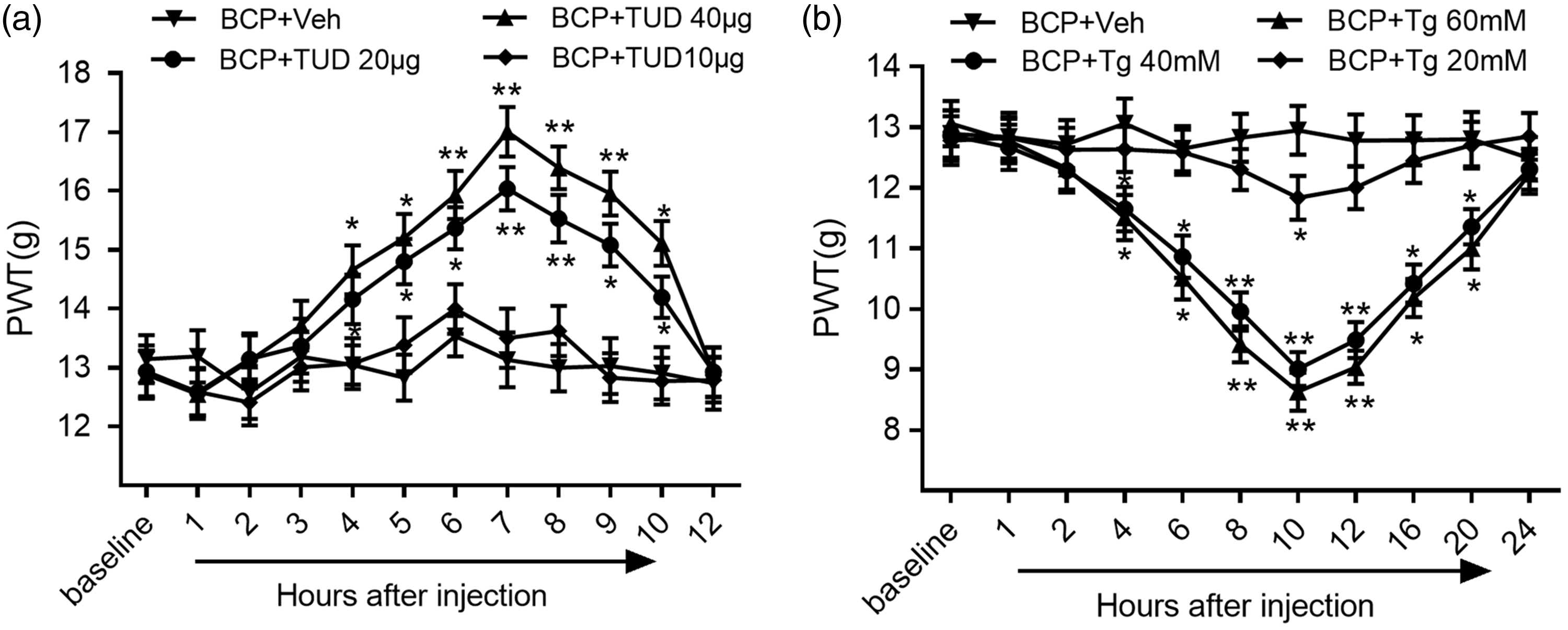
The effects of treatment with graded doses of an endoplasmic reticulum stress-specific inhibitor (TUD) or an ER stress inducer (Tg) on the PWT after tumor inoculation in rats. (a) The intrathecal injection of TUD downregulated the expression of ER stress markers in the spinal dorsal horn and attenuated abnormal mechanical pain in the BCP group compared with the vehicle group. (b) The effects of Tg on mechanical allodynia, as shown by the von Frey test (*P < 0.05, **P < 0.01; vs. BCP +Veh group; two-way or one-way ANOVA followed by the Bonferroni test; n = 10 rats in each group). BCP: bone cancer pain; PWT: paw withdrawal threshold; TUD: tauroursodeoxycholic acid; Tg: thapsigargin.
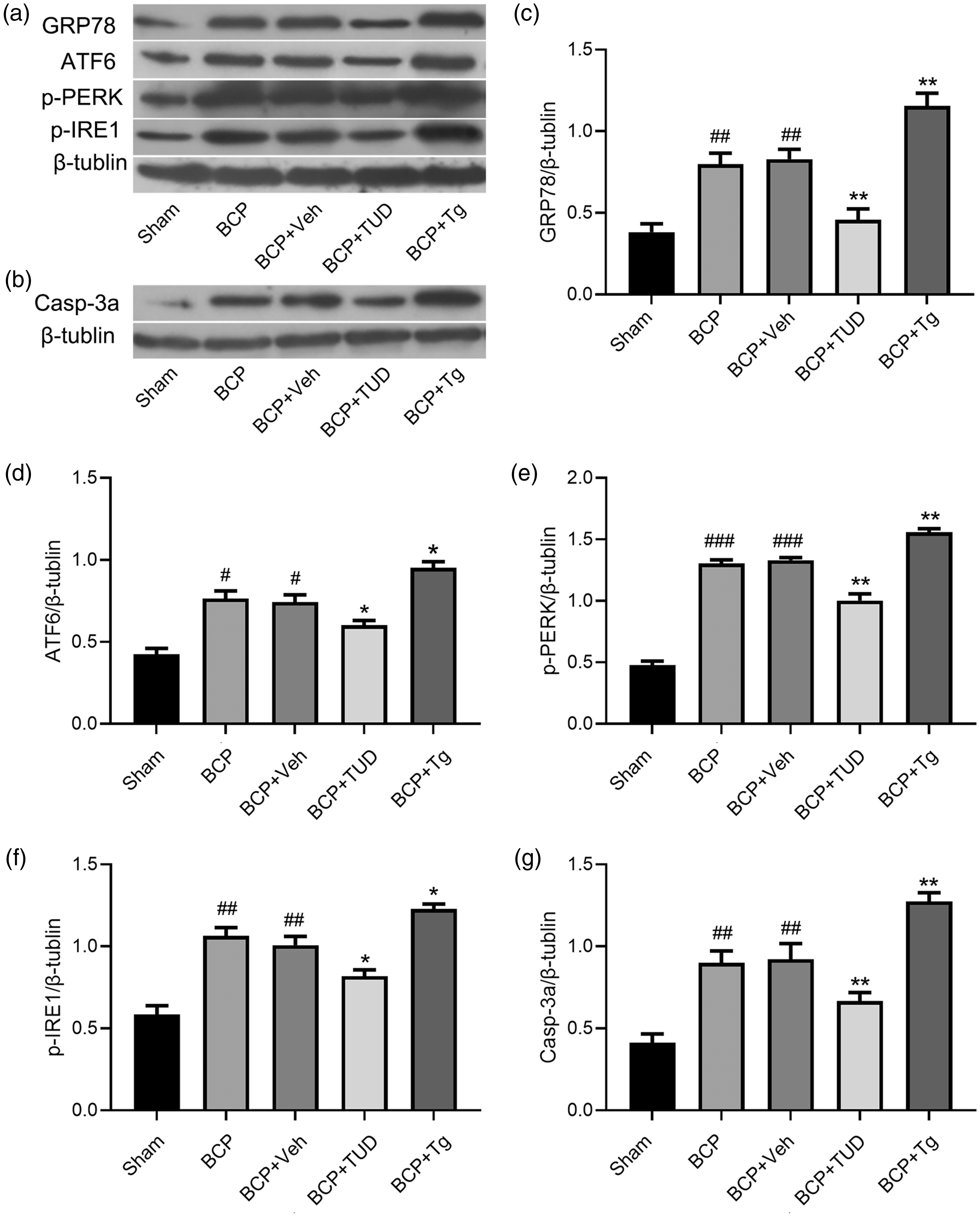
Western blot analysis of the changes in the expression of ER stress marker proteins (GRP78, ATF6, p-PERK, and p-IRE1) and the level of caspase-3a after the intrathecal injection of a specific inhibitor of endoplasmic reticulum stress (TUD) or an ER stress inducer (Tg) (#P < 0.05,
In addition, we examined behavioral changes in rats with BCP after the administration of an agonist of ER stress. The nociceptive behavior of rats with BCP was more significant after intrathecal administration (Figure 3(b)). Hyperalgesia occurred 4 h after the injection of thapsigargin (Tg), peaked at 10 h, and lasted for 20 h (P < 0.001; the BCP + Tg group vs. BCP + Veh group, One-way ANOVA; n = 10). Western blot indicated that the expression of ER stress markers in the spinal tissues of BCP rats was significantly upregulated after the intrathecal administration of Tg (Figure 4(a)).
Effects of intrathecal injections of TUD acid and thapsigargin on the activation of caspase-3a
After the intrathecal administration of an inhibitor and an agonist of ER stress, we also examined changes in caspase-3a expressed by western blot analysis at peak efficacy (Figure 4(b)). Treatment with TUD acid resulted in a lower expression level of caspase-3a in the BCP + TUD group than in the vehicle group (P < 0.05; BCP + TUD group vs. BCP + Veh group; One-way ANOVA; n = 10), caspase-3a was upregulated after the administration of thapsigargin (Tg) compared to after the administration of the solvent (P < 0.05; BCP + Tg group vs. BCP + Veh group; One-way ANOVA; n = 4).
Caspase-3a was involved in nociceptive pain, and Z-DEVD-FMK downregulated time‑ and dose‑dependent mechanical allodynia in BCP rats
To verify whether caspase-3a is involved in the development of BCP, 9 days after the establishment of the BCP model, we administered Z-DEVD-FMK intrathecally and described its effects on behavioral pain, specifically the PWT.
Behavioral assessments were performed 1 h prior to intrathecal injection. According to the results, increases in the PWT induced by the post-intrathecal injection of Z-DEVD-FMK (Figure 5(a)) lasted for 5 h, peaked at 7 h, and returned to a normal level at 10 h (P < 0.001; BCP + FMK group vs. BCP + Veh group; One-way ANOVA; n = 10). The role of Z-DEVD-FMK in the development of spinal nociceptive pain in the BCP model was then determined by using the experimental protocol shown in Figure 5(b). Compared with that in the vehicle group, the expression in the FMK groups was downregulated (P < 0.05; BCP + Tg group vs. BCP + Veh group; One-way ANOVA; n = 4).
ER stress and caspase-3a were both mainly co-localized with neurons
To the cell types of ER stress in the SDH, we selected p-eIF2α as a marker protein for ER stress and performed immunofluorescence double staining. 15 As shown in Figure 6, ER stress occurred in neuronal cells, as indicated by staining with p-eIF2α. As caspase-3a was also localized to neuronal cells, it was considered a detectable marker of apoptosis within neurons (Figure 7).

Effect of the intrathecal administration of a specific inhibitor of caspase-3 (Z-DEVD-FMK, FMK) nine days after the operation. (a) The expression of tumor cell inoculation-induced hypersensitivity and caspase-3a was reversed by Z-DEVD-FMK. Compared vehicle administration, the intrathecal administration of Z-DEVD-FMK significantly elevated the PWT of the BCP rats in a dose-dependent manner (***P < 0.001 vs. BCP + Veh group; n = 10). (b) Spinal dorsal horn tissues were collected 6 h after injection. FMK reversed the upregulation of caspase-3a in BCP rats (
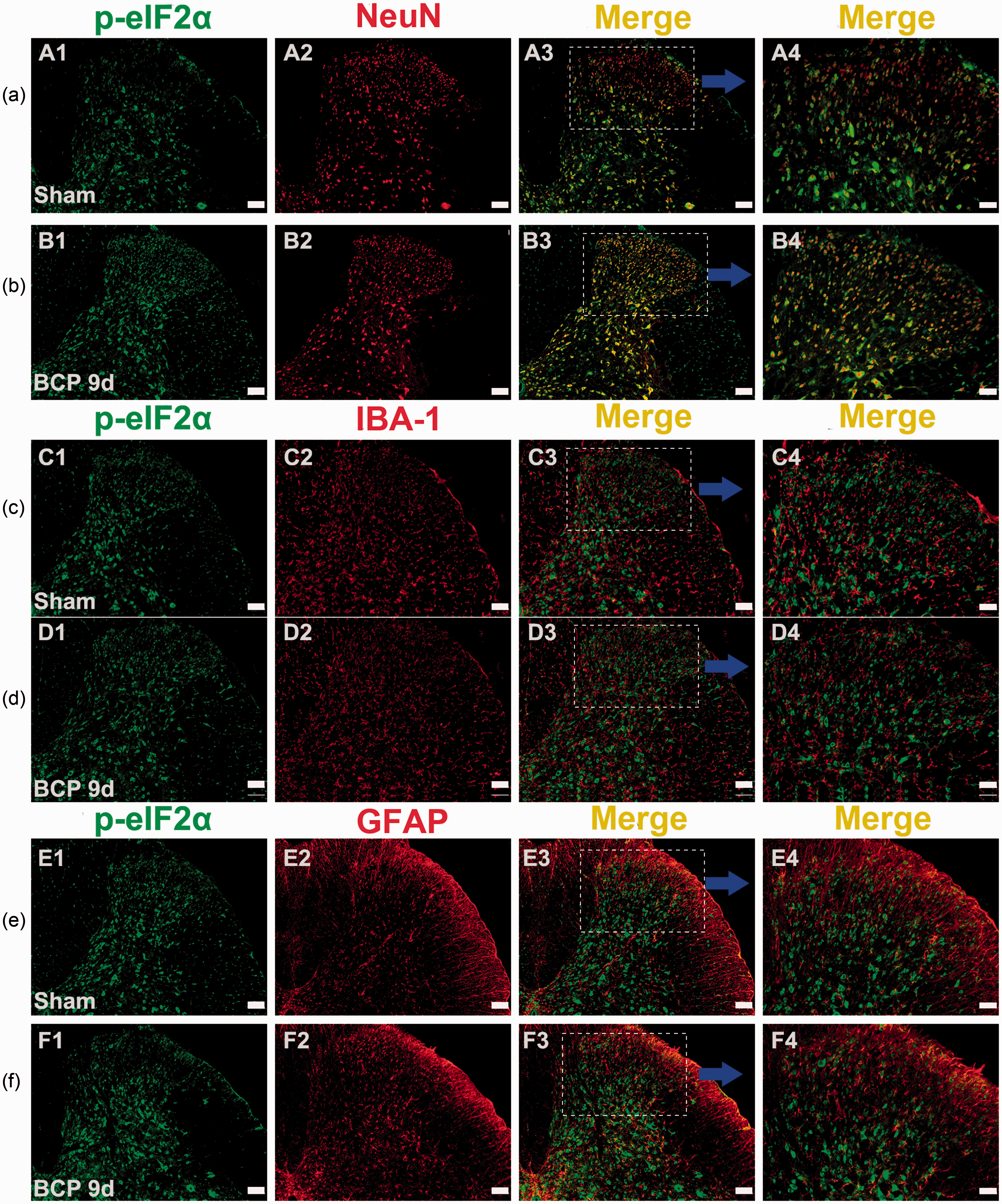
Immunofluorescence showing that p-eIF2α was localized to neurons in the spinal dorsal horn. (a and b) Double immunostaining revealed that p-eIF2α (green) was colocalized with a neuronal marker (red) but not with the microglia marker IBA-1 (red) (c and d) or the astrocyte marker GFAP (red) (e and f). Original magnification: 100 or 200×; scale bars (A1–A3, B1–B3, C1–C3, D1–D3, E1–E3, and F1–F3), 100 µm; scale bars (A4, B4, C4, D4, E4, and F4), 50 µm; n = 4 in each group. BCP: bone cancer pain; GFAP: glial fibrillary acidic protein.
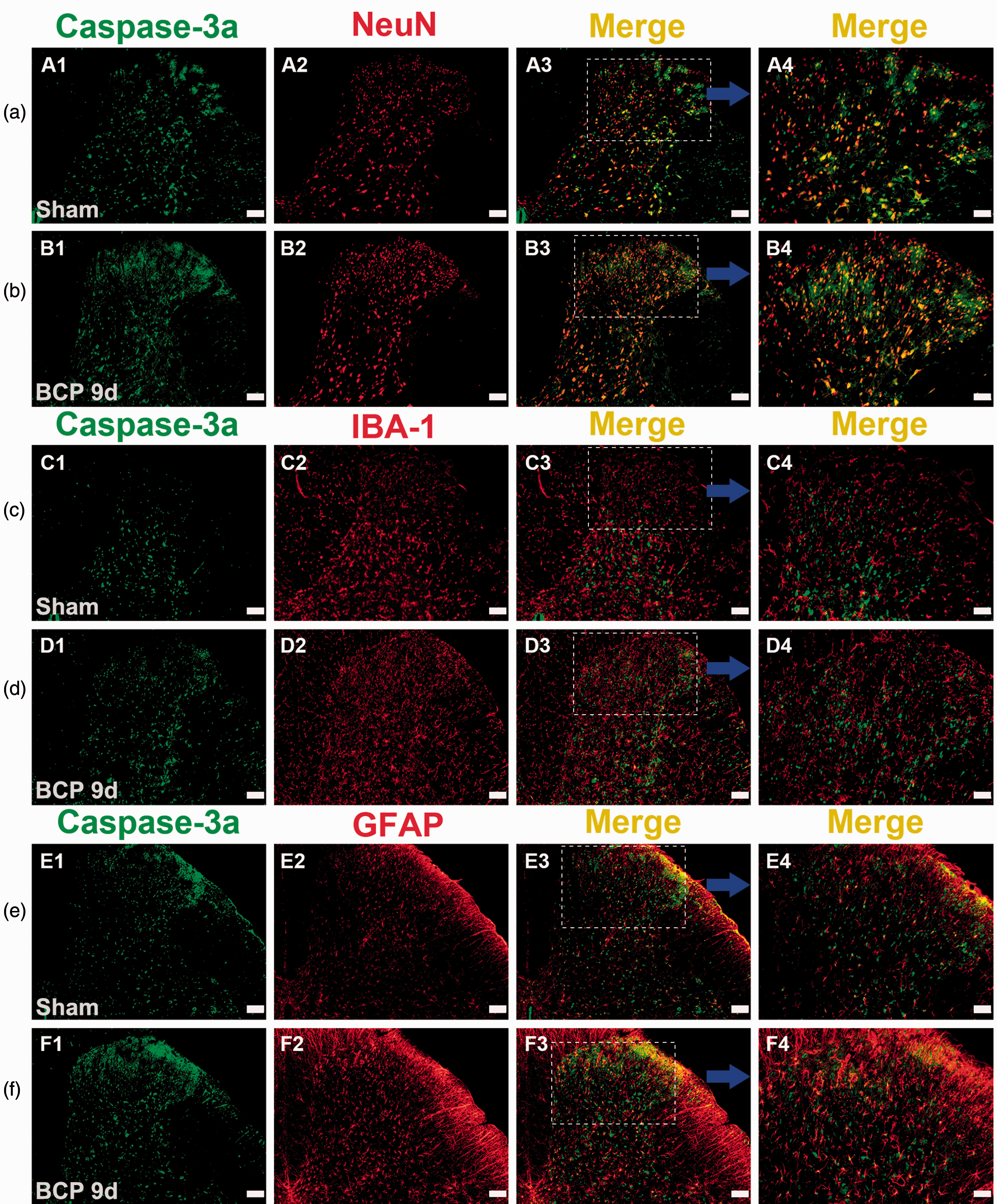
Immunofluorescence showed that caspase-3a was localized to the spinal dorsal horn. (a and b) Double immunostaining revealed that caspase-3a (green) was colocalized with a neuronal marker (red), (c and d) but not with the microglia marker IBA-1 (red) and rarely with (e and f) the astrocyte marker GFAP (red). Original magnification: 100× or 200×; scale bars (A1–A3, B1–B3, C1–C3, D1–D3, E1–E3, and F1–F3), 100 µm; scale bars (A4, B4, C4, D4, E4, and F4), 50 µm; n = 4 in each group. BCP: bone cancer pain; GFAP: glial fibrillary acidic protein.
Discussion
In this study, we successfully established a BCP model by injecting Walker 256 cells into the intramedullary space of the left leg of SD rats and examined the relationship between BCP and ER stress as well as the subsequent activation of its signaling pathways. First, BCP induced the persistent upregulation of ER stress in SDH neurons, and apoptosis was also detected during this pathological process. Second, the inhibition of ER stress not only alleviated BCP but also decreased apoptosis upregulation. Conversely, the activation of ER stress increased cancer pain hypersensitivity and apoptosis. Third, the inhibition of caspase-3a by Z-DEVD-FMK (FMK) dose-dependently alleviated BCP. Based on our data, ER stress was suggested to induce the occurrence of apoptosis, which plays an important role in the regulation of hyperalgesia in BCP rats.
As an important membranous organelle of eukaryotic cells, the ER carries out different functions, such as intracellular lipid metabolism and protein folding and aggregation after protein synthesis, which are important for the physiological regulation of cells. Therefore, ER function is a key to cell survival and the maintenance of the normal physiological functions of the body. 33 Prolonged ER stress in neurons may be the initial event underlying neurodegenerative disorders.34,35 Indeed, ER stress has been reported in various neuronal diseases, such as Parkinson’s disease, prion disease, amyotrophic lateral sclerosis (ALS), Alzheimer’s disease, and other disorders.36–39 Herein, we found that the suppression of spinal ER stress can significantly reduce BCP-related nociceptive behavior in rats. Simultaneously, the intrathecal ER stress inducer increased nociceptive behavior was noticed in BCP model. In addition, ER stress was activated in SDH neurons after the inoculation of Walker 256 cells. Recently, increasing research has suggested that ER stress is involved in the modulation of nociceptive signal transmission in neuropathic pain.12–16,34,39 It has been reported that ER stress is associated with oxidative stress in neuropathic pain and the disinhibition of the spinal nociceptive circuitry. 15 It has also been reported that ER stress is involved in central sensitization caused by N-methyl-D-aspartate receptor activity in formalin-induced pain. 12 The interesting outcome of the present study was that the intrathecal injection of drugs that regulate ER stress can affect the expression of cleaved caspase-3. To explore whether the activation of cleaved caspase-3 is involved in the regulation of hyperalgesia, BCP rats were injected with a caspase-3-specific inhibitor (FMK). The role of FMK in attenuating mechanical allodynia was represented by the PWT.
Caspase-3 is one of the key indicators of apoptosis as it is responsible for the proteolytic cleavage of many key proteins. Previous studies have reported that caspase inhibitors can prevent pain-related behavior,40–42 and conversely, caspase signaling pathways can contribute to pain,43,44 which is consistent with our findings. Traditionally, the basic function of neurons is to exchange information by accepting, integrating, transmitting, and presenting information. Recently, many studies have indicated that the loss of neurons is mainly involved in pain.45–47 In our BCP model, carcinoma cells stimulated local inflammatory mediators and created a highly acidic environment. 48 Due to the destruction of nerve endings caused by the microenvironment of bone tumor invasion, a long-lasting increase in the excitability of SDH neurons (central sensitization) is induced. 43 The process of central sensitization may cause neurons in the spinal cord to undergo pathologically high performance and high metabolism, 49 which may play an important role in ER stress and cell death (apoptosis). However, the causal relationship between central sensitization and apoptosis is still controversial. It is interesting that the disinhibitory process associated with excitotoxicity-induced neuronal death is considered the cause of the induction of dorsal horn neural sensitization. 50 Elevated levels of glutamate in the cancer invasion process may participate in nociceptive signaling. 51 Moreover, excitotoxicity caused by the excessive release of glutamate may impair mitochondrial function, rapidly increase the concentration of reactive oxygen species (ROS), and ultimately lead to apoptosis in neurons and the development of hypersensitivity.52–54 However, these contradictory views indicate that apoptosis in neurons is involved in the regulation of hyperalgesia, which is consistent with our results.
In addition, we detected that both ER stress and apoptosis mainly occur in SDH neurons. ER stress has been reported to occur simultaneously in neurons, microglia, and astrocytes, 15 which is inconsistent with our experimental results. This interesting difference may be due to the different pain models used, and our findings may represent a special mechanism of our model of BCP. Furthermore, studies have noted that high mobility group B-1 (HMGB-1) promotes apoptosis by inducing ER stress. 55 Our previous study showed that HMGB-1 is mainly co-localized with neurons, 24 and hence, we hypothesized that HMGB-1 may induce ER stress in neurons in our BCP model.
In conclusion, we confirmed that ER stress is involved in the development of BCP in rats, which has a strong connection with caspase signaling pathway-dependent neuronal apoptosis. Previous research has reported that non-dividing cells are more susceptible to ER stress, consequently making neurons a noticeable target for ER stress-induced cellular dysfunction. 34 Thus, protecting neurons from apoptosis may be a new strategy for treating BCP. The study reported here is important from a fundamental perspective because the association between bone cancer-induced pain and ER stress was notable. It is also important from an applied perspective because treatment for BCP remains a significant unmet clinical need. Investigating the specific mechanism underlying BCP in response to ER stress requires further investigation in the future.
Supplemental Material
MPX876150 Supplemental Material1 - Supplemental material for Endoplasmic reticulum stress promoting caspase signaling pathway-dependent apoptosis contributes to bone cancer pain in the spinal dorsal horn
Supplemental material, MPX876150 Supplemental Material1 for Endoplasmic reticulum stress promoting caspase signaling pathway-dependent apoptosis contributes to bone cancer pain in the spinal dorsal horn by Qiuli He, Tingting Wang, Huadong Ni, Qianying Liu, Kang An, Jiachun Tao, Yajing Chen, Longsheng Xu, Chunyan Zhu and Ming Yao in Molecular Pain
Supplemental Material
MPX876150 Supplemental Material2 - Supplemental material for Endoplasmic reticulum stress promoting caspase signaling pathway-dependent apoptosis contributes to bone cancer pain in the spinal dorsal horn
Supplemental material, MPX876150 Supplemental Material2 for Endoplasmic reticulum stress promoting caspase signaling pathway-dependent apoptosis contributes to bone cancer pain in the spinal dorsal horn by Qiuli He, Tingting Wang, Huadong Ni, Qianying Liu, Kang An, Jiachun Tao, Yajing Chen, Longsheng Xu, Chunyan Zhu and Ming Yao in Molecular Pain
Footnotes
Acknowledgments
The authors thank Renshan Ge (Wenzhou Medical University, China) for proofreading the paper.
Authors’ Contributions
MY conceived and designed the study. QLH performed the experiments. HDN, QYL, and LSX coordinated and supervised the experiments. JCT and CYZ participated in part of the behavioral testing and immunofluorescence staining experiments. YJC performed the data analysis. QLH, TTW, and HDN wrote the article. All authors read and approved the final manuscript.
Availability of data and materials
All data generated or analyzed during this study are included in this published article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics Approval
All animal procedures performed in this study were carried out in accordance with the guidelines of the International Association for the Study of Pain and were approved by the Zhejiang Chinese Medicine University Committee on Ethics in the Care and Use of Laboratory Animals.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was supported by the National Natural Science Foundation of China (81341035), Natural Science Foundation of Zhejiang Province (LY16H090016, LQ19H090007), Medicine and Health Care General Studies Program of Zhejiang Province (2015KYA217), Zhejiang Provincial Program for the Cultivation of High-Level Innovative Health Talents (2012-RC-22), the Construction Project of Anesthesiology Discipline Special Disease Center in Zhejiang North Region (201524), Jiaxing City Science and Technology Project (2017AY33008, 2018AY32012), and key disciplines of pain construction in the city and Zhejiang Province.
Supplemental Material
Supplemental material is available for this article online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
