Abstract
Background:
Chemokine, monocyte chemoattractant protein-1 (MCP-1), is a potential factor to cause cancer-induced bone pain (CIBP). NF-κB signaling is very important in mediating the expression of chemokines and may have a role in CIBP. However, the mechanism is still unclear. This study investigates the role of NF-κB in CIBP by regulating MCP-1/chemokine CC motif receptor-2 (CCR2) signaling pathway.
Methods:
A rat CIBP model was established by injecting Walker-256 cells into the tibia medullary cavity. Nine days later, animals were intrathecally administrated with MCP-1 neutralizing antibody, CCR2 antagonist (RS504393), or NF-кB inhibitor (BAY11–7081). Mechanical paw withdrawal threshold was used to assess pain behavior and sciatic functional index, and radiographic images were adopted to evaluate the damage of nerve and bone. The spinal cords were harvested for Western blot and quantitative reverse transcription polymerase chain reaction. The distribution of MCP-1, CCR2, and NF-кB was detected by double immunofluorescent staining.
Results:
CIBP caused remarkable bone destruction, injury of sciatic and femoral nerve, and persistent (>15 days) mechanical allodynia in rats. Tumor cell inoculation upregulate MCP-1 and NF-кB in activated neurons as well as CCR2 in neurons and microglia of the spinal cord. MCP-1 antibody, RS504393, and BAY11–7081 partially reversed CIBP-induced mechanical allodynia, and CIBP regulated the expression levels of pro-inflammatory cytokines, tumor necrosis factor-α and interferon-γ, and anti-inflammatory cytokine, interleukin 4, and BAY11–7081 lowered CIBP-induced MCP-1 and CCR2 expressions in a dose-dependent manner.
Conclusion:
In conclusion, NF-кB signaling pathway regulates the expressions of MCP-1/CCR2-induced inflammatory factors in the spinal cord of CIBP rats.
Introduction
Cancers have become the major threat to human health. About 70% patients with terminal cancers suffer from bone metastases, especially, spinal and long bone metastases. 1 Bone metastasis can cause severe bone pain. 1 Statistically, 60%–90% patients with advanced cancers have been reported to suffer from varying degrees of pain, and 30% of them have experienced severe pain.2,3 Surgery, radiotherapy, chemotherapy, nerve block technique, adoptive tumor immunotherapy, and drugs have been used to treat cancer-induced bone pain (CIBP). 4 However, current clinical therapies for persistent CIBP are limited, and some often cause serious side effects. 5 Therefore, new analgesic treatments are urgently needed.
Inflammation may be involved in CIBP. Recent evidence has shown that monocyte chemoattractant protein-1 (MCP-1) bound to chemokine CC motif receptor-2 (CCR2), taking parting in chronic pain.6–9 Several research groups have reported that MCP-1 was expressed in primary sensory neurons in the dorsal root ganglion (DRG) and spinal cord, and this expression increases following nerve injury and bone cancer.7,10–12 MCP-1 expression has also been demonstrated in astrocytes.13,14 Previous findings have suggested that MCP-1 may act at multiple sites causing the development of pain. Moreover, the precise sites of its action may differ in various models of pain, 15 such as neuropathic pain and CIBP. CIBP may result from the increased levels of MCP-1 in spinal cord; it is potentially released from DRG neurons at the dorsal horn of spinal cord or from spinal cord neurons and glial cells.16,17 CCR2 is a G protein-coupled receptor, which expressed in various cell types including microglia, 6 astrocytes, 18 and neurons in the spinal cord.17,19 Accumulated evidences indicate that the expression of CCR2 is upregulated in the spinal cord in several kinds of pain models, including peripheral nerve injury, 20 spinal nerve ligation, 17 spinal cord contusion injuries, 18 and bone cancer pain. 21
Nuclear factor kappa B (NF-κB) is a ubiquitous transcription factor. NF-κB signaling pathway serves as a transducer between extracellular signals and gene expression (e.g., cytokines, chemokines, iNOS) and is important for immunity, inflammation, central nervous system injury, and neuropathic pain.22,23 Emerging evidence has shown that NF-кB mediates chemokines such as CX3CL1, CXCL1 following tissue injury or nerve damage, and cause chronic pain and CIBP.24–26 The aim of this study is to investigate whether NF-кB signaling pathway mediates MCP-1 and CCR2 expression in the spinal cord of CIBP rats, and how to influence the expression of MCP-1/CCR2 signaling.
We initially detected the expression changes and cellular localization of spinal MCP-1, CCR2, and NF-кB in a rat model of CIBP by quantitative reverse transcription polymerase chain reaction (qRT-PCR), Western blot, and double immunofluorescence staining. Furthermore, we investigated whether NF-кB signaling pathway regulated the production of MCP-1/CCR2 signaling using qRT-PCR and Western blot. In addition, using Western blot, we investigated whether MCP-1/CCR2 signaling involved in CIBP influenced the production of downstream pro-inflammatory and anti-inflammatory factors.
Material and methods
Establishment of CIBP rat model
Female Sprague Dawley (SD) adult rats (nine weeks, 180–200 g) were purchased from ZheJiang Academy of Medical Sciences. Animals use license: scxk (Zhe) 20160011. All rats were allowed free access to food and water and were housed in an environment with temperature of 22 ± 1°C, relative humidity of 50 ± 1%, and a light/dark cycle of 12/12 h. All animal studies (including the mice euthanasia procedure) were done in compliance with the regulations and guidelines of Jiaxing University institutional animal care and conducted according to the Association for Assessment and Accreditation of Laboratory Animal Care and the Institutional Animal Care and Use Committee guidelines.
Rat breast carcinoma cell line, Walker-256 cell, was purchased from the Cell Bank of Type Culture Collection of Chinese Academy of Sciences (Shanghai, China). Walker-256 cells were grown in Dulbecco’s modified Eagle medium containing 4500 mg/L glucose, 100 mg/L penicillin, and 100 mg/L streptomycin and were supplemented with 10% fetal bovine serum at 37°C. Cells were collected following enzymatic digestion, centrifuged, and resuspended in phosphate-buffered saline (PBS) in a concentration of 5 × 107 cells/mL.27 The animals were anesthetized with 4% chloral hydrate (0.4 g/kg, i.p.). An arthrotomy was done to expose the condyles of the distal tibia. PBS containing 1 × 106 Walker-256 cells (10 μL) was injected into the intramedullary space of the lefttibia with a 30-gauge needle, and the injection site was sealed with bone wax. Sham control rats were injected with same quantity of heat-inactivated Walker-256 cells.
Radiographic imaging
Radiographic observation was performed to confirm cancer development in the tibia and to evaluate the destructive effect of the bone cancer. The rats were anesthetized with 4% chloral hydrate (0.4 g/kg, i.p.), placed in a prone position, and divided into several groups as follows: CIBP group and Sham group 1, 3, 5, 7, 9, 12, and 15 days post tumor cell injection, radiographic images were taken (exposure setting: 12 ms, 31 KVp) using a Philips Digital Radiographer System (Digital Diagnost VM; Philips Medical Systems DMC GmbH, Hamburg, Germany). The operator was blinded to the treatment of the samples.
Sciatic functional index
Damage of restriction-free mobile capability in the left hind limb was monitored by analysis of the free walking pattern according to de Medinaceli et al. 2 Following the example of Riffel et al. 28 and introducing slight changes, the rats’ footprints were measured as follows: (1) distance from the heel to the third toe, the print length (PL); (2) distance from the first to the fifth toe, the toe spread (TS); and (3) distance from the second to the fourth toe, the intermediate toe spread (ITS). These three measurements were both obtained from the surgical sides (E, experimental) and normal (N) sides. Several prints of each foot were adopted on each track, and only three prints were used to determine the mean of measurements on the experimental and normal sides. These mean values were included in the formula for the sciatic functional index (SFI) and femoral nerve functional index: SFI = −38.3 (EPL − NPL)/NPL + 109.5 (TS − NTS)/NTS + 13.3 (EIT − NIT)/NIT − 8.8. These data were regarded as an indication of the functional condition of the sciatic and femoral nerve, where zero (0±11) represented the normal function and about −100 represented the loss of function resulting from Walker-256 cell inoculation. The SFI was monitored in all rats at day 1, 3, 5, 7, 9, 12, 15, and 18 post inoculation. These tests were conducted at the same time of the day (09:00 a.m.) by the same operator.
Drugs and administration
The MCP-1 neutralizing antibody was purchased from Boster (Shanghai, China). RS504393, an effective and selective antagonist of CCR2, was purchased from Boster (Shanghai, China). BAY11–7082, a NF-кB inhibitor, was purchased from Abcam (Abcam, Cambridge, England). Intrathecal injection was performed between the L4 and L5 intervertebral space using 30-gauge needle to deliver the reagents to the cerebral spinal fluid. 29
Behavioral analysis
SD rats were daily habituated to testing environment for at least two days before baseline testing. The testing environment temperature remained stable at 22 ± 2°C for all experiments. For detecting mechanical sensitivity, animals were put into plastic boxes, which were set on an elevated metal mesh floor, and allowed 30 min for habituation before test. The plantar surface of left hind paws, stimulated with von Frey hairs (Stoelting, Wood Dale, IL), presented perpendicularly to the plantar surface (2–3 s for each time). The paw withdrawal threshold (PWT) was determined using Dixon’s up-down method; three values were obtained from each animal each time; and the mean was used as the value of PWT.30
Immunofluorescence
At peak effects, four rats were selected from each group. Briefly, animals were deeply anesthetized with sevoflurane and their chests were opened. The heart was punctured with an intravenous infusion needle toward ascending aorta, then perfused with PBS 500 ml followed by 250 ml 4% paraformaldehyde with 1.5% picric acid in 0.16 M PB. After the perfusion, L3–L5 spinal cord segments were removed and fixed in the same fixative solution overnight. On the following day, the spinal cord segments were put into 30% sucrose solution till they sunk to the bottom and then were put into 15% sucrose solution till they sunk to the bottom. Spinal cord sections (20 μm, free-floating) were cut in a cryostat microtome (Leica CM1900), and five nonadjacent sections were randomly selected for immunofluorescence staining. The sections were blocked with 2% donkey serum for 1 h at room temperature and then incubated at 4°C overnight with the following primary antibodies: MCP-1 antibody (Rabbit, 1:100; Boster), CCR2 antibody (Rabbit, 1:100; Boster), phospho-NF-кB p65 (Ser536) (pNF-кB) antibody (Rabbit, 1:500; Sigma), GFAP antibody (Mouse, 1:1,000; Abcam), OX42 antibody (Mouse, 1:500, Abcam), NeuN antibody (Mouse, 1:1,000, Abcam). On the next day, the sections were rinsed three times with PBS solution. Afterward, the sections were incubated for 1 h at room temperature with Alexa Fluor-488 (Donkey anti-rabbit, 1:500, Abcam) or Alexa Fluor-594 (Donkey anti-mouse, 1:500, Abcam) secondary antibodies. For double immunofluorescence, the spinal cord sections were incubated with a mixture of primary antibodies at 4°C overnight followed by 1-h incubation with a mixture of Alexa Fluor-488 or Alexa Fluor-594 secondary antibodies. The slides were then kept in dark place and rinsed three times with PBS. At last, the stained sections were observed with an Olympus fluorescence microscope, and images were captured with a CCD Spot camera. The specificity of MCP-1 and CCR2 primary antibodies were tested by preabsorption experiments. 31
Western blot
At the peak effects, four rats were selected from each group, animals were deeply anesthetized with intraperitoneal injection of 4% chloral hydrate (400 mg/kg). The L3–L5 spinal cord segments were removed and sectioned in a cryostat microtome. The spinal dorsal horn segments were collected using a 15-gauge cannula and then frozen at −80°C until further used. Samples were homogenized on ice in 15 mmol/l Tris buffer containing a cocktail of proteinase inhibitors and phosphatase inhibitors. Protein samples were separated by gel electrophoresis (sodium dodecyl sulfate-poly-acrylamide gel electrophoresis) and transferred onto a polyvinylidene fluoride membrane. The blots were placed in blocking buffer for 1 h at room temperature and incubated with primary antibody against IL-10 (Boster 1:1000), tumor necrosis factor-α (TNF-α; Boster 1:1000), interferon-γ (IFN-γ; Boster 1:1000), MCP-1 (Abcam 1:1000), CCR2 (Biorbyt 1:1000), NF-кB p65 (Abcam 1:1000), p-NF-кB p65 (Abcam 1:1000), and glyceraldehyde-3-phosphate dehydrogenase (GAPDH; Boster 1:2000) overnight at 4°C. The blots were then incubated with horseradish peroxidase–conjugated immunoglobulin G. Electro-chemiluminescence (Pierce, Rockford, IL) was used to detect the immune complex. The band intensity was quantified with computer-assisted imaging analysis system (ImageJ, National Institutes of Health, Bethesda, MD).
Quantitative reverse transcription PCR
Total RNAs were extracted from L3–L5 spinal cord with the Trizol reagent (Invitrogen, Carlsbad, CA). One microgram of total RNA was converted into cDNA using Prime Script RT reagent kit (Takara, Shiga, Japan). The cDNA was amplified using the following primers: MCP-1 forward, 5′-CTGTAGCATCCACGTGCTGT-3′; MCP-1 reverse, 5′-GGTGCTGAAGTCCTTAGGGT-3′; CCR2 forward, 5′-GGGCTTTATCACATTGGGTAT-3′; CCR2 reverse, 5′-CCACCACCCAAGTGACTACA-3′; β-actin forward, 5′-AGTACAACCTTCTTGCAGCTC-3′; β-actin reverse, 5′-TGACCCATACCCACCATCAC-3′. The SYBR Premix Ex Taq™ II kit (Takara) was used for all PCR reactions, which were run on a Rotor-Gene 6000 RT-PCR machine (Hamburg, Germany). The PCR amplifications were performed at 95°C for 30 s, followed by 45 cycles at 95°C for 5 s, 56°C for 30 s, and 72°C for 30 s. The melting curves were performed to validate the utility and specificity of each PCR product. The data were analyzed using Rotor-Gene 6000 series software and evaluated using the Comparative CT Method (2−ΔΔCT).
Quantification and statistics
Data were expressed as mean ± standard deviation, all statistical analyses were carried out using SPSS 17.0 (SPSS). Statistical comparisons were performed by one-way analysis of variance (ANOVA) followed by the Student–Newman–Keuls test. p < 0.05 was considered statistically significant. For Western blot, the intensity of specific bands was measured with Image J. The protein expression levels were normalized to loading control (GAPDH). 32 For the immunofluorescence, four to five sections from the L4–L5 spinal cord segments were randomly selected. An image in a square on the medial two-thirds of the superficial dorsal horn (laminae I–III) was captured under × 10 and × 20 objectives. 33
Results
Intramedullary inoculation of breast carcinoma cells Walker-256 induces the destruction of rats’ tibia cortical bone, mechanical allodynia, and nerve functional destruction
Walker-256 breast tumor cells were inoculated into the intramedullary space of SD rat’s left tibia metaphysis. Briefly, time-dependent destruction of cortical bone in the left tibia and gradual loss of medullary bone were observed on 7th to 17th day post cell injection (Figure 1). Bone destruction was assessed on a scale of 0 to 534,35: 0 = normal bone structure without any sign of deterioration; 1 = small radiolucent lesions in the proximal epiphysis (<3); 2 = increased number of radiolucent lesions (>3) indicating loss of medullary bone; 3 = loss of medullary bone, plus erosion of the cortical bone; 4 = full-thickness unicortical bone loss; and 5 = full-thickness bicortical bone loss and displaced fracture. Furthermore, no radiological changes were observed in the right tibia (Figure 1) or in the control animals injected with heat-inactivated Walker-256 cells. Pain behavioral test demonstrated that inoculated tumor cell induced progressive pain hypersensitivity, which was represented by mechanical allodynia (painful response to an uninjurious mechanical stimulation) in the left hind paws of inoculated rat. For mechanical sensitivity, the PWT of the ipsilateral paw, in response to von Frey hair stimulation, decreased from 32.9 ± 1.0 g (before inoculation) to 20.32 ± 1.68 g (day 7 post inoculation) (p < 0.01), 16.61 ± 1.34 g (day 9 post inoculation) (p < 0.01), 10.16 ± 0.79 g (day 15 post inoculation) (p < 0.01), and 7.01 ± 1.23 g (day 21 post inoculation) (p < 0.01, Figure 2(a)), indicating the progressive development of mechanical allodynia. Nevertheless, the paws of Sham-treated rats did not show conspicuous changes in mechanical allodynia (Figure 2(a)). In addition, tumor invasion caused the destruction of the cortical bone and neurological damage. The nerve functional destruction was evaluated by SFI. SFI values for baseline (BL) were about zero (0 ± 11), indicating normal sciatic nerve function (Figure 2(b)). The values were close to −100, indicating complete loss of sciatic nerve function. Briefly, from day 1 to day 5 after inoculation, the SFI values showed no significant difference among CIBP rats, and no significant change was found in the contralateral. However, five days after modeling, a decrease was observed in SFI values. Compared to the values before modeling (BL), the SFI value decreased approximately 70% in CIBP rats inoculated with Walker-256 cells, which in turn indicated that Walker-256 cells induced bone destruction and damaged the function of sciatic and femoral nerve.
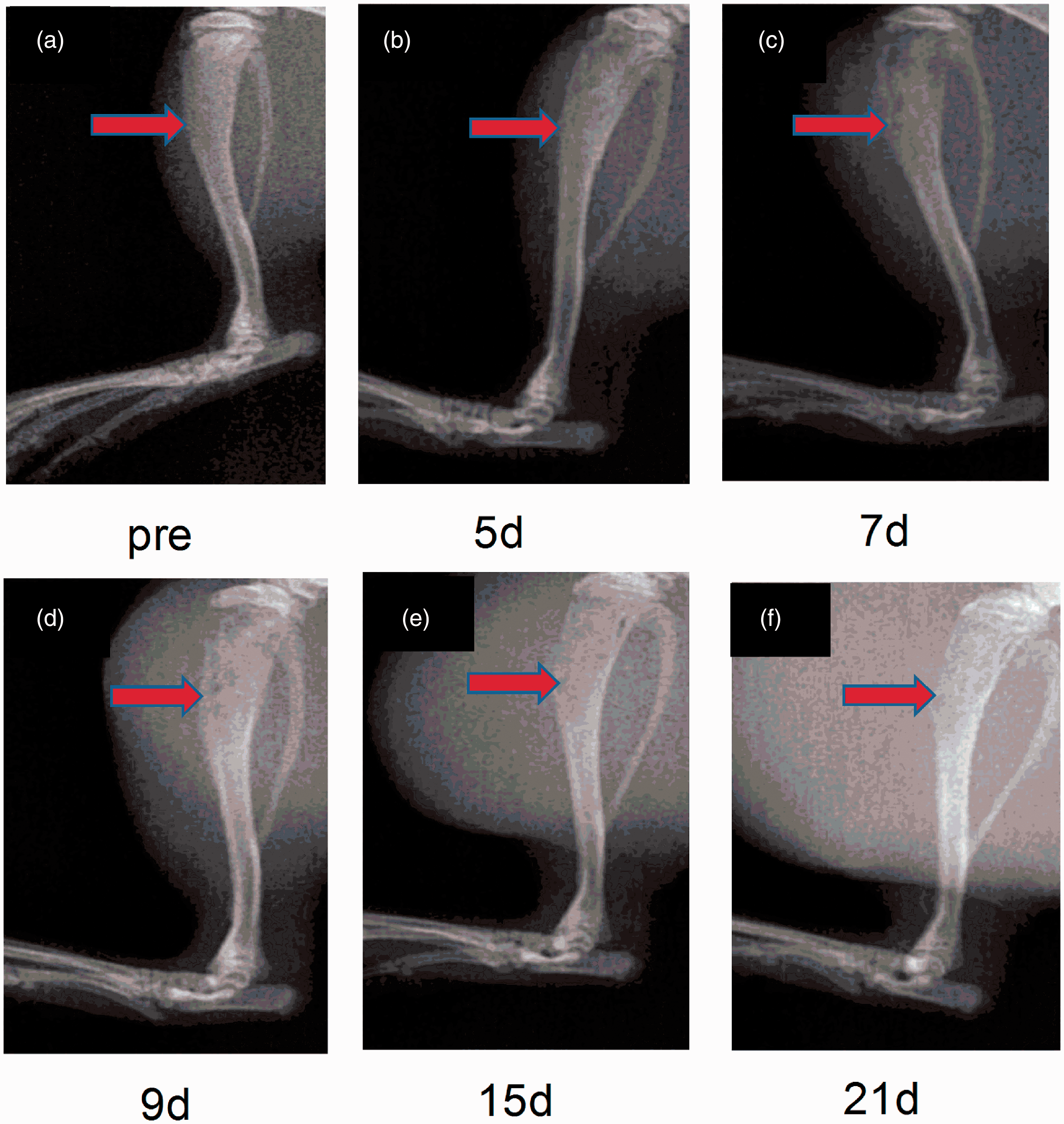
Walker-256 cell inoculation induced the destruction of rats’ tibia cortical bone. (a–f) Radiographic images of bone structure on day 0 (preinoculation), 5, 7, 9, 15, 17, 21 post cell injection. In the distal, one-third of the left tibia (arrows) bone destruction was assessed on a scale of 0–5: 0 = normal bone structure without any sign of deterioration; 1 = small radiolucent lesions in the proximal epiphysis (<3); 2 = increased number of radiolucent lesions (>3) indicating loss of medullary bone; 3 = loss of medullary bone, plus erosion of the cortical bone; 4 = full-thickness unicortical bone loss; and 5 = full-thickness bicortical bone loss and displaced fracture. (g) No radiological change was observed in the right tibia.

Walker-256 cells inoculation induced CIBP and the destruction of rats’ sciatic nerve and femoral nerve. (a) Behavioral tests showed that rats displayed mechanical allodynia in the ipsilateral paw after cell inoculation. The PWT progressively decreased from day 5 to day 15. *p < 0.05, **p < 0.01 vs. Sham-ipsi. One-way ANOVA followed by Newman–Keuls test. n = 10 rats per group. (b) Behavioral tests showed that rats displayed destruction of sciatic nerve and femoral nerve in the ipsilateral paw after cell inoculation. CIBP: cancer-induced bone pain.
MCP-1 and CCR2 were continuously increased in spinal cord after Walker-256 cell inoculation
qRT-PCR was performed to examine the MCP-1/CCR2, axis expression in the spinal cord. As shown in Figure 3(a) and (b), MCP-1 and CCR2 mRNA expression was significantly increased on day 9 (p < 0.01) and day 15 (p < 0.01) post cell inoculation, while no changes were observed in the Sham group. Second, MCP-1 and CCR2 protein expressions were examined using Western blot. Tumor cell inoculation induced a significant increase of MCP-1 and CCR2 protein expression in the affected side spinal cord on day 9 and day 15 post cell inoculation (Figure 3(c) to (g)). Furthermore, to understand the cellular distribution of MCP-1 and CCR2, we performed immunofluorescence double staining of MCP-1 and CCR2 with astrocyte, microglia or neuron cell, respectively. The results showed that MCP-1 was upregulated in the spinal cord of CIBP rats and colocalized with NeuN (neuronal marker) but neither with GFAP (astrocyte marker) nor with OX42 (microglial marker) (Figure 4(a) to (i)). While CCR2 was colocalized with NeuN and OX42 but not with GFAP (Figure 5(a) to (i)). To sum up, tumor cell inoculations induced spinal cord neurons to express MCP-1, while CCR2 was expressed by spinal cord neurons and microglial.

Walker-256 cell inoculation induces MCP-1 and CCR2 expressed increase in spinal cord. (a and b) RT-PCR results show the increase of MCP-1 mRNA and CCR2 mRNA level in the spinal cord after inoculation. (c and d) Western blotting results indicate the increase of MCP-1 and CCR2 protein expression in the spinal cord after inoculation. MCP-1 and CCR2 protein level was gradually increased from day 1 to day 15. *p < 0.05, **p < 0.01 vs. Sham control group. n = 4 rats per group. (e–g) Immunofluorescence staining showing the expression and distribution of MCP-1 (green). Original magnification: 200×. CCR2: chemokine CC motif receptor-2; CIBP: cancer-induced bone pain; GAPDH: glyceraldehyde-3-phosphate dehydrogenase; MCP-1: monocyte chemoattractant protein-1.

(a–i) Immunofluorescence double staining showed that MCP-1 (green) was upregulated in the spinal cord of CIBP rats and was colocalized with neuronal marker, NeuN (red), but neither with astrocyte marker GFAP (red) nor with microglial marker OX42 (red). Original magnification: 100×, scale bar: 200 µm.

Immunofluorescence double staining showed that CCR2 (green) was upregulated in the spinal cord of CIBP rats and was colocalized with neuronal marker, NeuN (red), and microglial marker OX42 (red) but not with astrocyte marker GFAP (red). Original magnification: 100×, scale bar: 200 µm.
MCP-1/CCR2 axis participates in CIBP and mediates the expression of TNF-α, IFN-γ, and IL-4
To investigate the role of endogenous MCP-1/CCR2 axis in CIBP, MCP-1 neutralizing antibody was intrathecally administered during day 9 post modeling and behavioral changes after pain were examined. MCP-1 neutralizing antibody at the dose of 2 μg partly relieved mechanical allodynia at 0.5 h (p < 0.05), 1 h (p < 0.01), 2 h (p < 0.01), and 3 h (p < 0.05) post injection. High dose (10 μg) of MCP-1 neutralizing antibody almost reversed the trend of mechanical allodynia for 0.5 h, 1 h, 2 h, and persistent to 3 h until 4 h post injection (Figure 6(a)). Consequently, an effective and highly selective CCR2 antagonist, RS504393 (25 μg and 50 μg) was intrathecally administered on day 9 post Walker-256 cell injection. Mechanical allodynia was modestly reduced by RS504393 at the dose of 25 μg (BR1), while it was significantly attenuated using higher dose (50 μg) of RS504393 (BR2) at 0.5 h to 3 h, lasting until 4 h post injection (Figure 7(a)). In addition, we detected the expression of TNF-α, IFN-γ, and interleukin-4 (IL-4) after intrathecal administration of MCP-1 neutralizing antibody and CCR2 antagonist (RS504393) on day 9 post modeling using Western blot. MCP-1 neutralizing antibody at the dose of 2 μg partly decreased the expression of TNF-α (p < 0.05) and IFN-γ (p < 0.05) but increased the expression of IL-4 (p < 0.05). High dose (10 μg) of MCP-1 neutralizing antibody significantly diminished the expression of TNF-α (p < 0.01) and IFN-γ (p < 0.01) but dramatically elevated the expression of IL-4 (p < 0.01) (Figure 6(b) to (d)). While CCR2 antagonist RS504393 at the dose of 25 μg (BR1) modestly decreased the expression of TNF-α (p < 0.05) and IFN-γ (p < 0.05), it upregulated the expression of IL-4 (p < 0.05). Moreover, higher dose (50 μg) of CCR2 antagonist RS504393 (BR2) downregulated the expression of TNF-α (p < 0.01), IFN-γ (p < 0.01) more significantly, and augment the expression of IL-4 (p < 0.01) more dramatically (Figure 7(b) to (d)). These results indicated that MCP-1/CCR2 axis was involved in CIBP, a mediator of TNF-α, IFN-γ, and IL-4 which in tumor cell inoculation induced pain hypersensitivity models.

Intrathecal injection of MCP-1 neutralizing antibody attenuated bone cancer pain and affected the expression of inflammation factors. (a) MCP-1 neutralizing antibody at a lower dose (2 μg) (BM1) had mild effect on Walker-256 cell inoculation-induced pain hypersensitivity, whereas the neutralizing antibody at a higher dose (10 μg) (BM2) reversed inoculation-induced mechanical allodynia for more than 4 h. *p < 0.05, **p < 0.01, vs. CIBP+PBS group. n = 12 rats per group. (b–d) A lower dose (2 μg) (BM1) of MCP-1 neutralizing antibody had a slight effect on pro-inflammation cytokine, and a higher dose (10 μg) (BM2) had a much more dramatic effect. #p < 0.05, ##p < 0.01, vs. Sham control *p < 0.05, **p < 0.01, vs. CIBP+PBS group. n = 4 rats per group. CIBP: cancer-induced bone pain; PBS: phosphate-buffered saline; GAPDH: glyceraldehyde-3-phosphate dehydrogenase; TNF-α: tumor necrosis factor-α; IL-4: interleukin-4; IFN-γ: interferon-γ.

Intrathecal injection of CCR2 inhibitor (RS504393) reduced bone cancer pain and affected the production of inflammation factors. (a) CCR2 antagonist (RS504393) at a lower dose (50 μg) had mild effect on Walker-256 cell inoculation-induced pain hypersensitivity, whereas the antagonist at a higher dose (100 μg) reversed inoculation-induced mechanical allodynia for more than 4 h. *p < 0.05, **p < 0.01, vs. control serum. n = 12 rats per group. (b–d) CCR2 antagonist (RS504393) at a lower dose (25 μg) had a slight effect on inflammation cytokine and at a higher dose (50 μg) had a much more dramatic effect. *p < 0.05, **p < 0.01, vs. control serum. n = 4 rats per group. CIBP: cancer-induced bone pain; DMSO: dimethyl sulfoxide; GAPDH: glyceraldehyde-3-phosphate dehydrogenase; TNF-α: tumor necrosis factor-α; IL-4: interleukin-4; IFN-γ: interferon-γ.
NF-кB signaling pathway is continuously upregulated and phosphorylated in spinal neurons
NF-κB is a ubiquitous transcription factor that serves as a transducer between extracellular signals and gene expression. Recent studies have shown that NF-κB signaling pathway is involved in CIBP; hence, we investigated the possible mechanisms. Western blotting and immunostaining staining showed that NF-κB and (p)NF-κB (pP65) expressions were persistently increased from day 1 to day 15 post Walker-256 cell injection (Figure 8(a) to (e)). Immunofluorescence double staining showed that (p)NF-κB (pP65) was increased in the spinal cord of CIBP rats and mainly colocalized with NeuN, which in turn suggested that CIBP induced NF-κB expression upregulation and phosphorylation (Figure 9).
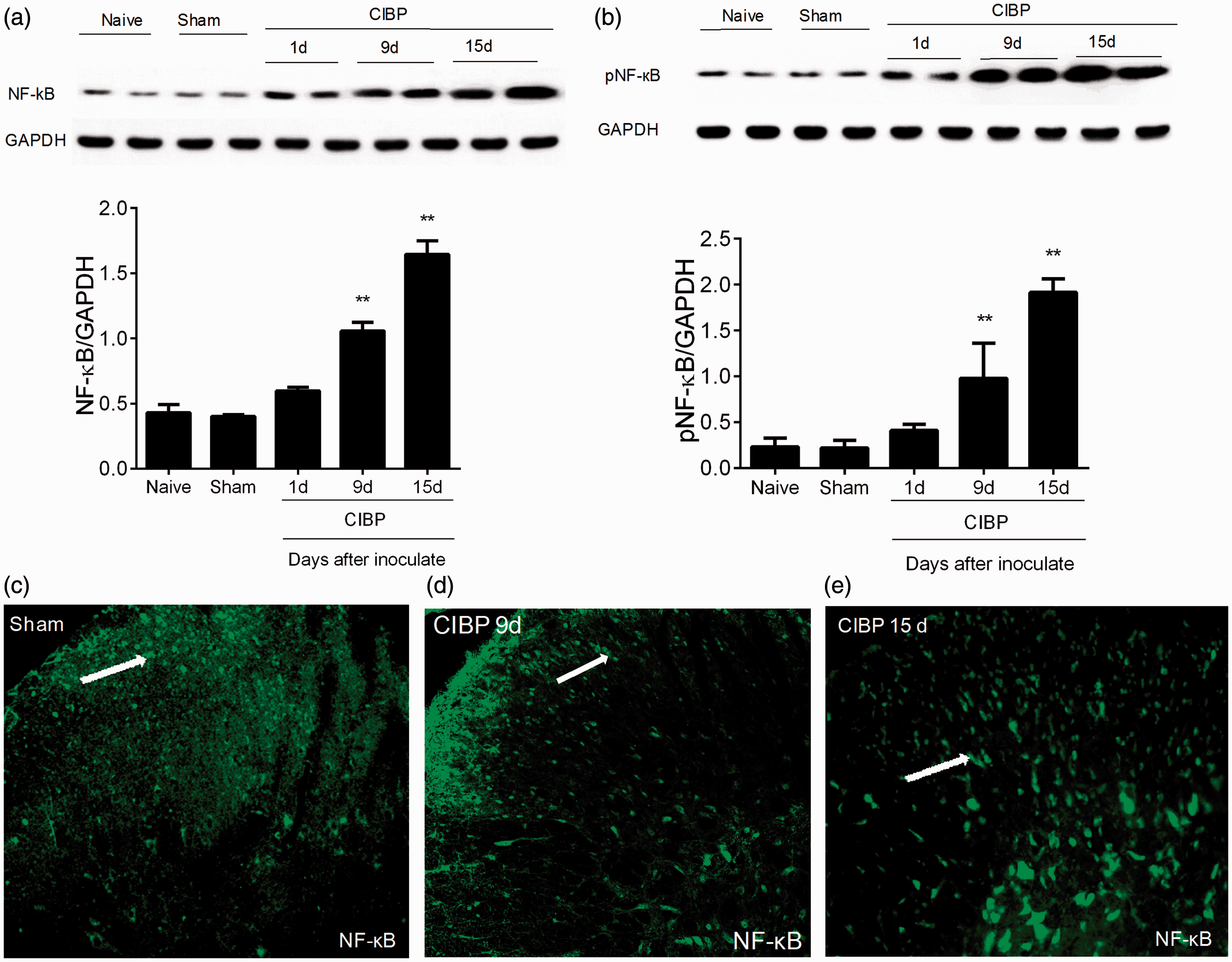
Walker-256 cell inoculation increased NF-кB and (p) NF-кB expression in spinal neutron. (a–e) Western blot showed that tumor cell inoculation increased NF-кB and (p) NF-кB expression in the spinal cord from day 1 to day 15 post cell injection. *p < 0.05, **p < 0.01 vs. Sham control group. One-way ANOVA followed by Newman–Keuls test was done for statistical analysis. n = 4 rats per group. (c–e) Immunostaining staining shows that (p) NF-кB (green) was expressed in the superficial dorsal horn in Sham animals. Original magnification: 200×. CIBP: cancer-induced bone pain; GAPDH: glyceraldehyde-3-phosphate dehydrogenase.
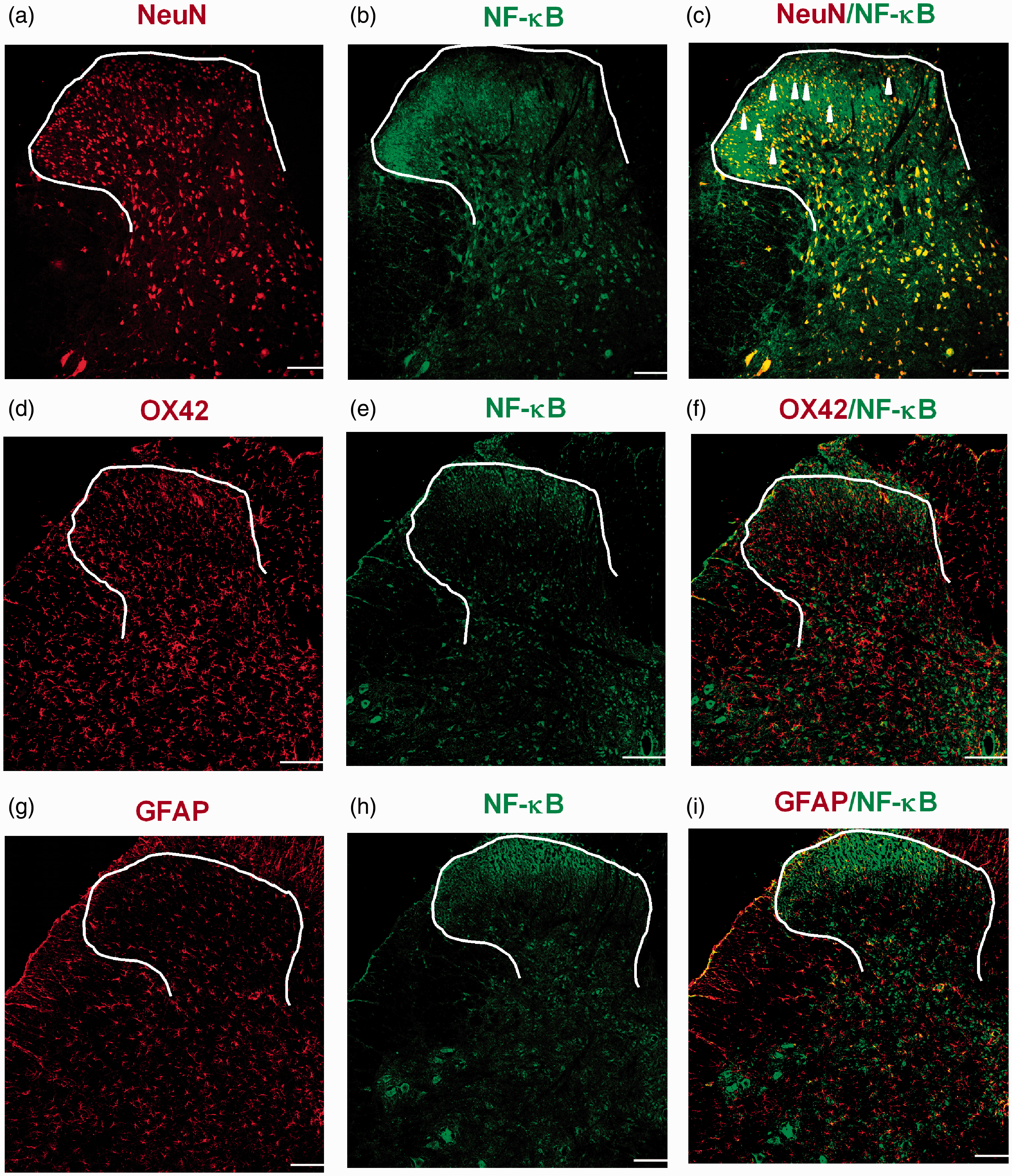
(a–i) Immunostaining double staining shows that (p) NF-кB (green) was colocalized with neuronal marker NeuN (red) but neither with astrocyte marker GFAP (red) nor with microglial marker OX42 (red). Original magnification: 100×, scale bar: 200 µm.
Inhibition of NF-кB attenuated CIBP and reduced MCP-1 and CCR2 upregulation in the spinal cord and influence the expression of TNF-α, IFN-γ, and IL-4
We further investigated the mechanism of NF-кB path way in CIBP. BAY11–7082, an effective and specific inhibitor of NF-кB, (25 μg, 50 μg, and 100 μg) was intrathecally injected on day 9 post tumor cell inoculation. Briefly, at the dose of 25 μg (BB1), BAY11–7082 had no effect on mechanical allodynia; 50 μg (BB2) of this inhibitor partly relieved mechanical allodynia at 0.5 h (p < 0.05) and 3 h (p < 0.05); while higher dose (100 μg) of BAY11–7082 (BB3) almost reversed the trend of mechanical allodynia for 0.5 h (p < 0.01), and the effect lasted to 4 h (Figure 10(a)). In addition, the production of MCP-1 and CCR2 in the spinal cord was reduced in dose-dependent way in animals injected with tumor cells: at the dose of 25 μg (BB1), BAY11–7082 had no effect on the expression of MCP-1 and CCR2 (p < 0.01) (Figure 10(b) to (e)); 50 μg (BB2) of this inhibitor partly reverse the upregulation of MCP-1 and CCR2; while higher dose (100 μg) of BAY11–7082 (BB3) almost reversed augmented levels of MCP-1 and CCR2. In addition, the inflammatory factors TNF-α, IFN-γ, and IL-4 were dose-dependently influenced by NF-кB inhibitor: BAY11–7082 at the dose of 25 μg (BB1) scarcely influenced the expression of TNF-α, IFN-γ, and IL-4; while at the dose of 50 μg (BB2) modestly reduced the expression of TNF-α and IFN-γ but increased the expression of IL-4 (p < 0.05); finally, high dose (100 μg) of BAY11–7082 (BB3) significantly decreased the expression of TNF-α and IFN-γ and dramatically facilitated the expression of IL-4 (p < 0.01) (Figure 10(f) to (h)). Furthermore, we detected the MCP-1 expression by immunostaining. Immunostaining result showed that NF-кB inhibitor (BAY11–7082) decreased the production of MCP-1 (Figure 10(i) and (j)). These data suggested that NF-кB signaling pathway participated in the pain hypersensitivity and had effect on the upregulation of MCP-1 and CCR2 expression as well the production of TNF-α, IFN-γ, and IL-4 in spinal cord induced by tumor cell inoculation.

NF-кB inhibitor BAY11–7082 decreased Walker-256 cell inoculation-induced pain hypersensitivity and downregulation of MCP-1, CCR2, and inflammation cytokines in the spinal cord. (a) Intrathecal injection of NF-кB inhibitor, BAY11–7082 at the dose of 25 μg (BB1) had no effect on mechanical allodynia, while higher doses, that is, 50 μg (BB2) and 100 μg (BB3) decreased mechanical allodynia at 1 h and 3 h. *p < 0.05, **p < 0.01, vs. CIBP+DMSO; n = 12 per group. (b–h) Intrathecal injection of NF-кB inhibitor, BAY11–7082 at the dose of 25 μg had no effect on MCP-1mRNA, MCP-1, CCR2, and inflammation cytokine IL-4, TNF-α, and α-IFN, while higher doses, that is, 50 μg and 100 μg downregulated MCP-1mRNA, MCP-1, CCR2, TNF-α, and IFN-γ, increased IL-4 level n = 4 per group. *p < 0.05, **p < 0.01, vs. CIBP+DMSO, #p < 0.05, ##p < 0.01, vs. Sham control group. (i and j) Immunostaining result showed that NF-кB inhibitor (BAY11–7082) decreased the production of MCP-1 (green). Original magnification: 200×. One-way ANOVA followed by Newman–Keuls test was used for statistical analysis. CIBP: cancer-induced bone pain; DMSO: dimethyl sulfoxide; GAPDH: glyceraldehyde-3-phosphate dehydrogenase; TNF-α: tumor necrosis factor-α; IL-4: interleukin-4; IFN-γ: interferon-γ; MCP-1: monocyte chemoattractant protein-1; CCR2: chemokine CC motif receptor-2.
Discussion
With global rising incidence of cancer, understanding the mechanisms behind CIBP and identifying novel therapeutic strategies are of crucial importance. To investigate the mechanisms behind CIBP, we established animal models by inoculating tumor cells (Walker-256 cell) derived from ascites in breast cancer rats into marrow cavity of tibia in female rats. 27 However, many other CIBP animal models were established using mice or male rats. In addition, in these studies, CIBP models were developed by injecting other tumor cell lines (e.g., osteolytic 2472 sarcoma, B16 melanoma, RM-1 prostate cancer) into bones (e.g., femur or tibia). Studies have shown that different animal models show different characteristics with reference to the extent of bone destruction, the severity, and the type of pain behaviors.36,37 Our approach, the use of animal models has certain advantages. First of all, Walker-256 cell induced obvious bone substance destruction of the tibia, and then serious and progressive mechanical allodynia, which showed its applicability for CIBP model. In addition, the tumor cells derived from the ascites of breast cancer rats were convenient and easy to save and spread. Moreover, they could successfully induce the success rate of modeling via inoculation of breast cancer cells into bones. Using this model, this study demonstrated that MCP-1 was dramatically increased in neurons of afferent nerve in the spinal cord of CIBP. Using MCP-1 neutralizing antibody attenuated inoculation-induced pain hypersensitivity. Second, CCR2, the major receptor of MCP-1 was also upregulated in spinal cord neurons and microglia after tumor cell injection. Intrathecal administration of CCR2 antagonist alleviated CIBP. Third, NF-кB was activated in spinal neurons and it was involved in MCP-1 production following inoculation. Inhibition of NF-кB attenuated CIBP and reduced MCP-1 upregulation in the spinal cord. These results revealed that NF-кB and MCP-1/CCR2 signal pathway played a momentous role in the generation and maintenance of CIBP by activating the neuron of afferent nerve in the spinal cord. Based on the results presented in the current study, the mechanisms for central MCP-1/CCR2 axis in the development of pain hypersensitivity in the CIBP model are proposed at Supplementary Figure 1.
MCP-1/CCR2 axis regulates CIBP via mediating the expression of the pro-inflammatory and the anti-inflammatory factors
Despite the high incidence of CIBP, the mechanisms that initiate and maintain the pain are still not completely understood. Majority of investigators have agreed that CIBP contains components of both neuropathic and inflammatory pain, as well as distinctive characteristics that make it a unique pain state.38,39 When the tumor grows in the bone, it damages the surrounding tissue; produces a wide range of cytokines and growth factors; releases inflammatory mediators; and injures the sensory nerve fiber terminals in bone. All these factors contribute to the inflammatory 40 as well as the neuropathic 41 components of cancer-induced pain. 42 In this study, we found that intrathecal administration of MCP-1 neutralizing antibody and CCR2 antagonist (RS504393) reduced the expression of TNF-α and IFN-γ but increased the expression of IL-4. In recent study, TNF-α was reported as a critical pro-inflammatory factor, involved in many pathological processes such as inflammatory response, nerve injury, neuropathic pain, and CIBP. Our results are consistent with these data. On the other hand, we found that MCP-1/CCR2 signal pathway regulated the expression of IL-4, which is an important anti-inflammatory factor. In the early stage of CIBP, tumor cells invaded the bone and slightly increased IL-4. When we administrated MCP-1 neutralizing antibody or CCR2 inhibitor (RS504393), its expression significantly increased. In addition, the expression of IFN-γ, which is a pro-inflammatory factor accompanied by other inflammatory effects, increased at early phase of CIBP, and it was downregulated following the administration of MCP-1 neutralizing antibody or CCR2 inhibitor (RS504393). These results indicated that MCP-1/CCR2 axis might mediate the expression of TNF-α, IFN-γ, and IL-4 that participate in CIBP.
Therefore, MCP-1/CCR2 pathway may regulate CIBP through upregulation of pain-related proteins. In addition, we observed that MCP-1 increases NMDA-induced currents on lamina II neurons of the spinal cord (unpublished observation), suggesting MCP-1/CCR2 pathway may also be involved in the maintenance of central sensitization through direct regulation of neuronal excitability.
NF-кB through mediating MCP-1/CCR2 axis and contributes to CIBP
NF-кB regulates the transcription of many inflammatory mediators, including chemokines, adhesion molecules, and pro-inflammatory cytokines.43,44 NF-кB has been found to regulate CXCL1 transcription in Hs294T malignant melanoma cells. 45 In our study, NF-кB regulated MCP-1 production in spinal neurons. Furthermore, the activation of NF-кB was persistently increased in the spinal cord induced by tumor cell inoculation. Accumulating evidences have suggested that the activation of NF-кB following tissue injury or nerve damage was related to the development and maintenance of neuropathic pain.43,46 For example, using potential inhibitor BAY11–7082 or transgenic inhibition to reduce NF-кB activation in the DRG level or spinal level reduces pain behavior and inflammation after peripheral nerve injury and attenuates neuropathic pain.47,48 Our study showed that intrathecal injection of NF-кB inhibitor at ninth day after modeling attenuated tumor induced hypersensitivity and decreased MCP-1 and CCR2 level in spinal cord, supporting the involvement of NF-кB in the development and maintenance of CIBP via MCP-1 and CCR2 produce in the spinal cord. Furthermore, our results showed NF-кB to be predominantly expressed in neurons of the spinal cord after modeling. Our results showed that BAY11–7082 reduced the expression of TNF-α and IFN-γ but increased the expression of IL-4. It indicated that NF-кB regulated the expression of TNF-α, IFN-γ, and IL-4 via MCP-1/CCR2 axis. In addition, the expression of (p)NF-кB was also increased. Accordingly, (p)NFκB expression was observed in the medullary dorsal horn after the inferior alveolar nerve injury and the astrocytes in the spinal cord after spinal nerve injury.48,49 Also, activated NF-кB is also found in macrophages/microglia in the spinal cord after spinal cord injury 50 or nerve-compression model rat. 51 The disparate distribution of NF-кB in the spinal cord may be due to different antibodies that contribute to biodiversity, which needs further investigation in the future.
NF-кB signaling pathway regulated the production of MCP-1/CCR2 axis in spinal cord of CIBP rats. Moreover, MCP-1/CCR2 may participate in the generation of CIBP by influencing the production of inflammatory factors.
Supplemental Material
Supplementary figure 1 -Supplemental material for Nuclear factor kappa B regulated monocyte chemoattractant protein-1/chemokine CC motif receptor-2 expressing in spinal cord contributes to the maintenance of cancer-induced bone pain in rats
Supplemental material, Supplementary figure 1 for Nuclear factor kappa B regulated monocyte chemoattractant protein-1/chemokine CC motif receptor-2 expressing in spinal cord contributes to the maintenance of cancer-induced bone pain in rats by Yungong Wang, Huadong Ni, Hongbo Li, Houshen Deng, Long S Xu, Shijie Xu, Ying Zhen, Hui Shen, Huan Pan and Ming Yao in Molecular Pain
Footnotes
Acknowledgment
We thank Professor Renshan Ge, a native speaker, the former head of the Endocrinology Lab of the CBR in Population Council (USA) for English language editing.
Author Contributions
Yungong Wang carried out the animal surgery, behavioral testing, immunofluorescence, Western blot experiments, PCR experiments, and X-ray scan. Ming Yao participated in designing the experiments, conceived the project, and coordinated and supervised the experiments. Huadong Ni, Shijie Xu, Long S Xu, and Ying Zhen participated in the animal surgery and behavioral testing. Hui Shen and Huan Pan participated in immunofluorescence. Huadong Ni, Hongbo Li, and Houshen Deng wrote the article. All authors read and approved the final manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the National Natural Science Foundation of China (81341035 and 81171057), Natural Science Foundation of Zhejiang Provincial (LY16H090016 and LY17H090019), Zhejiang provincial and Ministerial Cultivation Plan for Medicine and Health (2015PYA010), Zhejiang Provincial Program for the Cultivation of High-level Innovative Health Talents (2012-RC-22), the Construction Project of Anesthesiology Discipline Special Disease Center in Zhejiang North Region (201524), and Jiaxing city science and technology project (2017AY33008).
Supplemental Material
Supplementary material is available for this article online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
